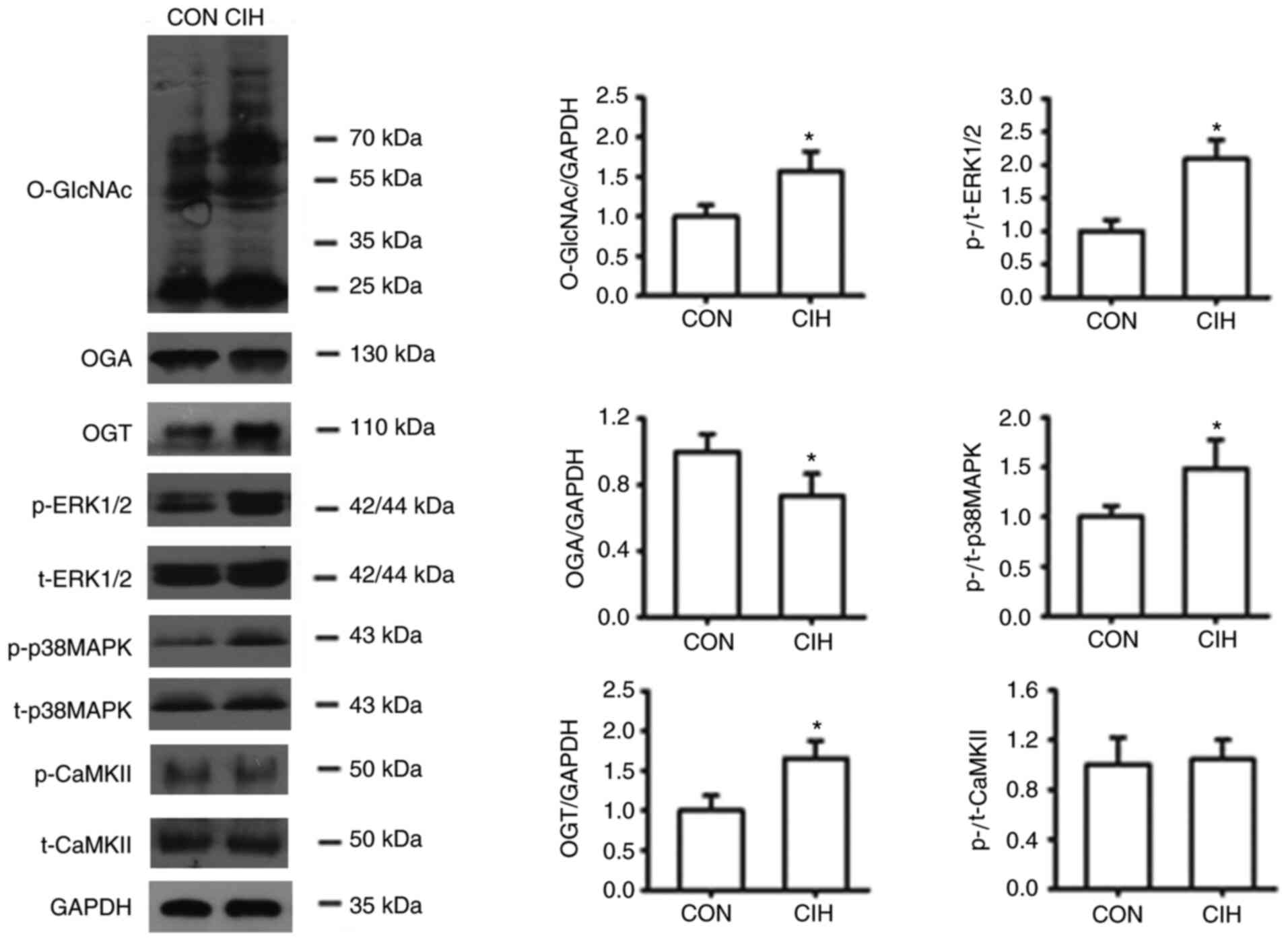
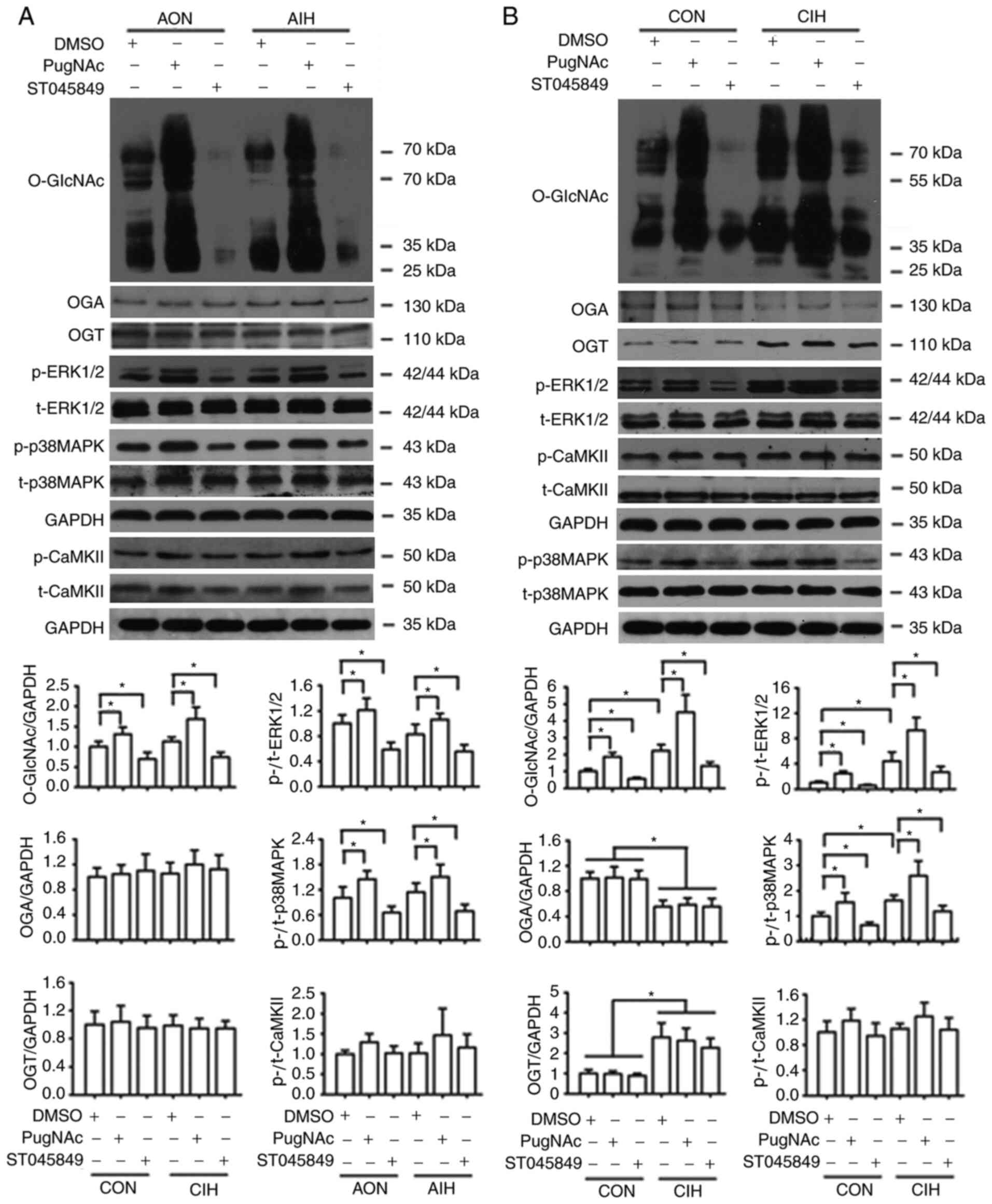
|
1
|
Foster GE, Poulin MJ and Hanly PJ:
Intermittent hypoxia and vascular function: Implications for
obstructive sleep apnoea. Exp Physiol. 92:51–65. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cai AP, Wang L and Zhou YL: Hypertension
and obstructive sleep apnea. Hypertens Res. 39:391–395. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Krause BJ, Casanello P, Dias AC, Arias P,
Velarde V, Arenas GA, Preite MD and Iturriaga R: Chronic
intermittent hypoxia-induced vascular dysfunction in rats is
reverted by-acetylcysteine supplementation and arginase inhibition.
Front Physiol. 9:901–912. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang YN, Zhang CL, Li HO and Hou JD:
Down-regulation of vascular PPAR-γ contributes to endothelial
dysfunction in high-fat diet-induced obese mice exposed to chronic
intermittent hypoxia. Biochem Biophys Res Commun. 492:243–248.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guo XL, Deng Y, Shang J, Liu K, Xu YJ and
Liu HG: ERK signaling mediates enhanced angiotensin II-induced rat
aortic constriction following chronic intermittent hypoxia. Chin
Med J (Engl). 126:3251–3258. 2013.PubMed/NCBI
|
|
6
|
Nicholl DDM, Hanly PJ, Zalucky AA, Mann
MC, MacRae JM, Poulin MJ, Handley GB, Sola DY and Ahmed SB: CPAP
therapy delays cardiovagal reactivation and decreases arterial
renin-angiotensin system activity in humans with obstructive sleep
apnea. J Clin Sleep Med. 14:1509–1520. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Allahdadi KJ, Walker BR and Kanagy NL: ROK
contribution to endothelin-mediated contraction in aorta and
mesenteric arteries following intermittent hypoxia/hypercapnia in
rats. Am J Physiol Heart Circ Physiol. 293:H2911–H2918. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Durgan DJ, Crossland RF, Lloyd EE,
Phillips SC and Bryan RM: Increased cerebrovascular sensitivity to
endothelin-1 in a rat model of obstructive sleep apnea: A role for
endothelin receptor B. J Cereb Blood Flow Metab. 35:402–411. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Snow JB, Norton CE, Sands MA, Weise-Cross
L, Yan S, Herbert LM, Sheak JR, Gonzalez Bosc LV, Walker BR, Kanagy
NL, et al: Intermittent hypoxia augments pulmonary vasoconstrictor
reactivity through PKCβ/mitochondrial oxidant signaling. Am J
Respir Cell Mol Biol. 62:732–746. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chatham JC, Zhang JH and Wende AR: Role of
O-linked N-acetylglucosamine (O-GlcNAc) protein modification in
cellular (patho)physiology. Physiol Rev. 101:427–493. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schwein PA and Woo CM: The O-GlcNAc
modification on kinases. ACS Chem Biol. 15:602–617. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wright JN, Collins HE, Wende AR and
Chatham JC: O-GlcNAcylation and cardiovascular disease. Biochem Soc
Trans. 45:545–553. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jensen RV, Andreadou I, Hausenloy DJ and
Bøtker HE: The role of O-GlcNAcylation for protection against
ischemia-reperfusion injury. Int J Mol Sci. 20:404–424. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guo XL, Shang J, Deng Y, Yuan X, Zhu D and
Liu HG: Alterations in left ventricular function during
intermittent hypoxia: Possible involvement of O-GlcNAc protein and
MAPK signaling. Int J Mol Med. 36:150–158. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lima VV, Giachini FR, Choi H, Carneiro FS,
Carneiro ZN, Fortes ZB, Carvalho MH, Webb RC and Tostes RC:
Impaired vasodilator activity in deoxycorticosterone acetate-salt
hypertension is associated with increased protein O-GlcNAcylation.
Hypertension. 53:166–174. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lima VV, Giachini FR, Hardy DM, Webb RC
and Tostes RC: O-GlcNAcylation: A novel pathway contributing to the
effects of endothelin in the vasculature. Am J Physiol Regul Integr
Comp Physiol. 300:R236–R250. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lima VV, Giachini FR, Carneiro FS,
Carvalho MH, Fortes ZB, Webb RC and Tostes RC: O-GlcNAcylation
contributes to the vascular effects of ET-1 via activation of the
RhoA/Rho-kinase pathway. Cardiovasc Res. 89:614–622. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
van der Laarse SAM, Leney AC and Heck AJR:
Crosstalk between phosphorylation and O-GlcNAcylation: friend or
foe. FEBS J. 285:3152–3167. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kumar GK and Prabhakar NR:
Post-translational modification of proteins during intermittent
hypoxia. Respir Physiol Neurobiol. 164:272–276. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hilgers RHP, Xing DQ, Gong KZ, Chen YF,
Chatham JC and Oparil S: Acute O-GlcNAcylation prevents
inflammation-induced vascular dysfunction. Am J Physiol Heart Circ
Physiol. 303:H513–H522. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lima VV, Rigsby CS, Hardy DM, Webb RC and
Tostes RC: O-GlcNAcylation: A novel post-translational mechanism to
alter vascular cellular signaling in health and disease: Focus on
hypertension. J Am Soc Hypertens. 3:374–387. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Erickson JR, Pereira L, Wang L, Han G,
Ferguson A, Dao K, Copeland RJ, Despa F, Hart GW, Ripplinger CM and
Bers DM: Diabetic hyperglycaemia activates CaMKII and arrhythmias
by O-linked glycosylation. Nature. 502:372–376. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Giachini FR, Sullivan JC, Lima VV,
Carneiro FS, Fortes ZB, Pollock DM, Carvalho MHC, Webb RC and
Tostes RC: Extracellular signal-regulated kinase 1/2 activation,
via downregulation of mitogen-activated protein kinase phosphatase
1, mediates sex differences in desoxycorticosterone acetate-salt
hypertension vascular reactivity. Hypertension. 55:172–179. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yousif MHM, Akhtar S, Walther T and Benter
IF: Role of Ca2+/calmodulin-dependent protein kinase II
in development of vascular dysfunction in diabetic rats with
hypertension. Cell Biochem Funct. 26:256–263. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Federici M, Menghini R, Mauriello A,
Hribal ML, Ferrelli F, Lauro D, Sbraccia P, Spagnoli LG, Sesti G
and Lauro R: Insulin-dependent activation of endothelial nitric
oxide synthase is impaired by O-linked glycosylation modification
of signaling proteins in human coronary endothelial cells.
Circulation. 106:466–472. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Goldberg H, Whiteside C and Fantus IG:
O-linked β-N-acetylglucosamine supports p38 MAPK activation by high
glucose in glomerular mesangial cells. Am J Physiol Endocrinol
Metab. 301:E713–E726. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jiang MZ, Qiu ZY, Zhang S, Fan X, Cai XQ,
Xu B, Li XW, Zhou JF, Zhang XY, Chu Y, et al: Elevated
O-GlcNAcylation promotes gastric cancer cells proliferation by
modulating cell cycle related proteins and ERK 1/2 signaling.
Oncotarget. 7:61390–61402. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang XL, Ma LN, Qi JQ, Shan H, Yu WG and
Gu YC: MAPK/ERK signaling pathway-induced hyper-O-GlcNAcylation
enhances cancer malignancy. Mol Cell Biochem. 410:101–110. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Institute for Laboratory Animal and
Research, . Guide for the Care and Use of Laboratory Animals. 8th
edition. National Academies Press; Washington, DC: 2011
|
|
30
|
Li JR, Zhao YS, Chang Y, Yang SC, Guo YJ
and Ji ES: Fasudil improves endothelial dysfunction in rats exposed
to chronic intermittent hypoxia through RhoA/ROCK/NFATc3 pathway.
PLoS One. 13:e01956042018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ishihata A, Tasaki K and Katano Y:
Involvement of p44/42 mitogen-activated protein kinases in
regulating angiotensin II- and endothelin-1-induced contraction of
rat thoracic aorta. Eur J Pharmacol. 445:247–256. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang CJ, Zhou ZG, Holmqvist A, Zhang H, Li
Y, Adell G and Sun XF: Survivin expression quantified by Image
Pro-Plus compared with visual assessment. Appl Immunohistochem Mol
Morphol. 17:530–535. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhu W, Mao Z, Zhu C, Li M, Cao C, Guan Y,
Yuan J, Xie G and Guan X: Adolescent exposure to cocaine increases
anxiety-like behavior and induces morphologic and neurochemical
changes in the hippocampus of adult rats. Neuroscience.
313:174–183. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang Y, Xu Y, Zhu Q, Zhao F, Luo J, Zhang
X and Wang X: Upregulation of dysbindin in temporal lobe epileptic
foci of human and experimental animals. Synapse. 66:622–629. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Feng J, Zhang D and Chen BY: Endothelial
mechanisms of endothelial dysfunction in patients with obstructive
sleep apnea. Sleep Breath. 16:283–294. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yang YY, Shang J and Liu HG: Role of
endoplasmic reticular stress in aortic endothelial apoptosis
induced by intermittent/persistent hypoxia. Chin Med J (Engl).
126:4517–4523. 2013.PubMed/NCBI
|
|
37
|
Ren J, Liu W, Deng Y, Li GC, Pan YY, Xie
S, Jin M and Liu HG: Losartan attenuates aortic endothelial
apoptosis induced by chronic intermittent hypoxia partly via the
phospholipase C pathway. Sleep Breath. 21:679–689. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yoon CK, Yoon SY, Hwang JS and Shin YJ:
O-GlcNAc signaling augmentation protects human corneal endothelial
cells from oxidative stress via AKT pathway activation. Curr Eye
Res. 45:556–562. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Friedman JK, Nitta CH, Henderson KM,
Codianni SJ, Sanchez L, Ramiro-Diaz JM, Howard TA, Giermakowska W,
Kanagy NL and Gonzalez Bosc LV: Intermittent hypoxia-induced
increases in reactive oxygen species activate NFATc3 increasing
endothelin-1 vasoconstrictor reactivity. Vascul Pharmacol.
60:17–24. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kim B, Kim J, Bae YM, Cho SI, Kwon SC,
Jung JY, Park JC and Ahn HY: p38 mitogen-activated protein kinase
contributes to the diminished aortic contraction by endothelin-1 in
DOCA-salt hypertensive rats. Hypertension. 43:1086–1091. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ding LL, Chapman A, Boyd R and Wang HD:
ERK activation contributes to regulation of spontaneous contractile
tone via superoxide anion in isolated rat aorta of angiotensin
II-induced hypertension. Am J Physiol Heart Circ Physiol.
292:H2997–H3005. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Escano CS, Keever LB, Gutweiler AA and
Andresen BT: Angiotensin II activates extracellular
signal-regulated kinase independently of receptor tyrosine kinases
in renal smooth muscle cells: Implications for blood pressure
regulation. J Pharmacol Exp Ther. 324:34–42. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Matrougui K, Eskildsen-Helmond YE,
Fiebeler A, Henrion D, Levy BI, Tedgui A and Mulvany MJ:
Angiotensin II stimulates extracellular signal-regulated kinase
activity in intact pressurized rat mesenteric resistance arteries.
Hypertension. 36:617–621. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhang K, Ma ZW, Song C, Duan XR, Yang Y
and Li GP: Role of ion channels in chronic intermittent
hypoxia-induced atrial remodeling in rats. Life Sci.
254:1177972020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yuan GX, Nanduri J, Bhasker CR, Semenza GL
and Prabhakar NR: Ca2+/calmodulin kinase-dependent
activation of hypoxia inducible factor 1 transcriptional activity
in cells subjected to intermittent hypoxia. J Biol Chem.
280:4321–4328. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Do KH, Kim MS, Kim JH, Rhim BY, Lee WS,
Kim CD and Bae SS: Angiotensin II-induced aortic ring constriction
is mediated by phosphatidylinositol 3-kinase/L-type calcium channel
signaling pathway. Exp Mol Med. 41:569–576. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Lima VV, Lobato NS, Filgueira FP, Webb RC,
Tostes RC and Giachini FR: Vascular O-GlcNAcylation augments
reactivity to constrictor stimuli by prolonging phosphorylated
levels of the myosin light chain. Braz J Med Biol Res. 47:826–833.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Matrougui K, Tankó LB, Loufrani L, Gorny
D, Levy BI, Tedgui A and Henrion D: Involvement of Rho-kinase and
the actin filament network in angiotensin II-induced contraction
and extracellular signal-regulated kinase activity in intact rat
mesenteric resistance arteries. Arterioscler Thromb Vasc Biol.
21:1288–1293. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Pedowitz NJ, Batt AR, Darabedian N and
Pratt MR: MYPT1 O-GlcNAc modification regulates
sphingosine-1-phosphate mediated contraction. Nat Chem Biol.
17:169–177. 2021. View Article : Google Scholar : PubMed/NCBI
|