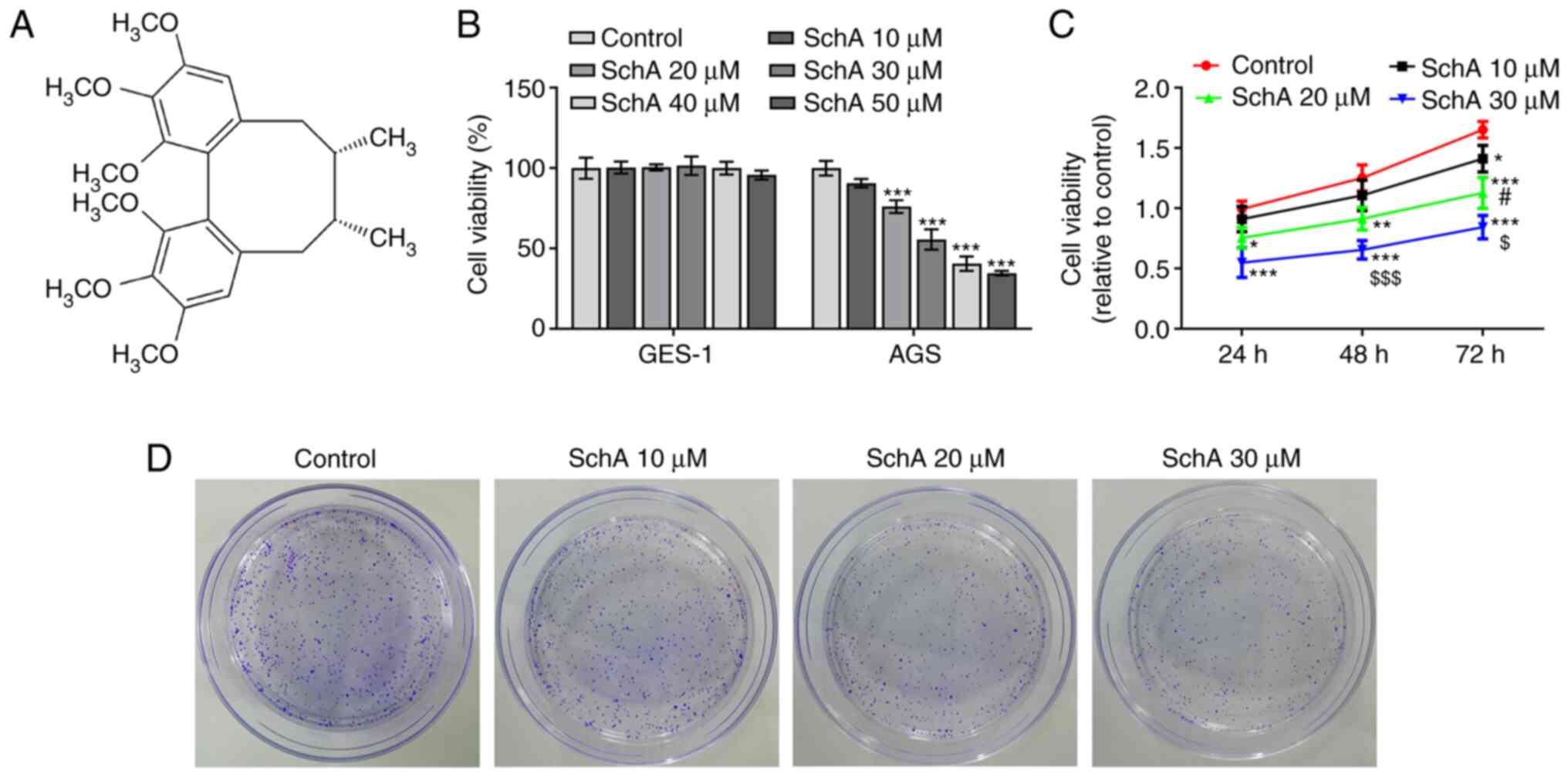
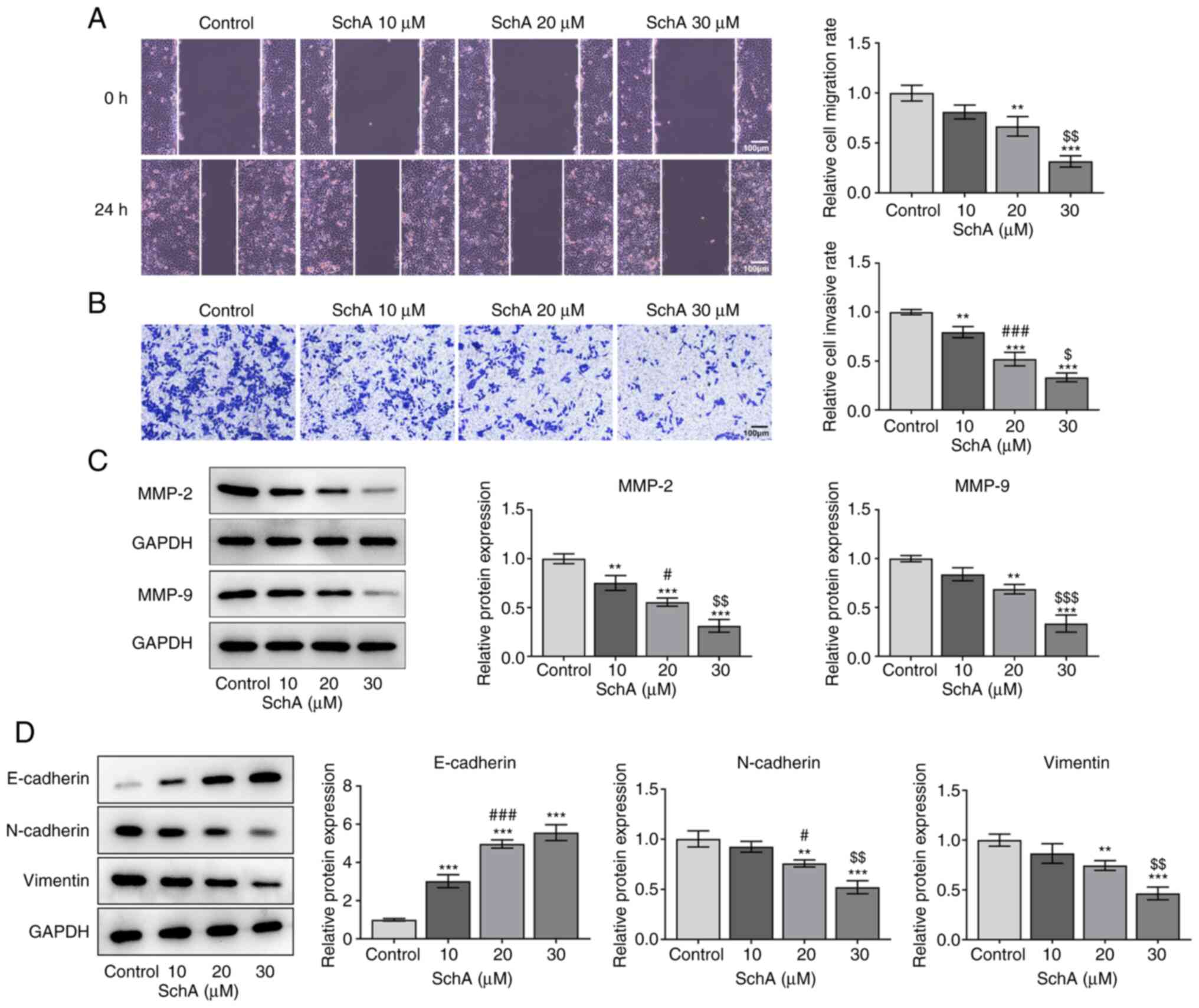
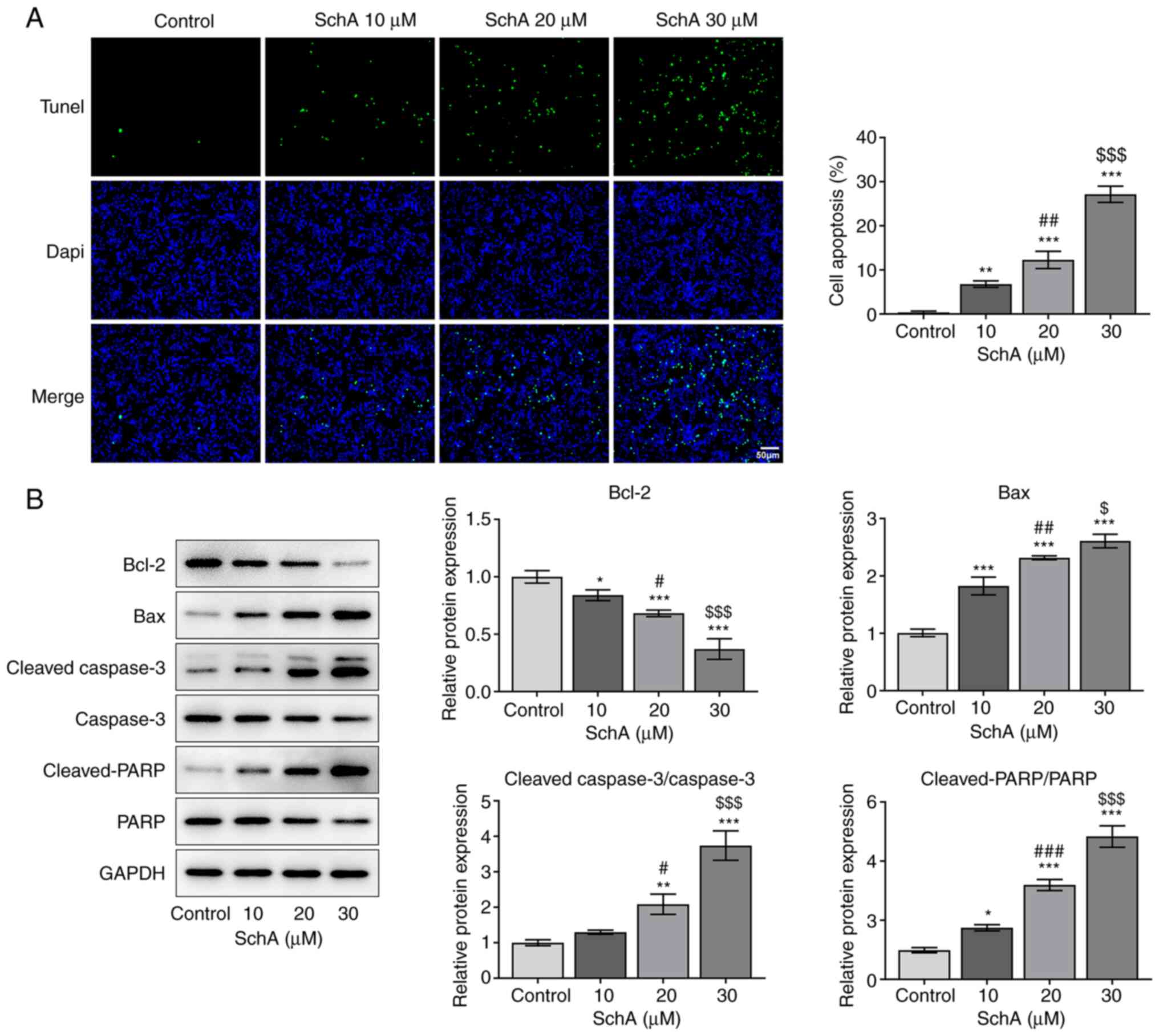
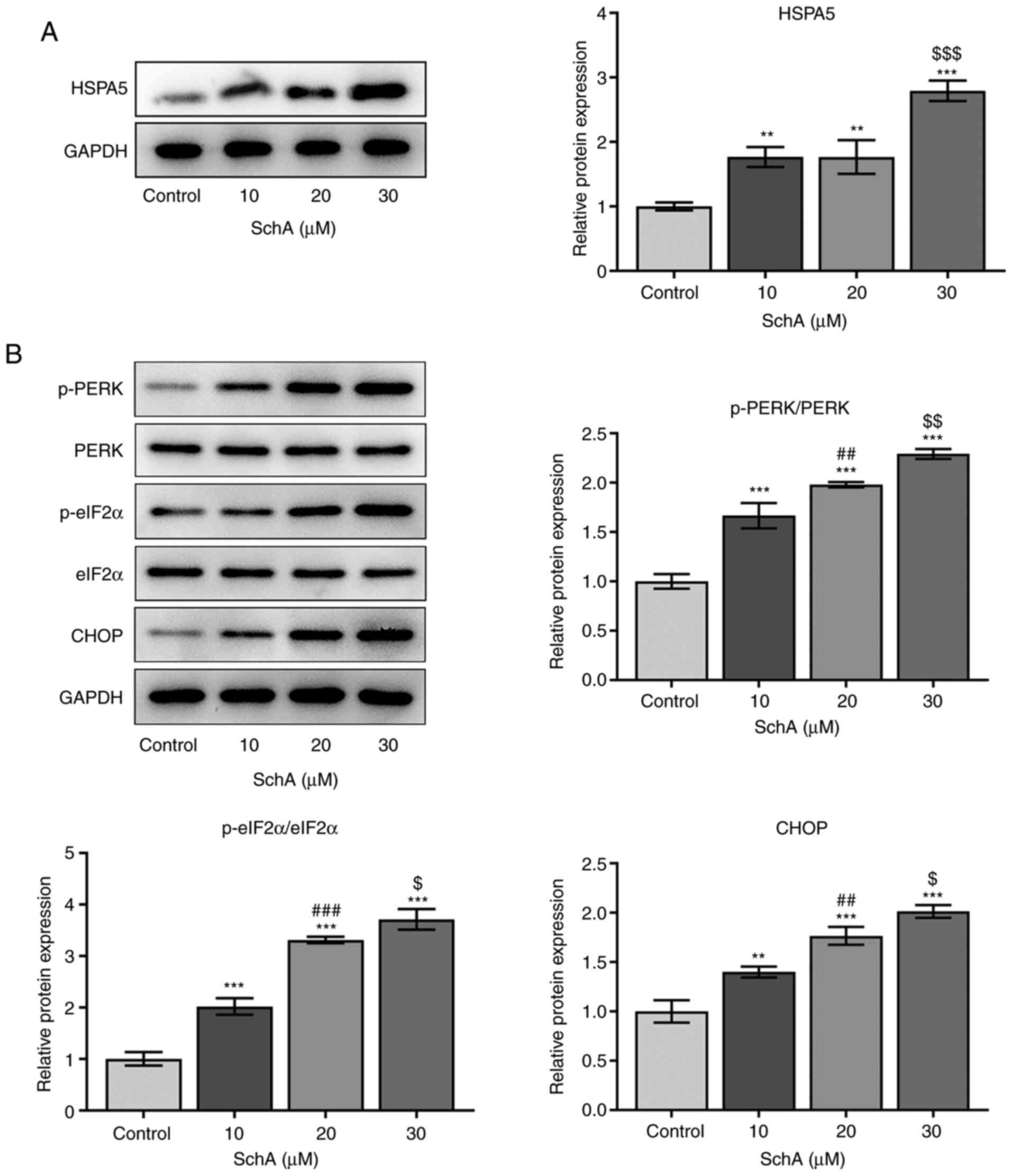
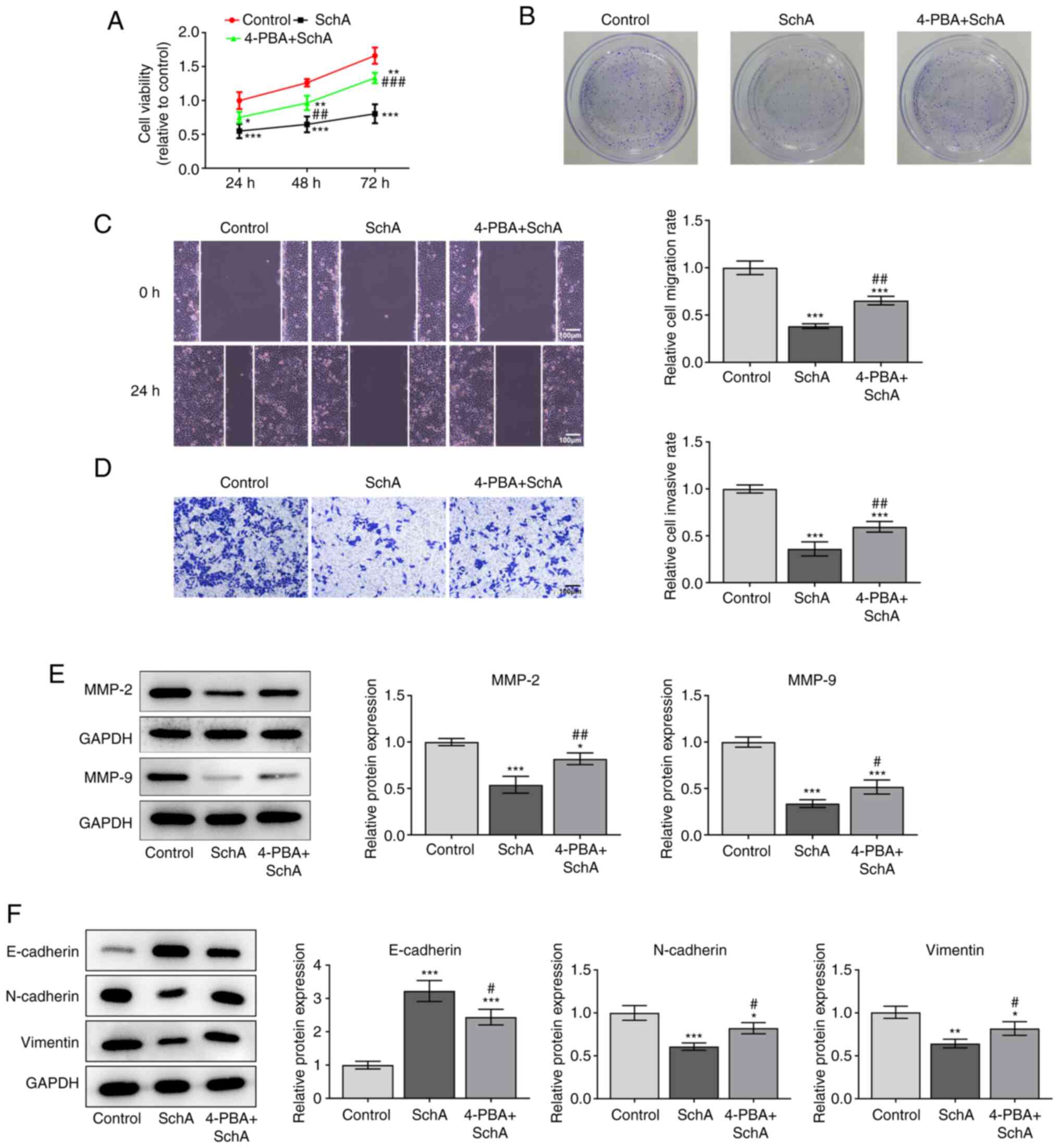
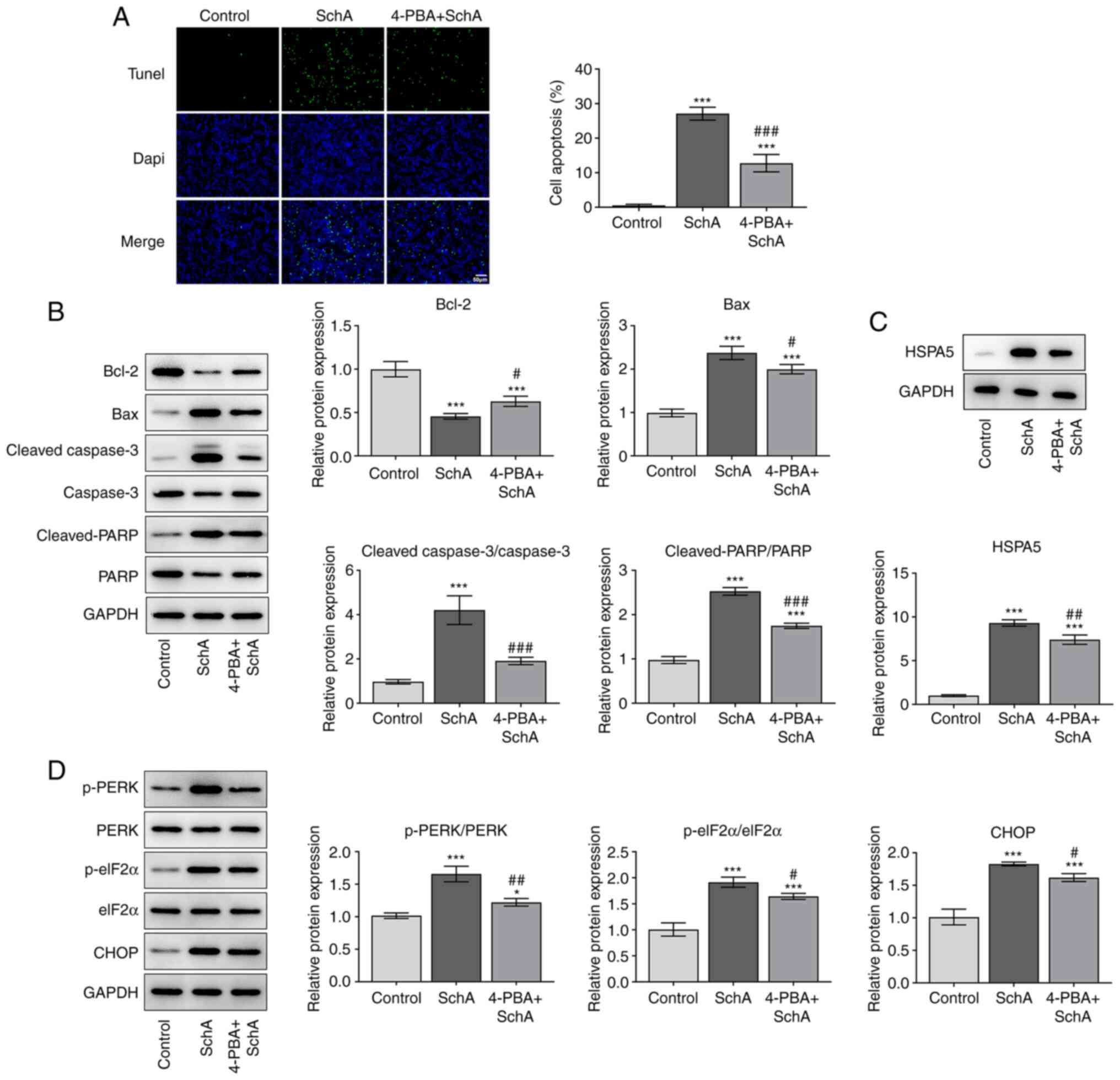
|
1
|
Shomali N, Mansoori B, Mohammadi A,
Shirafkan N, Ghasabi M and Baradaran B: MiR-146a functions as a
small silent player in gastric cancer. Biomed Pharmacother.
96:238–245. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Howson CP, Hiyama T and Wynder EL: The
decline in gastric cancer: Epidemiology of an unplanned triumph.
Epidemiol Rev. 8:1–27. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Van Cutsem E, Moiseyenko VM, Tjulandin S,
Majlis A, Constenla M, Boni C, Rodrigues A, Fodor M, Chao Y, Voznyi
E, et al: Phase III study of docetaxel and cisplatin plus
fluorouracil compared with cisplatin and fluorouracil as first-line
therapy for advanced gastric cancer: A report of the V325 study
group. J Clin Oncol. 24:4991–4997. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Xu X, Rajamanicham V, Xu S, Liu Z, Yan T,
Liang G, Guo G, Zhou H and Wang Y: Schisandrin A inhibits triple
negative breast cancer cells by regulating Wnt/ER stress signaling
pathway. Biomed Pharmacother. 115:1089222019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yan H and Guo M: Schizandrin A inhibits
cellular phenotypes of breast cancer cells by repressing miR-155.
IUBMB Life. 72:1640–1648. 2020. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen BC, Tu SL, Zheng BA, Dong QJ, Wan ZA
and Dai QQ: Schizandrin A exhibits potent anticancer activity in
colorectal cancer cells by inhibiting heat shock factor 1. Biosci
Rep. 40:BSR202002032020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bi Y, Fu Y, Wang S, Chen X and Cai X:
Schizandrin A exerts anti-tumor effects on A375 cells by
down-regulating H19. Braz J Med Biol Res. 52:e83852019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ding Q, Li X, Sun Y and Zhang X:
Schizandrin A inhibits proliferation, migration and invasion of
thyroid cancer cell line TPC-1 by down regulation of microRNA-429.
Cancer Biomark. 24:497–508. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ma YY, Di ZM, Cao Q, Xu WS, Bi SX, Yu JS,
Shen YJ, Yu YQ, Shen YX and Feng LJ: Xanthatin induces glioma cell
apoptosis and inhibits tumor growth via activating endoplasmic
reticulum stress-dependent CHOP pathway. Acta Pharmacol Sin.
41:404–414. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xian H, Feng W and Zhang J: Schizandrin A
enhances the efficacy of gefitinib by suppressing IKKβ/NF-κB
signaling in non-small cell lung cancer. Eur J Pharmacol.
855:10–19. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Urra H, Dufey E, Avril T, Chevet E and
Hetz C: Endoplasmic reticulum stress and the hallmarks of cancer.
Trends Cancer. 2:252–262. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Farina P, Tabouret E, Lehmann P, Barrie M,
Petrirena G, Campello C, Boucard C, Graillon T, Girard N and Chinot
O: Relationship between magnetic resonance imaging characteristics
and plasmatic levels of MMP2 and MMP9 in patients with recurrent
high-grade gliomas treated by Bevacizumab and Irinotecan. J
Neurooncol. 132:433–437. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shi Y, Shi H, Zhang B, Yan Y, Han X, Jiang
W, Qian H and Xu W: miR-373 suppresses gastric cancer metastasis by
downregulating vimentin. Mol Med Rep. 17:4027–4034. 2018.PubMed/NCBI
|
|
14
|
Wu D, Zhang P, Ma J, Xu J, Yang L, Xu W,
Que H, Chen M and Xu H: Serum biomarker panels for the diagnosis of
gastric cancer. Cancer Med. 8:1576–1583. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rona KA, Schwameis K, Zehetner J, Samakar
K, Green K, Samaan J, Sandhu K, Bildzukewicz N, Katkhouda N and
Lipham JC: Gastric cancer in the young: An advanced disease with
poor prognostic features. J Surg Oncol. 115:371–375. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu X, Xiu LJ, Jiao JP, Zhao J, Zhao Y, Lu
Y, Shi J, Li YJ, Ye M, Gu YF, et al: Traditional Chinese medicine
integrated with chemotherapy for stage IV non-surgical gastric
cancer: A retrospective clinical analysis. J Integr Med.
15:469–475. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lee K, Ahn JH, Lee KT, Jang DS and Choi
JH: Deoxyschizandrin, isolated from schisandra berries, induces
cell cycle arrest in ovarian cancer cells and inhibits the
protumoural activation of tumour-associated macrophages. Nutrients.
10:912018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Panossian A and Wikman G: Pharmacology of
Schisandra chinensis Bail.: An overview of Russian research
and uses in medicine. J Ethnopharmacol. 118:183–212. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li L, Pan Q, Sun M, Lu Q and Hu X:
Dibenzocyclooctadiene lignans: A class of novel inhibitors of
multidrug resistance- associated protein 1. Life Sci. 80:741–748.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen X and Cubillos-Ruiz JR: Endoplasmic
reticulum stress signals in the tumour and its microenvironment.
Nat Rev Cancer. 21:71–88. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bettigole SE and Glimcher LH: Endoplasmic
reticulum stress in immunity. Annu Rev Immunol. 33:107–138. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang S and Kaufman RJ: The impact of the
unfolded protein response on human disease. J Cell Biol.
197:857–867. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bertolotti A, Zhang Y, Hendershot LM,
Harding HP and Ron D: Dynamic interaction of BiP and ER stress
transducers in the unfolded-protein response. Nat Cell Biol.
2:326–332. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mohamed E, Cao Y and Rodriguez PC:
Endoplasmic reticulum stress regulates tumor growth and anti-tumor
immunity: A promising opportunity for cancer immunotherapy. Cancer
Immunol Immunother. 66:1069–1078. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang J, Hu X and Jiang H: ERS-PERK
signaling pathway-mediated Nrf2/ARE-HO-1 axis: A novel therapeutic
target for attenuating myocardial ischemia and reperfusion injury.
Int J Cardiol. 203:779–780. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sozen E, Karademir B and Ozer NK: Basic
mechanisms in endoplasmic reticulum stress and relation to
cardiovascular diseases. Free Radic Biol Med. 78:30–41. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Oakes SA and Papa FR: The role of
endoplasmic reticulum stress in human pathology. Annu Rev Pathol.
10:173–194. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kaira K, Toyoda M, Shimizu A, Shino M,
Sakakura K, Takayasu Y, Takahashi K, Asao T and Chikamatsu K:
Expression of ER stress markers (GRP78/BiP and PERK) in adenoid
cystic carcinoma. Acta Otolaryngol. 136:1–7. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zheng YZ, Cao ZG, Hu X and Shao ZM: The
endoplasmic reticulum stress markers GRP78 and CHOP predict
disease-free survival and responsiveness to chemotherapy in breast
cancer. Breast Cancer Res Treat. 145:349–358. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nagelkerke A, Bussink J, Sweep FC and Span
PN: The unfolded protein response as a target for cancer therapy.
Biochim Biophys Acta. 1846:277–284. 2014.PubMed/NCBI
|
|
31
|
Moon SY, Kim HS, Nho KW, Jang YJ and Lee
SK: Endoplasmic reticulum stress induces epithelial-mesenchymal
transition through autophagy via activation of c-Src kinase.
Nephron Exp Nephrol. 126:127–140. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang M and Kaufman RJ: The impact of the
endoplasmic reticulum protein-folding environment on cancer
development. Nat Rev Cancer. 14:581–597. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li Y, Liu H, Huang YY, Pu LJ, Zhang XD,
Jiang CC and Jiang ZW: Suppression of endoplasmic reticulum
stress-induced invasion and migration of breast cancer cells
through the downregulation of heparanase. Int J Mol Med.
31:1234–1242. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu R, Li X, Gao W, Zhou Y, Wey S, Mitra
SK, Krasnoperov V, Dong D, Liu S, Li D, et al: Monoclonal antibody
against cell surface GRP78 as a novel agent in suppressing PI3K/AKT
signaling, tumor growth, and metastasis. Clin Cancer Res.
19:6802–6811. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dong KN, Huang X, Xing WY, Guo WW and Feng
R: Effect of endoplasmic reticulum stress on gastric cancer cell
migration and invasion. World Chin J Dig. 24:14852016. View Article : Google Scholar
|