Introduction
The number of individuals >65 years old worldwide
is projected to increase from 524 million in 2010 to ~1.5 billion
by 2050 (1). Elderly individuals
are particularly susceptible to developing chronic diseases,
including hepatic fibrosis (2).
Liver fibrosis is a dynamic process associated with the continuous
deposition and resorption of the extracellular matrix, mainly
fibrillar collagen (3), which is
often the first step in architectural distortion and dysfunction
that prevents the normal functioning of the liver (4). If untreated, liver fibrosis may lead
to advanced liver cirrhosis and hepatoma (4). Accumulating evidence has suggested
that susceptibility to liver fibrosis and hepatitis significantly
increases with age (5). Thus, it
remains of high importance to study liver aging and the associated
underlying mechanisms to provide novel strategies to prevent
aging-related liver fibrosis.
The pathogenesis of aging is complex. For the past
40 years or so, oxidative stress has been increasingly recognized
as a contributing factor in aging-related diseases (6–8).
Oxidative stress is induced by an imbalance between the production
of reactive oxygen species (ROS) and the scavenging capacity of the
antioxidant system (9,10). It has been reported that excessive
production of ROS can cause damage to proteins, lipids and DNA,
resulting in a variety of disease types, such as Alzheimer's
disease, diabetes, heart failure, chronic fatigue syndrome and
cancer (11). NADPH oxidase (NOX)
is one of the major sources of ROS, and the family of NOX proteins
is composed of the membrane subunit (NOX1-5), p22phox and the
cytoplasmic subunits of p67phox, p47phox and Rac1 (12,13).
It is worth noting that NOX4 is a constitutively active enzyme and
has been found to be widely expressed in the liver, particularly
within hepatocytes, hepatic stellate cells and fibroblasts
(14). Increasing evidence has
indicated that oxidative stress caused by NOX4-derived ROS may play
a key role in liver fibrosis (15).
Chronic inflammation is another important process
that occurs during aging as a result of low-grade elevations of
circulating inflammatory mediators (16). Inflammation is a common feature of
several age-related pathologies, such as frailty and cardiovascular
disease (17), which contributes to
the progression of tissue dysfunction (18). It has been reported that
inflammation also played an important role in age-related liver
injury (19). Inflammasomes, which
are large cytoplasmic multiprotein complexes, consist of NLR family
pyrin domain containing (NLRP), a cytoplasmic pattern recognition
receptor of caspase-1 and apoptosis-associated speck-like protein
containing a C-terminal caspase recruitment domain (ASC). According
to the findings of previous studies, the NLRP3 inflammasome, a type
of inflammasome ubiquitously expressed in numerous tissues,
including the liver, was discovered to be involved in the evolution
of liver fibrosis and the progression of liver injury and
age-related liver disease (20–22).
When activated by diverse irritants, such as ATP and cholesterol
crystals, as well as bacterial, viral and fungal pathogens
(23,24), the NLRP3 inflammasome responds to
inflammation by promoting the maturation of a series of
proinflammatory cytokines, such as IL-1β and IL-18 (25). In addition, it has been reported
that excessive ROS accumulation activated the NLRP3 inflammasome in
the liver during the process of aging, eventually leading to
aging-associated liver disease (26).
The Senescence-accelerated mouse prone 8 (SAMP8) is
a model of accelerated aging solely of genetic origin, which has
not been subjected to any experimental manipulation (27). Previous studies have reported that
SAMP8 mice displayed extensive liver degeneration, including liver
steatosis, hepatocyte ballooning characterized by swollen cells,
focal necrosis and inflammation, and fibrosis (28,29).
Among these observations, liver fibrosis was the most common and
significant pathological change (5). Additionally, SAMP8 mice also have
abnormal liver function test results, such as significant increases
in alanine aminotransferase (ALT) and aspartate aminotransferase
(AST) levels (30). However, there
are still no effective methods and drugs for delaying liver aging
and aging-related liver injury and fibrosis.
Ginseng has been used for >2,000 years and
demonstrates several beneficial effects, such as improving liver
health and delaying aging (31).
Ginsenoside Rg1 (Rg1) is one of the active ingredients in ginseng
(32). It has been reported that
Rg1 exerted a protective effect on neuronal damage via inhibition
of oxidative stress-induced neuronal apoptosis (33). Moreover, Rg1 was found to ameliorate
diabetic cardiomyopathy by inhibiting endoplasmic reticulum (ER)
stress-induced apoptosis in diabetic rats (34). Our previous studies discovered that
Rg1 could protect against aging-related renal injuries and neuronal
senescence by inhibiting NOX4/2 (35,36).
However, to the best of our knowledge, it remains unknown whether
Rg1 protects against aging-related liver injury and fibrosis. The
present study aimed to investigate whether Rg1 treatment
ameliorates aging-related liver damage and fibrosis by inhibiting
NOX4/NLRP3 signaling to reduce oxidative stress and inflammation in
the liver during aging.
Materials and methods
Animals and treatment
In total, 9 male senescence-accelerated resistant
mouse 1 (SAMR1) and 45 male SAMP8 mice (both age, 6 months; weight,
30–40 g) were purchased from the Department of Experimental Animal
Science, Peking University Medical Science Center (Beijing, China).
The mice were maintained in an environmentally controlled room
(temperature, 22–25°C; relative humidity, 50–70%) under a 12-h
light/dark cycle with unlimited access to food and water. The SAMP8
mice were randomly divided into five groups (n=9 in each group): i)
SAMP8 model group; ii) SAMP8 + apocynin (50 mg/kg) group; iii)
SAMP8 + tempol (50 mg/kg) group; iv) SAMP8 + Rg1 (5 mg/kg) group;
v) SAMP8 + Rg1 (10 mg/kg) group; and vi) SAMR1 mice group, which
were used as the control group. The treatments were administered
intragastrically (0.1 ml/10 g body weight), and the mice treated
with either apocynin (MilliporeSigma), tempol (MilliporeSigma) or
Rg1 (content >98%; Chengdu Desite Biotechnology Co., Ltd.) once
a day for 9 weeks. The SAMP8 and SAMR1 groups were treated with
distilled water for 9 weeks. Following 9 weeks of treatment, six
mice in each group were sacrificed via cervical dislocation. The
livers were harvested and stored in at −80°C for subsequent use in
western blotting experiments, or placed in 4% paraformaldehyde for
24–48 h at room temperature for histological examination. The
experimental procedures were approved by the Animal Ethics
Committee of Anhui Medical University (approval no. LLSC20160183;
Hefei, China) and performed in accordance with the Guidelines for
the Care and Use of Laboratory Animals (37).
Detection of ROS
The levels of ROS production in the liver of the
remaining three mice in different groups of mice were detected
using dihydroethylene (DHE) staining. Briefly, 100 µM DHE (0.1
ml/10 g; Beyotime Institute of Biotechnology) was injected through
the tail vein in each group of mice (n=3). After 30 min, the
animals were sacrificed by cervical dislocation and the livers were
removed and embedded in optical cutting temperature compound
(Sakura Finetek USA, Inc.) at −20°C for 2 h. The liver tissues were
subsequently cut into 10-µm sections using a frozen microtome
(Leica CM3050; Leica Microsystems GmbH) at −20°C. The sections were
washed with PBS and incubated with 5 mg/l Hoechst 33258 solution
(Sigma-Aldrich; Merck KGaA) at room temperature for 5 min. Then,
the sections were sealed with anti-fluorescence quenching agent
(Beyotime Institute of Biotechnology) and visualized using a
fluorescence microscope (Olympus IX72; Olympus Corporation;
magnification, ×400). Image Pro Plus 6.0 software (Media
Cybernetics, Inc.) was used to detect the average density of red
fluorescence from three randomly selected fields of view in each
section to indicate ROS production.
Pathological examination of the liver
tissue
The morphological changes in the liver were examined
using H&E, periodic acid-Schiff (PAS) and Masson's trichrome
staining techniques. H&E staining is the most common method for
observing pathological changes in tissues (38). Briefly, liver specimens were fixed
in 4% paraformaldehyde for 24–48 h, dehydrated and paraffin
embedded, then cut into 5-µm thick sections. Liver sections (n=4)
were deparaffinized in xylene and rehydrated in graded alcohol
series (anhydrous ethanol, 85% ethanol, 75% ethanol), then stained
with hematoxylin for 3 min and eosin for 30 sec. All these steps
were carried out at room temperature. The sections were sealed with
neutral resin and observed using a light microscope (Olympus IX72;
Olympus Corporation; magnification, ×200).
PAS staining is often used to detect the
accumulation of acidic glycoproteins to evaluate liver injury
(39). For PAS staining, the tissue
sections (n=4) were deparaffinized and rehydrated, according to the
method described for H&E staining. Then, the sections were
stained with Schiff solution for 10 min (Beijing Solarbio Science
& Technology Co., Ltd.), followed by hematoxylin for 3 min. All
these steps were conducted at room temperature.
Masson's trichrome staining is an important method
for assessing collagen deposition in liver tissue (40). For Masson's trichrome staining, the
sections (n=4) were deparaffinized and rehydrated, according to the
method described for H&E staining. Then, the sections were
stained with hematoxylin, differentiated with acid ethanol, stained
in Masson's blue solution (Beijing Solarbio Science &
Technology Co., Ltd.), followed by staining with Fuchsin for 8 min.
The cells were then washed with phosphomolybdic acid for 2 min and
stained with aniline blue for 5 min. All these steps were performed
at room temperature. PAS- and Masson's trichrome-stained cells were
visualized using a light microscope (Olympus IX71; magnification,
×400). The positive areas of PAS staining appeared purple and the
Masson's trichrome stained collagen blue. The results of the PAS
and Masson's trichrome staining in the liver were analyzed in three
randomly selected fields of view in each section using Image-Pro
Plus software (Media Cybernetics, Inc.). The average densities of
PAS and Masson's positive areas were calculated to assess the
degree of liver fibrosis.
Immunohistochemistry staining
The paraffin-embedded sections (n=4) were
deparaffinized and rehydrated, according to the method described
for H&E staining. Then, the sections were incubated with 3%
H2O2 for 10 min at 37°C to block the
endogenous peroxidase activity prior to being immersed in boiling
sodium citrate buffer for 7 min in a microwave oven for antigen
retrieval. The sections were subsequently incubated with 10% goat
serum (cat. no. C0265; Beyotime Institute of Biotechnology) at 37°C
for 30 min to block non-specific binding. The sections were then
incubated with the following primary antibodies at 4°C overnight:
Rabbit polyclonal anti-collagen IV (1:100; Bioworld Biotechnology,
Inc.; cat. no. BS1072), rabbit polyclonal anti-NLRP3 (1:100;
Bioworld Biotechnology, Inc.; cat. no. BS90949) and rabbit
polyclonal anti-TGF-β1 (1:100; Abcam; cat. no. ab92486). Following
the primary antibody incubation, the sections were reheated to 37°C
for 30 min and washed three times with PBS. Then, the sections were
incubated with a polymer-coupled sheep anti-rabbit IgG
peroxidase-conjugated secondary antibody (1:500; Affinity
Biosciences; cat. no. S0001) at 37°C for 1 h, then washed three
time with PBS. Cells were subsequently incubated with DAB to
produce brown staining for 30 sec, then stained with hematoxylin
for 3 min both at room temperature, and sealed with neutral resin.
Stained cells were visualized under a microscope (Olympus IX71;
magnification, ×400). Image-Pro Plus software was used to analyze
the expression levels of collagen IV, TGF-β1 and NLRP3 in three
randomly fields of view from each section of liver tissue.
Western blotting
Total protein was extracted from liver tissues (n=3)
using RIPA lysis buffer (cat. no. P0013B; Beyotime Institute of
Biotechnology) and an automatic sample rapid grinding machine
(Jinxing Industrial Development Co., Ltd.) at 65 Hz for 60 sec at
4°C. Total protein was quantified using a BCA protein assay kit and
the proteins (20 µg) were separated via 8–15% SDS/PAGE. The
separated proteins were subsequently transferred onto PVDF
membranes (MilliporeSigma) and blocked with 5% skimmed milk in
TBS-0.05% Tween-20 (TBST) buffer for 1 h at room temperature. The
membranes were then incubated with the following primary antibodies
overnight at 4°C: Anti-NLRP3 (1:1,000; Bioworld Biotechnology,
Inc.; cat. no. BS90949), anti-ASC (1:1,000; BIOSS; cat. no.
bs-67412-R), anti-caspase-1 (1:1,000; Abcam; cat. no. ab1872),
anti-IL-1β (1:500; Abcam; cat. no. ab9722), anti-NOX4 (1:1,000;
Bioworld Biotechnology, Inc.; cat. no. BS60435), anti-p47phox
(1:1,000; Bioworld Biotechnology, Inc.; cat. no. BS4852),
anti-p22phox (1:1,000; Bioworld Biotechnology, Inc.; cat. no.
BS60290), anti-NF-κB p65 (1:1,000; Wuhan Servicebio Technology Co.,
Ltd.; cat. no. GB11142), anti-phosphorylated (p)-NF-κB p65
(1:1,000; Wuhan Servicebio Technology Co., Ltd.; cat. no.
GB11142-1) and anti-GAPDH (1:5,000; Affinity Biosciences; cat. no.
AF7021). Following the primary antibody incubation, the membranes
were washed with TBST three times (10 min each time) and incubated
with HRP-conjugated goat anti-rabbit IgG (1:10,000; Affinity
Biosciences; cat. no. S0001) and goat anti-mouse IgG (1:10,000
Affinity Biosciences; cat. no. S0002) secondary antibodies for 1 h
at room temperature. Protein bands were visualized using an ECL kit
(Bio-Rad Laboratories, Inc.) and a Bioshine Chemi Imaging System
(Q4600 Mini; Shanghai Bioshine Technology). The optical density of
each band was semi-quantified using ImageJ 1.53a software (National
Institutes of Health) and normalized to GAPDH expression.
Statistical analysis
All data are presented as the mean ± SD of ≥3
independent experiments. GraphPad Prism 8.0 software (GraphPad
Software, Inc.) was used to perform the statistical analyses.
One-way ANOVA followed by a Tukey's post hoc test was performed to
compare differences among groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
Rg1 treatment ameliorates liver
histopathological changes in the liver of SAMP8 mice
The results of the H&E staining revealed that in
the SAMR1 control group, the boundaries of cytoplasm and nuclei of
the hepatocytes were clear. The hepatocytes were arranged in cords
from the central vein to the surrounding area, and fat droplet
vacuoles were occasionally observed. Compared with the SAMR1 group,
the cells of the SAMP8 group were not clearly defined, the nuclei
were absent or deeply stained, and were squeezed to one side (black
arrows). The arrangement of hepatocytes was abnormal and disordered
and most of the cells showed vacuole-like degeneration in the SAMP8
group. However, the tempol (50 mg/kg), apocynin (50 mg/kg) and Rg1
(5 and 10 mg/kg) treatment groups showed a marked improvement in
liver histopathology compared with the SAMP8 group (Fig. 1A). The PAS staining results also
indicated that the hepatocytes were lightly stained, with a uniform
distribution of positive areas within the cells in the SAMR1 group.
Compared with the SAMR1 group, the accumulation of positive purple
staining in hepatocytes was significantly increased in the SAMP8
group, suggesting the presence of significant hepatocyte injury in
elderly mice (Fig. 1B and C).
Compared with the SAMP8 group, the accumulation of positive
staining was significantly reduced in the tempol, apocynin and Rg1
(5 and 10 mg/kg) treatment groups (Fig.
1B and C). These results suggested that Rg1 may significantly
ameliorate aging-induced liver injury in mice.
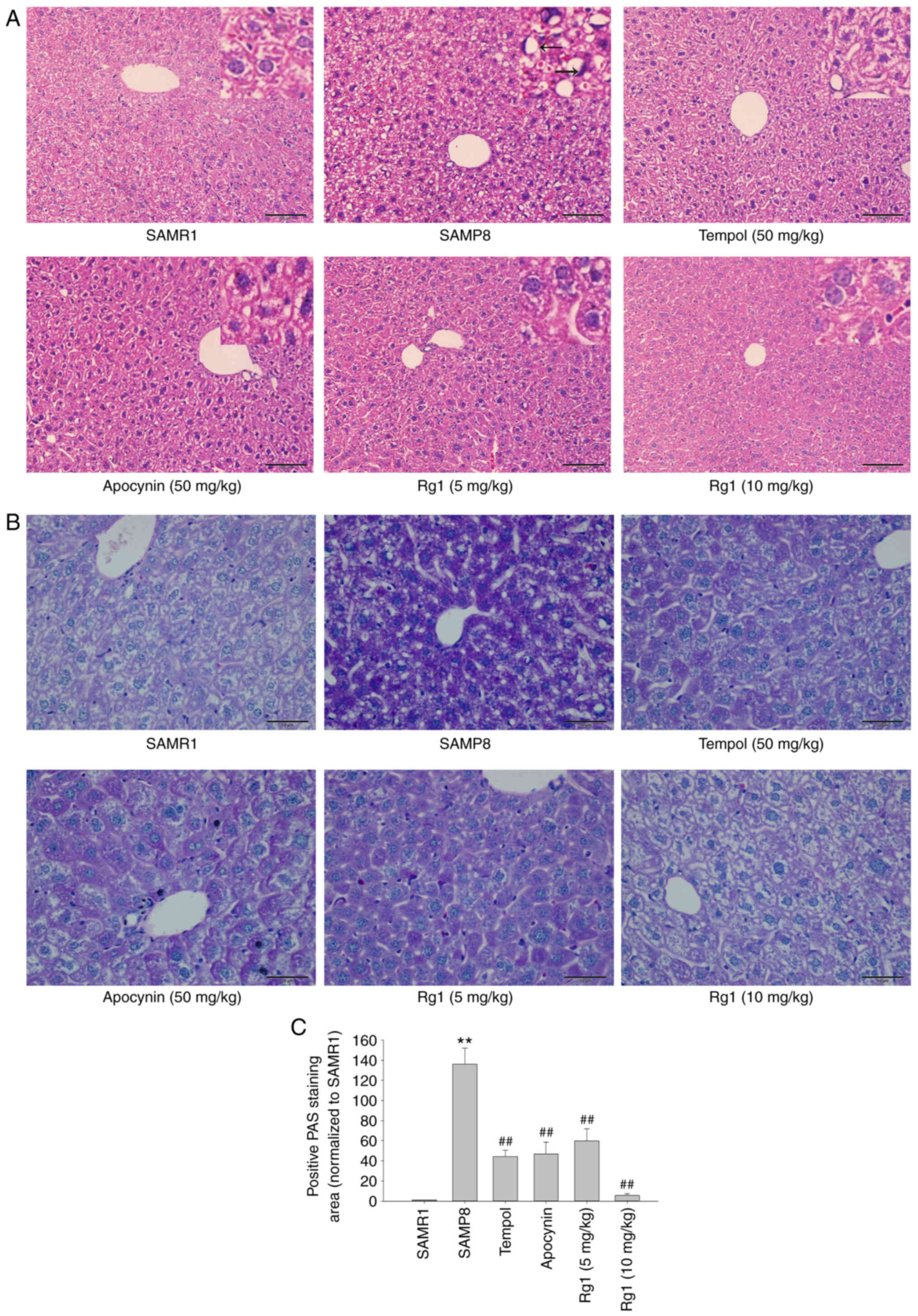 | Figure 1.Effects of Rg1 treatment on
histopathological changes in the liver in SAMP8 mice. (A) H&E
staining of the liver (Scale bar, 100 µm; Magnification, ×200). The
black arrows indicate that the cells of the SAMP8 group were not
clearly defined, the nuclei were absent or deeply stained, and were
squeezed to one side. (B) PAS staining of the liver (Scale bar, 50
µm; Magnification, ×400). (C) Positive PAS staining area
(normalized to SAMR1 group). Data are presented as the mean ± SD;
n=4. **P<0.01 vs. SAMR1; ##P<0.01 vs. SAMP8.
SAMP8, senescence-accelerated mouse prone 8; SAMR1,
senescence-accelerated resistant mouse 1; PAS, periodic
acid-Schiff; Rg1, ginsenoside Rg1. |
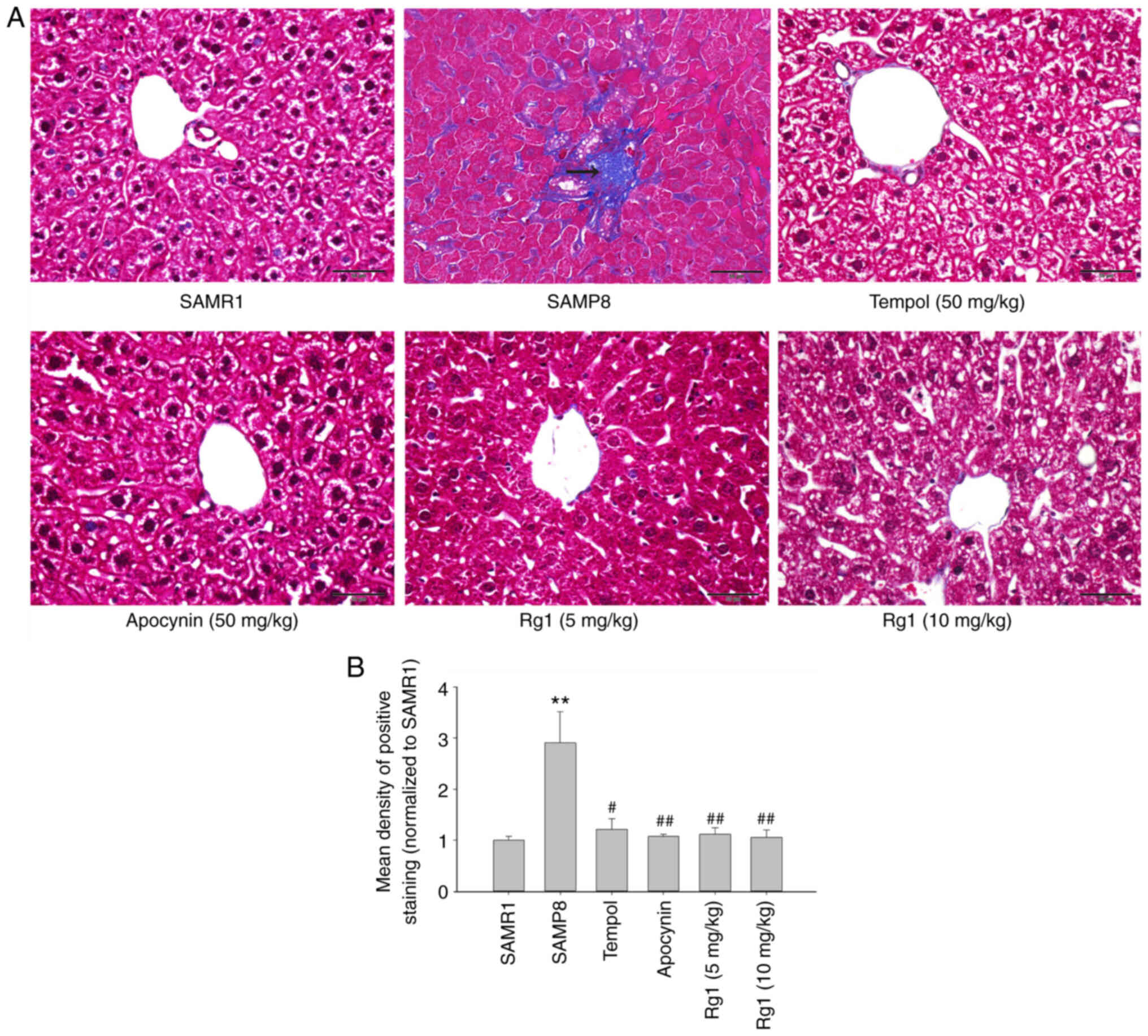
Rg1 treatment alleviates liver
fibrosis in SAMP8 mice
To explore whether Rg1 alleviates aging-related
liver fibrosis, collagen deposition was measured in liver tissues
by using Masson's staining. The results showed that the blue
positive areas were significantly increased in the liver tissues of
the SAMP8 group compared with the SAMR1 group (Fig. 2A and B). However, compared with the
SAMP8 model group, the levels of collagen deposition were
significantly reduced in the tempol, apocynin and Rg1 (5 and 10
mg/kg) treatment groups (Fig. 2A and
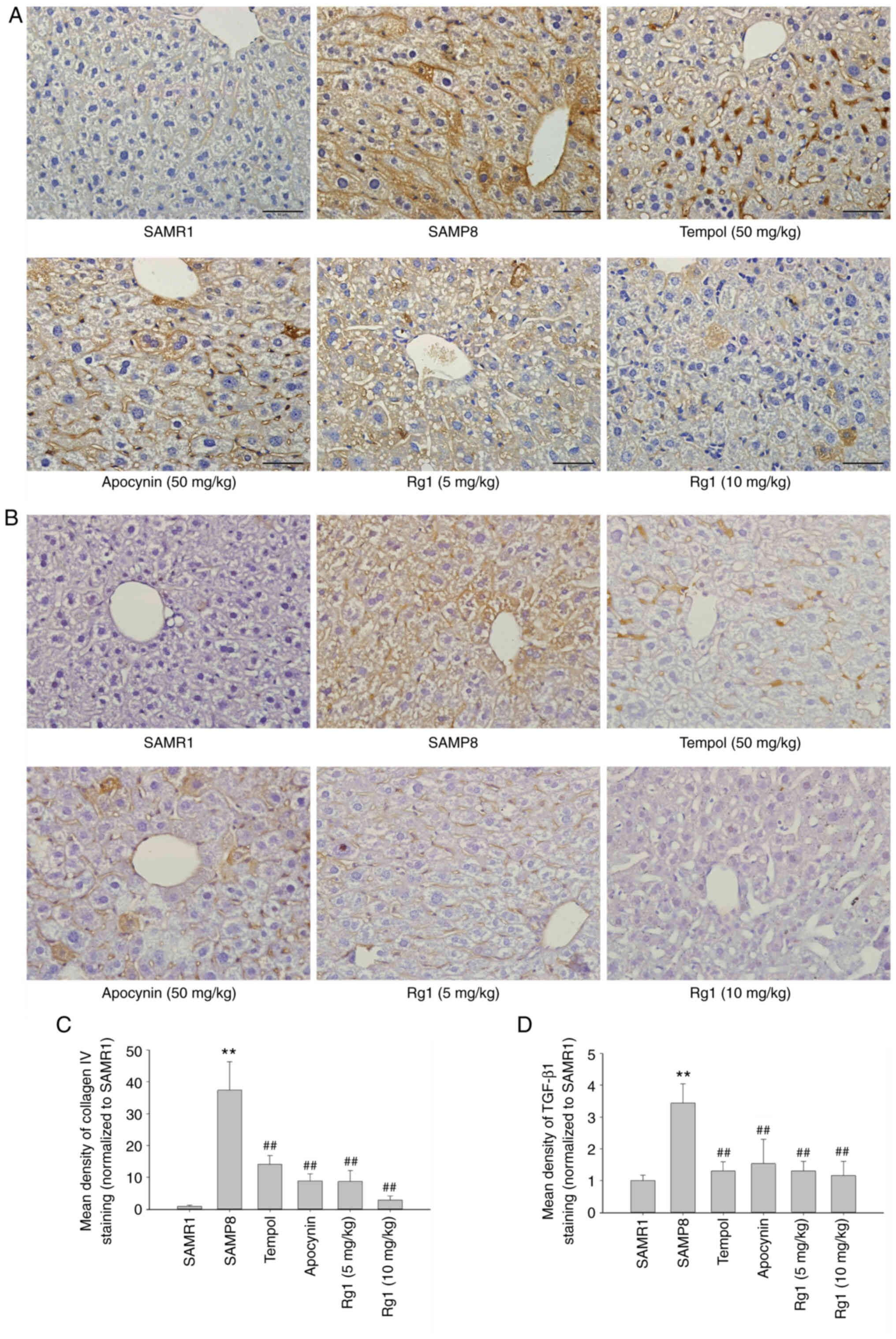
B). In addition, the expression levels of collagen IV and
TGF-β1 were measured in liver tissues by using immunohistochemical
staining. The results of the collagen IV staining revealed that
collagen IV was expressed at low levels in the liver tissues in the
SAMR1 group (Fig. 3A and C).
However, compared with the SAMR1 group, the expression levels of
collagen IV were significantly upregulated in the SAMP8 group
(Fig. 3A and C). Conversely,
compared with the SAMP8 group, treatment with tempol, apocynin and
Rg1 (5 and 10 mg/kg) significantly decreased collagen IV
deposition, especially in the Rg1 (10 mg/kg) group (Fig. 3A and C). Furthermore, similar to
collagen IV, the expression of TGF-β1 was also significantly
increased in the SAMP8 group compared with the SAMR1 group, and was
significantly decreased following treatment with tempol, apocynin
and Rg1 (5 and 10 mg/kg), especially in the Rg1 (10 mg/kg) group
(Fig. 3B and D). These results
suggested that aging may cause liver tissue fibrosis, and tempol,
apocynin and Rg1 treatment may significantly improve liver tissue
fibrosis during aging.
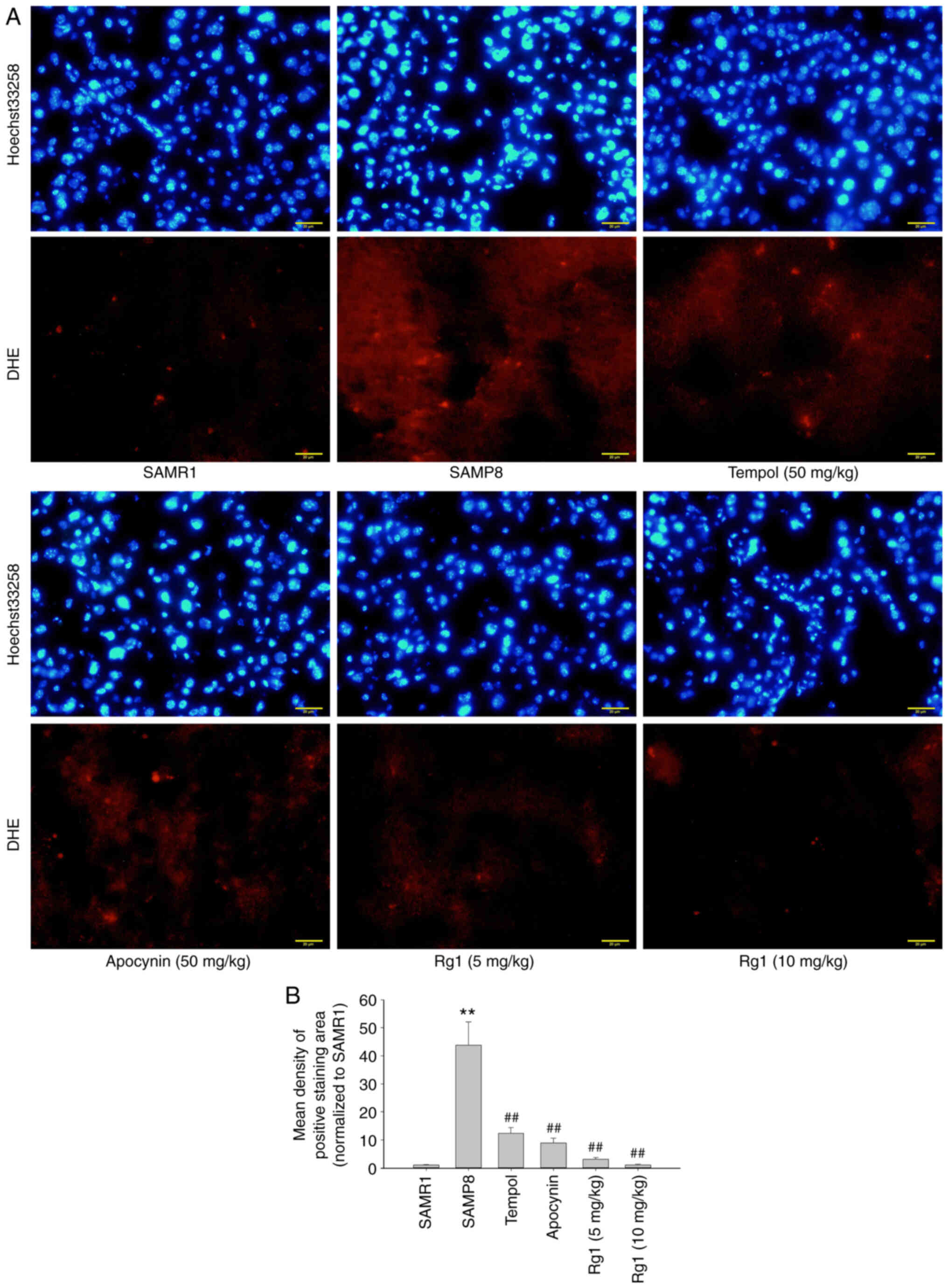
Rg1 treatment reduces ROS production
and NOX4 expression in the liver of SAMP8 mice
ROS is an important factor in the development of
hepatic fibrosis (41). In the
present study, a ROS probe, DHE, was used to detect the level of
ROS production in liver tissues. The results showed that there were
low levels of ROS production in the liver tissues of the SAMR1
group. However, compared with the SAMR1 group, the levels of ROS
production were significantly increased in the SAMP8 group
(Fig. 4A and B), while compared
with the SAMP8 group, tempol, apocynin and Rg1 (5 and 10 mg/kg)
treatment significantly reduced the levels of ROS production in the
liver tissues (Fig. 4A and B). To
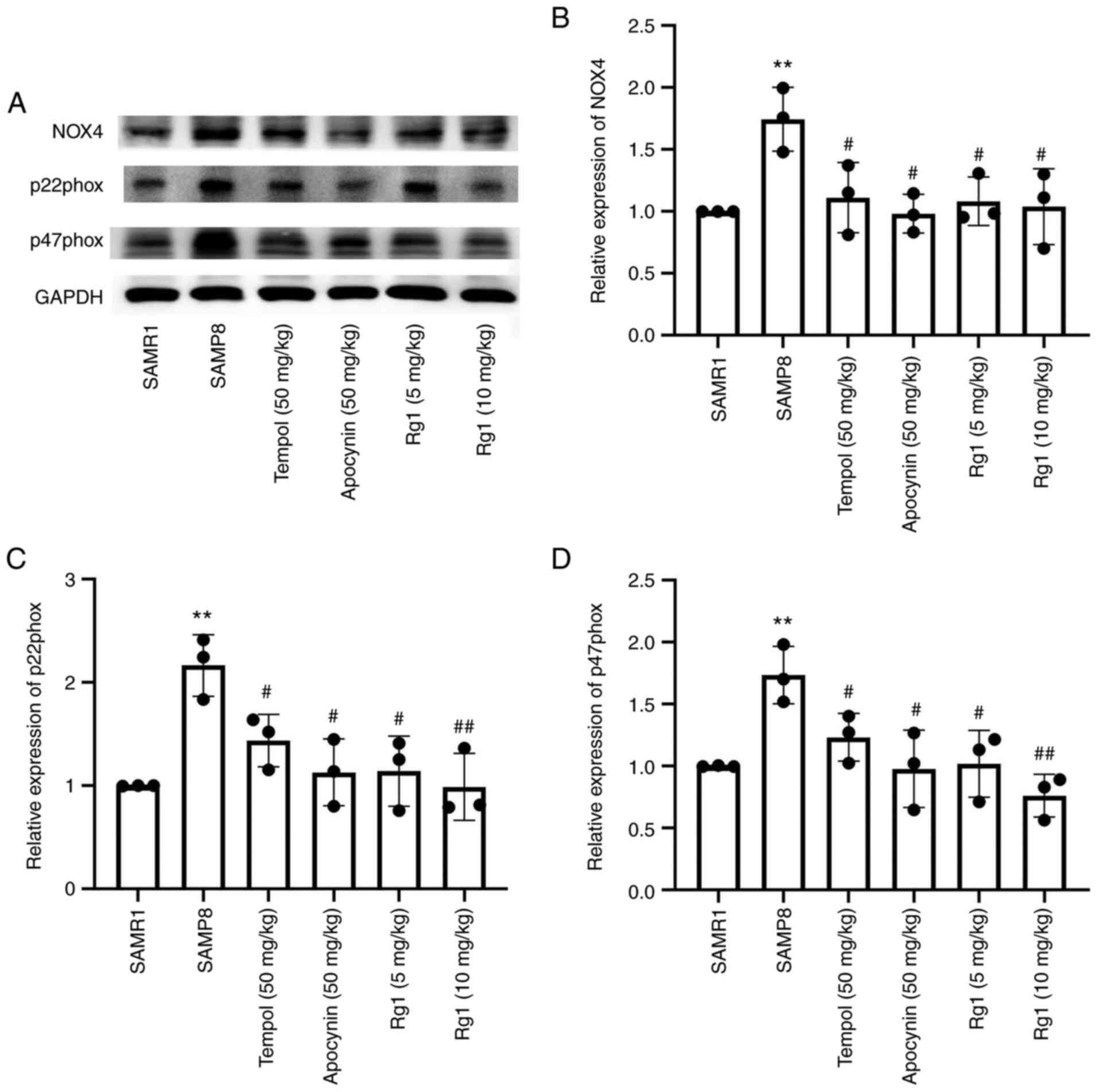
confirm the effect of NOX4 on ROS accumulation during aging, the
expression levels of NOX4-related proteins were analyzed. The
results demonstrated that, compared with the SAMR1 group, the
expression levels of NOX4, p22phox and p47phox in the liver tissues
were significantly upregulated in the SAMP8 group (Fig. 5A-D). However, compared with the
SAMP8 group, tempol, apocynin and Rg1 (5 and 10 mg/kg) treatment
significantly downregulated the expression levels of NOX4, p22phox
and p47phox in liver tissues during aging (Fig. 5A-D). These data suggested that Rg1
treatment may ameliorate ROS-induced oxidative stress injury in
liver tissues by inhibiting NOX4 during aging in mice.
Rg1 treatment downregulates the
expression levels of NLRP3, ASC, caspase-1 and IL-1β in the liver
of SAMP8 mice
It has been reported that activation of the NLRP3
inflammasome plays an important role in senescence-related renal
fibrosis (42). Therefore, the
present study further investigated the expression levels of
NLRP3-related proteins to confirm whether the NLRP3 inflammasome is
involved in age-related liver fibrosis. The results showed that the
expression levels of NLRP3, ASC, caspase-1 and IL-1β in liver
tissues were significantly upregulated in the SAMP8 group compared
with the SAMR1 group (Fig. 6A-E).
Compared with the SAMP8 group, the tempol, apocynin and Rg1 (5 and
10 mg/kg) treatment groups had significantly downregulated
expression levels of NLRP3, ASC, caspase-1 and IL-1β in the liver
tissues of SAMP8 mice (Fig. 6A-E).
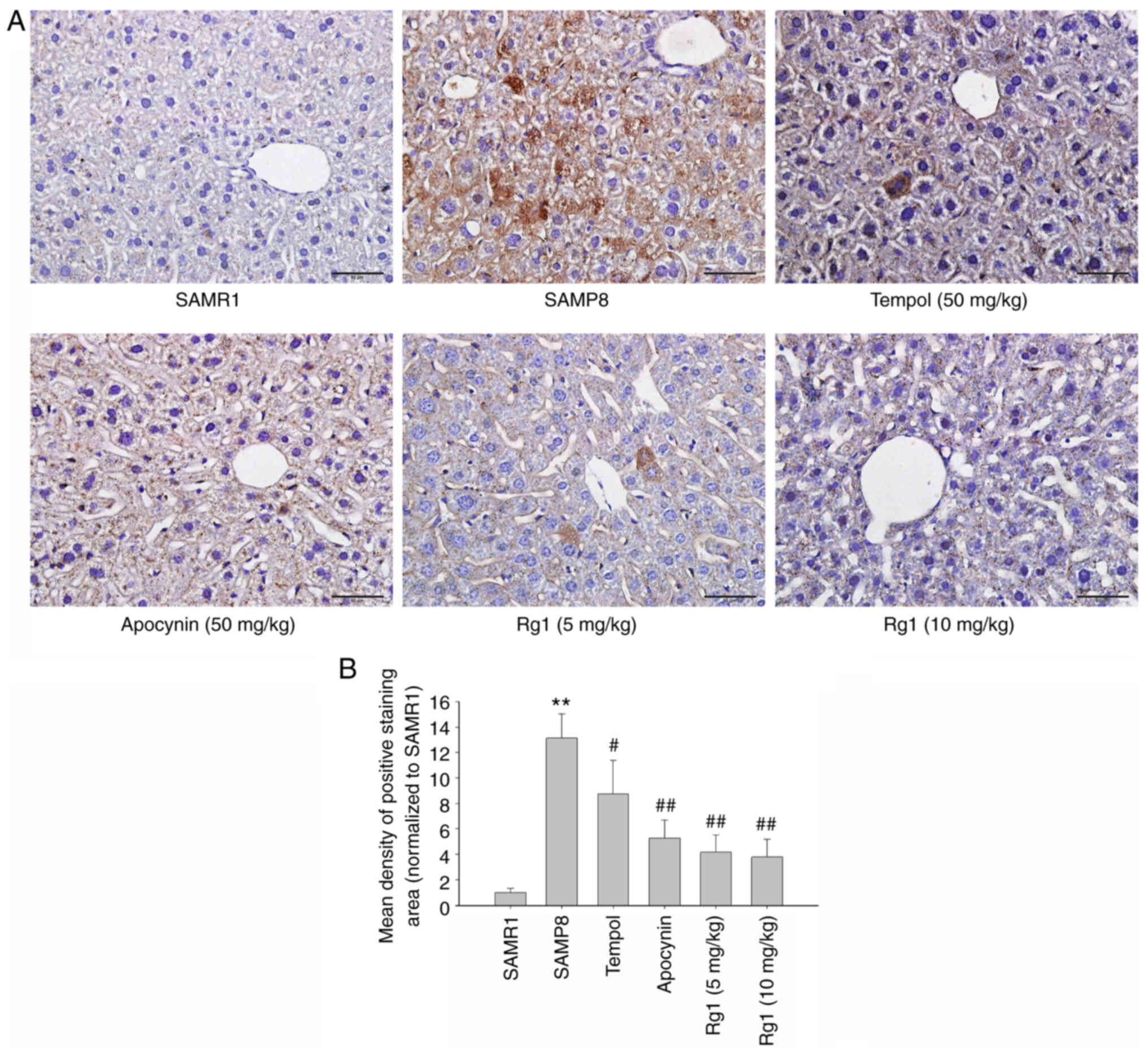
Immunohistochemical staining was performed to measure the
expression levels of NLRP3 in liver tissues. The results were
consistent with the expression of NLRP3 measured using western
blotting. The expression of NLRP3 was significantly upregulated in
the liver of the SAMP8 group compared with the SAMR1 group
(Fig. 7A and B), and tempol,
apocynin and Rg1 (5 and 10 mg/kg) treatment significantly
downregulated the expression levels of NLRP3 (Fig. 7A and B). These results suggested
that NLRP3 inflammasome activation may be closely related to liver
fibrosis during aging and Rg1 may improve liver fibrosis by
inhibiting the activation of the NLRP3 inflammasome.
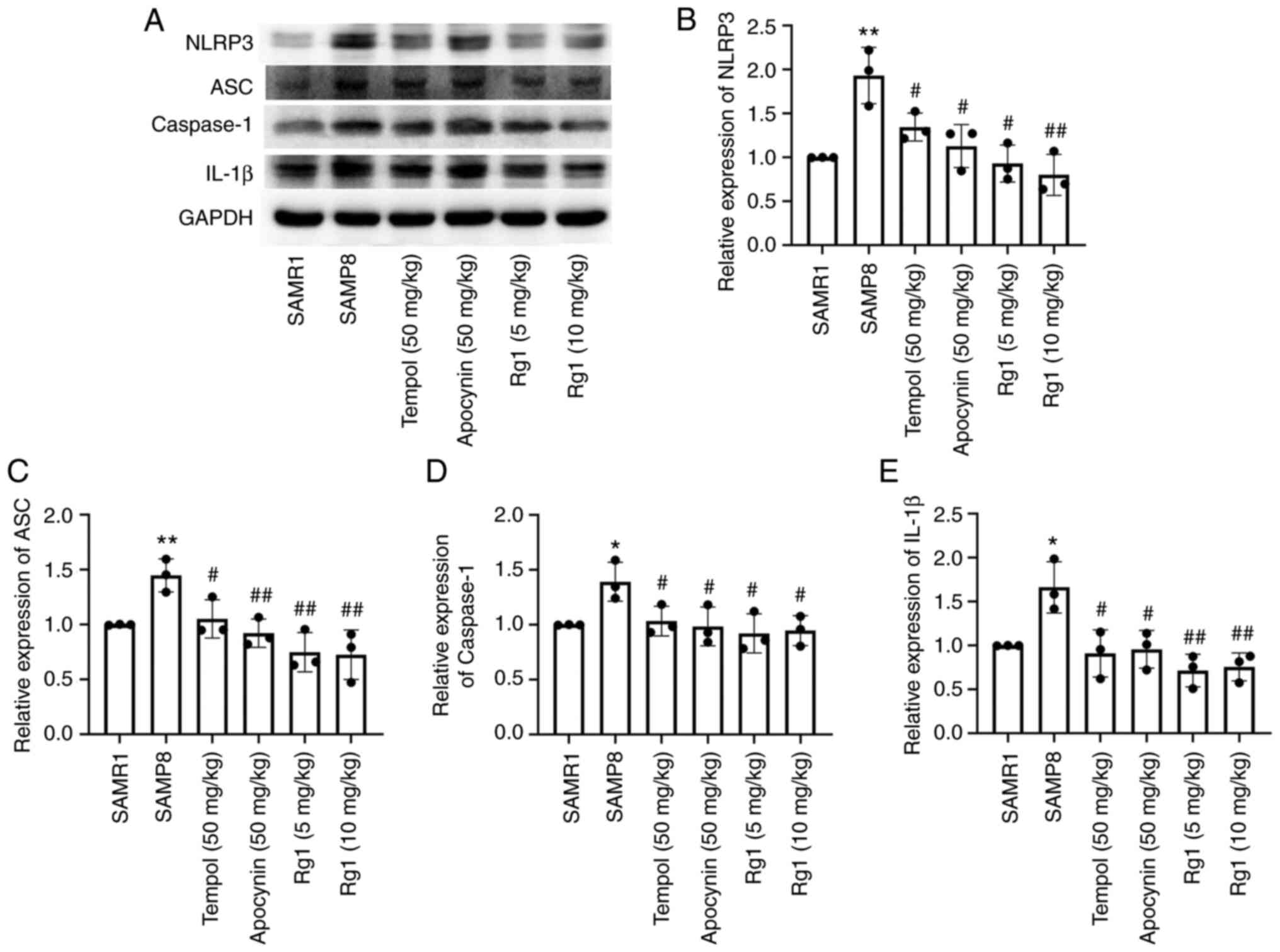 | Figure 6.Effects of Rg1 treatment on the
expression levels of NLRP3, ASC, caspase-1 and IL-1β in the liver
of SAMP8 mice. (A) Western blotting was used to analyze the
expression levels of NLRP3, ASC, caspase-1 and IL-1β in the liver
of SAMP8 mice. Semi-quantitative analysis of the relative
expression of (B) NLRP3, (C) ASC, (D) caspase-1 and (E) IL-1β.
Protein expression levels were normalized to GAPDH. Data are
presented as the mean ± SD; n=3. *P<0.05, **P<0.01 vs. SAMR1;
#P<0.05, ##P<0.01 vs. SAMP8. Rg1,
ginsenoside Rg1; SAMP8, senescence-accelerated mouse prone 8;
SAMR1, senescence-accelerated resistant mouse 1; NLRP3, NLR family
pyrin domain containing 3; ASC, apoptosis-associated speck-like
protein containing a C-terminal caspase recruitment domain. |
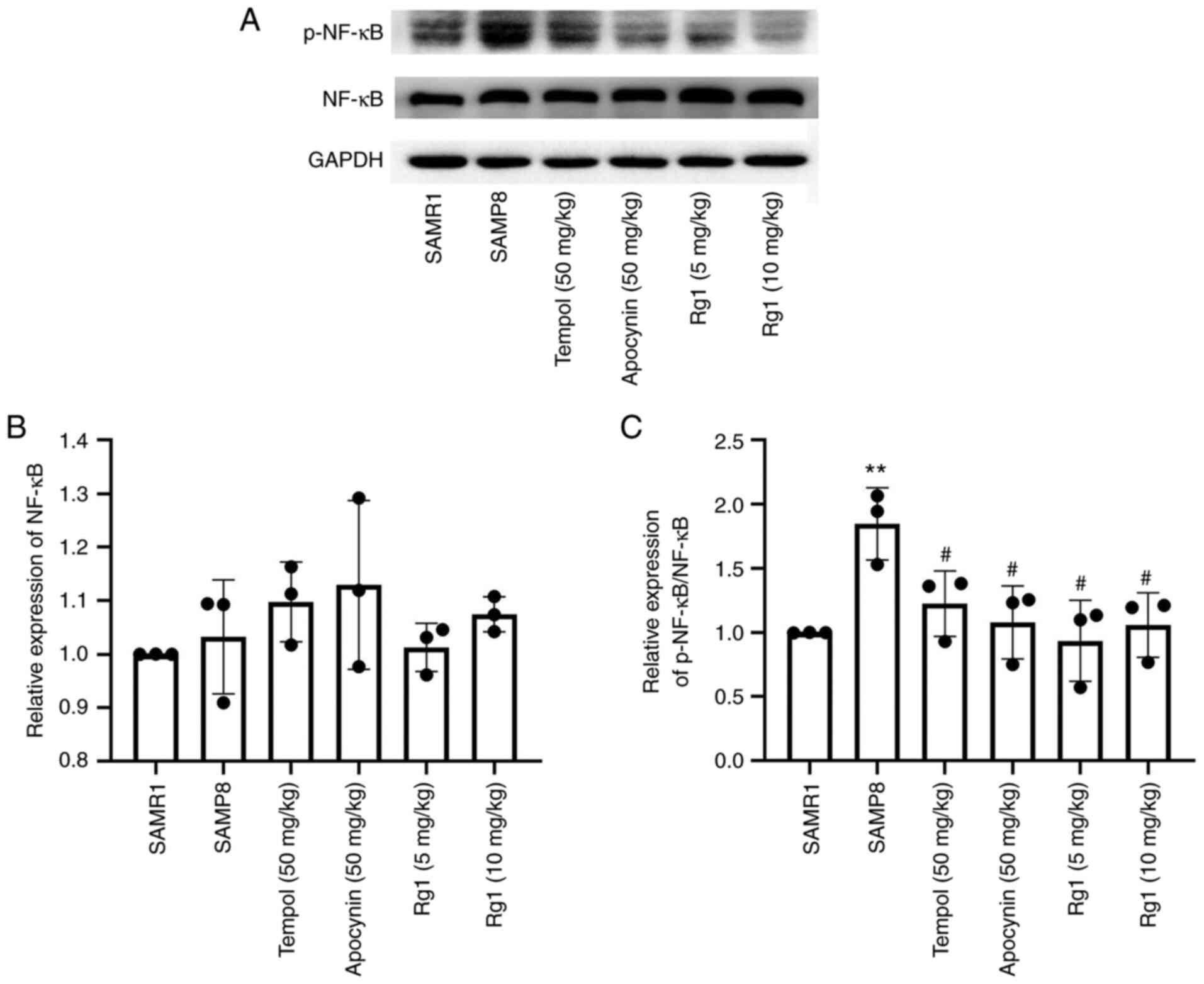
To further investigate whether the inflammatory
response was involved in liver fibrosis during aging, the
expression levels of NF-κB and p-NF-κB were measured in liver
tissues using western blotting. The results revealed that there
were no significant differences in the expression levels of NF-κB
among the groups (Fig. 8A and B).
However, the expression levels of p-NF-κB were significantly
increased in the SAMP8 group compared with the SAMR1 group
(Fig. 8A and C), and tempol,
apocynin and Rg1 (5 and 10 mg/kg) treatment significantly
downregulated the expression levels of p-NF-κB/NF-κB compared with
the SAMP8 group (Fig. 8A and C).
The results suggested that Rg1 may inhibit inflammation in the
liver during aging by decreasing the phosphorylation of NF-κB.
Discussion
Aging can promote the dysfunction of numerous
organs, such as the liver, kidney and brain (43). Aging has been demonstrated to
increase the susceptibility to hepatic inflammation or fibrosis
(5). Liver fibrosis destroys the
structure of the liver, leading to loss of liver cells and
disruption of liver function, ultimately resulting in liver failure
(44). However, the mechanisms of
aging-induced liver injury and fibrosis are still unclear and there
are no effective drugs for treating aging-related liver fibrosis.
Therefore, it is important to explore the mechanism of liver aging
and find appropriate medicines to prevent liver injury and fibrosis
at an early age to reduce the incidence of age-related liver
diseases.
Rg1, one of the main active components of ginseng,
has been previously investigated for its anti-inflammatory and
antioxidant properties and has been found to exert a protective
effect on the liver (45). Previous
studies have shown that Rg1 could inhibit the transformation of
hepatic stellate cells into myofibroblasts and inhibit liver
fibrosis in CCl4-induced mice (45,46).
Our previous study indicated that Rg1 (5 and 10 mg/kg) treatment
significantly improved aging-induced renal injury and fibrosis in
SAMP8 mice (35). The present study
was designed to study the effect and mechanism of Rg1 (5 and 10
mg/kg) treatment on aging-related liver injury and fibrosis in
SAMP8 mice during aging. The results indicated that Rg1 (5 and 10
mg/kg) treatment could protect against aging-induced liver injury
and fibrosis, especially the Rg1 (10 mg/kg) group. These results
also discovered that the arrangement of hepatocytes was abnormal
and disordered and most of the cells appeared to have vacuole-like
degeneration in the SAMP8 group, while Rg1 treatment showed a
significant improvement in liver histopathology. In addition, the
results discovered that NOX4/NLRP3 inflammasome signaling, which is
closely associated with age-related liver injury and liver fibrosis
(20), was significantly inhibited
by Rg1 (5 and 10 mg/kg) treatment, especially in the Rg1 (10 mg/kg)
group. These results suggested that Rg1 treatment may be effective
in preventing aged-related injury and fibrosis in a dose-dependent
manner, possibly by inhibiting NOX4 and the NLRP3 inflammasome.
Previous studies have shown that aging is an
important factor that is closely associated with the generation and
progression of liver fibrosis (5,47);
however, there is currently a lack of effective therapeutic
options. Ginseng is one of the most widely used natural products,
due to its wide range of pharmacological effects and biological
activities that can delay aging (48). The active ingredient, Rg1, has been
reported to exert protective effects on TGF-β-induced HSC-T6 cells
and CCl4-induced liver fibrosis in male Kunming mice
(31). In the present study, the
results indicated that the liver tissues were significantly damaged
and showed signs of fibrosis in 8-month-old SAMP8 mice, such as
disordered cell arrangement, vacuolar degeneration and excess
extracellular collagen IV deposition, sharing a number of
similarities with the findings of Dumeus et al (49). Notably, Rg1 treatment for 9 weeks
was found to significantly attenuate liver injury and extracellular
matrix accumulation in SAMP8 mice. It was previously shown that
TGF-β1 promoted liver fibrosis through Smad2 phosphorylation
(p-Smad2) and collagen synthesis, and depleting TGF-β1 could reduce
liver fibrosis (50). In the
present study, the results demonstrated that Rg1 treatment
significantly decreased the expression of TGF-β1 in the liver of
SAMP8 mice. Tempol is an active oxygen scavenger, which has been
reported to reduce the production of ROS in
H2O2-treated hippocampal neurons due to its
ROS scavenging capacity (51).
Apocynin is often used as an inhibitor of NADPH oxidase, and it has
been shown to possess a clear and obvious antioxidant effect on a
variety of central nervous system diseases, including Parkinson's
disease and Alzheimer's disease (52–54).
The current study also demonstrated that tempol and apocynin
exerted similar protective effects on aging-induced liver injury.
These data suggested that Rg1 treatment may significantly protect
against aging-induced liver injury and fibrosis in elderly
mice.
It is well known that excessive ROS-induced
oxidative stress plays a critical role in the pathogenesis of
aging, which is also closely related to age-related diseases,
including liver injury and fibrosis (7,55). The
imbalance between the production and clearance of ROS determines
the degree of oxidative stress (56). Multiple enzymes contribute to ROS
generation and oxidative stress in various tissues or cells. NOX
has been reported to be a major source of ROS in numerous types of
organ, such as kidney and liver, and previous research has shown
that NOX4 was a major ROS-producing enzyme in the liver (57). It has been reported that NOX4 plays
crucial roles in liver tissues under physiological conditions by
mediating ROS generation (14). The
accumulation of NOX4-derived ROS is also an important regulator in
promoting liver fibrosis (58). A
previous study showed that Rg1 could significantly improve the
survival rates of HepG2 and 293 cells in acetaminophen-induced
liver injury by reducing the excessive ROS production (59). Furthermore, evidence has shown that
Rg1 reduces ROS production by inhibiting NOX4 expression in obese
zebrafish induced by a high-fat diet (60). Our previous study found that Rg1
treatment could significantly downregulate the expression of NOX4
and p47phox in the kidney of aging SAMP8 mice (35). Similarly, the findings of the
present study found that the ROS production and the expression
levels of NOX4, p47phox and p22phox were significantly increased in
the liver of elderly SAMP8 mice compared with the control group,
suggesting that NOX4-mediated ROS accumulation is closely involved
in senescence-related liver fibrosis. Fernández-Garcia et al
(61) also demonstrated a
significant increase in ROS levels in the livers of SAMP8 mice. In
addition, the present results also indicated that tempol, apocynin
and Rg1 administration markedly reduced ROS production and the
expression levels of NOX4, p22phox and p47phox in the livers of
SAMP8 mice. These results suggested that Rg1 may improve
aging-induced liver injury by reducing NOX4-mediated ROS
accumulation.
Increasing evidence has shown that oxidative stress
may also be closely associated with the levels of inflammation,
which also plays a crucial role in aging-related diseases (62,63). A
previous study demonstrated that the accumulation of senescent
cells contributed to the development of fibrosis by secreting
proinflammatory cytokines and profibrotic mediators (64). ROS, as signaling molecules, have
been discovered to play important roles in numerous processes of
inflammation-related diseases. For example, deoxynivalenol induced
the inflammation of IPEC-J2 cells by promoting ROS production
(65,66). There has been a consensus that
inflammasome complexes play central roles in promoting inflammation
in a number of tissues, such as in the colon and brain (67–69).
Excessive ROS production is an important process for the activation
of inflammasome complexes (70–72).
The NLRP3 inflammasome has been reported to be extensively
expressed in the liver (20,73).
Previous studies have reported that the NLRP3 inflammasome was
activated in the progression of alcoholic liver disease (ALD), and
liver inflammation and steatosis were significantly improved in ALD
mice with knocked out NLRP3 or caspase-1 expression (74,75).
Activated NLRP3 can bind to ASC, causing procaspase-1 to be cleaved
into mature caspase-1. Caspase-1 further catalyzes the cleavage of
pro-IL-1β and pro-IL-18 into their mature forms, IL-1β and IL-18
(19). A previous study reported
that NLRP3 activation aggravated inflammation in liver disease and
subsequent fibrosis (76). In
addition, another study showed that Rg1 treatment improved
CCl4-induced acute liver injury by suppressing the
NF-κB/NLRP3 inflammasome signaling pathway (77). In the present study, the results
suggested that the expression levels of IL-1β, ASC, caspase-1 and
NLRP3 were upregulated in the liver tissues of SAMP8 mice. These
results are consistent with previous studies (35), and provide further support for the
role of the NLRP3 inflammasome in the occurrence of aging-related
liver injury in SAMP8 mice (78).
The improvement of inflammation and inhibition of NLRP3
inflammasome activation have been observed to exert protective
effects on target organs (76).
Similarly, the present study found that treatment with tempol,
apocynin and Rg1 significantly downregulated the expression levels
of IL-1β, ASC, caspase-1 and NLRP3 in the liver tissues of SAMP8
mice. It is well known that the NF-κB transcription factor family
is a central regulator of the inflammatory process (79). Phosphorylation of NF-κB at serine
536 is thought to be required for NF-κB activation and nuclear
translocation (80). It has been
reported that selective inhibition of NF-κB activity can suppress
CCl4-induced liver fibrosis (81). In this study, the results revealed
that the expression of p-NF-κB, which promotes the formation of the
NLRP3 inflammasome, was significantly upregulated in the liver
tissues of aging mice and was significantly decreased after Rg1
treatment for 9 weeks in SAMP8 mice. Altogether, these findings
indicated that the NLRP3 inflammasome may be an important target
and is closely involved in aging-induced liver injury. Furthermore,
inhibition of the NLRP3 inflammasome may be an important underlying
mechanism of Rg1 in preventing aging-related liver injury and
fibrosis.
In conclusion, the findings of the present study
suggested that Rg1 treatment may ameliorate aging-associated liver
injury and fibrosis in SAMP8 mice. Rg1 treatment significantly
decreased the levels of ROS accumulation and NOX4 expression. In
addition, Rg1 treatment significantly downregulated the expression
of the NLRP3 inflammasome in the liver of SAMP8 mice. These results
suggest that Rg1 may delay liver aging and reduce age-related liver
fibrosis by reducing NOX4-mediated ROS-induced oxidative stress and
inhibiting the activation of the NLRP3 inflammasome. Thus, Rg1 may
act as a potential treatment for the prevention of liver fibrosis
during aging.
However, the current study has several limitations.
For example, it only provided animal experimental results
demonstrating that Rg1 ameliorated aging-related liver fibrosis due
to the inhibition of NOX4 and NLRP3 inflammasome activation in
SAMP8 mice. In addition, the biochemical indicators and serological
indicators that represent liver function, inflammation and
oxidative stress levels were not measured. Future research will aim
to further study the effect of Rg1 treatment on aging-induced liver
function, inflammation and oxidative stress-related indicators in
the serum, such as aspartate aminotransferase, alanine
transaminase, IL-1β and superoxide dismutase. Furthermore, NOX4
inhibitors or NOX4 knockout experiments will be performed to
further investigate the exact underlying protective mechanism of
Rg1 against liver injury in vitro and in vivo.
Acknowledgements
We would like to thank Mrs. Zhirui Fang (Department
of Pharmacology, Anhui Medical University) for helping with
immunohistochemistry staining and Mr. Dake Huang (Synthetic
Laboratory of Basic Medicine College, Anhui Medical University) for
helping with frozen section.
Funding
The present study was supported by grants from the
National Natural Science Foundation of China (grant no. 81970630)
and the Major Science and Technology Projects in Anhui Province
(grant no. 201903a07020025).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
WZL and WPL conceived and designed the study; YL, DZ
and LL performed the experiments and statistical analysis and wrote
the manuscript; YH and XD confirmed the authenticity of all the raw
data. YH, XD, LY and XL helped to perform the experiments and wrote
part of the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The experimental procedures were approved by the
Animal Ethics Committee of Anhui Medical University (approval no.
LLSC20160183; Hefei, China) and performed in accordance with the
Guidelines for the Care and Use of Laboratory Animals.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Stahl EC, Haschak MJ, Popovic B and Brown
BN: Macrophages in the aging liver and age-related liver disease.
Front Immunol. 9:27952018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tominaga K and Suzuki HI: TGF-β signaling
in cellular senescence and aging-related pathology. Int J Mol Sci.
20:50022019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Iwaisako K, Brenner DA and Kisseleva T:
What's new in liver fibrosis? The origin of myofibroblasts in liver
fibrosis. J Gastroenterol Hepatol. 27 (Suppl 2):S65–S68. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Aydin MM and Akçalı KC: Liver fibrosis.
Turk J Gastroenterol. 29:14–21. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kim IH, Xu J, Liu X, Koyama Y, Ma HY,
Diggle K, You YH, Schilling JM, Jeste D, Sharma K, et al: Aging
increases the susceptibility of hepatic inflammation, liver
fibrosis and aging in response to high-fat diet in mice. Age
(Dordr). 38:291–302. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stefanatos R and Sanz A: The role of
mitochondrial ROS in the aging brain. FEBS Lett. 592:743–758. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Davalli P, Mitic T, Caporali A, Lauriola A
and D'Arca D: ROS, cell senescence, and novel molecular mechanisms
in aging and age-related diseases. Oxid Med Cell Longev.
2016:35651272016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kudryavtseva AV, Krasnov GS, Dmitriev AA,
Alekseev BY, Kardymon OL, Sadritdinova AF, Fedorova MS, Pokrovsky
AV, Melnikova NV, Kaprin AD, et al: Mitochondrial dysfunction and
oxidative stress in aging and cancer. Oncotarget. 7:44879–44905.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Luangmonkong T, Suriguga S, Mutsaers HAM,
Groothuis GMM, Olinga P and Boersema M: Targeting oxidative stress
for the treatment of liver fibrosis. Rev Physiol Biochem Pharmacol.
175:71–102. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tao Y, Qiu T, Yao X, Jiang L, Wang N,
Jiang J, Jia X, Wei S, Zhang J, Zhu Y, et al: IRE1α/NOX4 signaling
pathway mediates ROS-dependent activation of hepatic stellate cells
in NaAsO2-induced liver fibrosis. J Cell Physiol.
236:1469–1480. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Valko M, Leibfritz D, Moncol J, Cronin MT,
Mazur M and Telser J: Free radicals and antioxidants in normal
physiological functions and human disease. Int J Biochem Cell Biol.
39:44–84. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ushio-Fukai M: VEGF signaling through
NADPH oxidase-derived ROS. Antioxid Redox Signal. 9:731–739. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ghosh R, Siddharth M, Singh N, Kare PK,
Banerjee BD, Wadhwa N and Tripathi AK: Organochlorine
pesticide-mediated induction of NADPH oxidase and nitric-oxide
synthase in endothelial cell. J Clin Diagn Res. 11:BC09–BC12.
2017.PubMed/NCBI
|
|
14
|
Cheng Q, Li C, Yang CF, Zhong YJ, Wu D,
Shi L, Chen L, Li YW and Li L: Methyl ferulic acid attenuates liver
fibrosis and hepatic stellate cell activation through the
TGF-β1/Smad and NOX4/ROS pathways. Chem Biol Interact. 299:131–139.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen Y, Zhao C, Liu X, Wu G, Zhong J, Zhao
T, Li J, Lin Y, Zhou Y and Wei Y: Plumbagin ameliorates liver
fibrosis via a ROS-mediated NF-κB signaling pathway in vitro and in
vivo. Biomed Pharmacother. 116:1089232019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fougère B, Boulanger E, Nourhashémi F,
Guyonnet S and Cesari M: Chronic inflammation: Accelerator of
biological aging. J Gerontol A Biol Sci Med Sci. 72:1218–1225.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Soysal P, Arik F, Smith L, Jackson SE and
Isik AT: Inflammation, frailty and cardiovascular disease. Adv Exp
Med Biol. 1216:55–64. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Neves J and Sousa-Victor P: Regulation of
inflammation as an anti-aging intervention. FEBS J. 287:43–52.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mangan MSJ, Olhava EJ, Roush WR, Seidel
HM, Glick GD and Latz E: Targeting the NLRP3 inflammasome in
inflammatory diseases. Nat Rev Drug Discov. 17:588–606. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gallego P, Castejón-Vega B, Del Campo JA
and Cordero MD: The absence of NLRP3-inflammasome modulates hepatic
fibrosis progression, lipid metabolism, and inflammation in KO
NLRP3 mice during aging. Cells. 9:21482020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sun L, Ma W, Gao W, Xing Y, Chen L, Xia Z,
Zhang Z and Dai Z: Propofol directly induces caspase-1-dependent
macrophage pyroptosis through the NLRP3-ASC inflammasome. Cell
Death Dis. 10:5422019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Stancu IC, Cremers N, Vanrusselt H,
Couturier J, Vanoosthuyse A, Kessels S, Lodder C, Brône B, Huaux F,
Octave JN, et al: Aggregated tau activates NLRP3-ASC inflammasome
exacerbating exogenously seeded and non-exogenously seeded tau
pathology in vivo. Acta Neuropathol. 137:599–617. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yin Y, Pastrana JL, Li X, Huang X,
Mallilankaraman K, Choi ET, Madesh M, Wang H and Yang XF:
Inflammasomes: Sensors of metabolic stresses for vascular
inflammation. Front Biosci (Landmark Ed). 18:638–649. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Guo H, Callaway JB and Ting JP:
Inflammasomes: Mechanism of action, role in disease, and
therapeutics. Nat Med. 21:677–687. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Malik A and Kanneganti TD: Inflammasome
activation and assembly at a glance. J Cell Sci. 130:3955–3963.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang M, Zhu X, Tong H, Lou A, Li Y, Li Y,
Su L and Li X: AVE 0991 attenuates pyroptosis and liver damage
after heatstroke by inhibiting the ROS-NLRP3 inflammatory
signalling pathway. Biomed Res Int. 2019:18062342019.PubMed/NCBI
|
|
27
|
Baltanás A, Solesio ME, Zalba G, Galindo
MF, Fortuño A and Jordán J: The senescence-accelerated mouse
prone-8 (SAM-P8) oxidative stress is associated with upregulation
of renal NADPH oxidase system. J Physiol Biochem. 69:927–935. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Maroni L, Pinto C, Giordano DM, Saccomanno
S, Banales JM, Spallacci D, Albertini MC, Orlando F, Provinciali M,
Milkiewicz M, et al: Aging-related expression of twinfilin-1
regulates cholangiocyte biological response to injury. Hepatology.
70:883–898. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ye X, Meeker HC, Kozlowski PB, Wegiel J,
Wang KC, Imaki H and Carp RI: Pathological changes in the liver of
a senescence accelerated mouse strain (SAMP8): A mouse model for
the study of liver diseases. Histol Histopathol. 19:1141–1151.
2004.PubMed/NCBI
|
|
30
|
Xiong X, Ren Y, Cui Y, Li R, Wang C and
Zhang Y: Obeticholic acid protects mice against
lipopolysaccharide-induced liver injury and inflammation. Biomed
Pharmacother. 96:1292–1298. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wei X, Chen Y and Huang W: Ginsenoside Rg1
ameliorates liver fibrosis via suppressing epithelial to
mesenchymal transition and reactive oxygen species production in
vitro and in vivo. Biofactors. 44:327–335. 2018. View Article : Google Scholar
|
|
32
|
Kim DH: Chemical diversity of Panax
ginseng, Panax quinquifolium, and Panax notoginseng. J
Ginseng Res. 36:1–15. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Xu TZ, Shen XY, Sun LL, Chen YL, Zhang BQ,
Huang DK and Li WZ: Ginsenoside Rg1 protects against
H2O2-induced neuronal damage due to
inhibition of the NLRP1 inflammasome signalling pathway in
hippocampal neurons in vitro. Int J Mol Med. 43:717–726.
2019.PubMed/NCBI
|
|
34
|
Schmucker DL: Age-related changes in liver
structure and function: Implications for disease ? Exp Gerontol.
40:650–659. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shen X, Dong X, Han Y, Li Y, Ding S, Zhang
H, Sun Z, Yin Y and Li W and Li W: Ginsenoside Rg1 ameliorates
glomerular fibrosis during kidney aging by inhibiting NOX4 and
NLRP3 inflammasome activation in SAMP8 mice. Int Immunopharmacol.
82:1063392020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chen Y, Ding S, Zhang H, Sun Z, Shen X,
Sun L, Yin Y, Qun S and Li W: Protective effects of ginsenoside Rg1
on neuronal senescence due to inhibition of NOX2 and NLRP1
inflammasome activation in SAMP8 mice. J Funct Foods.
65:1037132020. View Article : Google Scholar
|
|
37
|
Jones-Bolin S: Guidelines for the care and
use of laboratory animals in biomedical research. Curr Protoc
Pharmacol. 4:4B2012.PubMed/NCBI
|
|
38
|
Wick MR: The hematoxylin and eosin stain
in anatomic pathology-an often-neglected focus of quality assurance
in the laboratory. Semin Diagn Pathol. 36:303–311. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lefkowitch JH: Special stains in
diagnostic liver pathology. Semin Diagn Pathol. 23:190–198. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Iezzoni JC: Diagnostic histochemistry in
hepatic pathology. Semin Diagn Pathol. 35:381–389. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yu H, Zhen J, Yang Y, Gu J, Wu S and Liu
Q: Ginsenoside Rg1 ameliorates diabetic cardiomyopathy by
inhibiting endoplasmic reticulum stress-induced apoptosis in a
streptozotocin-induced diabetes rat model. J Cell Mol Med.
20:623–631. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liu Q, Zhang FG, Zhang WS, Pan A, Yang YL,
Liu JF, Li P, Liu BL and Qi LW: Ginsenoside Rg1 inhibits
glucagon-induced hepatic gluconeogenesis through Akt-FoxO1
interaction. Theranostics. 7:4001–4012. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Childs BG, Durik M, Baker DJ and van
Deursen JM: Cellular senescence in aging and age-related disease:
From mechanisms to therapy. Nat Med. 21:1424–1435. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kisseleva T and Brenner DA: Mechanisms of
fibrogenesis. Exp Biol Med (Maywood). 233:109–122. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Mo C, Xie S, Zeng T, Lai Y, Huang S, Zhou
C, Yan W, Huang S, Gao L and Lv Z: Ginsenoside-Rg1 acts as an IDO1
inhibitor, protects against liver fibrosis via alleviating
IDO1-mediated the inhibition of DCs maturation. Phytomedicine.
84:1535242021. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Li JP, Gao Y, Chu SF, Zhang Z, Xia CY, Mou
Z, Song XY, He WB, Guo XF and Chen NH: Nrf2 pathway activation
contributes to anti-fibrosis effects of ginsenoside Rg1 in a rat
model of alcohol- and CCl4-induced hepatic fibrosis. Acta Pharmacol
Sin. 35:1031–1044. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Jin CJ, Baumann A, Brandt A, Engstler AJ,
Nier A, Hege M, Schmeer C, Kehm R, Höhn A, Grune T, et al:
Aging-related liver degeneration is associated with increased
bacterial endotoxin and lipopolysaccharide binding protein levels.
Am J Physiol Gastrointest Liver Physiol. 318:G736–G747. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Qin Q, Lin N, Huang H, Zhang X, Cao X,
Wang Y and Li P: Ginsenoside Rg1 ameliorates cardiac oxidative
stress and inflammation in streptozotocin-induced diabetic rats.
Diabetes Metab Syndr Obes. 12:1091–1103. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Dumeus S, Shibu MA, Lin WT, Wang MF, Lai
CH, Shen CY, Lin YM, Viswanadha VP, Kuo WW and Huang CY: Bioactive
peptide improves diet-induced hepatic fat deposition and hepatocyte
proinflammatory response in SAMP8 ageing mice. Cell Physiol
Biochem. 48:1942–1952. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xu F, Liu C, Zhou D and Zhang L:
TGF-β/SMAD pathway and its regulation in hepatic fibrosis. J
Histochem Cytochem. 64:157–167. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang Y, Liu Q, Xu Y, Zhang Y, Lv Y, Tan Y,
Jiang N, Cao G, Ma X, Wang J, et al: Ginsenoside Rg1 protects
against oxidative stress-induced neuronal apoptosis through myosin
IIA-actin related cytoskeletal reorganization. Int J Biol Sci.
12:1341–1356. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hou L, Sun F, Huang R, Sun W, Zhang D and
Wang Q: Inhibition of NADPH oxidase by apocynin prevents learning
and memory deficits in a mouse Parkinson's disease model. Redox
Biol. 22:1011342019. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Nayernia Z, Jaquet V and Krause KH: New
insights on NOX enzymes in the central nervous system. Antioxid
Redox Signal. 20:2815–2837. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Langley M, Ghosh A, Charli A, Sarkar S, Ay
M, Luo J, Zielonka J, Brenza T, Bennett B, Jin H, et al:
Mito-apocynin prevents mitochondrial dysfunction, microglial
activation, oxidative damage, and progressive neurodegeneration in
MitoPark transgenic mice. Antioxid Redox Signal. 27:1048–1066.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hsieh CC and Papaconstantinou J:
Thioredoxin-ASK1 complex levels regulate ROS-mediated p38 MAPK
pathway activity in livers of aged and long-lived Snell dwarf mice.
FASEB J. 20:259–268. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wojtovich AP and Foster TH: Optogenetic
control of ROS production. Redox Biol. 2:368–376. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Huang C, Gan D, Luo F, Wan S, Chen J, Wang
A, Li B and Zhu X: Interaction mechanisms between the NOX4/ROS and
RhoA/ROCK1 signaling pathways as new anti-fibrosis targets of
ursolic acid in hepatic stellate cells. Front Pharmacol.
10:4312019. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lan T, Kisseleva T and Brenner DA:
Deficiency of NOX1 or NOX4 prevents liver inflammation and fibrosis
in mice through inhibition of hepatic stellate cell activation.
PLoS One. 10:e01297432015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Gao Y, Chu SF, Zhang Z, Ai QD, Xia CY,
Huang HY and Chen NH: Ginsenoside Rg1 prevents
acetaminophen-induced oxidative stress and apoptosis via Nrf2/ARE
signaling pathway. J Asian Nat Prod Res. 21:782–797. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Koh EJ, Kim KJ, Choi J, Jeon HJ, Seo MJ
and Lee BY: Ginsenoside Rg1 suppresses early stage of adipocyte
development via activation of C/EBP homologous protein-10 in 3T3-L1
and attenuates fat accumulation in high fat diet-induced obese
zebrafish. J Ginseng Res. 41:23–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Fernández-Garcia C, Rancan L, Paredes SD,
Montero C, de la Fuente M, Vara E and Tresguerres JAF: Xanthohumol
exerts protective effects in liver alterations associated with
aging. Eur J Nutr. 58:653–663. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Pinto C, Ninfole E, Benedetti A, Maroni L
and Marzioni M: Aging-related molecular pathways in chronic
cholestatic conditions. Front Med (Lausanne). 6:3322020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Stakos DA, Stamatelopoulos K, Bampatsias
D, Sachse M, Zormpas E, Vlachogiannis NI, Tual-Chalot S and Stellos
K: The Alzheimer's disease amyloid-beta hypothesis in
cardiovascular aging and disease: JACC focus seminar. J Am Coll
Cardiol. 75:952–967. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Sun X, Nkennor B, Mastikhina O, Soon K and
Nunes SS: Endothelium-mediated contributions to fibrosis. Semin
Cell Dev Biol. 101:78–86. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Zhu H and Li YR: Oxidative stress and
redox signaling mechanisms of inflammatory bowel disease: Updated
experimental and clinical evidence. Exp Biol Med (Maywood).
237:474–480. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Kang R, Li R, Dai P, Li Z, Li Y and Li C:
Deoxynivalenol induced apoptosis and inflammation of IPEC-J2 cells
by promoting ROS production. Environ Pollut. 251:689–698. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ralston JC, Lyons CL, Kennedy EB, Kirwan
AM and Roche HM: Fatty acids and NLRP3 inflammasome-mediated
inflammation in metabolic tissues. Annu Rev Nutr. 37:77–102. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Elinav E, Strowig T, Kau AL, Henao-Mejia
J, Thaiss CA, Booth CJ, Peaper DR, Bertin J, Eisenbarth SC, Gordon
JI and Flavell RA: NLRP6 inflammasome regulates colonic microbial
ecology and risk for colitis. Cell. 145:745–757. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Song AQ, Gao B, Fan JJ, Zhu YJ, Zhou J,
Wang YL, Xu LZ and Wu WN: NLRP1 inflammasome contributes to chronic
stress-induced depressive-like behaviors in mice. J
Neuroinflammation. 17:1782020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Yu X, Lan P, Hou X, Han Q, Lu N, Li T,
Jiao C, Zhang J, Zhang C and Tian Z: HBV inhibits LPS-induced NLRP3
inflammasome activation and IL-1β production via suppressing the
NF-κB pathway and ROS production. J Hepatol. 66:693–702. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Cheng YC, Chu LW, Chen JY, Hsieh SL, Chang
YC, Dai ZK and Wu BN: Loganin attenuates high glucose-induced
schwann cells pyroptosis by inhibiting ROS generation and NLRP3
inflammasome activation. Cells. 9:19482020. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Sho T and Xu J: Role and mechanism of ROS
scavengers in alleviating NLRP3-mediated inflammation. Biotechnol
Appl Biochem. 66:4–13. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Wree A, McGeough MD, Inzaugarat ME, Eguchi
A, Schuster S, Johnson CD, Peña CA, Geisler LJ, Papouchado BG,
Hoffman HM and Feldstein AE: NLRP3 inflammasome driven liver injury
and fibrosis: Roles of IL-17 and TNF in mice. Hepatology.
67:736–749. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Mridha AR, Wree A, Robertson AAB, Yeh MM,
Johnson CD, Van Rooyen DM, Haczeyni F, Teoh NC, Savard C, Ioannou
GN, et al: NLRP3 inflammasome blockade reduces liver inflammation
and fibrosis in experimental NASH in mice. J Hepatol. 66:1037–1046.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Cao Z, Fang Y, Lu Y, Tan D, Du C, Li Y, Ma
Q, Yu J, Chen M, Zhou C, et al: Melatonin alleviates
cadmium-induced liver injury by inhibiting the TXNIP-NLRP3
inflammasome. J Pineal Res. 62:e123892017. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Wree A, Eguchi A, McGeough MD, Pena CA,
Johnson CD, Canbay A, Hoffman HM and Feldstein AE: NLRP3
inflammasome activation results in hepatocyte pyroptosis, liver
inflammation, and fibrosis in mice. Hepatology. 59:898–910. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Zhao J, He B, Zhang S, Huang W and Li X:
Ginsenoside Rg1 alleviates acute liver injury through the induction
of autophagy and suppressing NF-κB/NLRP3 inflammasome signaling
pathway. Int J Med Sci. 18:1382–1389. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Cuesta S, Kireev R, Forman K, García C,
Escames G, Ariznavarreta C, Vara E and Tresguerres JA: Melatonin
improves inflammation processes in liver of senescence-accelerated
prone male mice (SAMP8). Exp Gerontol. 45:950–956. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Lawrence T: The nuclear factor NF-kappaB
pathway in inflammation. Cold Spring Harb Perspect Biol.
1:a0016512009. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Dolcet X, Llobet D, Pallares J and
Matias-Guiu X: NF-kB in development and progression of human
cancer. Virchows Arch. 446:475–482. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Zhou D, Huang W, Wei J, Zhang J, Liu Z, Ji
R, Ge S, Xiao M, Fan Y and Lu C: RelB promotes liver fibrosis via
inducing the release of injury-associated inflammatory cytokines. J
Cell Mol Med. 24:6008–6014. 2020. View Article : Google Scholar : PubMed/NCBI
|