Introduction
Breast cancer is the leading cause of
cancer-associated mortality (mortality rate, 11.6%) in women
worldwide (1). A total of ~80% of
breast tumors are estrogen receptor (ER) positive and often respond
to chemotherapeutic agents, such as tamoxifen, which is a selective
ER modulator (2). Tamoxifen
induces cell cycle arrest and inhibits tumor growth (3,4);
therefore, tamoxifen therapy reduces breast cancer recurrence
(5). However, tamoxifen
resistance limits its clinical applications (6) and the potential mechanisms
underlying tamoxifen resistance remain unclear.
Autophagy can cause drug resistance, and thus lead
to a reduction in the benefits of chemotherapy (7). Alterations in autophagy have been
considered to be a mechanism of tamoxifen resistance (8). The synthetic ER antagonist
4-hydroxytamoxifen (4-OHT) induces autophagy and decreases
apoptosis in ER-positive breast cancer cells (9).
To date, >30 autophagy-related genes have been
identified (10). Among them,
autophagy-related (ATG) protein 4 (ATG4), a cysteine protease,
serves an important role in the ATG8/LC3 lipid conjugation system,
which is essential for the late step of autophagosome formation
(11). The human genome contains
four ATG4 homologous genes: ATG4A, ATG4B, ATG4C and ATG4D (12). Previous studies have reported that
ATG4A cleaves gamma-aminobutyric acid receptor-associated protein
subfamily isoforms (a subfamily of the ATG8 proteins) (11,13). ATG4A is overexpressed in multiple
human cancers, such as osteosarcoma (14) and gastric cancer (15), and its overexpression is often
associated with the progression of malignancy. A previous study has
reported that ATG4A participates in stem cell maintenance during
breast cancer (16). Therefore,
an improved understanding of the role of ATG4A during tamoxifen
resistance would be beneficial in treatment planning for patients
undergoing chemotherapy to treat breast cancer.
In the present study, it was aimed to investigate
whether interference with ATG4A could affect autophagy and 4-OHT
sensitivity in breast cancer cell lines and elucidate its
underlying molecular mechanisms.
Materials and methods
Cell culture and treatment
Human breast cancer MCF7 cells (The Cell Bank of
Type Culture Collection of The Chinese Academy of Sciences) were
cultured in DMEM: F-12 (HyClone; Cytiva) containing 10% FBS
(HyClone; Cytiva), 100 U/ml penicillin (Thermo Fisher Scientific,
Inc.), 100 mg/ml streptomycin (Thermo Fisher Scientific, Inc.), 2
mM GlutaMax (HyClone; Cytiva) and 6 ng/ml insulin (HyClone; Cytiva)
at 37°C and 5% CO2. MCF7/TAMR-7 cells (MCF7/R; Ximbio),
which are tamoxifen-resistant, were cultured in the presence of 1
µM 4-OHT (Sigma-Aldrich; Merck KGaA) under the same conditions as
aforementioned.
MCF7 and MCF7/R cells were divided into control,
4-OHT and 4-OHT+3-methyladenine (3MA) groups. Cells in the 4-OHT
group were treated with 10 µM 4-OHT with DMSO as the vehicle. Cells
in 4-OHT+3-MA group were treated with 10 µM 4-OHT and 1 mM 3MA
(Sigma-Aldrich; Merck KGaA) with DMSO as the vehicle. MCF7/R cells
in rapamycin group were treated with 10 nM rapamycin (Selleck
Chemicals) with DMSO as the vehicle. Cells in control group were
treated with DMSO in follow up experiments.
Lentiviral infection and
treatment
Lentiviral plasmid (GV248; GeneChem, Inc.) carrying
the knockdown vector of ATG4A [short hairpin (sh)-ATG4A] and empty
vector were purchased from Shanghai GenePharma Co., Ltd. The
sequences of sh-ATG4A and sh-negative control (NC) were as follows:
sh-ATG4A sense, 5′-GGATGTTTGAATTAGTTCA-3′ and antisense,
5′-TGAACTAATTCAAACATCC-3′; and sh-NC sense,
5′-TTCTCCGAACGTGTCACGTTT-3′ and antisense,
5′-ACGTGACACGTTCGGAGAATT-3′. A second-generation system was used in
this experiment. Briefly, 293T cells (The Cell Bank of Type Culture
Collection of The Chinese Academy of Sciences) were seeded into
six-well plates (4×105 cells/well) and transfected with
lentiviral plasmid, psPAX2 (Biovector NTCC Inc.) and PMD2G
(Biovector NTCC Inc.) in the following ratio: 10:7.5:2.5 µg.
Transfection was conducted using lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C for 48 h.
Next, lentiviral particles were collected after centrifugation at
50,000 × g for 2 h. MCF7/R cells (5×104 cells/well) were
seeded into 24-well plates and then infected with lentivirus
supernatant (1×108 TU/ml) at a multiplicity of infection
of 20 in the presence of 8 mg/ml polybrene (MilliporeSigma). After
48 h of incubation, stably transfected cell lines were selected
with 2.5 µg/ml puromycin (Sigma-Aldrich; Merck KGaA) for 1 week.
The stably transfected cell lines were maintained under 0.5 µg/ml
puromycin. After transfection for 24 h, subsequent experiments were
performed. Cells were treated with 4-OHT (10 µM). Additionally, 20
µM SKL2001 (MedChemExpress) was added to the 4-OHT + sh-ATG4A
group.
Cell Counting Kit-8 (CCK-8) assay
MCF7/R cells (2×103 cells/well) were
incubated at 37°C and 5% CO2 for 24 h after exposure to
tamoxifen (10 µM). The cells were then cultured with 10 µl CCK-8
solution (Beyotime Institute of Biotechnology) for 2 h at 37°C. The
absorbance was measured at 450 nm using a microplate reader (Dynex
Technologies, Inc.).
Flow cytometric analysis of
apoptosis
According to the manufacturer's instructions, cell
apoptosis was detected using an Annexin V-FITC kit (Beyotime
Institute of Biotechnology). Briefly, MCF-7/R cells
(5×104 cells/well) were harvested and centrifuged at
1,000 × g for 5 min at room temperature. Cells were then stained
with Annexin V-FITC and propidium iodide for 15 min at room
temperature in the dark. Finally, apoptosis was analyzed using a BD
Accuri™ C6 Plus flow cytometer (BD Biosciences) and the rate of
apoptosis was calculated using BD Accuri C6 Software (BD
Biosciences).
Green fluorescent protein (GFP)-LC3
analysis
MCF7 and MCF7/R cells were seeded into 12-well
plates (6×104 cells/well) at 37°C overnight, and then
transfected with the GFP-LC3 expression plasmids (2 µg/l; LC3;
Hanbio Biotechnology Co., Ltd.) using lipofectamine®
2000 at 37°C for 24 h. After transfection for 24 h, cells were
treated with 4-OHT (10 µM), 3MA (1 mM) or rapamycin (10 nM) for 24
h. Cells were then fixed with 4% formaldehyde for 10 min at room
temperature and observed under a confocal microscope (Leica
Microsystems GmbH). The number of LC3 puncta was counted using at
least 20 cells from six randomly selected fields, and each
experiment was repeated three times.
Western blot analysis
MCF7 and MCF7/R cells were lysed in a RIPA lysis
buffer (Sigma-Aldrich; Merck KGaA). According to the manufacturer's
protocol, NE-PER nuclear and cytoplasmic extraction reagents
(Thermo Scientific Pierce) was used to extract nuclear and
cytoplasmic protein from MCF7/R cells. Equal amounts of protein
samples (30 µg per lane) were separated via 12% SDS-PAGE and
transferred to PVDF membranes (EMD Millipore). After blocking using
TBS with 0.05% Tween-20 (TBST) containing 5% non-fat milk at room
temperature for 2 h, the membranes were incubated with the
following primary antibodies: ATG4A (cat. no. ab108322; 1:1,000;
Abcam), LC3 (cat. no. 3868, 1:1,000), p62 (cat. no. 8025; 1:1,000),
phosphorylated (p)-GSK-3β (cat. no. 9322; 1:1,000; all from Cell
Signaling Technology, Inc.), GSK-3β (cat. no. ab32391; 1:1,000;
Abcam), β-catenin (cat. no. 8480; 1:1,000; Cell Signaling
Technology, Inc.), cyclin D1 (cat. no. 55506, 1:1,000; Cell
Signaling Technology, Inc.), c-myc (cat. no. ab32072; 1:1,000;
Abcam), Bax (cat. no. 5023; 1:1,000), Bcl-2 (cat. no. 3498;
1:1,000), GAPDH (cat. no. 5174; 1:1,000), histone H3 (cat. no.
4499; 1:1,000; all from Cell Signaling Technology, Inc.) and
β-actin (cat. no. ab179467; 1:1,000; Abcam) at 4°C overnight. After
washing with TBST, the membranes were incubated with horseradish
peroxidase-labelled secondary antibody (cat. no. 7074; 1:2,000;
Cell Signaling Technology, Inc.) at room temperature for 1 h. The
bands were visualized using Pierce™ ECL Plus Western Blotting
Substrate (Pierce; Thermo Fisher Scientific, Inc.) and detected
with ImageJ v1.8.0 (National Institutes of Health).
Immunofluorescence
MCF7/R cells were fixed with 4% paraformaldehyde for
20 min at room temperature and incubated with 0.3% Triton X-100 for
30 min at room temperature. The cells were incubated with primary
antibodies targeting β-catenin (1:200; cat. no. 8480; Cell
Signaling Technology, Inc.) overnight at 4°C and then incubated
with goad anti-rabbit Alexa Fluor 488 (1:300; cat. no. A11008;
Thermo Fisher Scientific, Inc.) for 1 h at 37°C. The cells were
then incubated with 5 µg/ml DAPI for 20 min at room temperature and
observed under a fluorescence microscope (Leica Microsystems
GmbH).
The Cancer Genome Atlas (TCGA)
database analysis
ATG4A expression data were obtained from the TCGA
database (https://tcga-data.nci.nih.gov/tcga/) using the R
package TCGAbiolinks (17). These
data included 1,097 breast cancer samples and 114 adjacent normal
samples. Subsequently, the clinical information of 439 patients
with breast cancer were downloaded from the TCGA database. After
excluding 8 patients with breast cancer with incomplete relevant
information, 431 patients were divided into two groups based on the
median value of ATG4A levels. The clinical characteristics (age and
TNM stage) of the patients were analyzed. After excluding 73
patients with breast cancer with an overall survival ≤30 days, 358
patients with breast cancer were divided into two groups (high and
low) based on a group cut-off of ‘quartile’. Survival analysis was
performed with the R package ‘survival’ (18) using the Kaplan-Meier method.
Gene set enrichment analysis
(GSEA)
To further explore the pathway in which ATG4A may be
involved, the Molecular Signatures Database (v7.4; http://www.gsea-msigdb.org/gsea/msigdb/index.jsp)
hallmark gene set was used to perform GSEA. GSEA parameters were
set as follows: Number of permutations=1,000; permutation
type=gene_set; enrichment statistic=weighted; metric for ranking
genes=Signal2Noise. Gene sets with a positive (or negative)
enrichment score and P<0.05 after 1,000 permutations were
considered as significantly enriched gene sets.
Statistical analysis
Data are presented as the mean ± standard deviation
of three experiments. Computer-based calculations were conducted
using GraphPad Prism 7 (GraphPad Software, Inc.). Statistical
analysis was performed using unpaired Student's t-test and one-way
analysis of variance with Tukey's post hoc test. χ2 test
was used to assess the relationship between ATG4A expression and
clinicopathological features in patients with breast cancer.
Kaplan-Meier analysis was performed using log-rank test. P<0.05
was considered to indicate a statistically significant
difference.
Results
ATG4A levels are associated with the
survival of patients with breast cancer in The Cancer Genome Atlas
(TCGA) database
To investigate the clinical relevance of ATG4A
expression, ATG4A expression in TCGA database was firstly analyzed.
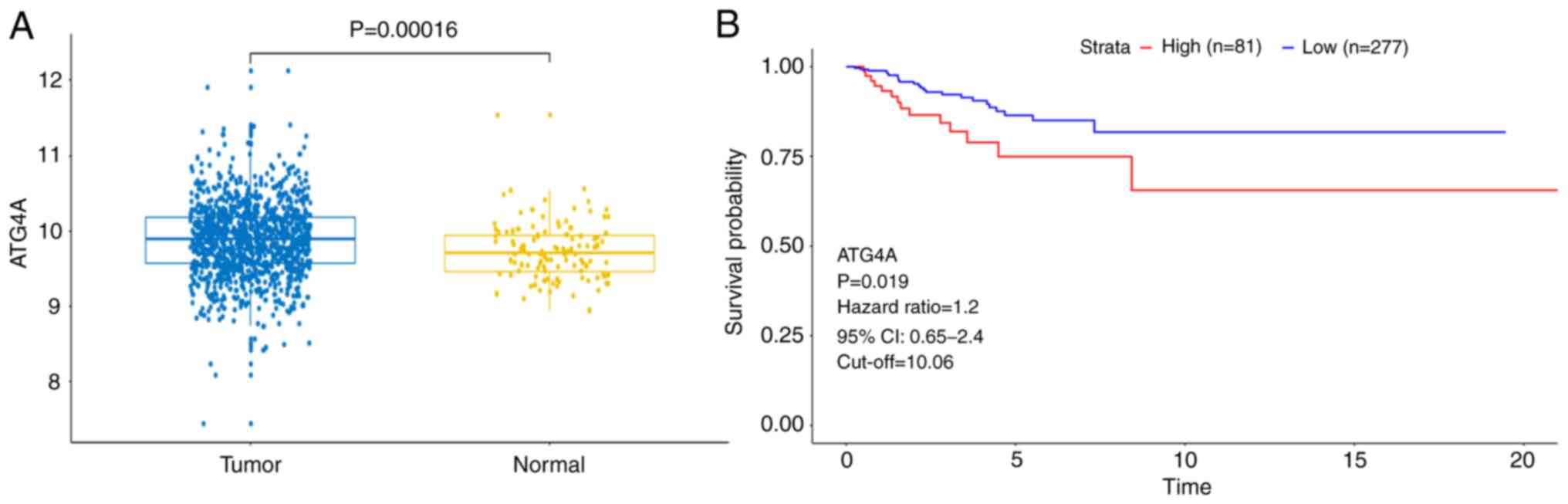
The results revealed that the expression of ATG4A was higher in
breast cancer tissues than in a number of adjacent normal tissues
(Fig. 1A). Subsequently, the
clinical characteristics (age and TNM stage) of the patients were
analyzed. As demonstrated in Table
I, high expression of ATG4A was significantly associated with
age, but not with TNM stage. In addition, the results of
Kaplan-Meier survival analysis revealed that patients with a higher
ATG4A expression exhibited poor disease-free survival (Fig. 1B). These results indicated that
ATG4A may be involved in the pathogenesis of breast cancer.
 | Table I.Relationship between ATG4A expression
and clinicopathological features in patients with breast
cancer. |
Table I.
Relationship between ATG4A expression
and clinicopathological features in patients with breast
cancer.
|
| ATG4A |
|
|---|
|
|
|
|
|---|
| Characteristic | Low expression,
n | High expression,
n | P-value |
|---|
| Age, years |
|
| 0.0006 |
|
<50 | 89 | 55 |
|
|
≥50 | 127 | 160 |
|
| TNM stage |
|
| 0.5786 |
|
I/II | 166 | 170 |
|
|
III/IV | 50 | 45 |
|
ATG4A expression and autophagy are
enhanced in MCF7/R cells
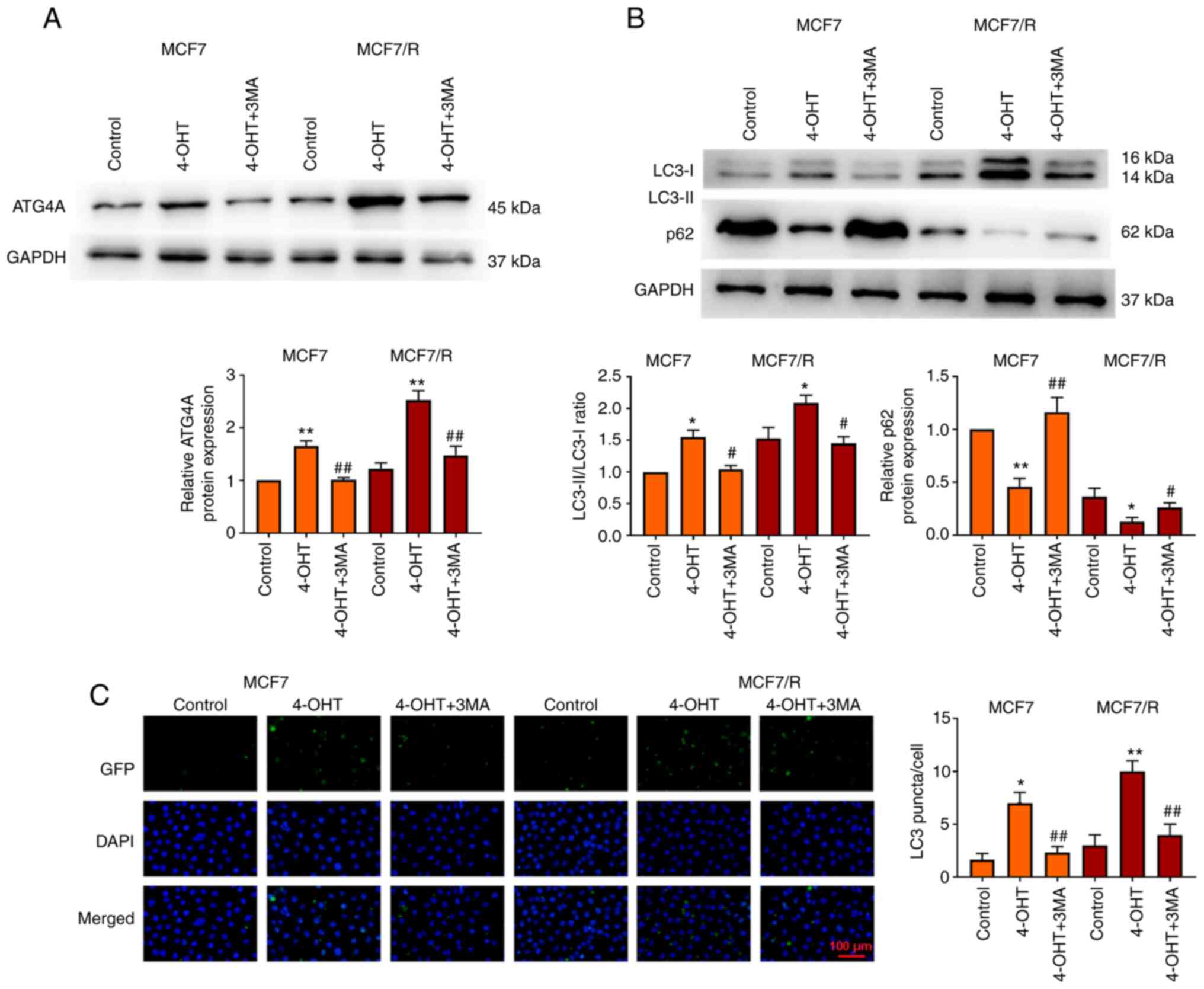
To explore the molecular mechanism of 4-OHT
resistance in breast cancer cells, a 4-OHT-resistant breast cancer
cell line (MCF7/R) was used. It was investigated whether ATG4A
mediated the regulatory effect of autophagy on 4-OHT resistance in
breast cancer. MCF7 and MCF7/R cells were treated with the
autophagy inhibitor 3MA and the expression of ATG4A and autophagy
genes in vitro was detected. The results revealed that
treatment with 4-OHT increased ATG4A expression in MCF7 and MCF7/R
cells compared with control group, while treatment with 3MA
decreased ATG4A expression compared with 4-OHT group (Fig. 2A). The LC3-II/LC3-I ratio
increased and the level of p62 decreased in MCF7/R cells compared
with that in MCF7 cells. Compared with control group, treatment
with 4-OHT significantly increased the LC3-II/LC3-I ratio and
decreased the level of p62, particularly in MCF7/R cells; however,
this effect of 4-OHT was reversed by 3MA (Fig. 2B). In addition, the number of LC3
puncta increased in MCF7 and MCF7/R cells after treatment with
4-OHT, but decreased after treatment with 3MA (Fig. 2C).
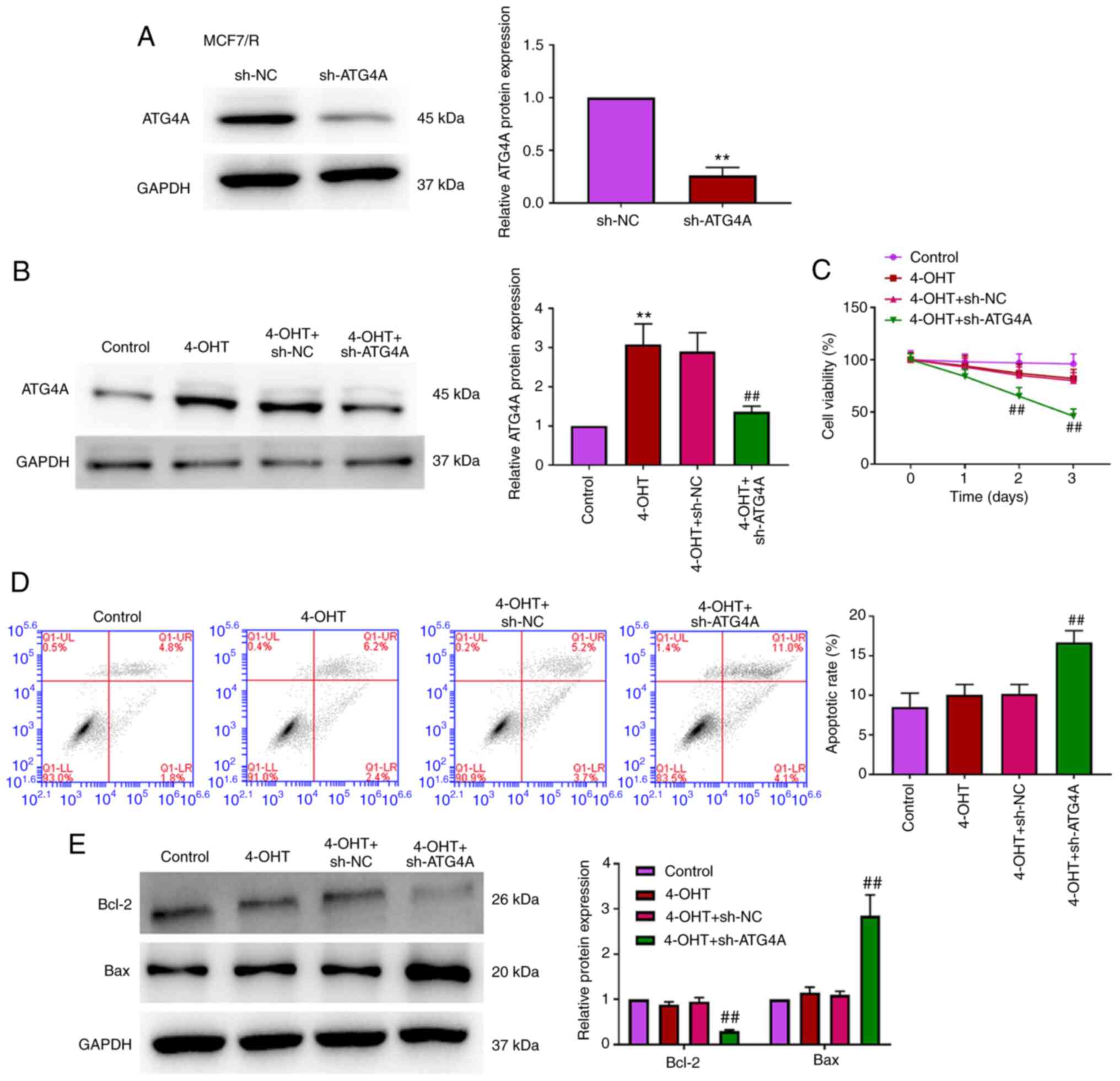
ATG4A knockdown enhances the
sensitivity of breast cancer cells to 4-OHT
To investigate the role of ATG4 in 4-OHT resistance
in breast cancer cells, MCF7/R cells were transfected with sh-ATG4
to establish a stable knockdown ATG4A cell line. As demonstrated in
Fig. 3A and B, ATG4A expression
was significantly inhibited in MCF7/R cells transfected with
sh-ATG4. CCK-8 and flow cytometric apoptosis analysis revealed that
there was no significant difference in cell viability and apoptosis
between the control and 4-OHT groups, while the knockdown of ATG4A
reduced viability and apoptosis in MCF7/R cells treated with 4-OHT
(Fig. 3C and D). Additionally,
the expression of apoptosis-related proteins (Bcl-2 and Bax) was
analyzed via western blot analysis. The results demonstrated that
the knockdown of ATG4A decreased Bcl-2 expression and increased Bax
expression (Fig. 3E).
ATG4A knockdown attenuates
tamoxifen-induced autophagy
Previous studies have demonstrated that ATG4A is
closely related to autophagy (19–21). However, the role of ATG4A during
autophagy in breast cancer cells remains unclear. To further
confirm the role of ATG4A in autophagy, MCF7/R cells were
transfected with sh-ATG4 to perform loss-of-function analysis. As
revealed in Fig. 4A and C, after
treatment with 4-OHT, the knockdown of ATG4A decreased the
LC3-II/LC3-I ratio, increased the level of p62, and decreased the
number of LC3 puncta compared with 4-OHT + sh-NC group. After
treatment with rapamycin (an autophagy activator), the LC3II/LC3I
ratio increased, p62 level decreased, and the number of LC3 puncta
increased compared with control group. The knockdown of ATG4A
reversed the effect of rapamycin on the LC3-II/LC3-I ratio, level
of p62 and number of LC3 puncta (Fig.
4B and D).
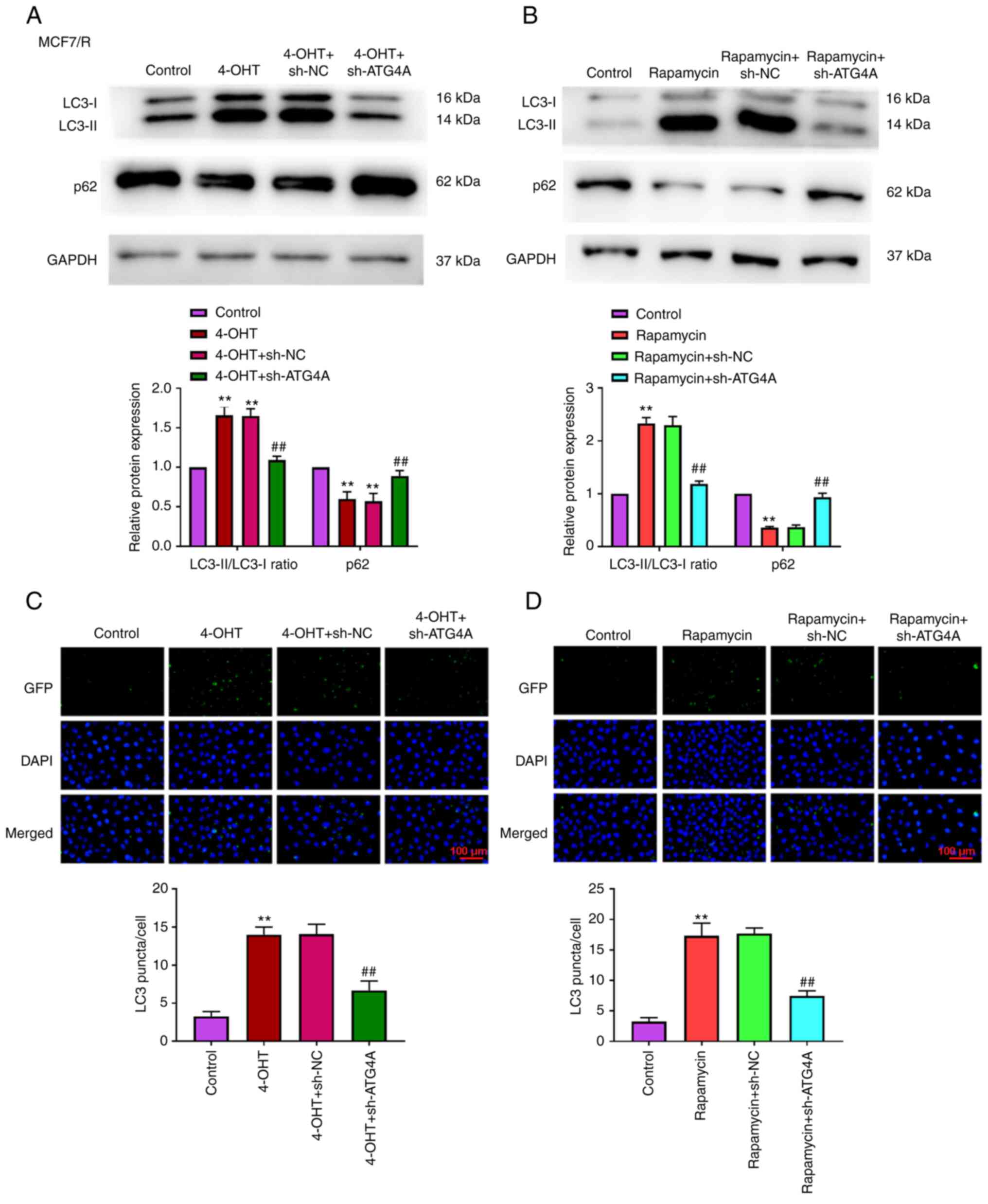 | Figure 4.ATG4A knockdown attenuates
tamoxifen-induced autophagy in MCF7/R cells. (A) After treatment
with 4-OHT, the expression of LC3 and p62 in MCF7/R cells was
determined using western blot analysis. (B) After treatment with
rapamycin, the expression of LC3 and p62 in MCF7/R cells was
determined using western blot analysis. (C) After treatment with
4-OHT, the number of LC3-puncta in MCF7/R cells was counted (scale
bar, 100 µm). (D) After treatment with rapamycin, the number of
LC3-puncta in MCF7/R cells was counted (scale bar, 100 µm).
**P<0.01 vs. control group; ##P<0.01 vs. 4-OHT +
sh-NC or rapamycin + sh-NC group. ATG4A, autophagy-related 4A;
4-OHT, 4-hydroxytamoxifen; sh-, short hairpin; NC, negative
control. |
ATG4A knockdown inhibits the
Wnt/β-catenin pathway
To further explore the mechanism of ATG4A in breast
cancer cells, GSEA was performed to identify the pathway
responsible for the effects of ATG4A. The results indicated that
ATG4A was enriched in the Wnt/β-catenin pathway (Fig. 5A). The Wnt/β-catenin pathway
serves an important role in cancer progression and chemoresistance
(22). Compared with the
4-OHT+sh-NC group, the knockdown of ATG4A increased p-GSK3β
expression and decreased the expression of β-catenin, cyclin D1 and
c-myc in MCF7/R cells (Fig. 5B).
Compared with the 4-OHT+sh-NC group, the knockdown of ATG4A
decreased the expression of β-catenin in the nucleus (Fig. 5C and D). SKL2001 is an activator
of the Wnt/β-catenin signaling pathway (23). Treatment with SKL2001 reversed the
effect of ATG4A knockdown on cell viability, apoptosis and the
Wnt/β-catenin signaling pathway in MCF7/R cells (Fig. 5E-G).
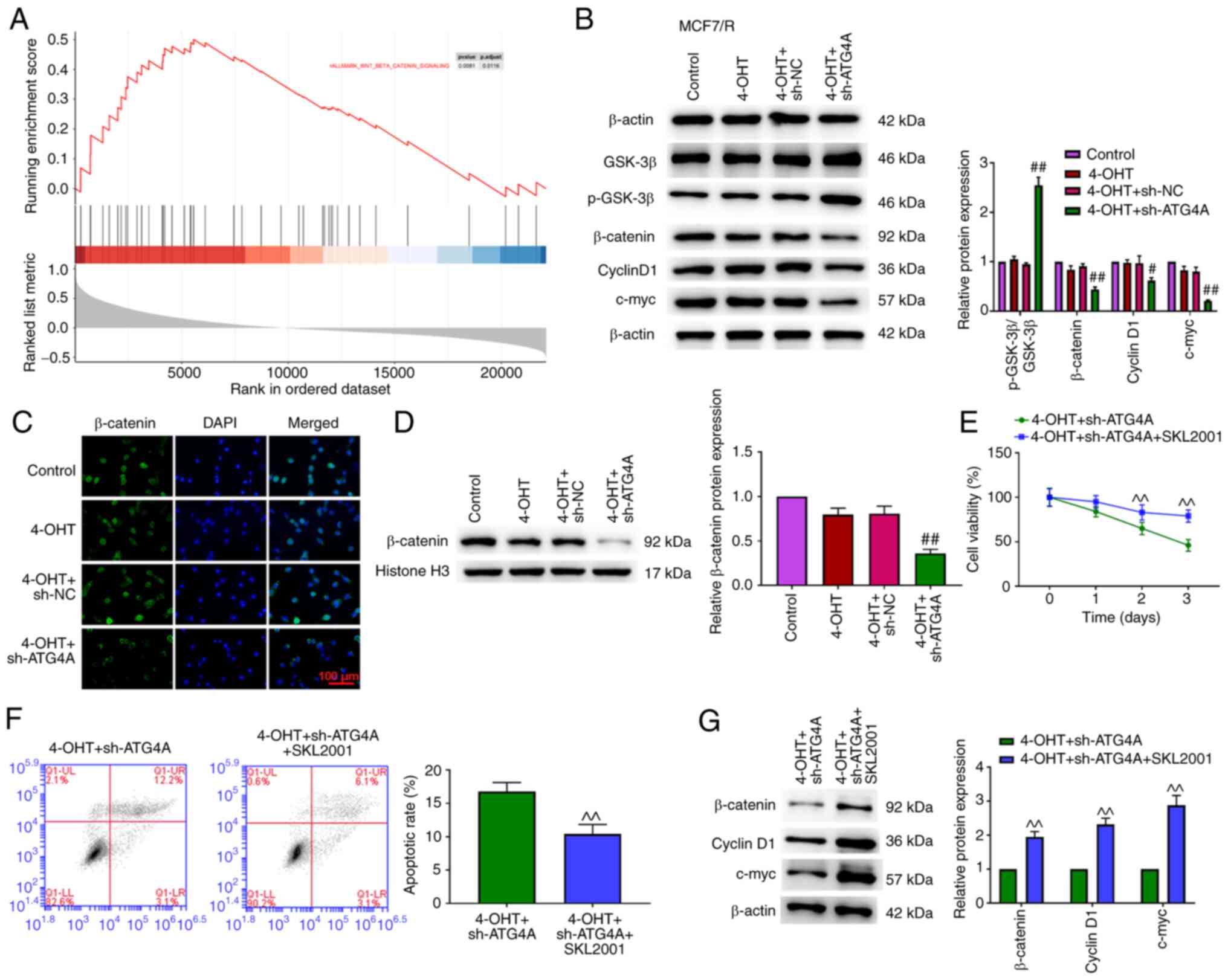 | Figure 5.ATG4A knockdown dysregulates the
Wnt/β-catenin pathway in MCF7/R cells. (A) Gene set enrichment
analysis indicated the association between ATG4A and Wnt/β-catenin
pathway. (B) The protein expression of p-GSK-3β, GSK-3β, β-catenin,
cyclin D1 and c-myc was examined using western blot analysis. (C)
ATG4A knockdown promoted the translocation of β-catenin from the
nucleus to the cytoplasm in MCF7/R cells (scale bar, 100 µm). (D)
ATG4A knockdown decreased β-catenin protein level in MCF7/R cells.
(E) Cell viability was detected using Cell Counting Kit-8 assay.
(F) Apoptosis was detected using flow cytometric analysis. (G)
Western blot analysis was performed to detect the protein
expression of β-catenin, cyclin D1 and c-myc.
^^P<0.01 vs. 4-OHT + sh-ATG4A group;
#P<0.05 and ##P<0.01 vs. 4-OHT + sh-NC
group. ATG4A, autophagy-related 4A; 4-OHT, 4-hydroxytamoxifen; sh-,
short hairpin; NC, negative control; p, phosphorylated. |
Discussion
Breast cancer is the most common malignant cancer in
women (24). Although surgery and
radiotherapy have been indicated to be curative for certain early
stage cases, chemotherapy remains a common and essential
therapeutic method in breast cancer (25,26). Long-term clinical observations
have revealed that patients with breast cancer are more responsive
to endocrine therapy agents, such as selective ER modulators
(SERMs), aromatase inhibitors and selective ER downregulators
(27,28). Tamoxifen, which is one of the
SERMs, is a commonly used drug for treating ER positive breast
cancer (4). However, the efficacy
of tamoxifen is limited due to primary or acquired resistance
(5,29–31). Thus, it is important to increase
the sensitivity of breast cancer to tamoxifen. The present study
suggested that downregulation of ATG4A inhibits the proliferation
of breast cancer cells and overcomes tamoxifen resistance.
ATG4A, which is an autophagy-regulating molecule, is
overexpressed during malignant progression of cancer (32). For instance, ATG4A expression is
increased in osteosarcoma tissues, and its overexpression enhances
the proliferation of osteosarcoma cell lines (14). Yang et al (15) identified that the expression of
ATG4A is upregulated in gastric cancer tissues and its
overexpression promotes cell migration and invasion. The present
study revealed that a higher expression of ATG4A was associated
with a poor prognosis in breast cancer. In addition, an increase in
ATG4A was observed in tamoxifen-resistant cells compared with the
control group. Autophagy is a protective housekeeping mechanism
that maintains cellular metabolism under nutrient-limiting
conditions, which is implicated in tumor development (33). LC3 is commonly used as an
indicator for detecting autophagy (34). During the formation of
autophagosomes, LC3 is cleaved by ATG4 to form LC3-I, then
cleaved-LC3 is conjugated with phosphatidylethanolamine to regulate
the conversion of LC3-I to LC3-II. (35) Several studies suggested that ATG4
family members promote tumor progression. For example, the
knockdown of ATG4B suppresses the survival of chronic myeloid
leukemia cells, and promotes imatinib mesylate chemosensitivity by
suppressing autophagy (36). The
knockdown of ATG4C impairs autophagy, inhibits glioma progression,
and sensitizes these cells to temozolomide (37). Studies have reported that 4-OHT
stimulates autophagy in breast cancer, and inhibition of autophagy
enhances the sensitivity of breast cancer cells to 4-OHT (2,38,39). In the present study, treatment
with 4-OHT significantly increased the LC3-II/LC3-I ratio and
decreased the level of p62 (a marker of autophagy), while the
knockdown of ATG4A enhanced the sensitivity of breast cancer cells
to 4-OHT by inhibiting autophagy. Consistent with the findings of
the present study, another study reported that overexpression of
microRNA-24-3p led to reduction in ATG4A expression, allowing
etoposide/cisplatin-resistant small-cell lung cancer cells to
become sensitive to etoposide and cisplatin (21).
The Wnt/β-catenin pathway is one of the most crucial
pathways in regulating proliferation, cell cycle and death
(40). When the Wnt/β-catenin
pathway is inactivated, nuclear β-catenin is transferred into the
cytoplasm (41). Inhibition of
GSK-3β phosphorylation promotes β-catenin degradation and changes
the expression of cyclin D1 and c-myc (42). The results of the present study
confirmed that ATG4A knockdown inhibited the Wnt/β-catenin pathway
in MCF7/R cells treated with 4-OHT. Consistent with the present
research, overexpression of protein crumbs homolog 3 enhances
tamoxifen sensitivity of tamoxifen-resistant cells by inhibiting
β-catenin signaling (43). MS-275
(a histone deacetylase inhibitor) combined with cisplatin has been
indicated to inhibit cell proliferation and migration and increase
cell apoptosis by regulating the Wnt/β-catenin pathway in
esophageal squamous cell carcinoma cells (44). In addition, SKL2001, an agonist of
the Wnt/β-catenin pathway, effectively rescued the ATG4A
knockdown-mediated anti-proliferative effect of tamoxifen-resistant
cells. Consistent with the current study, a previous study
demonstrated that SKL2001 rescued the WSB2-WD repeat and SOCS box
containing 2 knockdown-mediated inhibition of cell proliferation in
melanoma (45).
In summary, it was demonstrated that knockdown of
ATG4A in tamoxifen-resistant cells reduced autophagy and suppressed
the Wnt/β-catenin pathway. ATG4A may affect the response to
tamoxifen therapy of patients with breast cancer.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
QL designed the experiments. LZ performed the
research. QL analyzed the data and wrote the manuscript. QL and LZ
confirmed the authenticity of all the raw data. Both authors read
and approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Schoenlein PV, Periyasamy-Thandavan S,
Samaddar JS, Jackson WH and Barrett JT: Autophagy facilitates the
progression of ERalpha-positive breast cancer cells to antiestrogen
resistance. Autophagy. 5:400–403. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jennings CJ, Zainal N, Dahlan IM, Kay EW,
Harvey BJ and Thomas W: Tamoxifen suppresses the growth of
malignant pleural mesothelioma cells. Anticancer Res. 36:5905–5913.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shagufta and Ahmad I: Tamoxifen a
pioneering drug: An update on the therapeutic potential of
tamoxifen derivatives. Eur J Med Chem. 143:515–531. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Early Breast Cancer Trialists'
Collaborative Group (EBCTCG), ; Davies C, Godwin J, Gray R, Clarke
M, Cutter D, Darby S, McGale P, Pan HC, Taylor C, et al: Relevance
of breast cancer hormone receptors and other factors to the
efficacy of adjuvant tamoxifen: Patient-level meta-analysis of
randomised trials. Lancet. 378:771–784. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nass N and Kalinski T: Tamoxifen
resistance: From cell culture experiments towards novel biomarkers.
Pathol Res Pract. 211:189–197. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li YJ, Lei YH, Yao N, Wang CR, Hu N, Ye
WC, Zhang DM and Chen ZS: Autophagy and multidrug resistance in
cancer. Chin J Cancer. 36:522017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu J, Yue W and Chen H: The correlation
between autophagy and tamoxifen resistance in breast cancer. Int J
Clin Exp Pathol. 12:2066–2074. 2019.PubMed/NCBI
|
|
9
|
Samaddar JS, Gaddy VT, Duplantier J,
Thandavan SP, Shah M, Smith MJ, Browning D, Rawson J, Smith SB,
Barrett JT and Schoenlein PV: A role for macroautophagy in
protection against 4-hydroxytamoxifen-induced cell death and the
development of antiestrogen resistance. Mol Cancer Ther.
7:2977–2987. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Klionsky DJ: Citing recent declines in the
discovery of new ATG genes, some scientists now suggest that the
end of autophagy research may be within sight. Autophagy.
10:715–716. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang L, Li J, Ouyang L, Liu B and Cheng
Y: Unraveling the roles of Atg4 proteases from autophagy modulation
to targeted cancer therapy. Cancer Lett. 373:19–26. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mariño G, Uría JA, Puente XS, Quesada V,
Bordallo J and López-Otín C: Human autophagins, a family of
cysteine proteinases potentially implicated in cell degradation by
autophagy. J Biol Chem. 278:3671–3678. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Betin VM and Lane JD: Atg4D at the
interface between autophagy and apoptosis. Autophagy. 5:1057–1059.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Su H, Zhu G, Rong X, Zhou Y, Jiang P and
Chen P: Upregulation of ATG4A promotes osteosarcoma cell
epithelial-to-mesenchymal transition through the Notch signaling
pathway. Int J Clin Exp Pathol. 10:7975–7982. 2017.PubMed/NCBI
|
|
15
|
Yang SW, Ping YF, Jiang YX, Luo X, Zhang
X, Bian XW and Yu PW: ATG4A promotes tumor metastasis by inducing
the epithelial-mesenchymal transition and stem-like properties in
gastric cells. Oncotarget. 7:39279–39292. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wolf J, Dewi DL, Fredebohm J,
Müller-Decker K, Flechtenmacher C, Hoheisel JD and Boettcher M: A
mammosphere formation RNAi screen reveals that ATG4A promotes a
breast cancer stem-like phenotype. Breast Cancer Res. 15:R1092013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Colaprico A, Silva TC, Olsen C, Garofano
L, Cava C, Garolini D, Sabedot TS, Malta TM, Pagnotta SM,
Castiglioni I, et al: TCGAbiolinks: An R/Bioconductor package for
integrative analysis of TCGA data. Nucleic Acids Res. 44:e712016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Therneau TM: Survival Analysis [R package
survival version 2.41-3]. Technometrics. 46:111–112. 2015.
|
|
19
|
Yan C, Zhao J, Qin Y, Zhao F, Ji L and
Zhang J: Overexpression of ATG4a promotes autophagy and
proliferation, and inhibits apoptosis in lens epithelial cells via
the AMPK and Akt pathways. Mol Med Rep. 22:1295–1302. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Guo L, Zhou L, Gao Q, Zhang A, Wei J, Hong
D, Chu Y, Duan X, Zhang Y and Xu G: MicroRNA-144-3p inhibits
autophagy activation and enhances Bacillus Calmette-Guérin
infection by targeting ATG4a in RAW264.7 macrophage cells. PLoS
One. 12:e01797722017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pan B, Chen Y, Song H, Xu Y, Wang R and
Chen L: Mir-24-3p downregulation contributes to VP16-DDP resistance
in small-cell lung cancer by targeting ATG4A. Oncotarget.
6:317–331. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Uppada SB, Gowrikumar S, Ahmad R, Kumar B,
Szeglin B, Chen X, Smith JJ, Batra SK, Singh AB and Dhawan P: MASTL
induces colon cancer progression and chemoresistance by promoting
Wnt/β-catenin signaling. Mol Cancer. 17:1112018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ohashi W, Yamamine N, Imura J and Hattori
Y: SKL2001 suppresses colon cancer spheroid growth through
regulation of the E-cadherin/β-Catenin complex. Biochem Biophys Res
Commun. 493:1342–1348. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gianni L, Baselga J, Eiermann W, Porta VG,
Semiglazov V, Lluch A, Zambetti M, Sabadell D, Raab G, Cussac AL,
et al: Phase III trial evaluating the addition of paclitaxel to
doxorubicin followed by cyclophosphamide, methotrexate, and
fluorouracil, as adjuvant or primary systemic therapy: European
cooperative trial in operable breast cancer. J Clin Oncol.
27:2474–2481. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Erol K, Baltali E, Altundag K, Guler N,
Ozisik Y, Onat DA, Sayek I, Cengiz M, Atahan L and Tekuzman G:
Neoadjuvant chemotherapy with cyclophosphamide, mitoxantrone, and
5-fluorouracil in locally advanced breast cancer. Onkologie.
28:81–85. 2005.PubMed/NCBI
|
|
27
|
Harbeck N and Gnant M: Breast cancer.
Lancet. 389:1134–1150. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ignatiadis M and Sotiriou C: Luminal
breast cancer: From biology to treatment. Nat Rev Clin Oncol.
10:494–506. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mansouri S, Feizi N, Mahdi A,
Majidzadeh-AK and Farahmand L: A review on the role of VEGF in
tamoxifen resistance. Anticancer Agents Med Chem. 18:2006–2009.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Szostakowska M, Trębińska-Stryjewska A,
Grzybowska EA and Fabisiewicz A: Resistance to endocrine therapy in
breast cancer: Molecular mechanisms and future goals. Breast Cancer
Res Treat. 173:489–497. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ali S, Mondal N, Choudhry H, Rasool M,
Pushparaj PN, Khan MA, Mahfooz M, Sami GA, Jarullah J, Ali A and
Jamal MS: Current management strategies in breast cancer by
targeting key altered molecular players. Front Oncol. 6:452016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Du JX, Chen C, Luo YH, Cai JL, Cai CZ, Xu
J, Ni XJ and Zhu W: Establishment and validation of a novel
autophagy-related gene signature for patients with breast cancer.
Gene. 762:1449742020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bhat P, Kriel J, Shubha Priya B, Basappa,
Shivananju NS and Loos B: Modulating autophagy in cancer therapy:
Advancements and challenges for cancer cell death sensitization.
Biochem Pharmacol. 147:170–182. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mizushima N: Methods for monitoring
autophagy using GFP-LC3 transgenic mice. Methods Enzymol.
452:13–23. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ravikumar B, Sarkar S, Davies JE, Futter
M, Garcia-Arencibia M, Green-Thompson ZW, Jimenez-Sanchez M,
Korolchuk VI, Lichtenberg M, Luo S, et al: Regulation of mammalian
autophagy in physiology and pathophysiology. Physiol Rev.
90:1383–1435. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Rothe K, Lin H, Lin KB, Leung A, Wang HM,
Malekesmaeili M, Brinkman RR, Forrest DL, Gorski SM and Jiang X:
The core autophagy protein ATG4B is a potential biomarker and
therapeutic target in CML stem/progenitor cells. Blood.
123:3622–3634. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wen ZP, Zeng WJ, Chen YH, Li H, Wang JY,
Cheng Q, Yu J, Zhou HH, Liu ZZ, Xiao J and Chen XP: Knockdown ATG4C
inhibits gliomas progression and promotes temozolomide
chemosensitivity by suppressing autophagic flux. J Exp Clin Cancer
Res. 38:2982019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Pawlik A, Słomińska-Wojewódzka M and
Herman-Antosiewicz A: Sensitization of estrogen receptor-positive
breast cancer cell lines to 4-hydroxytamoxifen by isothiocyanates
present in cruciferous plants. Eur J Nutr. 55:1165–1180. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yu X, Luo A, Liu Y, Wang S, Li Y, Shi W,
Liu Z and Qu X: MiR-214 increases the sensitivity of breast cancer
cells to tamoxifen and fulvestrant through inhibition of autophagy.
Mol Cancer. 14:2082015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yu J, Liu D, Sun X, Yang K, Yao J, Cheng
C, Wang C and Zheng J: CDX2 inhibits the proliferation and tumor
formation of colon cancer cells by suppressing Wnt/β-catenin
signaling via transactivation of GSK-3β and Axin2 expression. Cell
Death Dis. 10:262019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xu C, Liu F, Xiang G, Wang S, Liu J, Meng
Q, Xu D, Lv S, Jiao J and Niu Y: β-Catenin nuclear localization
positively feeds back on EGF/EGFR-attenuated AJAP1 expression in
breast cancer. J Exp Clin Cancer Res. 38:2382019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liu S, Wang L, Ding W, Wang D, Wang X, Luo
Q, Lu Y and Zhu L: Cleistanthin A inhibits the invasion of
MDA-MB-231 human breast cancer cells: Involvement of the β-catenin
pathway. Pharmacol Rep. 72:188–198. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li P, Feng C, Chen H, Jiang Y, Cao F, Liu
J and Liu P: Elevated CRB3 expression suppresses breast cancer
stemness by inhibiting β-catenin signalling to restore tamoxifen
sensitivity. J Cell Mol Med. 22:3423–3433. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu T, Wang Y, Zhang Z, Zhang Z, Li Y, Cui
Y, Li Z, Liu H, Zhang Y, Wang Y and Ma S: MS-275 combined with
cisplatin exerts synergistic antitumor effects in human esophageal
squamous cell carcinoma cells. Toxicol Appl Pharmacol.
23:1149712020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang Y, Li Z, Zhao W, Hu H, Zhao L, Zhu
Y, Yang X, Gao B, Yang H, Huang Y and Song X: WD repeat and SOCS
box containing protein 2 in the proliferation, cycle progression,
and migration of melanoma cells. Biomed Pharmacother.
116:1089742019. View Article : Google Scholar : PubMed/NCBI
|