Introduction
Gene methylation is an important epigenetic
modification that involves both DNA and RNA molecules. DNA
methylation mainly affects gene expression at the transcriptional
level (1), whereas RNA
methylation primarily regulates gene expression at the
post-transcriptional level by influencing RNA stability,
maturation, localization, transportation, transcription and
translation (2,3). Overall, methylation modification
affects numerous cellular physiological behaviors, such as
neurodevelopment, immunoregulation, and cellular differentiation,
and contributes to the development of various diseases (4,5).
Circular RNA (circRNA/circ) molecules are novel
noncoding RNAs formed by reverse splicing of their corresponding
host genes and can therefore be regulated via methylation (6). Furthermore, circRNAs can regulate
gene expression and bind proteins, among other functions, and may
modulate gene expression by controlling the methylation
modification process (7).
Therefore, fully understanding the effects of methylation
modifications on the expression and function of circRNAs and an
investigation into the regulation of methylation via circRNAs, are
essential to determining the molecular mechanisms underlying
circRNA activity and disease progression. The aim of the present
review was to define the relationship between circRNAs and
methylation, as well as the functions and mechanisms of their
interactions during disease progression.
Literature search strategy
The PubMed database (pubmed.ncbi.nlm.nih.gov/) was
searched using the following terms in the title or abstract: i)
circRNA or circular RNA and ii) methylation or N6-methyladenosine
(m6A) or m6A. The inclusion criteria for the articles selected were
that they must have been published from January 1, 2010, to
September 1, 2021 and the language was English. The exclusion
criteria for the articles selected were that non-original study and
studies contained any duplicated data. A total of 375 papers were
screened and a researcher conducted a full-text search of these
publications to assess the relevance of each study. In total, 53
studies were selected for the present review.
DNA methylation
DNA methylation is among the earliest-discovered and
most intensively studied epigenetic regulatory mechanisms. It
refers to the chemical modification process, in which specific
bases in DNA sequences obtain a methyl group from S-adenosyl
methionine by covalent bonding via DNA methyltransferase (DNMT)
activity (8,9). DNA base sites that undergo
methylation include the C-5 position of cytosine, the N-6 position
of adenine and the N-7 position of guanine (8,9).
In mammals, the most important and only DNA methylation
modification is that of the carbon atom at position C-5 of cytosine
in CpG dinucleotides (8,9). DNA methylation controls gene
expression by altering DNA conformation, DNA stability and DNA
interactions with protein, as well as chromatin structure (8,10,11).
The methylation of DNA can be divided into de
novo and maintenance forms, whereby the former indicates that a
DNA duplex is not methylated before undergoing methylation, whereas
the latter describes methylation of DNA duplexes that have been
methylated previously. The methylation process is mainly mediated
by DNMTs (12,13). In mammals, DNMT3a and DNMT3b
perform de novo methylation, whereas maintenance methylation
occurs via DNMT1 activity (12,13). Other proteins, such as Alpha
Thalassemia/Mental Retardation Syndrome X-Linked, also contribute
to methylation, assisted by accessory factors, such as the
retinoblastoma protein, which function in chromosome remodeling and
DNA helicase activity (14,15).
Methylated DNA can also be demethylated, which can
be either a passive or an active process. In the passive process,
no methylation occurs during semi-retained replication and DNA
gradually becomes demethylated via ‘dilution’. The active process
involves demethylation via demethylases, such as DNA glycosidase
and methylated CpG-binding proteins, such as methyl-CpG-binding
domain protein 2 (16–18).
DNA methylation can also interact with the
epigenetic regulation of histones. For example, DNA methylation
leads to the removal of histone acetylation marks near the
methylated site via the recruitment of histone deacetylases
(19), and lysine 9 methylation
on histone H3 can promote DNA methylation processes (20).
DNA methylation has important functions in mammalian
development and disease processes. In particular, embryonic
development involves the continuous establishment of methylation
and demethylation (21,22). Methylation of CpG dinucleotide
sequences in gene promoter regions can lead to silencing of gene
expression (1), which can also
serve an important role in disease pathogenesis. For example, via
the inactivation of the tumor suppressor gene, cyclin-dependent
kinase inhibitor 2A, following methylation (23–25), which is an important cause of
tumorigenesis.
RNA methylation
RNA methylation refers to the methyltransferase
catalysis chemical modification of methyl groups via selective
addition of methyladenine to RNA, which primarily regulates gene
expression at the post-transcriptional level (2). To date, more than 170 cellular RNA
modifications have been identified in various coding and noncoding
RNAs (26). m6A is the most
abundant and important RNA modification in eukaryotic mRNAs, which
is mainly located near stop codons and in the 3′noncoding region
and is an important epigenetic modification of both mRNA and
noncoding RNA (27). Methylation
can affect the stability, localization, transportation,
transcription, maturation and translation of RNA molecules at the
post-transcriptional level, which thereby regulates gene expression
(2,3).
M6A methylation of RNA occurs mainly on adenines in
the DRACH sequence (m6A consensus motifs) and is regulated by
methyltransferase-encoders, demethylase-decoders and m6A binding
protein-readers (28).
Methyltransferases, including methyltransferase-like (METTL)3,
METTL14, WT1 associated protein and vir-like m6A
methyltransferase-associated (KIAA1429), mainly promote methylation
to regulate gene expression and function (28). Demethylases, primarily AlkB
homolog 5 RNA demethylase (ALKBH5) and fat mass and
obesity-associated protein (FTO), regulate gene function via
demethylation (28). m6A binding
proteins regulate the translation, degradation and splicing, as
well as other gene functions modified by m6A, via recognizing and
binding to m6A methylation sites (28).
RNA methylation has important biological functions
in mammals, with roles in germ cell development, immunity and cell
differentiation (29). RNA
methylation is also closely related to numerous malignant diseases,
including liver cancer, lung cancer, breast cancer and glioma
(30).
Effects of methylation on circRNA biological
function
Methylation is closely related to
circRNAs
Ferreira et al (31) reported that enhancing CpG island
methylation in the promoter regions of genes in human tumor cells
leads to the significant downregulation of corresponding linear and
circRNA transcript expression, including of tumor suppressor
candidate 3, protein O-mannosyltransferase 1, attractin-like 1 and
sterile α-motif domain containing 4A. These data indicated that
methylation in gene promoter regions also affects circRNA
expression. The most abundant epigenetic modification in eukaryotes
of RNAs is m6A and numerous studies have confirmed that m6A is
common in circRNAs. For example, 360 m6A-circRNAs have been
detected in lung tissue in a hypoxia-induced pulmonary hypertension
rat model and control tissues (32). A total of 9,382 circRNAs have been
identified in lens epithelium cells from age-related cataract and
control cells, 4,646 of which had m6A peaks with differing
abundances (33). Furthermore, in
a study systematically describing global circRNA m6A modification
patterns in glioblastoma (GBM), researchers demonstrated that
compared with the normal control group, there were 1,370 new m6A
peaks and 1,322 missing m6A peaks in circRNAs from GBM. This study
also demonstrated that m6A levels were positively correlated with
circRNA expression (34).
Moreover, in an analysis of 10 upregulated and 10 significantly
downregulated differentially expressed circRNAs, selected by
analysis of the circRNA expression profiles from
poorly-differentiated adenocarcinoma of the stomach, 6 upregulated
and 8 downregulated circRNAs with high scores for prediction of m6A
sites were identified, respectively (35). Analysis of m6A modification
patterns in human ameloblastoma, relative to normal oral tissues,
also detected 364 differentially methylated m6A sites within
circRNAs, of which 22.5% were hypermethylated (36). In a study of circRNA expression
profiles and m6A modification in poorly-differentiated gastric
adenocarcinoma, a total of 65 differentially expressed circRNAs
were detected, most of which had m6A modification, whereby the
trend in m6A modification changes was generally consistent with
that of circRNA expression levels (35). In another study, the number of
circRNA reads recovered in m6A immunoprecipitation experiments was
compared with the total input of circRNA sequence reads, leading to
an estimation that ~13% of total circRNAs possess an m6A
modification (37). m6A tends to
occur in larger exonic regions of circRNAs and is concentrated
upstream and in the middle of exonic regions, and m6A exhibits
cell-specificity, for example, more than half of m6A-circRNAs
detected in HeLa cells were not detected in human embryonic stem
cells (38,39).
Effects of methylation on the
biogenesis, transport, stability, translation and degradation of
circRNA
Fig. 1 presents
the use of three small interfering (si) RNAs to downregulate METTL3
expression led to the strong and specific downregulation of
circ-zinc finger protein 609 (ZNF609) levels, accompanied by an
increase in its unspliced precursor RNA, which indicated that
decreased methylation may influence the conversion of precursor
mRNA to circRNA (40). Similarly,
when two siRNAs were used to downregulate YTH domain containing 1
(YTHDC1) expression levels, circ-ZNF609 levels decreased and those
of its precursor transcript increased. Moreover, corresponding
linear mRNA levels remained unchanged, with this phenomenon
occurring regardless of cell type, which indicated that YTHDC1
influences circ-ZNF609 splicing (40). Further results from this study
demonstrated that YTHDC1 expression does not affect circ-ZNF609
subcellular localization, excluding the possibility of circRNA
degradation due to nuclear retention (40). In general, METTL3 and YTHDC1
influence circRNA formation by regulating m6A modification, a
process that is driven by m6A modification at specific sites
(39).
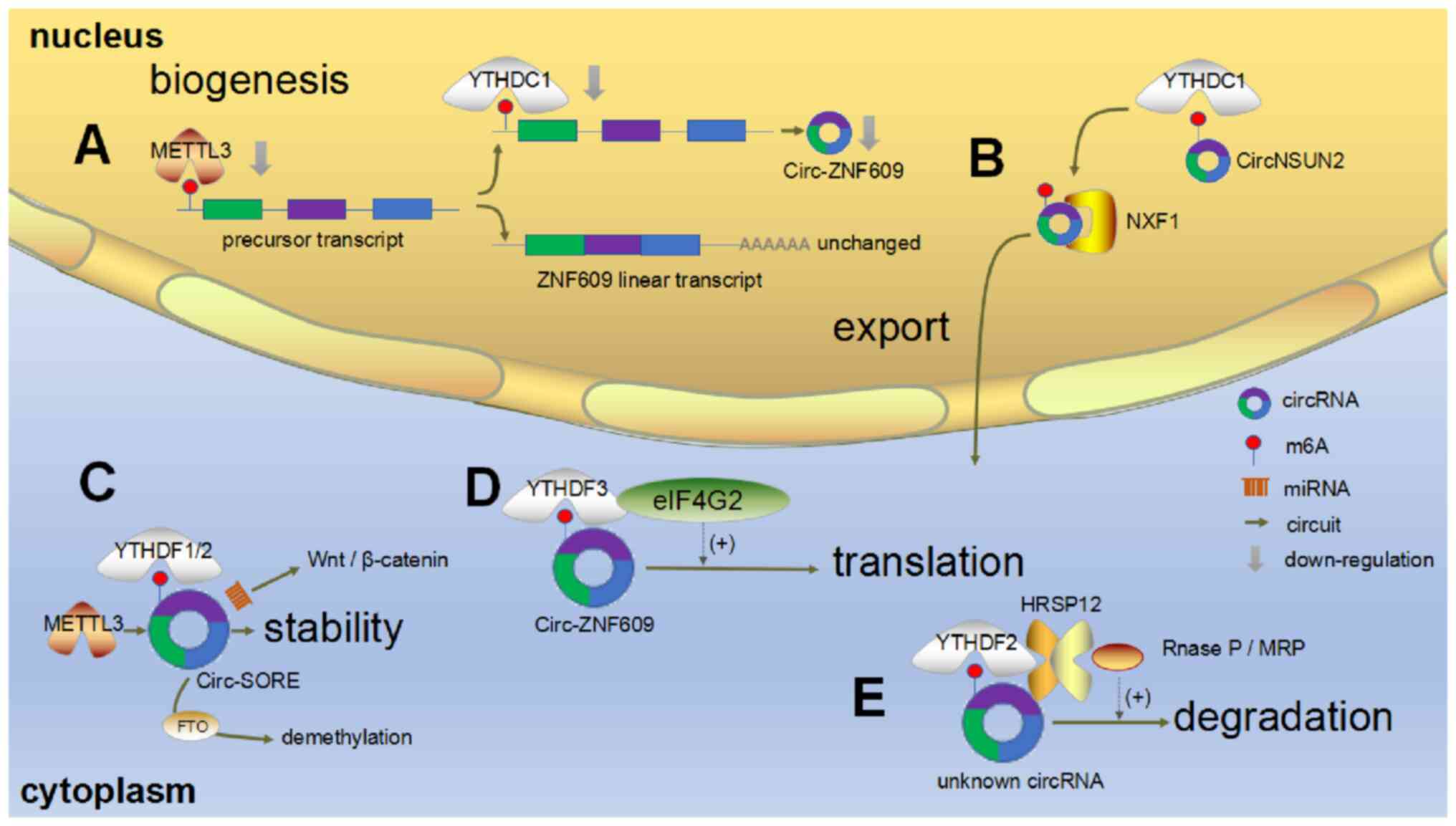 | Figure 1.Effects of methylation on circRNA
biogenesis, transport, stability, translation and degradation. (A)
METTL3 or YTHDC1 affect circRNA generation by regulating m6A
modification. (B) m6A modification regulates circNSUN2 export to
the cytoplasm. (C) m6A modification improves circRNA-SORE
stability. (D) YTHDF3 and eIF4G2 physically interact with
endogenous circ-ZNF609 and can affect circ-ZNF609 translation
levels. (E) m6A modification is involved in circRNA degradation via
the YTHDF2/HRSP12/RNase P/MRP complex signaling pathway. Gray
arrows represent downregulation. circRNA, circular RNA; METTL3,
methyltransferase-like 3; YTHDC, YTH domain containing; m6A,
N6-methyladenosine; NSUN2, NOP2/Sun RNA methyltransferase 2; YTHDF,
YTH domain-containing family protein; eIF4G2, eukaryotic
translation initiation factor 4-γ2; ZNF609, zinc finger protein
609; HRSP12, heat-responsive protein 12; MRP, mitochondrial RNA
processing enzyme; miRNA, microRNA; FTO, fat mass and
obesity-associated protein. |
A study on the effects of circNOP2/Sun RNA
methyltransferase 2 (NSUN2) in colorectal cancer (CRC) liver
metastasis, reported that it underwent m6A modification, whereas
RNA pull-down and mass spectrometry analysis indicated that there
was an interaction between circNSUN2, YTHDC1, serine and arginine
rich splicing factor 3 (SRSF3) and nuclear RNA export factor 1
(NXF1) (41). YTHDC1, an m6A
reader, interacts with the splicing factor, SRSF3, to regulate mRNA
splicing and promote binding of mRNA to SRSF3 and the canonical
export receptor, NXF1, to mediate m6A-modified mRNA export and
metabolism (38). This therefore
indicates that m6A modification may influence circNSUN2 subcellular
localization. Silencing of YTHDC1 expression significantly
increases circNSUN2 levels in the nucleus, whereas overexpression
of wild-type YTHDC1 can restore circNSUN2 export from the nucleus
to the cytoplasm (41).
RNA-fluorescence in situ hybridization analysis has
previously demonstrated that silencing of the m6A
methyltransferase, METTL3, leads to a significant increase in
circNSUN2 levels in the nucleus and restores defective circNSUN2
cytoplasmic export following the overexpression of wild-type METTL3
(41). Fig. 1 presents that m6A modification of
circNSUN2 regulates its cytoplasmic export.
Recognition of m6A-circRNAs via YTH
domain-containing family protein (YTHDF)2 regulates the stability
of the corresponding parental genes (39,42). Whether YTHDF2 can regulate circRNA
stability via the recognition of m6A modified circRNAs warrants
further exploration. circRNA-SORE (a circRNA upregulated in
sorafenib-resistant HCC cells) activates the Wnt/β-catenin
signaling pathway and induces sorafenib resistance via a microRNA
(miRNA/miR) sponge mechanism, while increased circRNA-SORE levels
benefit from improved stability. RNA pull-down assays demonstrated
that circRNA-SORE interacts with METTL3, FTO and YTHDF1/2; an
encoder, decoder and reader of m6A, respectively (43), which indicated that circRNA-SORE
is associated with m6A modifications. A set of sequence-specific
morpholino antisense oligonucleotides designed to target the m6A
site in circRNA-SORE have been used to reduce m6A-modified
circRNA-SORE in HepG2-SR cells, which led to a reduction in
circRNA-SORE stability (43).
Furthermore, it has been demonstrated that wild-type circRNA-SORE
is more stable than m6A-mutated circRNA-SORE during actinomycin D
treatment (43), indicating that
circRNA-SORE undergoes m6A modification and that this improves
mRNA-SORE stability, which is presented in Fig. 1. These findings led to the
proposal of a novel mechanism of sorafenib resistance in patients
with hepatocellular carcinoma (HCC) and suggested that both
circRNA-SORE and its m6A modification may be potential drug
intervention targets in patients with advanced HCC.
The m6A reader, YTHDF3, interacts with ribosomal
proteins to promote mRNA translation as m6A modification enables
cap-independent translation (44–46). Furthermore, a recent study has
reported that circRNAs can be translated into proteins (47) and therefore m6A modification may
promote circRNA translation. Jakobi et al (48) compared the expression profiles of
circRNAs from human cardiovascular cell models and cardiac tissue
with those of m6A-methylated circRNAs described in previous studies
(49). This aforementioned study
concluded that certain m6A-modified circRNAs function in protein
translation. Furthermore, Yang et al (37) reported that the negative control
may also initiate translation when exploring internal ribosome
entry site-mediated circRNA translation. Further experiments
confirmed that the short sequence containing the m6A site has an
important role in this process, as well as the m6A encoders, METTL3
and METTL14, and the readers, YTHDF1, YTHDF2 and YTHDF3. Moreover,
circRNA translation was demonstrated to be reduced by the m6A
demethylase decoder, FTO, whereas YTHDF3 was determined to strongly
interact with the translation initiation factor, eukaryotic
translation initiation factor 4-γ2 (eIF4G2), thereby promoting
translation (37). These
aforementioned studies demonstrated that the transcription
initiation factors, eIF4G2, YTHDF1, YTHDF2 and YTHDF3, may be
involved in m6A-induced translation of circRNAs and that this
process is regulated by METTL3 and METTL14. These results were
confirmed by a further study on the effect of m6A modification on
circ-ZNF609 translation. Fig. 1
presents although YTHDF1/2 consumption was demonstrated to have no
effect on circ-ZNF609 transformation, both YTHDF3 and eIF4G2 were
determined to physically interact with endogenous circ-ZNF609, with
increases or decreases in these factors being reported to affect
the circ-ZNF609 translation level (40).
There are two main mechanisms of mRNA degradation of
m6A modification: i) The YTHDF2/C-C motif chemokine receptor 4
signaling pathway (50); and ii)
the YTHDF2/heat-responsive protein 12 (HRSP12; adaptor
protein)/RNase P/mitochondrial RNA processing enzyme (MRP) complex
signaling pathway (51). YTHDF2
can recognize m6A and HRSP12 is an adaptor that can connect YTHDF2
and the RNase P/MRP complex. Fig.
1 presents circRNAs containing m6A are recognized by YTHDF2 in
an HRSP12-dependent manner, this is followed by selective
downregulation by RNase P/MRP, which indicates that m6A
modification is involved in circRNA degradation via the
YTHDF2/HRSP12/RNase P/MRP complex signaling pathway (52).
Study of methylation-modified circRNAs in
disease
Studies of methylation-modified circRNA molecules in
disease are summarized in Table
I.
 | Table I.M6A-modified circRNAs in disease. |
Table I.
M6A-modified circRNAs in disease.
| First author,
year | circRNA | Disease | Mechanism | Function | (Refs.) |
|---|
| Chen C, 2021 | circ1662 | Colorectal
cancer |
METTL3/circ1662/YAP1/SMAD3 | Cell invasion and
migration | (53) |
| Chen RX, 2019 | circNSUN2 | Colorectal
cancer | Not clear | Invasion | (41) |
| Liu H, 2021 | circDLC1 | Hepatocellular
carcinoma |
KIAA1429/circDLC1 | Cell proliferation
and migration | (54) |
| Rao X, 2021 | circ-ARL3 | Hepatocellular
carcinoma |
HBx/METTL3/circ-ARL3/miR-1305 | Cell proliferation
and invasion | (55) |
| Zhao J, 2019 | circE7 | Cervical
cancer | METTL3/14/circE7/E7
protein | Cell
transformation | (56) |
| Chen Z, 2021 | circ0000069 | Cervical
cancer |
METTL3/circ0000069/miR-4426 | Cell proliferation
and migration | (57) |
| Li B, 2021 | circNDUFB2 | Non-small cell lung
cancer |
TRIM25/circNDUFB2/IGF2BPs ternary
complex | Cell growth and
metastasis | (58) |
| Nan A, 2019 | circNOL10 | Lung carcinoma |
circNOL10/SCML1/HN/p53 | Cell growth | (59) |
| Wu P, 2021 | circCUX1 | Hypopharyngeal
squamous cell carcinoma |
METTL3/circCUX1 | Radiotherapy
resistance | (61) |
| Li Z, 2021 | circMETTL3 | Breast cancer |
METL14/circMETTL3/miR-31-5p-CDK1 | Cell proliferation,
migration and invasion | (62) |
| Su H, 2020 | circXpo6 and
circTmtc3 | Pulmonary
hypertension | Not clear | Not clear | (32) |
| Guo M, 2020
macrophage pyroptosis |
hsa_circ_0029589 |
Atherosclerosis |
IRF-1/METTL3/hsa_circ_0029589 |
| (66) |
| Li X, 2021 | circGFRα1 | Infertility |
METTL14/circGFRα1 | Female germline
stem cell renewal and maintenance | (67) |
Cancer
CRC
In CRC cells, circ1662 expression increases in
response to overexpression of METTL3 (53), which mediates m6A modification of
the reverse complementary sequence of circ1662 to induce circ1662
expression. Furthermore, circ1662 can induce yes-associated protein
1 to translocate from the cytoplasm to the nucleus, where it
activates SMAD3 to promote CRC cell invasion and migration
(53).
circNSUN2 levels are elevated in tissues and sera
from patients with liver metastases from CRC, indicating that it
may serve a role in cancer cell metastasis. circNSUN2 levels are
elevated in both the nucleus and cytoplasm of CRC cells, but
particularly in the cytoplasm. Increased m6A methylation levels can
promote CRC cell invasion. Moreover, a previous study demonstrated
that the mutation of an m6A modification site in a plasmid for
overexpression of circNUSN2, leads to the downregulation of m6A
modification levels and a consequent reduction in CRC cell invasive
activity. Overall, these results indicated that m6A modification of
circNSUN2 is important for CRC cell invasive ability (41).
HCC
circ-deleted in liver cancer 1 (DLC1) is expressed
at low levels in HCC tissues and cells and inhibits HCC
proliferation and migration when it is overexpressed, which is a
good prognostic indicator in patients with HCC (54). The circDLC1 molecule was
identified by methylated RNA immunoprecipitation sequencing
experiments, indicating that it undergoes m6A modification.
Furthermore, circDLC1 is differentially expressed in response to
the knockdown of the m6A methyltransferase complex, KIAA1429, which
indicates that KIAA1429 regulates HCC progression via the
regulation of m6A modification of circDLC1 in HCC cells (54).
circ-ADP ribosylation factor-like GTPase 3 [ARL3;
also referred to as human serum albumin (hsa)_circ_0092493] is
highly expressed in hepatitis B virus (HBV)-positive HCC cells and
promotes their proliferation and invasion (55). The main underlying mechanism
involves HBV X protein upregulation of METTL3, which increases
circ-ARL3 m6A modification (55).
YTHDC1A binds to m6A modified circ-ARL3 and supports its reverse
splicing and biogenesis (55).
Furthermore, the high expression of circ-ARL3 antagonizes the
inhibitory effects of miR-1305 on oncogenes (55).
Cervical cancer (CC)
Human papillomavirus type 16-derived circE7 can be
translated into protein and promote the transformation of CC cells
(56). The translation of this
protein is independent of corresponding linear molecules and circE7
has a specific level of m6A modification (56). Silencing expression of the RNA
methyltransferases, METTL3/14, can significantly inhibit circE7
expression. Mutation of a potential m6A site in the circE7
untranslated region can also significantly decrease its expression
and E7 protein expression levels. However, the expression of
corresponding linear molecules does not significantly change, which
indicates that m6A modification promotes the expression of circE7
and its protein products, which therefore promotes CC disease
progression (56).
circ0000069 promotes CC cell proliferation and
migration by inhibiting miR-4426 (57). METTL3 knockdown significantly
inhibits circ0000069 m6A levels and expression and m6A modification
can improve circ0000069 stability (57).
Lung cancer
In a previous study of non-small cell lung cancer,
circ-NADH:ubiquinone oxidoreductase subunit B2 (circ-NDUFB2) was
reported to act as a scaffold during formation of the tripartite
motif-containing (TRIM) 25/circNDUFB2/insulin-like growth factor-2
mRNA-binding proteins (IGF2BPs) ternary complex, which promotes
ubiquitination and degradation of IGF2BPs and inhibits non-small
cell lung cancer progression (58). Levels of circNDUFB2 m6A
modification affect the strength of circNDUFB2 binding to IGF2BPs,
which influences the ubiquitin-ligase activity of TRIM25 for
IGF2BPs (58). Furthermore, the
combination of circ-nucleolar protein 10 (circNOL10) and the
transcription factor Scm polycomb group protein-like 1 (SCML1) in
lung cancer cells promotes SCML1 transcriptional regulation of
humanin (HN), regulates the HN polypeptide family, alters various
tumor signaling pathways, including p53, and inhibits lung cancer
cell growth (59). Moreover, m6A
methylation of pre-NOL10 in H460 and A549 cells inhibits circNOL10.
Epithelial splicing regulatory protein 1 and methylated pre-NOL10
regulate circNOL10 expression together in lung cancer cells
(59).
Hypopharyngeal squamous cell carcinoma
(HPSCC)
HPSCC is among the most common malignancies of the
head and neck and is one of the malignant tumors that exhibits the
worst prognosis (60). circRNAs
are potential biomarkers and/or therapeutic targets in numerous
types of tumor. However, the expression and function of circRNA
regulation via m6A in HPSCC remains unclear. Wu et al
(61) demonstrated that
circ-CUT-like homeobox 1 (circCUX1) is upregulated in patients with
HPSCC resistant to radiotherapy and that it is a predictor of poor
survival. Furthermore, this study reported that METTL3 mediates
circCUX1 m6A methylation and stabilizes its expression, whereas
circCUX1 knockdown promotes hypopharyngeal cancer cell sensitivity
to radiotherapy (61).
Breast cancer
circMETTL3 is highly expressed in breast cancer and
methylated RNA immunoprecipitation (MeRIP) analysis has
demonstrated that it is highly enriched in the m6A precipitation
component (62). This previous
study demonstrated that the downregulation of METTL14 can reduce
circMETTL3 expression levels, whereas FTO reduction has the
opposite effect, which indicates that circMETTL3 expression may be
regulated by m6A modification levels. Furthermore, circMETTL3 was
determined to promote the proliferation, migration and invasion of
breast cancer cells via the circMETTL3/miR-31-5p/cyclin dependent
kinase 1 (CDK1) axis (62).
Immunity and non-neoplastic disease
Immunity
Immune responses can be controlled by m6A
modification (63). Chen et
al (64) reported that
m6A-modified endogenous circRNAs have important functions in
dampening innate immune responses by inhibiting retinoic
acid-inducible gene I (RIG-I) activation. m6A-modified exogenous
circRNAs contribute to immune function by activating RIG-I-mediated
innate immune responses. Furthermore, exogenous circRNAs can induce
antigen-specific T and B cell activation, antibody production and
antitumor immune capacity in vivo (64). YTHDF2 may also be essential for
innate immunity suppression via recognition of m6A-modification and
promotion of m6A-modified circRNA degradation (64).
Cardiovascular diseases
A previous study reported that m6A modification
levels of 166 and 191 circRNAs were significantly up- and
downregulated, respectively, in lung tissues in a rat model of
hypoxia-induced pulmonary hypertension (HPH) compared with the
controls. Moreover, m6A levels of circRNAs, which are mainly
derived from coding regions, were also lower overall in HPH rats
than those in the control groups (32). Gene ontology and Kyoto
Encyclopedia of Genes and Genomes analyses of 76 circRNAs
demonstrated their upregulation with increased m6A levels and 107
circRNAs were downregulated with reduced m6A levels. The results
also demonstrated that circ-exportin 6 and circ-transmembrane
O-mannosyltransferase-targeting cadherins 3 were selected as
significant factors because of their enriched biding sites with
target miRNAs of interest (32)
and their m6A modification and downregulation were confirmed as
markers of HPH in pulmonary artery smooth muscle cells and
pulmonary artery endothelial cells. This aforementioned study
therefore provided evidence for the potential use of m6A-circRNA as
a diagnostic marker and therapeutic target in HPH (32).
Macrophage pyroptosis is among the causes of
atherosclerosis and interferon regulatory factor-1 (IRF-1) can
effectively promote this process (65). In one study, compared with
patients with clinical presentation of chest pain and those with
stable angina, hsa_circ_0029589 levels were significantly lower in
patients with unstable angina and acute myocardial infarction.
IRF-1 was reported to induce disease progression by downregulating
hsa_circ_0029589 expression and promoting macrophage pyroptosis via
METTL3 upregulation to promote hsa_circ_0029589 m6A modification
(66).
Infertility
Dysregulation of m6A-modified circRNAs may affect
female fertility. For example, circ-GDNF family receptor α1
(circGFRα1) is stably and abundantly expressed in female germline
stem cells (FGSC) and its overexpression promotes FGSC self-renewal
and maintenance (67).
Furthermore, m6A modification of circGFRα1 promotes its export from
the nucleus to the cytoplasm. Levels of circGFRα1 m6A modification
in FGSC are also high and are regulated by METTL14 (67).
Lack of circRNAs may result in male infertility as
sperm carry a wealth of evolutionarily conserved circRNAs.
Considering the continuous degradation of linear RNA, the
continuous enrichment of circRNAs during sperm development may
provide genetic information that is translated into functional
proteins (68). circRNA
accumulation is associated with enhanced splicing at the m6A site
and m6A modification may interfere with sperm motility by
influencing circRNA expression levels (68). Lack of METTL3 is among a
combination of factors in spermatogenic cells that leads to a
decline in m6A levels, causing spermatogenic arrest at the meiotic
phase and male infertility (68).
circRNAs influence disease progression via
methylation regulation
The mechanisms by which circRNAs influence disease
progression via methylation regulation are summarized in Table II.
 | Table II.Mechanisms of circRNA influencing
disease progression via methylation regulation. |
Table II.
Mechanisms of circRNA influencing
disease progression via methylation regulation.
| First author,
year | circRNA | Disease | Methylation
modification | Mechanism | Function | (Refs.) |
|---|
| Zhao Z, 2020 | circSOD2 | Hepatocellular
carcinoma | DNA methylation of
SOCS3 |
circSOD2/DNMT3a/SOCS3/JAK2/STAT3 | Cell proliferation
and tumorigenesis | (70) |
| Liu Z, 2019 | circRNA-5692 | Hepatocellular
carcinoma | DNA methylation of
DAB2IP |
circRNA-5692/miR-328/DAB2IP | Cell proliferation,
wound healing and invasion | (71) |
| Zhang PF, 2019 | circTRIM33-12 | Hepatocellular
carcinoma | Demethylation of
WWC3, TP53INP1,ULBP1 and JHDM1D mRNA |
circTRIM33-12/miR-191/TET1/WWC3,TP53INP1,
ULBP1 and JHDM1D | Cell proliferation,
migration, invasion and immune escape | (75) |
| Dong ZR, 2021 | circMEMO1 | Hepatocellular
carcinoma | DNA methylation of
TCF21 |
circMEMO1/miR-106b/TCF21 | Cell proliferation,
migration and invasion and the sensitivity to sorafenib
treatment | (76) |
| Sun S, 2020 | circ-LRIG3 | Hepatocellular
carcinoma | Methylation of
STAT3 mRNA |
circ-LRIG3/EZH2/STAT3 | Cell proliferation,
migration, invasion and apoptosis | (77) |
| Chi F, 2021 | circmap2k4 | Hepatocellular
carcinoma | Not clear |
circmap2k4/miR-139-5p/YTHDF1 | Cell
proliferation | (78) |
| Chen N, 2018 | FECR1 | Breast cancer | DNA methylation of
FLI1 | FECR1/FLI1/TET1 or
FECR1/DNMT1/FLI1 | Cell
metastasis | (80) |
| Xie F, 2021 | circPTPRA | Bladder cancer | m6A of FSCN1/MYC
mRNA |
circPTPRA/IGF2BP1/FSCN1/MYC | Cell proliferation,
migration and invasion | (81) |
| Huang ZM, 2021 | circRNA-100284 | Bladder cancer | Demethylation of
HSP70 |
circRNA-100284/miR-217/HSP70/Aurora-B-cell | Cell cycle
transition and cell proliferation | (82) |
| Mo WL, 2021 |
hsa_circ_0072309 | Non-small cell lung
carcinoma | Methylation of
downstream genes of FTO |
hsa_circ_0072309/miR-607/FTO | Cell tumorigenesis
and invasion | (83) |
| Zhang Z, 2021 | circRAB11FIP1 | Ovarian cancer | m6A of ATG5 and
ATG7 mRNA |
circRAB11FIP1/FTO/ATG5/ATG7 | Autophagy flux | (84) |
| Wan H, 2021 | circRIMS | Esophageal squamous
cell carcinoma | DNA methylation of
miR-613 |
circRIMS/miR-613 | Cell
proliferation | (86) |
| Yang P, 2021 | circ-ATAD1 | Endometrial
cancer | DNA methylation of
miR-10a |
circ-ATAD1/miR-10a | Cell invasion and
migration | (87) |
| Wu W, 2021 | circFAT1 | Endometrial
cancer | DNA methylation of
miR-21 |
circFAT1/miR-21 | Cell stemness | (88) |
| Du WW, 2020 | circSKA3 | Glioblastoma | DNA methylation of
miR-1 | circSKA3/miR-1 | Cell
proliferation | (89) |
| Yang ZG, 2017 | circ-Amotl1 | Wound repair | DNA methylation of
miR-17 |
circ-Amotl1/DNMT3a/miR-17/STAT | Cell proliferation,
survival, Migration and wound repair | (90) |
| Wang X, 2018 | circIBTK | Systemic lupus
erythematosus | DNA demethylation
of AKT |
circIBTK/miR-29b/AKT | Not clear | (93) |
| Zhao M, 2010 |
hsa_circ_0012919 | Systemic lupus
erythematosus | DNA methylation of
CD11a and CD70 |
hsa_circ_0012919/DNMT1/CD11a/CD70 | Autoantibody
production | (94) |
| Zhou LY, 2019 | circRNA ACR | Myocardial ischemia
reperfusion injury | DNA methylation of
Pink1 | circRNA
ACR/DNMT3b/Pink | Cell autophagy | (97) |
| Huang R, 2020 | circSTAG1 | Depression | m6A of FAAH
mRNA |
circSTAG1/ALKBH5/FAAH | Depression-like
behavior | (98) |
| Zhang H, 2021 | circFADS2 | Osteoarthritis | DNA methylation of
miR-195 |
circFADS2/miR-195-5p | Cell apoptosis | (99) |
Cancer
HCC
A previous study identified a total of 1,012
upregulated and 747 downregulated circRNAs in tumor samples
compared with normal adjacent samples, based on genome-wide DNA
methylation and RNA sequencing data from 20 patients with HCC. No
significant change in the expression levels of the corresponding
parental genes of 46 upregulated and 31 downregulated circRNAs in
HCC tumors was detected. It was also determined that 34 (44.2%) of
these 77 differentially expressed circRNAs were significantly
related to changes in DNA methylation in HCC, which indicated that
abnormal DNA methylation may regulate circRNA expression in HCC
(69). In the same study,
MeRIP-sequencing (seq) was used to identify 5-methylcytosine (m5C)
sites in circRNAs in HCC and paired adjacent non-tumor tissues, and
the relationship between m5C and HCC was analyzed (69). The results indicated that there
may be a correlation between HCC and m5C in circRNAs. In liver
cancer tissues, 51.7% of circRNAs with methylation sites had only
one methylation peak, whereas the proportion was higher in adjacent
tissues (69). Furthermore,
hypermethylated genes were reported to exhibit lower
transcriptional expression in both liver cancer and paired adjacent
non-tumor tissues (69) which was
confirmed in another study (70).
circ-superoxide dismutase 2 is expressed at higher levels in HCC
tumors than in normal liver tissues, inhibits miR-502-5p expression
and rescues levels of the miR-502-5p target gene, DNMT3a, by acting
as a sponge. Upregulation of DNMT3a inhibits the suppressor of
cytokine signaling 3 (SOCS3) by increasing DNA methylation of the
SOCS3 promoter (70), which
accelerates Janus kinase 2/STAT3 signaling pathway activation,
downstream of SOCS3, thereby promoting disease progression
(70). Similarly, arrest of HCC
disease progression caused by the overexpression of circRNA-5692
may be due to the interaction of circRNA-5692 with a
methyltransferase, which reduces the methylation level of the
promoter region of the tumor suppressor DAB2 interacting protein,
which enhances its expression in HCC. This process is primarily
mediated by circRNA-5692 acting as sponge for miR-328-5p (71).
In addition to regulating methyltransferases,
circRNAs can also control gene promoter methylation by influencing
the expression of other proteins associated with methylation, which
can affect tumor progression. Tet methylcytosine dioxygenase 1
(TET1) mediates DNA demethylation by converting 5-methylcytosine to
5-hydroxymethylcytosine (5hmC), 5-formylcytosine and
5-carboxycytosine (72). TET1
exerts antitumor functions in cancer cells via a variety of
mechanisms, including increasing the expression of demethylated Wnt
antagonists to inhibit the Wnt/β-catenin signaling pathway, or
demethylating CpG sites in the promoter regions of tumor suppressor
genes to promote tumor suppressor gene expression (73,74). In HCC, TET1 knockdown promotes
tumor cell proliferation, migration and invasion, and circTRIM33-12
can regulate TET1 levels by acting as an miR-191 sponge.
Furthermore, overexpression of circTRIM33-12 and TET1 can promote
the mRNA and protein expression levels of WWC family member 3,
tumor protein p53 inducible nuclear protein 1, UL16 binding protein
1 and lysine demethylase 7A via demethylation to suppress cancer.
This process may also be associated with immune escape (75).
Cirrhosis is a recognized risk factor for HCC
development. circ-mediator of cell motility 1 (circMEMO1) levels
are significantly downregulated in HCC samples compared with
cirrhotic nodules and are closely associated with overall survival
and disease-free survival of patients with HCC (76). Analysis of the underlying
mechanism has demonstrated that circMEMO1 can target the TET gene
family by acting as a sponge of miR-106b-5p and increasing 5hmC
levels, thereby regulating transcription factor 21 promoter
methylation and gene expression to influence HCC progression
(76). circMEMO1 can also
increase HCC cell sensitivity to rafenib treatment (76).
circ-leucine rich repeats and immunoglobulin-like
domains 3 (LRIG3) is significantly upregulated in liver cancer and
promotes liver cell proliferation, migration and invasion and
reduces apoptosis (77).
circ-LRIG3 forms a ternary complex with enhancer of zeste 2
polycomb repressive complex 2 subunit (EZH2) and STAT3 to promote
EZH2-induced STAT3 methylation and subsequent phosphorylation,
activating the STAT3 signaling pathway (77). Subsequently, activated STAT3 can
directly bind to the circ-LRIG3 promoter, which increases
circ-LRIG3 transcription and therefore forms a positive feedback
loop (77).
Chi et al (78) screened for microRNAs and circRNAs
differentially expressed between paired HCC tumor and normal
tissues. The study using The Cancer Genome Atlas and Gene
Expression Ontology databases and conducted bioinformatics analysis
to construct a regulatory network of circRNA/miRNA/prognostic m6A
RNA methylation modulators, which was used to screen for target
circRNAs. Experimental verification demonstrated that
circ-mitogen-activated protein kinase 4 promotes HCC proliferation
by modulating the hsa-miR-139-5p/YTHDF1 axis (78). This aforementioned study provided
a research strategy to identify circRNA targets that function in
disease mechanisms via methylation regulation.
Breast cancer
Leukemia virus complex factor 1 (FLI1) promotes
solid tumor progression and is highly expressed in advanced and
metastatic breast cancer (79).
Its role in breast cancer progression and metastasis is related to
circRNA FLI1 exonic circular RNA (FECR1), which is formed from
exons 2, 3 and 4 of FLI1, and can both bind to the FLI1 promoter in
cis configuration and recruit TET1 via a positive feedback
mechanism. It also activates FLI1 expression by binding to DNMT1 in
trans configuration, thereby downregulating DNMT1 expression and
inducing DNA hypomethylation to promote breast cancer metastasis
(80).
Bladder cancer
IGF2BP1 can function as an m6A reader and has
carcinogenic effects in cancer cells by stabilizing the methylation
of oncogene mRNA molecules, including Fascin actin-bundling protein
1 (FSCN1) and MYC (81).
circ-protein tyrosine phosphatase receptor type A interacts with
the K homology domain of IGF2BP1 and prevents it from recognizing
m6A-modified FSCN1 and MYC mRNAs, to inhibit bladder cancer cell
proliferation, migration and invasion (81). A previous study of bladder cancer
demonstrated that circ-100284 expression increases and acts as a
sponge for miR-217 to induce demethylation of heat shock protein 70
(HSP70) and enhance Aurora-B activity, which accelerates cell cycle
transitions and cell proliferation (82). However, the aforementioned study
did not determine how miR-217 caused HSP70 demethylation (82).
Non-small cell lung carcinoma
Hsa_circ_0072309 acts as a sponge for miR-607 to
upregulate FTO and promote tumorigenesis in non-small cell lung
carcinoma (83). Therefore,
hsa_circ_0072309 regulates downstream gene expression levels
through methylation modification via the regulation of the
miR-607/FTO axis to promote non-small cell lung carcinoma
tumorigenesis and invasion (83).
Ovarian cancer
circ-RAB11 family interacting protein 1 induces and
promotes autophagy flux in ovarian cancer cells to increase tumor
cell proliferation and migration (84). This circRNA can bind to FTO mRNA
and promote FTO expression, which downregulates autophagy-related
gene (ATG)5 and ATG7 mRNA m6A methylation and therefore increases
ATG5 and ATG7 expression levels, promoting disease progression
(84).
Esophageal squamous cell carcinoma
(ESCC)
circ-regulating synaptic membrane exocytosis protein
(circRIMS) can promote gastric cancer progression (85) and may also serve a similar role in
ESCC. It has previously been reported that circRIMS was
overexpressed in 60 patients with ESCC, predicting poor survival,
and its levels were negatively correlated with those of miR-613,
which is under-expressed in ESCC (86). In ESCC cells, overexpression of
circRIMS was demonstrated to increase miR-613 methylation and
reduce the inhibitory effect of miR-613 on cell proliferation
(86). Furthermore, animal
experiments demonstrated that circRIMS promotes tumor growth by
downregulating miR-613 via methylation (86).
Endometrial cancer (EC)
In an analysis comparing 60 EC and paired non-tumor
tissue samples, circ-ATPase family AAA domain containing 1
(circ-ATAD1) expression was downregulated in EC tissues, whereas
miR-10a was upregulated and its levels were correlated with those
of circ-ATAD1 (87). Reverse
transcription-quantitative PCR and methylation-specific PCR
analysis, determined that circ-ATAD1 overexpression promotes
miR-10a methylation and downregulates its expression, completely
reversing the effect of miR-10a in enhancing EC cell invasion and
migration. These results indicated that circ-ATAD1 is downregulated
in EC and may function to downregulate miR-10a via methylation to
inhibit EC cell invasion and migration (87). Using a similar methodology,
circ-FAT atypical cadherin 1 (circFAT1) was shown to be upregulated
in EC and its levels were positively correlated with those of
miR-21 in EC tissues (88).
Overexpression of circFAT1 increased miR-21 expression levels by
reducing miR-21 gene methylation to increase cancer stem cell
characteristics and promote tumor progression (88).
GBM
circ-spindle and kinetochore-associated complex
subunit 3 (circSKA3) is highly expressed in breast cancer and
exhibits a carcinogenic effect (89). It is also highly expressed in GBM
tissues, where its levels are negatively correlated with those of
miR-1 (34). Furthermore, high
circSKA3 levels are significantly associated with poor survival
rates of patients with GBM (34).
Analysis of the underlying mechanism confirmed that overexpression
of circSKA3 in GBM cells increases miR-1 methylation and decreases
its expression, limiting the inhibitory effect of miR-1 on cell
proliferation (34).
Wound repair, immunity and non-neoplastic
disease
Wound repair
Wound healing is enhanced in mice with increased
circ-angiomotin-like 1 (circ-Amotl1) expression levels. This is a
result of the ectopic expression of circ-Amotl1, which leads to
increased levels of the transcription factor, STAT3, and promotes
its nuclear translocation, which enhances DNMT3a expression leading
to miR-17 gene promoter methylation (90). Decreased miR-17-5p levels increase
fibronectin, DNMT3a and STAT3 expression levels, generating a
positive feedback loop (90).
Together, these activities promote cell proliferation, survival and
migration and enhance wound repair (90).
Immunity and immune diseases
Type three innate lymphocytes (ILC3) have key roles
in innate immunity and intestinal homeostasis (91). Nuclear receptor subfamily 4 group
A member 1 (Nr4a1) initiates Notch2 signal activation to help
maintain ILC3 homeostasis, which depends on m6A modification of
Nr4a1 mRNA by the highly expressed molecule, circ-zinc finger and
BTB domain-containing protein 20 (circ-Zbtb20). Circ-Zbtb20
enhances the interaction between ALKBH5 and Nr4a1 mRNA, which
indicates that circRNA and methylation may have a unique role in
immune system diseases (92). For
example, the expression levels of circ-inhibitor of Bruton tyrosine
kinase (circ-IBTK) and miR-29b are down- and upregulated,
respectively, in systemic lupus erythematosus (SLE). These
expression levels correlate with SLE disease activity index score,
as well as anti-double-stranded DNA and complement C3 levels in
patients with SLE (93).
Mechanistic studies have demonstrated that circ-IBTK may reverse
miR-29b-induced DNA methylation in SLE by binding to miR-29b, which
activates the AKT signaling pathway and promotes disease occurrence
and development (93). Similarly,
the overexpression of CD11a and CD70 in CD4+ T cells
contributes to the production of large amounts of autoantibodies,
which induce SLE (94).
Hsa_circ_0012919 upregulation is higher in CD4+ T cells
from patients with active SLE than in those with inactive SLE or
healthy controls. Moreover, CD11a and CD70 expression and
methylation levels are inversely proportional to hsa_circ_0012919
expression levels (95).
Furthermore, the overexpression of DNMT1 can reverse this
phenomenon, which indicates that the upregulation of
hsa_circ_0012919 reduces CD11a and CD70 methylation levels by
downregulating DNMT1. This induces high expression levels of these
two proteins to promote SLE occurrence and progression (95).
Cardiovascular diseases
Reducing autophagy can ameliorate cardiomyocyte
damage (96) and
autophagy-related circRNAs (ACRs) have been identified in a
myocardium ischemia-reperfusion injury model (97). PTEN-induced kinase 1 (Pink1)
expression levels are significantly altered via ACR overexpression,
which indicates that ACR may regulate autophagy in cardiomyocytes
via Pink1. Furthermore, RNA immunoprecipitation and biotinylated
probe pull-down assays demonstrated an interaction between ACR and
DNMT3b, in which DNMT3b regulates Pink1 protein expression levels
(97). The results of this study
also demonstrated that binding of DNMT3b to the CpG region of the
Pink1 promoter increases following ACR knockdown, which
demonstrated that ACR can reduce the methylation level of the CpG
region of the Pink1 promoter and promote Pink1 expression. This
reduced the excessive activation of the autophagy signaling pathway
(97).
Neurological diseases
circ-stromal antigen 1 (circ-STAG1) levels are
significantly lower in hippocampus tissue from mice exposed to
chronically unpredictable stress and in the peripheral blood of
patients with depression. Furthermore, circ-STAG1 overexpression
significantly alleviates astrocyte dysfunction and depression-like
behavior caused by chronic unpredictable stress (98). circSTAG1 binds to ALKBH5 and
reduces its nuclear transport, resulting in increased m6A
methylation of fatty acid amide hydrolase (FAAH) mRNA and the
downregulation of FAAH expression levels in astrocytes, which
subsequently alleviates depression-like behavior (98).
Osteoarthritis
Lipopolysaccharide (LPS) can induce chondrocyte
damage, leading to osteoarthritis. circ-fatty acid desaturase 2
(FADS2) can protect against this effect (99). circFADS2 overexpression and
miR-195-5p downregulation are observed in chondrocytes following
LPS treatment and the overexpression of circFADS2 increases
miR-195-5p methylation, reduces miR-195-5p expression levels and
downregulates apoptosis, which therefore protects chondrocytes from
LPS-induced damage (99).
Databases related to circRNA methylation
research
Databases relevant to circRNA methylation research
are presented in Table III. The
TRCirc (http://licpathway.net/TRCirc)
database mainly focuses on the retrieval and visualization of
circRNA regulatory information (100). TRCirc content includes uniform
transcription factor binding site data, and H3K27ac, RNA-seq and
450k chromatin immunoprecipitation-seq array data sets. The current
release of TRCirc includes data from more than 100 cell types,
involving 92,375 circRNAs and 161 transcription factors, covering
circRNA-related genetic and epigenetic information from 151 cell
lines, including DNA methylation and super-enhancer H3K27ac
signaling and expression, and supports user downloads. Currently,
only human data searches are supported, providing an effective
screening tool for downstream analysis of circRNAs.
 | Table III.Databases for circRNA methylation
research. |
Table III.
Databases for circRNA methylation
research.
| First author,
year | Database | Type of data | URL | (Refs.) |
|---|
| Tang Z, 2019 | TRCirc | DNA
methylation |
http://www.licpathway.net/TRCirc | (100) |
| Huang W, 2021 | TransCirc | circRNAs with m6A
modification site information |
https://www.biosino.org/transcirc/ | (101) |
| Zheng Y, 2018 | m6AVar | Single nucleotide
polymorphism sites that influence m6A modification |
http://m6avar.renlab.org/ | (102) |
| Luo X, 2021 | RMVar | Single nucleotide
polymorphism sites that influence m6A modification |
http://rmvar.renlab.org/ | (103) |
| Zhou Y, 2016 | SRAMP | m6A modification
site prediction on the target RNA sequence |
http://www.cuilab.cn/sramp | (104) |
| Liu H, 2015 | MeT-DB | Transcriptome
methylation in mammalian cells |
http://compgenomics.utsa.edu/methylation/ | (105) |
| Liu M, 2019 | Circbank | circRNA m6A
modification information |
http://www.circbank.cn/ | (106) |
The TransCirc database (http://www.biosino.org/transcirc/) (101) integrates a variety of
translation-related evidence and search results intuitively present
relevant evidence related to translation products. The data
includes analysis of the translation potential of 328,080 known
human circRNAs, of which there are 39,397 circRNAs with m6A
modification site information.
The m6AVar (http://m6avar.renlab.org/) or RMVar (http://rmvar.renlab.org/) (102,103) databases can be used to identify
single nucleotide polymorphism (SNP) sites that influence m6A
modification. These databases include information on SNPs
implicated in post-transcriptional regulation, miRNA binding
predictions and SNPs with disease effects.
The sequence-based RNA adenosine methylation site
predictor (SRAMP) database (http://www.cuilab.cn/sramp) (104) is used to predict m6A
modification sites in target RNA sequences, as well as m6A
modification sites in mammals. SRAMP only needs RNA sequences to
run predictions, without the need to load external omics data, and
is a powerful tool for m6A locus analysis where sequencing data are
insufficient, such as those for long non-coding RNA and circRNA
molecules.
The MethylTranscriptome (MeT-DB) database
(http://compgenomics.utsa.edu/methylation/) (105) was constructed by collecting data
from the Photoactivatable ribonucleoside-enhanced crosslinking and
immunoprecipitation (PAR-CLIP)-seq and MeRIP-seq databases relating
to eight m6A-related regulatory factors (FTO, KIAA1429, METTL14,
METTL3, WT1-associated protein, heterogenous nuclear
ribonucleoprotein C, YTHDC1 and YTHDF1) and allows visualization of
specific regulatory positions using relevant sequencing data.
The Circbank database (circbank.cn/) (106) contains more than 140,000 human
circRNA records, each of which has a separate detailed information
page, which includes the following: i) circRNA sequence data; ii)
mouse circRNAs with high homology to human circRNAs and their
corresponding sequences; iii) predictive analysis of miRNA binding;
iv) predictive open reading frame analysis; v) summary of mutations
and polymorphism sites recorded in the Catalogue of Somatic
Mutations in Cancer database (https://cancer.sanger.ac.uk/cosmic) (107); and vi) m6A modification
information.
Conclusion
RNA methylation serves an important role in the
expression, transport, stability, translation and degradation of
circRNAs (108); however, to the
best of our knowledge no studies have determined whether DNA
methylation affects circRNA biological function. circRNAs can also
regulate disease processes and may participate in immune responses
by regulating proteins related to methylation modification or being
modified by methylation. The interaction between circRNAs and
methylation illustrates the synergistic role of epigenetics and
noncoding RNA molecules in disease. Therefore, future studies
focusing on the relationship between methylation and circRNAs will
be important for study of mammalian development and disease.
Although progress has been made in this area, further exploration
is required. For example, the mechanisms underlying the effects of
methylation modification on circRNA localization and expression,
warrant further investigation. Moreover, to the best of our
knowledge there are no relevant studies on the methylation of other
basic sites in RNA, for example, the uridine modification of
circRNA (another common mode of RNA methylation modification).
Furthermore, the effects of circRNAs on additional cellular
biological functions via methylation modification requires further
elucidation, such as the metabolism, development and morphological
changes of cells.
In addition to DNA methylation and RNA methylation,
histone modification is also an important epigenetic process. At
present, reports involving histones, methylation and circRNA are
rare. For example, one study reported that circ histone-lysine
N-methyltransferase eggless (Egg) inhibits histone H3 lysine 9
methylation via encoding the circEgg-P122 protein in the silkworm,
Bombyx mori (Bm/bmo). Moreover, these results demonstrated
that circEgg also promotes histone H3 lysine 9 acetylation and
positively regulates gene expression of histone deacetylase Rpd, by
acting as a sponge for the miRNA, bmo-miR-3391-5p (109). Furthermore, differentially
expressed circRNA may control pheochromocytoma/paragangliomas
pathogenesis by regulating histone methylation, highlighting the
potential role of circRNAs as biomarkers (110). However, studies concerning
epigenetic modifications, such as histone methylations and
circRNAs, in the context of disease, remain very limited.
The study of combination of circRNA and methylation
modification faces certain future challenges. First, there is a
lack of bioinformatics methods that can efficiently identify and
analyze the types, sites and patterns of circRNA methylation
modifications. Second, circRNAs function in brain development, due
to their unique, highly stable structures (111) and methylation is also related to
mammalian development (112).
How to effectively study and determine the roles and underlying
mechanisms of methylation and circRNA in such developmental
processes will be a challenge for future research. Finally, the
goal of all biomedical research is translation into clinical
application. Although methylation-modified circRNAs and methylation
regulated by circRNAs have been shown to influence disease
processes, translation of these findings into targeted drug
treatments for patients with diseases is a difficulty that needs to
be overcome in the future.
Acknowledgements
Not applicable.
Funding
This work was supported by the Natural Science Foundation of
Gansu Province (grant no. 20JR5RA601) and the In-hospital project
of The 940 Hospital of Joint Logistics Support Force of Chinese
People's Liberation Army (grant no. 2021yxky057 and
2021yxky070).
Availability of data and materials
Not applicable.
Authors' contributions
CZ, HC, WZ and DC conceived the ideas for the
article and helped draft the manuscript. QY, PM and ZC performed
the literature search and acquisition of data. CH and FK
contributed to the analysis and interpretation of all the
literature. QY, CZ and DC provided funding support. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Razin A and Cedar H: DNA methylation and
gene expression. Microbiol Rev. 55:451–458. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Covelo-Molares H, Bartosovic M and
Vanacova S: RNA methylation in nuclear pre-mRNA processing. Wiley
Interdiscip Rev RNA. 9:e14892018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang X, Zhao BS, Roundtree IA, Lu Z, Han
D, Ma H, Weng X, Chen K, Shi H and He C: N(6)-methyladenosine
modulates messenger RNA translation efficiency. Cell.
161:1388–1399. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ehrlich M: DNA hypermethylation in
disease: Mechanisms and clinical relevance. Epigenetics.
14:1141–1163. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yang C, Hu Y, Zhou B, Bao Y, Li Z, Gong C,
Yang H, Wang S and Xiao Y: The role of m6A modification
in physiology and disease. Cell Death Dis. 11:9602020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen LL and Yang L: Regulation of circRNA
biogenesis. RNA Biol. 12:381–388. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kristensen LS, Andersen MS, Stagsted LVW,
Ebbesen KK, Hansen TB and Kjems J: The biogenesis, biology and
characterization of circular RNAs. Nat Rev Genet. 20:675–691. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jones PA: Functions of DNA methylation:
Islands, start sites, gene bodies and beyond. Nat Rev Genet.
13:484–492. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Breiling A and Lyko F: Epigenetic
regulatory functions of DNA modifications: 5-methylcytosine and
beyond. Epigenetics Chromatin. 8:242015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sobiak B and Leśniak W: The effect of
single CpG demethylation on the pattern of DNA-protein binding. Int
J Mol Sci. 20:9142019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cedar H and Bergman Y: Programming of DNA
methylation patterns. Ann Rev Biochem. 81:97–117. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Okano M, Bell DW, Haber DA and Li E: DNA
methyltransferases Dnmt3a and Dnmt3b are essential for de novo
methylation and mammalian development. Cell. 99:247–257. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Goll MG and Bestor TH: Eukaryotic cytosine
methyltransferases. Ann Rev Biochem. 74:481–514. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gibbons RJ, McDowell TL, Raman S, O'Rourke
DM, Garrick D, Ayyub H and Higgs DR: Mutations in ATRX, encoding a
SWI/SNF-like protein, cause diverse changes in the pattern of DNA
methylation. Nat Genet. 24:368–371. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shamma A, Suzuki M, Hayashi N, Kobayashi
M, Sasaki N, Nishiuchi T, Doki Y, Okamoto T, Kohno S, Muranaka H,
et al: ATM mediates pRB function to control DNMT1 protein stability
and DNA methylation. Mol Cell Biol. 33:3113–3124. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bhutani N, Burns DM and Blau HM: DNA
demethylation dynamics. Cell. 146:866–872. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dean W: Pathways of DNA demethylation. Adv
Exp Med Biol. 945:247–274. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bochtler M, Kolano A and Xu GL: DNA
demethylation pathways: Additional players and regulators.
Bioessays. 39:1–13. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nan X, Ng HH, Johnson CA, Laherty CD,
Turner BM, Eisenman RN and Bird A: Transcriptional repression by
the methyl-CpG-binding protein MeCP2 involves a histone deacetylase
complex. Nature. 393:386–389. 1998. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zheng Y, He L, Wan Y and Song J:
H3K9me-enhanced DNA hypermethylation of the p16INK4a gene: An
epigenetic signature for spontaneous transformation of rat
mesenchymal stem cells. Stem Cells Dev. 22:256–267. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gujar H, Weisenberger DJ and Liang G: The
roles of Human DNA methyltransferases and their isoforms in shaping
the epigenome. Genes (Basel). 10:1722019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen Z and Zhang Y: Role of mammalian DNA
methyltransferases in development. Ann Rev Biochem. 89:135–158.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang FL, Yang Y, Liu ZY, Qin Y and Jin T:
Correlation between methylation of the p16 promoter and cervical
cancer incidence. Eur Rev Med Pharmacol Sci. 21:2351–2356.
2017.PubMed/NCBI
|
|
24
|
Allameh A, Moazeni-Roodi A, Harirchi I,
Ravanshad M, Motiee-Langroudi M, Garajei A, Hamidavi A and
Mesbah-Namin SA: Promoter DNA methylation and mRNA expression level
of p16 gene in oral squamous cell carcinoma: Correlation with
Clinicopathological characteristics. Pathol Oncol Res.
25:1535–1543. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li Y, Zhang T, Zhang H, Wang X, Liu X,
Huang Q and Li L: Clinical significance of P16 gene methylation in
lung cancer. Adv Exp Med Biol. 1255:133–142. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Huang H, Weng H, Deng X and Chen J: RNA
modifications in cancer: Functions, mechanisms, and therapeutic
implications. Ann Rev Cancer Biol. 4:221–240. 2020. View Article : Google Scholar
|
|
27
|
Karthiya R and Khandelia P: m6A RNA
methylation: Ramifications for gene expression and human health.
Mol Biotechnol. 62:467–484. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zaccara S, Ries RJ and Jaffrey SR:
Reading, writing and erasing mRNA methylation. Nat Rev Mol Cell
Biol. 20:608–624. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen X, Sun YZ, Liu H, Zhang L, Li JQ and
Meng J: RNA methylation and diseases: Experimental results,
databases, Web servers and computational models. Brief Bioinform.
20:896–917. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lan Q, Liu PY, Haase J, Bell JL,
Hüttelmaier S and Liu T: The critical role of RNA m6A
methylation in cancer. Cancer Res. 79:1285–1292. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ferreira HJ, Davalos V, de Moura MC, Soler
M, Perez-Salvia M, Bueno-Costa A, Setien F, Moran S, Villanueva A
and Esteller M: Circular RNA CpG island hypermethylation-associated
silencing in human cancer. Oncotarget. 9:29208–29219. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Su H, Wang G, Wu L, Ma X, Ying K and Zhang
R: Transcriptome-wide map of m6A circRNAs identified in a rat model
of hypoxia mediated pulmonary hypertension. BMC Genomics.
21:392020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li P, Yu H, Zhang G, Kang L, Qin B, Cao Y,
Luo J, Chen X, Wang Y, Qin M, et al: Identification and
characterization of N6-methyladenosine circRNAs and
methyltransferases in the lens epithelium cells from age-related
cataract. Invest Ophthalmol Vis Sci. 61:132020. View Article : Google Scholar
|
|
34
|
Zhou M, Li H, Chen K, Ding W, Yang C and
Wang X: CircSKA3 downregulates miR-1 through methylation in
glioblastoma to promote cancer cell proliferation. Cancer Manag
Res. 13:509–514. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang C, Wang J, Geng X, Tu J, Gao H, Li
L, Zhou X, Wu H, Jing J, Pan W and Mou Y: Circular RNA expression
profile and m6A modification analysis in poorly differentiated
adenocarcinoma of the stomach. Epigenomics. 12:1027–1040. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Niu X, Xu J, Liu J, Chen L, Qiao X and
Zhong M: Landscape of N6-methyladenosine modification
patterns in human ameloblastoma. Front Oncol. 10:5564972020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yang Y, Fan X, Mao M, Song X, Wu P, Zhang
Y, Jin Y, Yang Y, Chen LL, Wang Y, et al: Extensive translation of
circular RNAs driven by N6-methyladenosine. Cell Res.
27:626–641. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Roundtree IA, Luo GZ, Zhang Z, Wang X,
Zhou T, Cui Y, Sha J, Huang X, Guerrero L, Xie P, et al: YTHDC1
mediates nuclear export of N6-methyladenosine methylated
mRNAs. Elife. 6:e313112017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhou C, Molinie B, Daneshvar K, Pondick
JV, Wang J, Van Wittenberghe N, Xing Y, Giallourakis CC and Mullen
AC: Genome-wide maps of m6A circRNAs identify widespread and
cell-type-specific methylation patterns that are distinct from
mRNAs. Cell Rep. 20:2262–2276. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Di Timoteo G, Dattilo D, Centrón-Broco A,
Colantoni A, Guarnacci M, Rossi F, Incarnato D, Oliviero S, Fatica
A, Morlando M and Bozzoni I: Modulation of circRNA Metabolism by
m6A modification. Cell Rep. 31:1076412020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chen RX, Chen X, Xia LP, Zhang JX, Pan ZZ,
Ma XD, Han K, Chen JW, Judde JG, Deas O, et al:
N6-methyladenosine modification of circNSUN2 facilitates
cytoplasmic export and stabilizes HMGA2 to promote colorectal liver
metastasis. Nat Commun. 10:46952019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang X, Lu Z, Gomez A, Hon GC, Yue Y, Han
D, Fu Y, Parisien M, Dai Q, Jia G, et al:
N6-methyladenosine-dependent regulation of messenger RNA stability.
Nature. 505:117–120. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Xu J, Wan Z, Tang M, Lin Z, Jiang S, Ji L,
Gorshkov K, Mao Q, Xia S, Cen D, et al:
N6-methyladenosine-modified circRNA-SORE sustains
sorafenib resistance in hepatocellular carcinoma by regulating
β-catenin signaling. Mol Cancer. 19:1632020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Coots RA, Liu XM, Mao Y, Dong L, Zhou J,
Wan J, Zhang X and Qian SB: m6A Facilitates
eIF4F-independent mRNA translation. Mol Cell. 68:504–514.e7. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Li A, Chen YS, Ping XL, Yang X, Xiao W,
Yang Y, Sun HY, Zhu Q, Baidya P, Wang X, et al: Cytoplasmic
m6A reader YTHDF3 promotes mRNA translation. Cell Res.
27:444–447. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Shi H, Wang X, Lu Z, Zhao BS, Ma H, Hsu
PJ, Liu C and He C: YTHDF3 facilitates translation and decay of
N6-methyladenosine-modified RNA. Cell Res. 27:315–328.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Lei M, Zheng G, Ning Q, Zheng J and Dong
D: Translation and functional roles of circular RNAs in human
cancer. Mol Cancer. 19:302020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jakobi T, Siede D, Eschenbach J, Heumüller
AW, Busch M, Nietsch R, Meder B, Most P, Dimmeler S, Backs J, et
al: Deep characterization of circular RNAs from human
cardiovascular cell models and cardiac tissue. Cells. 9:16162020.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kmietczyk V, Riechert E, Kalinski L,
Boileau E, Malovrh E, Malone B, Gorska A, Hofmann C, Varma E,
Jürgensen L, et al: m6A-mRNA methylation regulates
cardiac gene expression and cellular growth. Life Sci Alliance.
2:e2018002332019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Du H, Zhao Y, He J, Zhang Y, Xi H, Liu M,
Ma J and Wu L: YTHDF2 destabilizes m(6)A-containing RNA through
direct recruitment of the CCR4-NOT deadenylase complex. Nat Commun.
7:126262016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Park OH, Ha H, Lee Y, Boo SH, Kwon DH,
Song HK and Kim YK: Endoribonucleolytic cleavage of
m6A-containing RNAs by RNase P/MRP complex. Mol Cell.
74:494–507.e8. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Lee Y, Choe J, Park OH and Kim YK:
Molecular mechanisms driving mRNA degradation by m6A
modification. Trends Genet. 36:177–188. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Chen C, Yuan W, Zhou Q, Shao B, Guo Y,
Wang W, Yang S, Guo Y, Zhao L, Dang Q, et al:
N6-methyladenosine-induced circ1662 promotes metastasis of
colorectal cancer by accelerating YAP1 nuclear localization.
Theranostics. 11:4298–4315. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Liu H, Lan T, Li H, Xu L, Chen X, Liao H,
Chen X, Du J, Cai Y, Wang J, et al: Circular RNA circDLC1 inhibits
MMP1-mediated liver cancer progression via interaction with HuR.
Theranostics. 11:1396–1411. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Rao X, Lai L, Li X, Wang L, Li A and Yang
Q: N6-methyladenosine modification of circular RNA
circ-ARL3 facilitates Hepatitis B virus-associated hepatocellular
carcinoma via sponging miR-1305. IUBMB Life. 73:408–417. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhao J, Lee EE, Kim J, Yang R, Chamseddin
B, Ni C, Gusho E, Xie Y, Chiang CM, Buszczak M, et al: Transforming
activity of an oncoprotein-encoding circular RNA from human
papillomavirus. Nat Commun. 10:23002019. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Chen Z, Ling K, Zhu Y, Deng L, Li Y and
Liang Z: circ0000069 promotes cervical cancer cell proliferation
and migration by inhibiting miR-4426. Biochem Biophys Res Commun.
551:114–120. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Li B, Zhu L, Lu C, Wang C, Wang H, Jin H,
Ma X, Cheng Z, Yu C, Wang S, et al: circNDUFB2 inhibits non-small
cell lung cancer progression via destabilizing IGF2BPs and
activating anti-tumor immunity. Nat Commun. 12:2952021. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Nan A, Chen L, Zhang N, Jia Y, Li X, Zhou
H, Ling Y, Wang Z, Yang C, Liu S and Jiang Y: Circular RNA
circNOL10 inhibits lung cancer development by promoting
SCLM1-mediated transcriptional regulation of the humanin
polypeptide family. Adv Sci (Weinh). 6:18006542019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Marur S and Forastiere AA: Head and neck
squamous cell carcinoma: Update on epidemiology, diagnosis, and
treatment. Mayo Clinic Proc. 91:386–396. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wu P, Fang X, Liu Y, Tang Y, Wang W, Li X
and Fan Y: N6-methyladenosine modification of circCUX1 confers
radioresistance of hypopharyngeal squamous cell carcinoma through
caspase1 pathway. Cell Death Dis. 12:2982021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Li Z, Yang HY, Dai XY, Zhang X, Huang YZ,
Shi L, Wei JF and Ding Q: CircMETTL3, upregulated in a
m6A-dependent manner, promotes breast cancer progression. Int J
Biol Sci. 17:1178–1190. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Wang H, Hu X, Huang M, Liu J, Gu Y, Ma L,
Zhou Q and Cao X: Mettl3-mediated mRNA m6A methylation
promotes dendritic cell activation. Nat Commun. 10:18982019.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Chen YG, Chen R, Ahmad S, Verma R, Kasturi
SP, Amaya L, Broughton JP, Kim J, Cadena C, Pulendran B, et al:
N6-methyladenosine modification controls circular RNA immunity. Mol
Cell. 76:96–109.e9. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Guo M, Yan R, Yao H, Duan L, Sun M, Xue Z
and Jia Y: IFN regulatory Factor 1 mediates macrophage pyroptosis
induced by oxidized low-density lipoprotein in patients with acute
coronary syndrome. Mediators Inflamm. 2019:29171282019. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Guo M, Yan R, Ji Q, Yao H, Sun M, Duan L,
Xue Z and Jia Y: IFN regulatory Factor-1 induced macrophage
pyroptosis by modulating m6A modification of circ_0029589 in
patients with acute coronary syndrome. Int Immunopharmacol.
86:1068002020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Li X, Tian G and Wu J: Novel circGFRα1
promotes self-renewal of female germline stem cells mediated by
m6A Writer METTL14. Front Cell Dev Biol. 9:6404022021.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Tang C, Xie Y, Yu T, Liu N, Wang Z,
Woolsey RJ, Tang Y, Zhang X, Qin W, Zhang Y, et al:
m6A-dependent biogenesis of circular RNAs in male germ
cells. Cell Res. 30:211–228. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
He Y, Zhang Q, Zheng Q, Yu X and Guo W:
Distinct 5-methylcytosine profiles of circular RNA in human
hepatocellular carcinoma. Am J Transl Res. 12:5719–5729.
2020.PubMed/NCBI
|
|
70
|
Zhao Z, Song J, Tang B, Fang S, Zhang D,
Zheng L, Wu F, Gao Y, Chen C, Hu X, et al: CircSOD2 induced
epigenetic alteration drives hepatocellular carcinoma progression
through activating JAK2/STAT3 signaling pathway. J Exp Clin Cancer
Res. 39:2592020. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Liu Z, Yu Y, Huang Z, Kong Y, Hu X, Xiao
W, Quan J and Fan X: circRNA-5692 inhibits the progression of
hepatocellular carcinoma by sponging miR-328-5p to enhance DAB2IP
expression. Cell Death Dis. 10:9002019. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
An J, Rao A and Ko M: TET family
dioxygenases and DNA demethylation in stem cells and cancers. Exp
Mol Med. 49:e3232017. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Pei YF, Tao R, Li JF, Su LP, Yu BQ, Wu XY,
Yan M, Gu QL, Zhu ZG and Liu BY: TET1 inhibits gastric cancer
growth and metastasis by PTEN demethylation and re-expression.
Oncotarget. 7:31322–31335. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Fan J, Zhang Y, Mu J, He X, Shao B, Zhou
D, Peng W, Tang J, Jiang Y, Ren G and Xiang T: TET1 exerts its
anti-tumor functions via demethylating DACT2 and SFRP2 to
antagonize Wnt/β-catenin signaling pathway in nasopharyngeal
carcinoma cells. Clin Epigenetics. 10:1032018. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Zhang PF, Wei CY, Huang XY, Peng R, Yang
X, Lu JC, Zhang C, Gao C, Cai JB, Gao PT, et al: Circular RNA
circTRIM33-12 acts as the sponge of MicroRNA-191 to suppress
hepatocellular carcinoma progression. Mol Cancer. 18:1052019.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Dong ZR, Ke AW, Li T, Cai JB, Yang YF,
Zhou W, Shi GM and Fan J: CircMEMO1 modulates the promoter
methylation and expression of TCF21 to regulate hepatocellular
carcinoma progression and sorafenib treatment sensitivity. Mol
Cancer. 20:752021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Sun S, Gao J, Zhou S, Li Y, Wang Y, Jin L,
Li J, Liu B, Zhang B, Han S, et al: A novel circular RNA circ-LRIG3
facilitates the malignant progression of hepatocellular carcinoma
by modulating the EZH2/STAT3 signaling. J Exp Clin Cancer Res.
39:2522020. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Chi F, Cao Y and Chen Y: Analysis and
validation of circRNA-miRNA network in regulating m6A
RNA methylation modulators reveals CircMAP2K4/miR-139-5p/YTHDF1
axis involving the proliferation of hepatocellular carcinoma. Front
Oncol. 11:5605062021. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Miao B, Bauer AS, Hufnagel K, Wu Y,
Trajkovic-Arsic M, Pirona AC, Giese N, Taipale J, Siveke JT,
Hoheisel JD and Lueong S: The transcription factor FLI1 promotes
cancer progression by affecting cell cycle regulation. Int J
Cancer. 147:189–201. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Chen N, Zhao G, Yan X, Lv Z, Yin H, Zhang
S, Song W, Li X, Li L, Du Z, et al: A novel FLI1 exonic circular
RNA promotes metastasis in breast cancer by coordinately regulating
TET1 and DNMT1. Genome Biol. 19:2182018. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Xie F, Huang C, Liu F, Zhang H, Xiao X,
Sun J, Zhang X and Jiang G: CircPTPRA blocks the recognition of RNA
N6-methyladenosine through interacting with IGF2BP1 to
suppress bladder cancer progression. Mol Cancer. 20:682021.
View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Huang ZM, Wang H and Ji ZG: circRNA-100284
activates aurora kinase B by inducing methylation of HSP70 via
microRNA-217 to promote proliferation of bladder cancer cells. J
Cancer Res Clin Oncol. 147:703–712. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Mo WL, Deng LJ, Cheng Y, Yu WJ, Yang YH
and Gu WD: Circular RNA hsa_circ_0072309 promotes tumorigenesis and
invasion by regulating the miR-607/FTO axis in non-small cell lung
carcinoma. Aging. 13:11629–11645. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Zhang Z, Zhu H and Hu J: circRAB11FIP1
promoted autophagy flux of ovarian cancer through DSC1 and miR-129.
Cell Death Dis. 12:2192021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Lin J, Zhang Y, Zeng X, Xue C and Lin X:
circRNA circRIMS acts as a MicroRNA sponge to promote gastric
cancer metastasis. ACS Omega. 5:23237–23246. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Wan H, Yuan B, Jiang K, Wei J, Feng X, Sun
B and Wang F: circRNA circRIMS is overexpressed in esophageal
squamous cell carcinoma and downregulate miR-613 through
methylation to increase cell proliferation. Cancer Manag Res.
13:4587–4595. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Yang P, Yun K and Zhang R: circRNA
circ-ATAD1 is downregulated in endometrial cancer and suppresses
cell invasion and migration by downregulating miR-10a through
methylation. Mamm Genome. 32:488–494. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Wu W, Zhou J, Wu Y, Tang X and Zhu W:
Overexpression of circRNA circFAT1 in endometrial cancer cells
increases their stemness by upregulating miR-21 through
methylation. Cancer Biother Radiopharm. Jul 27–2021.(Epub ahead of
print). doi: 10.1089/cbr.2020.4506. View Article : Google Scholar
|
|
89
|
Du WW, Yang W, Li X, Fang L, Wu N, Li F,
Chen Y, He Q, Liu E, Yang Z, et al: The circular RNA circSKA3 binds
integrin β1 to induce invadopodium formation enhancing breast
cancer invasion. Mol Ther. 28:1287–1298. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Yang ZG, Awan FM, Du WW, Zeng Y, Lyu J, Wu
D, Gupta S, Yang W and Yang BB: The circular RNA interacts with
STAT3, increasing its nuclear translocation and wound repair by
modulating Dnmt3a and miR-17 function. Mol Ther. 25:2062–2074.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Domingues RG and Hepworth MR:
Immunoregulatory sensory circuits in group 3 innate lymphoid cell
(ILC3) function and tissue homeostasis. Front Immunol. 11:1162020.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Liu B, Liu N, Zhu X, Yang L, Ye B, Li H,
Zhu P, Lu T, Tian Y and Fan Z: Circular RNA circZbtb20 maintains
ILC3 homeostasis and function via Alkbh5-dependent m6A
demethylation of Nr4a1 mRNA. Cell Mol Immunol. 18:1412–1424. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Wang X, Zhang C, Wu Z, Chen Y and Shi W:
CircIBTK inhibits DNA demethylation and activation of AKT signaling
pathway via miR-29b in peripheral blood mononuclear cells in
systemic lupus erythematosus. Arthritis Res Ther. 20:1182018.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Zhao M, Sun Y, Gao F, Wu X, Tang J, Yin H,
Luo Y, Richardson B and Lu Q: Epigenetics and SLE: RFX1
downregulation causes CD11a and CD70 overexpression by altering
epigenetic modifications in lupus CD4+ T cells. J
Autoimmun. 35:58–69. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhang C, Wang X, Chen Y, Wu Z, Zhang C and
Shi W: The down-regulation of hsa_circ_0012919, the sponge for
miR-125a-3p, contributes to DNA methylation of CD11a and CD70 in
CD4+ T cells of systemic lupus erythematous. Clin Sci
(Lond). 132:2285–2298. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Corsetti G, Chen-Scarabelli C, Romano C,
Pasini E, Dioguardi FS, Onorati F, Knight R, Patel H, Saravolatz L,
Faggian G and Scarabelli TM: Autophagy and Oncosis/Necroptosis are
enhanced in cardiomyocytes from heart failure patients. Med Sci
Monit Basic Res. 25:33–44. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zhou LY, Zhai M, Huang Y, Xu S, An T, Wang
YH, Zhang RC, Liu CY, Dong YH, Wang M, et al: The circular RNA ACR
attenuates myocardial ischemia/reperfusion injury by suppressing
autophagy via modulation of the Pink1/FAM65B pathway. Cell Death
Differ. 26:1299–1315. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Huang R, Zhang Y, Bai Y, Han B, Ju M, Chen
B, Yang L, Wang Y, Zhang H, Zhang H, et al:
N6-Methyladenosine modification of fatty acid amide
hydrolase messenger RNA in circular RNA STAG1-regulated astrocyte
dysfunction and depressive-like behaviors. Biol Psychiatry.
88:392–404. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Zhang H, Ge J and Lu X: CircFADS2 is
downregulated in osteoarthritis and suppresses LPS-induced
apoptosis of chondrocytes by regulating miR-195-5p methylation.
Arch Gerontol Geriatr. 96:1044772021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Tang Z, Li X, Zhao J, Qian F, Feng C, Li
Y, Zhang J, Jiang Y, Yang Y, Wang Q and Li C: TRCirc: A resource
for transcriptional regulation information of circRNAs. Brief
Bioinform. 20:2327–2333. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Huang W, Ling Y, Zhang S, Xia Q, Cao R,
Fan X, Fang Z, Wang Z and Zhang G: TransCirc: An interactive
database for translatable circular RNAs based on multi-omics
evidence. Nucleic Acids Res. 49:D236–D242. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Zheng Y, Nie P, Peng D, He Z, Liu M, Xie
Y, Miao Y, Zuo Z and Ren J: m6AVar: A database of functional
variants involved in m6A modification. Nucleic Acids Res.
46:D139–D145. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Luo X, Li H, Liang J, Zhao Q, Xie Y, Ren J
and Zuo Z: RMVar: An updated database of functional variants
involved in RNA modifications. Nucleic Acids Res. 49:D1405–D1412.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Zhou Y, Zeng P, Li YH, Zhang Z and Cui Q:
SRAMP: Prediction of mammalian N6-methyladenosine (m6A) sites based
on sequence-derived features. Nucleic Acids Res. 44:e912016.
View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Liu H, Flores MA, Meng J, Zhang L, Zhao X,
Rao MK, Chen Y and Huang Y: MeT-DB: A database of transcriptome
methylation in mammalian cells. Nucleic Acids Res. 43:(Database
Issue). D197–D203. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Liu M, Wang Q, Shen J, Yang BB and Ding X:
Circbank: A comprehensive database for circRNA with standard
nomenclature. RNA Biol. 16:899–905. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Tate JG, Bamford S, Jubb HC, Sondka Z,
Beare DM, Bindal N, Boutselakis H, Cole CG, Creatore C, Dawson E,
et al: COSMIC: The catalogue of somatic mutations in cancer.
Nucleic Acids Res. 47:D941–D947. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Zhang L, Hou C, Chen C, Guo Y, Yuan W, Yin
D, Liu J and Sun Z: The role of N 6-methyladenosine (m 6A)
modification in the regulation of circRNAs. Mol Cancer. 19:1052020.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Wang Z, Zhang Y, Dai K, Liang Z, Zhu M,
Zhang M, Pan J, Hu X, Zhang X, Xue R, et al: circEgg regulates
histone H3K9me3 by sponging bmo-miR-3391-5p and encoding
circEgg-P122 protein in the silkworm, Bombyx mori. Insect Biochem
Mol Biol. 124:1034302020. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Yu A, Li M, Xing C, Chen D, Wang C, Xiao
Q, Zhang L, Pang Y, Wang Y, Zu X and Liu L: A Comprehensive
analysis identified the key differentially expressed circular
ribonucleic acids and methylation-related function in
pheochromocytomas and paragangliomas. Front Genet. 11:152020.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Mehta SL, Dempsey RJ and Vemuganti R: Role
of circular RNAs in brain development and CNS diseases. Prog
Neurobiol. 186:1017462020. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Smith ZD and Meissner A: DNA methylation:
Roles in mammalian development. Nat Rev Genet. 14:204–220. 2013.
View Article : Google Scholar : PubMed/NCBI
|















