|
1
|
Kerschan-Schindl K: Prevention and
rehabilitation of osteoporosis. Wien Med Wochenschr. 166:22–27.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ensrud KE and Crandall CJ: Osteoporosis.
Ann Intern Med. 167:ITC17–ITC32. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Christiansen C: Osteoporosis: Diagnosis
and management today and tomorrow. Bone. 17 (Suppl 5):513S–516S.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Miller PD: Management of severe
osteoporosis. Expert Opin Pharmacother. 17:473–488. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Anthamatten A and Parish A: Clinical
update on osteoporosis. J Midwifery Womens Health. 64:265–275.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lee WC, Guntur AR, Long F and Rosen CJ:
Energy metabolism of the osteoblast: Implications for osteoporosis.
Endocr Rev. 38:255–266. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Verhaeghe J, Thomsen JS, van Bree R, van
Herck E, Bouillon R and Mosekilde L: Effects of exercise and disuse
on bone remodeling, bone mass, and biomechanical competence in
spontaneously diabetic female rats. Bone. 27:249–256. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Whiting DR, Guariguata L, Weil C and Shaw
J: IDF diabetes atlas: Global estimates of the prevalence of
diabetes for 2011 and 2030. Diabetes Res Clin Pract. 94:311–321.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ying X, Chen X, Liu H, Nie P, Shui X, Shen
Y, Yu K and Cheng S: Silibinin alleviates high glucose-suppressed
osteogenic differentiation of human bone marrow stromal cells via
antioxidant effect and PI3K/Akt signaling. Eur J Pharmacol.
765:394–401. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Botolin S and McCabe LR: Chronic
hyperglycemia modulates osteoblast gene expression through osmotic
and non-osmotic pathways. J Cell Biochem. 99:411–424. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gopalakrishnan V, Vignesh RC, Arunakaran
J, Aruldhas MM and Srinivasan N: Effects of glucose and its
modulation by insulin and estradiol on BMSC differentiation into
osteoblastic lineages. Biochem Cell Biol. 84:93–101. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Saliminejad K, Khorram Khorshid HR,
Soleymani Fard S and Ghaffari SH: An overview of microRNAs:
Biology, functions, therapeutics, and analysis methods. J Cell
Physiol. 234:5451–5465. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mohr AM and Mott JL: Overview of microRNA
biology. Semin Liver Dis. 35:3–11. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Awasthi R, Rathbone MJ, Hansbro PM, Bebawy
M and Dua K: Therapeutic prospects of microRNAs in cancer treatment
through nanotechnology. Drug Deliv Transl Res. 8:97–110. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Papaioannou G, Mirzamohammadi F and
Kobayashi T: MicroRNAs involved in bone formation. Cell Mol Life
Sci. 71:4747–4761. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pan BL, Tong ZW, Li SD, Wu L, Liao JL,
Yang YX, Li HH, Dai YJ, Li JE and Pan L: Decreased microRNA-182-5p
helps alendronate promote osteoblast proliferation and
differentiation in osteoporosis via the Rap1/MAPK pathway. Biosci
Rep. 38:BSR201806962018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hassan MQ, Maeda Y, Taipaleenmaki H, Zhang
W, Jafferji M, Gordon JA, Li Z, Croce CM, van Wijnen AJ, Stein JL,
et al: MiR-218 directs a Wnt signaling circuit to promote
differentiation of osteoblasts and osteomimicry of metastatic
cancer cells. J Biol Chem. 287:42084–42092. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kamiya N, Kobayashi T, Mochida Y, Yu PB,
Yamauchi M, Kronenberg HM and Mishina Y: Wnt inhibitors Dkk1 and
Sost are downstream targets of BMP signaling through the type IA
receptor (BMPRIA) in osteoblasts. J Bone Miner Res. 25:200–210.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xu R, Zhao M, Yang Y, Huang Z, Shi C, Hou
X, Zhao Y, Chen B, Xiao Z, Liu J, et al: MicroRNA-449c-5p inhibits
osteogenic differentiation of human VICs through Smad4-mediated
pathway. Sci Rep. 7:87402017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu Q, Guo Y, Wang Y, Zou X and Yan Z:
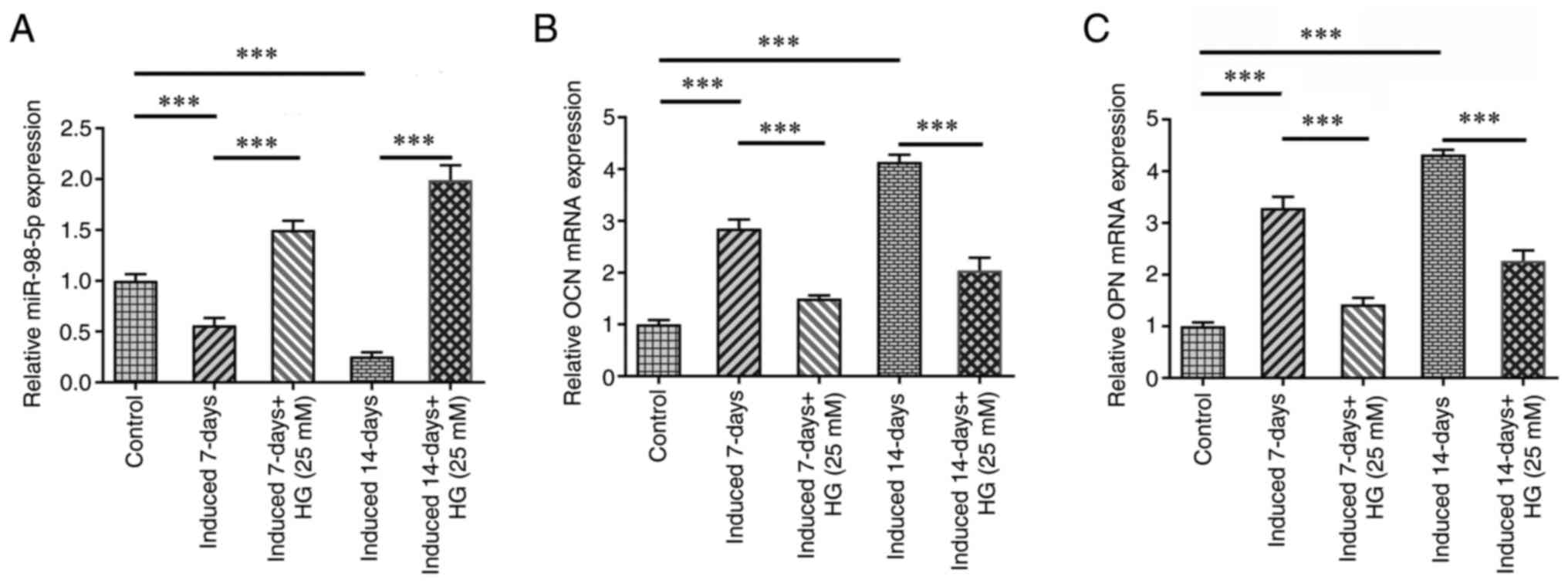
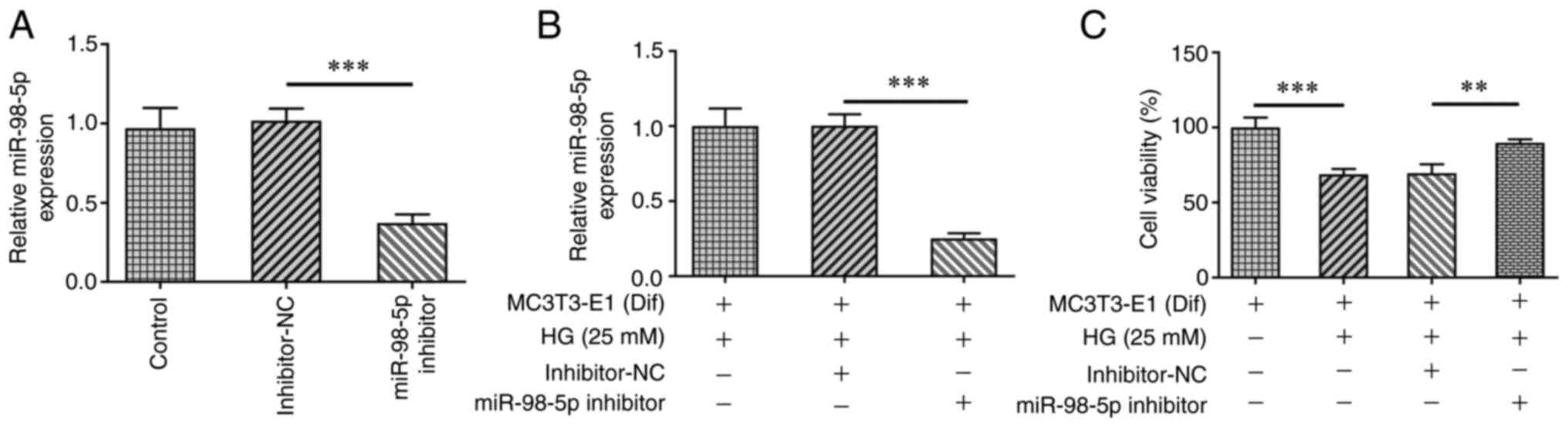
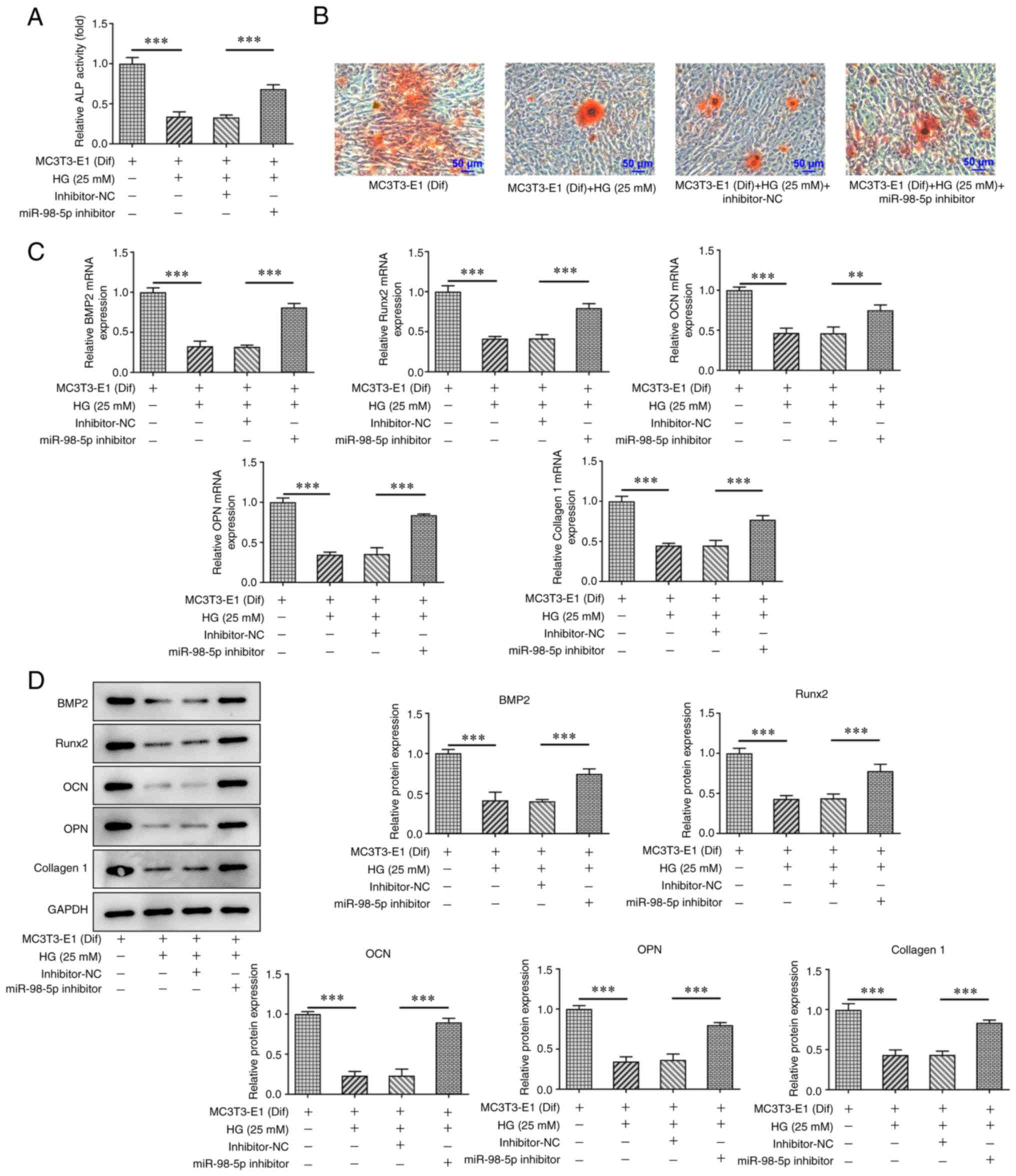
MiR-98-5p promotes osteoblast differentiation in MC3T3-E1 cells by
targeting CKIP-1. Mol Med Rep. 17:4797–4802. 2018.PubMed/NCBI
|
|
21
|
Jafari M, Ghadami E, Dadkhah T and
Akhavan-Niaki H: PI3k/AKT signaling pathway: Erythropoiesis and
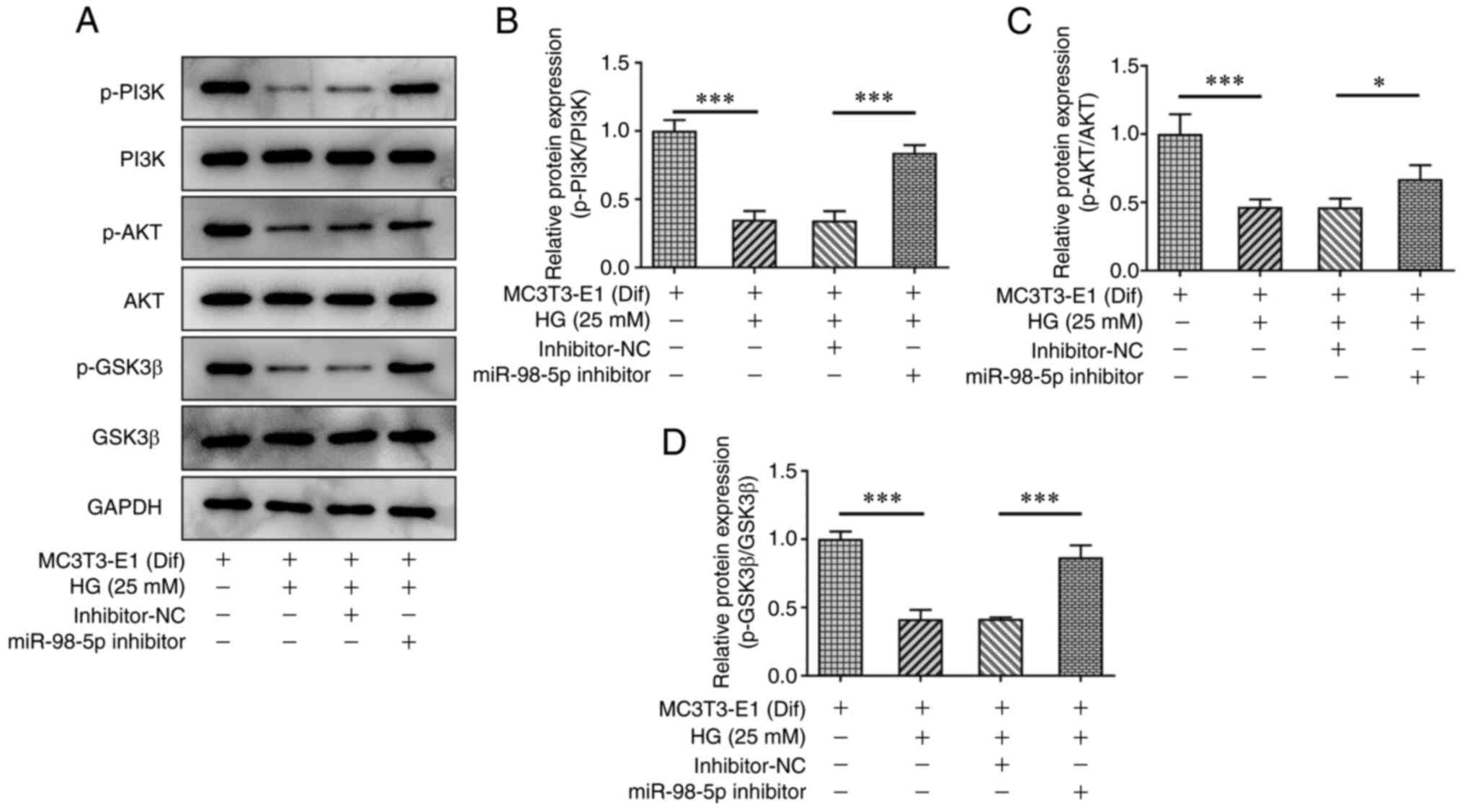
beyond. J Cell Physiol. 234:2373–2385. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xia P and Xu XY: P13K/Akt/mTOR signaling
pathway in cancer stem cells: From basic research to clinical
application. Am J Cancer Res. 5:1602–1609. 2015.PubMed/NCBI
|
|
23
|
Ediriweera MK, Tennekoon KH and Samarakoon
SR: Role of the P13K/AKT/mTOR signaling pathway in ovarian cancer:
Biological and therapeutic significance. Semin Cancer Biol.
59:147–160. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sun K, Luo J, Guo J, Yao X, Jing X and Guo
F: The PI3K/AKT/mTOR signaling pathway in osteoarthritis: A
narrative review. Osteoarthritis Cartilage. 28:400–409. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Method. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tong W, Li J, Feng X, Wang C, Xu Y, He C
and Xu W: Kaiso regulates osteoblast differentiation and
mineralization via the Itga10/P13K/AKT signaling pathway. Int Mol
Med. 47:412021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hou QC, Wang JW, Yuan G, Wang YP, Xu KQ,
Zhang L, Xu XF, Mao WJ and Liu Y: AGEs promote calcification of
HASMCs by mediating P13k/AKT-GSK3β signaling. Front Biosci
(Landmark Ed). 26:125–134. 2021. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tang P, Xiong Q, Ge W and Zhang L: The
role of microRNAs in osteoclasts and osteoporosis. RNA Biol.
11:1355–1363. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu B, Li JY and Cairns MJ: Identifying
miRNAs, targets and functions. Brief Bioinform. 15:1–19. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gamez B, Rodriguez-Carballo E and Ventura
F: MicroRNAs and post-transcriptional regulation of skeletal
development. J Mol Endocrinol. 52:R179–R197. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lian JB, Stein GS, van Wijnen AJ, Stein
JL, Hassan MQ, Gaur T and Zhang Y: MicroRNA control of bone
formation and homeostasis. Nat Rev Endocrinol. 8:212–227. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li H, Xie H, Liu W, Hu R, Huang B, Tan YF,
Xu K, Sheng ZF, Zhou HD, Wu XP and Luo XH: A novel microRNA
targeting HDAC5 regulates osteoblast differentiation in mice and
contributes to primary osteoporosis in humans. J Clin Invest.
119:3666–3677. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kim KM, Park SJ, Jung SH, Kim EJ, Jogeswar
G, Ajita J, Rhee Y, Kim CH and Lim SK: MiR-182 is a negative
regulator of osteoblast proliferation, differentiation, and
skeletogenesis through targeting FoxO1. J Bone Miner Res.
27:1669–1679. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li W, Zhang S, Liu J, Liu Y and Liang Q:
Vitamin K2 stimulates MC3T3E1 osteoblast differentiation and
mineralization through autophagy induction. Mol Med Rep.
19:3676–3684. 2019.PubMed/NCBI
|
|
35
|
Janus JM, O'Shaughnessy RFL, Harwood CA
and Maffucci T: Phosphoinositide 3-Kinase-dependent signalling
pathways in cutaneous squamous cell carcinomas. Cancers (Basel).
9:862017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shen ZH, Hu XQ, Hu MJ, Pan XK, Lu HG, Chen
B, Wu B and Chen G: Activation of AKT signaling via small molecule
natural compound prevents against osteoblast apoptosis and
osteonecrosis of the femoral head. Am J Transl Res. 12:7211–7222.
2020.PubMed/NCBI
|
|
37
|
Ersahin T, Tuncbag N and Cetin-Atalay R:
The PI3K/AKT/mTOR interactive pathway. Mol Biosyst. 11:1946–1954.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhao R, Tao L, Qiu S, Shen L, Tian Y, Gong
Z, Tao ZB and Zhu Y: Melatonin rescues glucocorticoid-induced
inhibition of osteoblast differentiation in MC3T3-E1 cells via the
PI3K/AKT and BMP/Smad signalling pathways. Life Sci.
257:1180442020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ren Q, Zhou J, Wang MG and Chen KM: Pulsed
electromagnetic fields stimulating osteogenic differentiation and
maturation involves primary cilia-PI3K/AKT pathway. Beijing Da Xue
Xue Bao Yi Xue Ban. 51:245–251. 2019.(In Chinese). PubMed/NCBI
|
|
40
|
Price LC, Shao D, Meng C, Perros F,
Garfield BE, Zhu J, Montani D, Dorfmuller P, Humbert M, Adcock IM
and Wort SJ: Dexamethasone induces apoptosis in pulmonary arterial
smooth muscle cells. Respir Res. 16:1142015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Neis VB, Moretti M, Rosa PB, Dalsenter YO,
Werle I, Platt N, Kaufmann FN, Rosado AF, Besen MH and Rodrigues
ALS: The involvement of PI3K/Akt/mTOR/GSK3β signaling pathways in
the antidepressant-like effect of AZD6765. Pharmacol Biochem Behav.
198:1730202020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Donnelly H, Smith CA, Sweeten PE,
Gadegaard N, Meek RD, D'Este M, Mata A, Eglin D and Dalby MJ: Bone
and cartilage differentiation of a single stem cell population
driven by material interface. J Tissue Eng. May 15–2017.(Epub ahead
of print). View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hart CG and Karimi-Abdolrezaee S: Bone
morphogenetic proteins: New insights into their roles and
mechanisms in CNS development, pathology and repair. Exp Neurol.
334:1134552020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lauzon MA, Drevelle O, Daviau A and
Faucheux N: Effects of BMP-9 and BMP-2 on the PI3K/Akt pathway in
MC3T3-E1 preosteoblasts. Tissue Eng Part A. 22:1075–1085. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen G, Deng C and Li YP: TGF-β and BMP
signaling in osteoblast differentiation and bone formation. Int J
Biol Sci. 8:272–288. 2012. View Article : Google Scholar : PubMed/NCBI
|