Diabetic kidney disease (DKD), recognized as a
severe chronic microvascular complication of diabetes, has emerged
as the leading cause of chronic kidney disease, surpassing chronic
glomerulonephritis in incidence (1). Clinically, DKD is characterized by
progressive proteinuria, edema and hypertension. Pathologically, it
affects multiple renal structures, including glomeruli, tubules and
microvessels, ultimately culminating in end-stage renal disease
(2). DKD diagnosis is performed
through biopsy; however, the filtration rate and albumin excretion
rate enable initial identification (3). Notably, accumulating evidence has
underscored pre-biopsy biomarker screening as important for curbing
disease progression (4–6). Therefore, the identification of
important therapeutic targets and molecular biomarkers in
early-stage DKD holds notable clinical implications. Shifting away
from established DKD research models, the present review identified
endoplasmic reticulum (ER) stress (ERS) as a prospective
therapeutic focus, including a systematic dissection of its role in
disease progression and a thorough re-evaluation of herbal
treatment approaches.
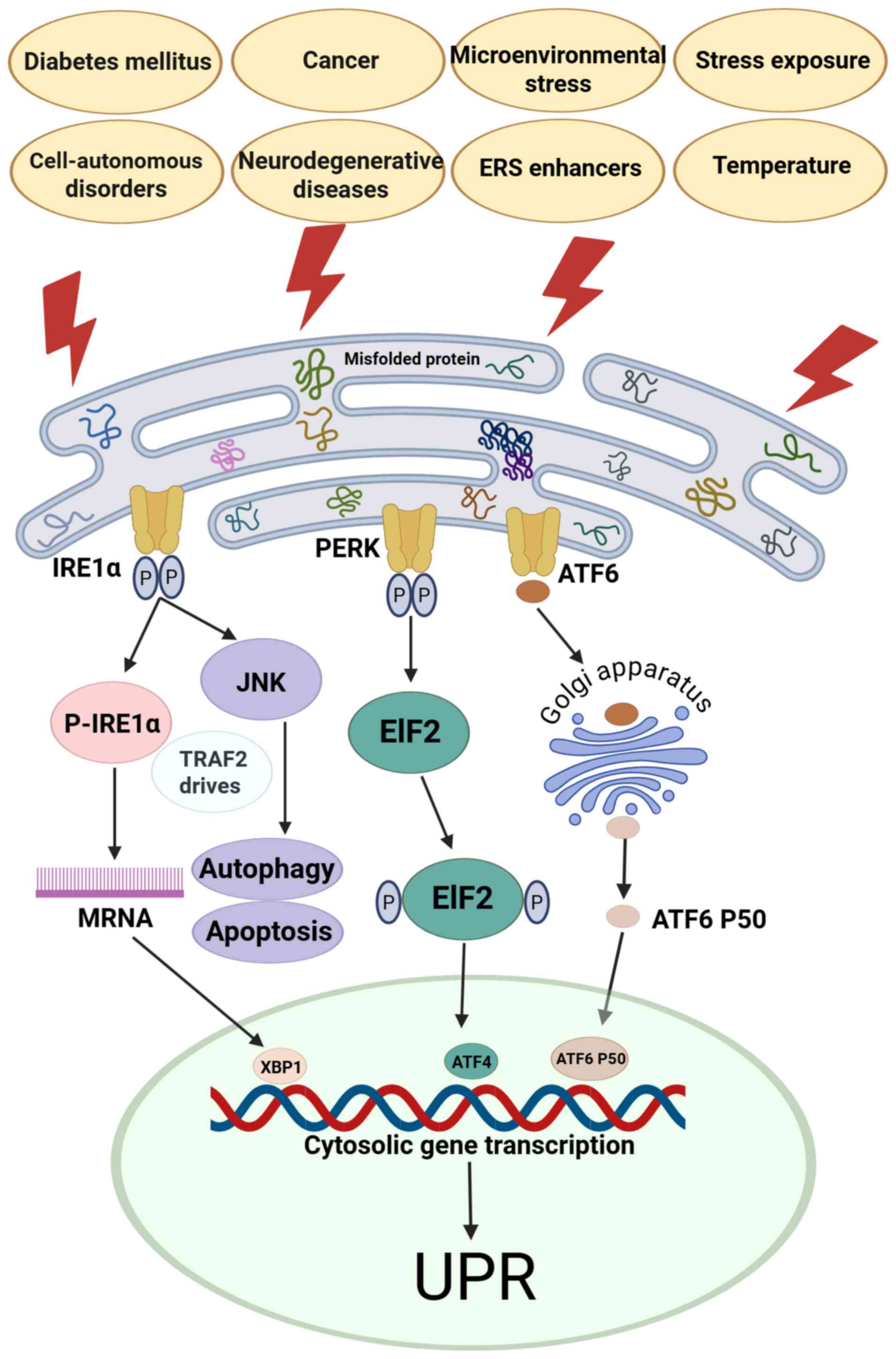
The ER, a notable cellular organelle, orchestrates
polypeptide folding and protein processing, functions that underpin
its roles in calcium storage and regulation, lipid metabolism, and
glucose homeostasis (7). ERS can
be initiated by two distinct mechanisms: i) Endogenous factors such
as cancer and neurodegenerative diseases, which disrupt cellular
processes (8); and ii) exogenous
stressors such as microenvironmental alterations and chemical
insults that induce ERS responses (9). Microenvironmental perturbations and
diverse stressors, including metabolic dysregulation,
cancer-associated stress, drug toxicity and radiation, elicit the
unfolded protein response (UPR) in the ER, a hallmark of ERS
(10). ERS serves as one of the
fundamental mechanisms underlying metabolic dysfunction across
renal organs and tissues. As a trophic signaling hub in metabolic
disorders such as obesity and type 2 diabetes, the ER integrates
inflammation, autophagy and apoptosis pathways, while modulating
cytokine release (11). ERS
induces dynamic changes in the expression of inositol-requiring
enzyme 1α (IRE1α), PKR-like ER kinase (PERK), activating
transcription factor (ATF)6 and glucose-regulated protein
(GRP)78/binding immunoglobulin protein (BiP). Following energy
metabolism fluctuations, these metabolism sensors (IRE1α, PERK and
ATF6) dissociate from GRP78/BiP, triggering increased metabolic
activity and protective responses such as autophagy (10). Failure to restore homeostasis via
translational arrest and chaperone upregulation leads to the
initiation of CHOP-dependent apoptosis, a determinant of stressed
cell fate (2,12).
ERS-related protective responses activate three
stress protein pathways. First, starting with the IRE1α-spliced
X-box binding protein 1 (XBP1s) pathway, phosphorylated (p-)IRE1α
dimerization catalyzes XBP1 mRNA splicing, generating
nuclear-translocated XBP1 that activates ER-associated degradation
(ERAD) or promotes mRNA decay during adaptive UPR. Concurrently,
tumor necrosis factor receptor-associated factor 2 (TRAF2)-mediated
JNK activation contributes to dual-function UPR signaling,
balancing adaptive and apoptotic responses. Second, p-PERK
dimerization in the PERK/eukaryotic initiation factor 2 (eIF2)
pathway drives eIF2 phosphorylation, which enhances ATF4
translation to elicit adaptive UPR. During severe ERS, ATF4 induces
CHOP expression, activating the apoptotic arm of the UPR. Third, in
the ATF6 axis, chaperone dissociation of GRP78/BiP promotes Golgi
trafficking, where proteolytic processing generates ATF6 p50. This
nuclear effector drives the adaptive UPR via chaperone and foldase
transcription or the apoptotic UPR via ERAD activation (13).
Collectively, the IRE1α/XBP1s, PERK/eIF2 and ATF6
pathways balance ERS-induced survival and apoptosis through UPR
modulation: Moderate UPR restores homeostasis, while persistent ERS
activates apoptotic signals such as caspase-12, leading to cell
death (Fig. 1) (8–14).
Preclinical and clinical investigations have
consistently revealed ERS marker upregulation in renal tissues of
patients with DKD and relevant animal models (15–17).
Pathological changes in DKD tend to start early with thickening of
the basement membrane, gradually spreading to glomeruli,
microvessels and tubules. By contrast, changes in glomerular
hyaluronan appear in the late stages of DKD (18,19).
Persistent urinary protein abnormalities stem predominantly from
four sources: i) Renal filtration barrier defects, including those
affecting the basement membrane, podocytes and associated cells
(20); ii) damage across renal
tubular segments (21); iii) renal
interstitial functional impairment (22); and iv) immune cell population
dysregulation (23). In
conclusion, renal histopathology reveals that ERS impacts DKD in
three notable manners: Glomerular filtration membrane injury, renal
tubular reabsorptive dysfunction and renal interstitial remodeling
with immune cell dysregulation (24).
Studies in DKD model mice have revealed that ERS
contributes to glomerular injury, as demonstrated by increased
GRP78/caspase-12 expression and reduced Bcl-2 levels (25–27)
Notably, genetic deletion of ER protein 44 in db/db mice
upregulated ATF6, XBP1 and CHOP, further indicating ERS involvement
in glomerular filtration membrane injury (28). Composed of endothelial cells,
basement membrane and podocytes, the glomerular filtration membrane
is important for blood filtration. Damage to these components
drives early DKD progression and proteinuria (27). Surface-bound GRP78 drives
extracellular matrix (ECM) accumulation through PI3K/Akt
activation, while the AMP-activated protein kinase (AMPK)/mTOR
pathway promotes late-stage PERK/CHOP upregulation to enhance
autophagy, and the IRE1/NF-κB pathway mediates ERS-induced
inflammation in podocytes (29–31).
Compared with individuals without DKD, renal biopsy analyses of
patients with DKD have revealed increased GRP78 and caspase-12
expression in human glomerular podocytes (28,32).
The elevation of ATF6 and PERK in human podocytes triggers lipid
metabolism disorders and inflammation, which are suppressed by
JNK-mediated insulin activity (33). Mechanistically, the high
glucose-induced elevation of PERK, ATF6 and IRE1α in rat podocytes
is linked to protein arginine methyltransferase 1 (PRMT1) (34). Concurrently, the activation of
XBP1s in glomerular mesangial cells triggers histone-lysine
N-methyltransferase SETD7-mediated histone methylation, leading to
the upregulation of DKD-associated monocyte chemoattractant protein
1 (35,36). These findings indicate that ERS
influences the expression of UPR-induced transcription factors,
which in turn contribute to fibrotic and inflammatory pathological
changes, thereby regulating the balance between adaptive and
apoptotic states in podocytes.
The depletion of terminally differentiated
podocytes, which form the glomerular filtration barrier, is
characteristic of chronic kidney diseases, including DKD. Numerous
studies have revealed an association between ERS and podocyte
injury in DKD, as ERS markers are upregulated in human and murine
podocytes exposed to high glucose; this process is induced by the
dysregulation of hyperglycemia, lipid metabolism and insulin
signaling (37,38). In mouse podocytes, the accumulation
of advanced glycation end-products (AGEs) and high-glucose exposure
activate GRP78, CHOP and caspase-12 through distinct pathways,
ultimately driving podocyte apoptotic processes (39). PERK-eIF2α activation induces
CHOP-mediated podocyte apoptosis, and PERK phosphorylation triggers
a CHOP-reticulon 1A (RTN1A) positive feedback loop for synergistic
apoptotic enhancement (40).
Dysfunction in the insulin PI3K/Akt pathway may underlie
GRP78/CHOP-mediated ERS and podocyte apoptosis. Reduced activity of
PTEN, a downstream inhibitory regulator, exacerbates ERS, while
MEK/ERK signaling counteracts this pathological effect (41). The GRP78-mediated modulation of the
MEK kinase 1 (MEKK1)/JNK signaling cascade induces podocyte
apoptosis through MEKK1-dependent Ser280 phosphorylation (42).
The ERAD pathway is a specialized mechanism within
the ER that employs the ubiquitin-proteasome system to degrade
misfolded or aberrantly modified proteins, serving an important
role in eliminating these faulty proteins from cells (43). Derlin-2 expression is elevated in
podocytes from patients with DKD biopsies, triggering ERAD to
sustain the cellular balance (44,45).
Similarly, ERS serves an important role in balancing podocyte
survival and apoptosis through autophagy, given that compared with
DKD, podocytes inherently exhibit a higher basal autophagic
activity (46) High glucose levels
increase eIF2α, CHOP, caspase-3/12, GRP78, ATF6 and PERK levels in
podocytes through the inhibition of autophagy, whereas
sarcoendoplasmic reticulum calcium-ATPase (SERCA)2b-mediated ERS
attenuation is associated with AMPKα-induced autophagy (46,47).
The intercellular transmission of ERS signals, coupled with
podocyte-intraglomerular cell interactions, suppresses ERAD through
derlin-2 downregulation, inducing podocyte apoptosis in DKD
(48). Additional evidence has
shown that GRP78/CHOP activation in endothelial cells drives
epithelial-mesenchymal transition (EMT) (49), implying a potential role for
glomerular endothelial cells in DKD pathogenesis, although the
underlying mechanisms remain ambiguous.
UPR-activated transcription factors drive fibrosis
and inflammation in glomerular podocytes at the early stage of
diabetic kidney injury. Three ERS pathways collectively influence
disease progression, including: i) Regulation of podocyte apoptosis
through the PI3K/Akt, PTEN and MEK/ERK signaling pathways; ii)
maintenance of cellular homeostasis through the activation of ERAD;
and iii) modulation of podocyte autophagy via AMPK (50,51).
In addition, key ERS factors also contribute to the DKD process by
affecting intraglomerular mesangial cells and podocytes (52).
Composed of Henle's loop, proximal and distal
tubules and collecting ducts, renal tubules reabsorb small
molecules and lipids, with their epithelium serving as a key
DKD-targeted cell type susceptible to injury (53,54).
Extensive research has established that ERS contributes to
DKD-related renal tubular injury, as evidenced by the upregulation
of differential ERS markers in human biopsies, animal tissues and
in vitro models (15,55–57).
This process is driven by the AGE/receptor for AGEs (RAGE) axis,
PRMT1 and histone H4 deacetylation (15,58,59),
while it is alleviated by calbindin-D28k, transmembrane BAX
inhibitor motif-containing protein 6, sestrin 2 and dapagliflozin
(60,61). Renal tubular ERS markers (notably
including GRP78) can modulate the autophagic processes and drive
inflammatory responses by regulating mitochondrial function and
reactive oxygen species (ROS) generation (62,63).
Saturated fatty acid-associated factors, such as
carbohydrate-responsive element-binding protein, cholesterol and
palmitic acid (PA), promote ERS marker upregulation in renal
tubular cells, causing lipid accumulation and apoptotic cell death
(64,65). Therefore, the comprehensive role of
ERS in renal tubules is closely intertwined with autophagy,
inflammation and lipid peroxidation.
In the renal tubular system, the proximal tubule
reabsorbs glucose, amino acids, HCO3−,
Cl− and Na+, while secreting H+.
By contrast, the distal tubule and collecting ducts mainly mediate
Na+ and water reabsorption alongside K+
secretion. The proximal and distal tubule of the kidney functional
distinction partially explain the structural variations in ERS
signaling, with GRP78-governed ATF4/p16/p21 axes promoting
AGE/RAGE-driven premature cellular senescence in mouse proximal
tubular cells (66). GRP78 and
protein disulfide isomerase exhibit distinct regulatory dynamics
between proximal and distal tubules, varying across early and late
stages of diabetes-induced ERS. Early-stage diabetes triggers PERK
activation, while late-stage disease promotes ATF6 activation in
renal tubules, resulting in stage-dependent differential ERS
responses between proximal and distal segments in DKD (67).
ERS notably regulates renal cell function by
suppressing inflammation, inhibiting abnormal proliferation and
fibrosis, and combating oxidative damage through the precise
modulation of DKD pathways, including the PERK/CHOP and caspase-12
pathways (53,68,69).
The mechanistic exploration of ERS in DKD pathway regulation sheds
light on DKD pathogenesis and facilitates the identification of
novel therapeutic targets (Fig.
2).
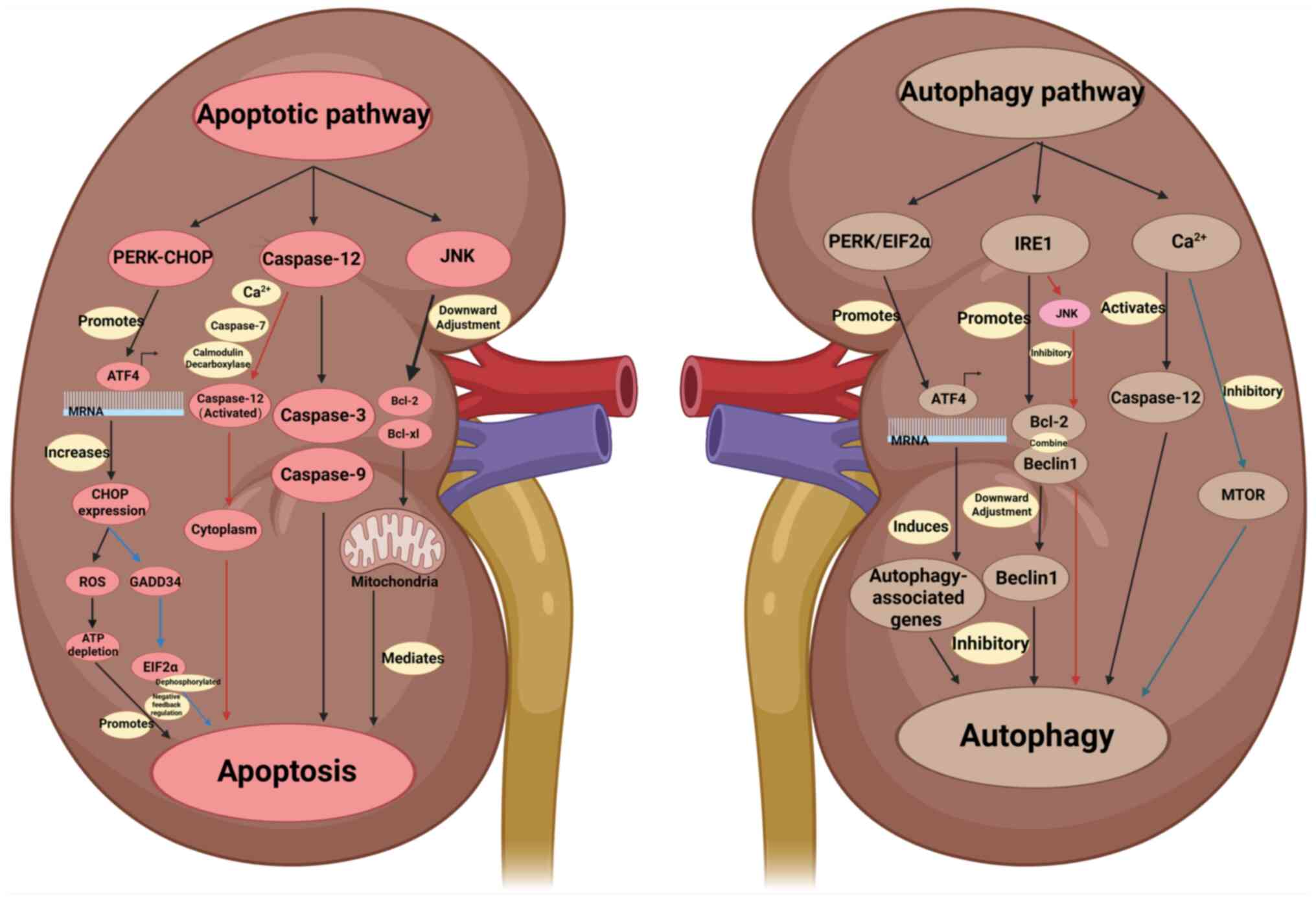
The PERK/CHOP pathway regulates ERS and contributes
to DKD. The core aspects of the PERK/CHOP pathway comprise the
following mechanisms: Initially, ERS is triggered when high glucose
levels, oxidative stress and inflammation lead to misfolded protein
accumulation in the ER, prompting GRP78/BiP dissociation from PERK
and subsequent kinase activation (70). Subsequently, the PERK/eIF2α/ATF4
axis is activated; PERK phosphorylates eIF2α, globally inhibiting
translation to reduce ERS burden. Simultaneously, p-eIF2α
selectively promotes ATF4 translation, leading to CHOP expression
(71) Finally, CHOP-mediated
apoptotic signaling occurs (72)
CHOP, a transcription factor, upregulates pro-apoptotic genes, such
as Bcl-2-interacting mediator of cell death (Bim), p53 upregulated
modulator of apoptosis and Bax, and downregulates Bcl-2, ultimately
inducing apoptosis (73).
The PERK/CHOP pathway mediates DKD podocyte injury
through high glucose-activated PERK/eIF2α/ATF4/CHOP signaling,
inducing apoptosis, foot process effacement, proteinuria and
glomerulosclerosis, which are exacerbated by pathway
hyperactivation (74). The
PERK/CHOP pathway drives mesangial cell proliferation, fibrosis and
ECM deposition, mediated via pathways such as the TGF-β1/connective
tissue growth factor pathway, in DKD, additionally serving as a
central mediator of proteinuria and glomerulosclerosis (75). In DKD, the CHOP pathway mediates
renal tubular injury. High glucose-induced ERS promotes tubular
apoptosis through the PERK/CHOP axis and activates pro-inflammatory
cytokines such as IL-6 and TNF-α to exacerbate renal interstitial
inflammation (68). Renal
tubulointerstitial fibrosis is an important step in the progression
of DKD to end-stage renal disease. Sustained PERK activation
enhances ATF4 transcription to upregulate CHOP, which downregulates
Bcl-2 and promotes apoptosis through ROS production and ATP
depletion (76). Concurrently,
CHOP triggers growth arrest and DNA-damage-inducible 34 activation
to facilitate eIF2α dephosphorylation, a negative feedback
mechanism that restores transiently-arrested protein synthesis in
the PERK pathway (77).
The PERK/CHOP regulatory axis balances adaptive and
apoptotic responses to ERS through integrated positive and negative
feedback mechanisms (78). PERK
and its downstream effector CHOP exhibit segment-specific
regulation across different regions of the renal tubule. Renal
tubular cells secrete CHOP-induced fibronectin, driving mesenchymal
fibrosis in DKD, a process implying that PERK/CHOP signaling
controls the synthesis and release of ECM proteins in these cells
(79). The RTN1A/PERK/CHOP
positive feedback loop exacerbates ERS-induced podocyte apoptosis,
highlighting the dual role of the PERK/CHOP pathway in maintaining
ERS balance and promoting pathological cell death (80).
Caspase-12 acts as a key executor of apoptotic
signaling, with its activation closely linked to ERS-induced cell
death (81). Upon ER misfolded
protein accumulation, for example, due to high blood glucose
levels, oxidative stress or calcium dyshomeostasis, GRP78/BiP
dissociates from ER membrane sensors such as PERK, IRE1 and ATF6 to
bind misfolded proteins, triggering the activation of caspase-12 as
a downstream responder to ERS signals (82,83).
In non-stressed cells, GRP78 associates with pro-caspase-12 to
suppress its activity. During ERS, GRP78 dissociation exposes the
active site for proteolytic processing by upstream enzymes,
including IRE1α and calpain, generating active caspase-12 (p35/p12
subunits) that cleaves caspase-3 to initiate apoptosis (84,85).
In patients with DKD, high glucose induces ERS,
which activates caspase-12 and promotes the apoptosis of podocytes
and tubular epithelial cells, accelerating proteinuria and renal
interstitial fibrosis (86). In
the DKD mouse model, CHOP overexpression upregulates caspase-12 to
promote apoptosis, whereas caspase-12 inhibition attenuates high
glucose-induced renal cell injury (87). ERS also induces calcium
dyshomeostasis and mitochondrial calcium overload, indirectly
activating the mitochondrial apoptotic pathway via the
ER-mitochondrial axis (88).
ER-released Ca2+ activates peripheral calpains,
prompting their translocation to the cytoplasm. Calpain activation
results in translocation to the ER outer membrane, triggering
caspase-7 translocation and activation (87,89).
Both factors process pro-caspase-12 into an active form that
translocates to the cytoplasm, activates caspase-9 and −3 through
the cytochrome c (CytC)-independent pathway and induces
apoptosis (90). In addition, the
IRE1α-TRAF2 complex activates caspase-12 through calmodulin
decarboxylase, thereby initiating the caspase-mediated apoptotic
pathway (91).
Caspase-12-dependent signaling contributes to severe podocyte and
tubular cell injury in DKD. Therapeutic targeting of this pathway
mitigates ERS-mediated apoptosis, slowing DKD progression (92,93).
Therefore, caspase-12 signaling is important for ERS-induced
apoptosis. Despite genetic polymorphisms limiting its activity in
humans, caspase-12 retains diagnostic and therapeutic value for ERS
(94). Targeting the caspase-12
pathway may provide novel targets for the treatment of DKD.
JNK, originally identified in its capacity to
phosphorylate the amino terminus of the transcription factor c-Jun
(95), is also referred to as
stress-activated protein kinase. Extracellular stimuli, such as
stress signals, cytokines and growth factors, activate JNK, with
the activation relying on the cellular microenvironment. JNK
inhibition reduces podocyte apoptosis and matrix deposition, and
combination with PERK/CHOP inhibitors may enhance therapeutic
efficacy (96,97). As an important branch of the MAPK
cascade, the JNK pathway orchestrates multiple physiological
processes (98). During ERS,
activated IRE1α engages TRAF2 to trigger apoptosis
signal-regulating kinase 1 (ASK1) activation, generating a trimeric
complex that sequentially activates JNK, CHOP and pro-apoptotic
factors such as BH3-interacting domain death agonist and Bim, a
process that suppresses anti-apoptotic genes, including Bcl-2 and
Bcl-xL, and initiates mitochondrial apoptotic pathways (99–102). In addition, phosphorylation at
the MEKK1 Ser280 residue activates the JNK signaling pathway,
thereby promoting ERS-induced podocyte apoptosis (38,103). High glucose triggers JNK
activation in renal cells, inducing apoptosis and fibrosis. These
processes are alleviated by JNK inhibitors, which reduce
CHOP/caspase-3-dependent apoptosis in ERS-impaired podocytes to
mitigate DKD-related renal injury (33). Acting as a central stress response
hub, the JNK pathway integrates external stimuli with intracellular
signaling to govern inflammation and disease progression, with
functional complexity emerging from stimulus subtype specificity,
stimulus intensity and cellular context (104,105). Despite the challenges of drug
development targeting JNK (such as poor specificity and high
concentrations required to inhibit c-Jun phosphorylation), its
potential applications in areas such as DKD and neurodegenerative
diseases make it a promising therapeutic target (106–109).
The PERK/eIF2α signaling axis, a key branch of ERS
signaling, maintains intracellular proteostasis by regulating
protein synthesis and gene expression (110). Following the accumulation of
misfolded or unfolded proteins in the ER, this pathway is activated
to promote either stress adaptation or apoptosis through global
translation inhibition and induction of stress-responsive genes
(111). The key component of this
pathway, PERK, is an ER transmembrane protein with a kinase domain
on the cytoplasmic side, while only its stress-sensing domain is
present within the lumen of the ER) (112). In unstressed cells, PERK stays
inactive by associating with the ER chaperone BiP that has
dissociated from GRP78 (113). By
contrast, eIF2α is a key factor in the initiation of protein
translation, and phosphorylation at its Ser51 site is a central
event in the activation of the PERK/eIF2α pathway. This pathway
coordinates cellular homeostasis through multifaceted mechanisms,
such as translation repression for energy conservation, amino acid
transporter induction for survival and proteostasis regulation
through reduced synthesis, enhanced folding through mechanisms such
as BiP upregulation, or misfolded protein degradation, for example,
via ERAD (72).
The PERK/eIF2α branch of the UPR signaling pathway
induces autophagy-associated gene expression through the
transcriptional regulation of ATF4, thereby promoting the
transcription of autophagy-associated genes (114). Downstream genetic effectors
include beclin-1, which mediates autophagosome biogenesis and
maturation, the ubiquitin-like system and transport receptor genes
such as p62 and next to BRCA1 gene 1 protein (115). These transport receptor genes
undergo selective degradation via ubiquitinated cargoes. Vaspin, a
serine protease inhibitor, suppresses p62 aggregation by forming
complexes with GRP78 and heat shock 70 kDa protein 1L (116,117). This mechanism protects organelles
from metabolic stress-induced damage through endocytosis-driven
autophagy facilitation, maintaining cellular homeostasis under
stress (118). The PERK/eIF2α
pathway is a ‘regulatory hub’ for ERS through precise regulation at
the translational level and reprogramming of gene expression to
maintain a balance between cell adaptation and death (119).
The IRE1 signaling cascade acts as a core ERS
effector, governing adaptive or apoptotic cell fates predominantly
by modulating mRNA splicing and gene transcription (120,121). The pathway is highly
evolutionarily conserved from yeast to mammals and is a key
mechanism for maintaining ER protein homeostasis. The IRE1/XBP1
axis preserves cellular homeostasis during acute stress by boosting
protein folding, degradation and transport, while governing the
activity of secretory cells, such as plasma and pancreatic β-cells,
for efficient antibody and insulin production (122,123). Conversely, chronic ERS in
pancreatic β-cells engages the IRE1/JNK pathway, causing β-cell
death and diminishing insulin release (124,125).
The IRE1 pathway engages in crosstalk with the other
two UPR arms: The PERK/eIF2α and ATF6 pathways. The PERK branch
suppresses global translation through eIF2α phosphorylation to
reduce protein synthesis load, while ATF6 activates BiP, ERAD and
other chaperone genes through nuclear translocation (126,127). The IRE1 pathway promotes protein
folding and degradation through XBP1 splicing, concurrently
regulating cell fate through JNK activation and the regulated
IRE1-dependent decay (RIDD) pathway (111,128). Collectively, these three pathways
dictate cellular outcomes during stress, promoting survival under
acute stress conditions and triggering apoptosis under prolonged or
severe stress (129). In the ERS
state, IRE1/XBP1 signaling directly promotes the expression of
Bcl-2, increases the binding of Bcl-2 to beclin-1 and downregulates
the activity of beclin-1, thus inhibiting autophagy (2). During early ERS, IRE1α assembles with
TRAF2 and ASK1 to activate the downstream JNK signaling cascade,
which phosphorylates Bcl-2 to promote its dissociation from
beclin-1 and encourages subsequent autophagy initiation (92,93).
In addition, XBP1, a downstream effector of IRE1α, promotes
autophagy through beclin-1 activation (43). Blocking the kinase and RNase
activities of IRE1α has been shown to intensify PA-induced
cytotoxicity in renal tubular cells, indicating that IRE1α protects
against PA-mediated kidney injury in DKD (130). The IRE1 signaling pathway is a
‘multifunctional hub’ of the ERS response, responsible for shutting
down and splicing of the mRNA that encodes secretory proteins in a
process called regulated RIDD to decrease ER protein load and
spliced mRNA of XBP1 that activated the transcription of ER
hemostatic factors as chaperone and endoplasmic
reticulum-associated degradation components that degraded unfolded
and misfolded proteins in an attempt to resolve endoplasmic
reticulum stress; if this stress was unresolved, XBP1 upregulated
the expression of JNK that activated podocytes, tubular apoptosis,
and inflammation (131), and its
aberrant activation is closely associated with a variety of
diseases(such as Sepsis, DKD, breast cancer et.) (131–133).
Calcium channels are the core molecular machinery
regulating the intracellular flow of Ca2+ across the
membrane (129). DKD marked by
glomerular mesangial matrix dilation, basement membrane thickening,
tubulointerstitial fibrosis and renal unit loss involves
Ca2+ as a key signaling molecule; calcium homeostatic
imbalance markedly drives disease pathogenesis (134,135). The ER functions as a primary
intracellular Ca2+ store, regulating calcium homeostasis
(136). During stress,
Ca2+ influx sustains cellular homeostasis by activating
the MAPK pathway, thereby promoting intracellular protein folding
(137,138). Excessive release of
Ca2+ into the cytoplasm initiates ERS, thereby
activating caspase-12-dependent autophagy (139). The transient ER surface calcium
concentration is a key signal that determines the activation of ER
autophagy (140). In addition,
ATP and other Ca2+ mobilizers antagonize mTOR-mediated
autophagy inhibition via Ca2+-dependent pathways,
thereby promoting beclin-1- or autophagy-related gene 7-dependent
autophagosome biogenesis (141,142). In addition, Ca2+ can
activate both protein kinase C (PKC) and death-associated protein
kinase (DAPK). PKC induces autophagy through an mTOR-independent
pathway, whereas DAPK promotes beclin-1 phosphorylation and
disrupts the beclin-1/Bcl-2 complex, thereby activating autophagy
(143,144). The ERS-associated inhibition of
PERK and IRE1 protects against kidney injury, mitochondrial
dysfunction and renal cell apoptosis by maintaining Ca2+
homeostasis, which blocks cytosolic release of CytC and
apoptosis-inducing factor, inhibits caspase-9 activation and
restores mitochondrial membrane integrity, thereby suppressing
pathological cell death pathways (145). Calcium channels are ‘signaling
hubs’ for cellular functions, and their diversity and sophisticated
regulatory mechanisms serve key roles in physiological and
pathological processes (129,134).
Research on AS modulation of DKD through ERS
intervention has primarily focused on DKD rodent models and
meta-analyses (26,47,150–152). In DKD rat models (47,150) induced by a high-fat diet combined
with streptozotocin (STZ), intragastric administration of AS-IV
effectively mitigated apoptosis in renal tubular epithelial cells.
This protective effect involved two primary mechanisms: i)
Downregulation of ERS-associated proteins, including p-PERK, ATF4
and CHOP; and ii) restoration of the homeostatic balance between
the pro-apoptotic protein Bax and anti-apoptotic protein Bcl-2
(47,150). In STZ-induced DKD rats (26,151,152), AS-IV alleviated ERS-driven
podocyte apoptosis through three coordinated pathways: i)
Suppression of the PERK/ATF4/CHOP signaling cascade; ii) inhibition
of oxysterol-binding protein-related protein 150 (ORP150) and GRP78
expression; and iii) reduction of phosphorylation levels of PERK,
eIF2α and JNK (26,151). Similarly, in db/db mice and
podocytes exposed to high glucose or PA, two important pathological
stimuli in DKD, AS-IV mitigated ERS and reduced podocyte apoptosis
by restoring SERCA2 expression and activity (152). Collectively, AS-IV exerted
anti-ERS effects through a multi-targeted strategy: Blocking three
important ERS signaling pathways, including the PERK/ATF4/CHOP,
PERK/eIF2α and IRE1/JNK pathways, increasing SERCA2 expression and
decreasing ORP150 and GRP78 expression (Table I) (26,151,152).
Huangkui is a TCM approved by the Chinese National
FDA for nephritis treatment. Its therapeutic effect on DKD is
primarily mediated by active ingredients, particularly flavonoid
derivatives such as quercetin glycosides and kaempferol glycosides.
These components intervene in the core pathological mechanisms of
DKD, including ERS, oxidative stress, the inflammatory response,
apoptosis and renal interstitial fibrosis, thereby forming a
multi-dimensional protective network (159–164). Huangkui (158,159) can restore ER protein homeostasis:
The upregulation of molecular chaperone proteins GRP78 and GRP94
promotes correct protein folding, enhances ERAD of misfolded
proteins, and attenuates ER loading (165).
In summary, Huangkui capsules exert protective
effects against both glomerular and tubular injuries by regulating
multiple mechanisms, including signal transduction pathways,
nuclear receptor activities, inflammatory responses, mitochondrial
function and gut microbiota, thus providing novel drugs and
approaches for the treatment of kidney disease. Ge et al
(33) demonstrated that Huangkui
ameliorated renal damage by attenuating ERS in DKD model rats.
Through its multi-targeted mechanism, encompassing ERS inhibition,
and antioxidant, anti-inflammatory, antifibrotic and
podocyte-protective effects, Huangkui mediates multi-level
regulatory effects in DKD progression, with notable promise in
proteinuria control and delaying renal interstitial fibrosis
(33,171,172). The natural medicinal properties
of Huangkui provide a novel strategy for early intervention in DKD,
and the combination of Chinese and Western medicine, which is
particularly suitable for the long-term management of patients with
mild to moderate proteinuria (170)
OA, as a monounsaturated fatty acid, serves an
important role in the development of DKD, and its involvement in
kidney injury through the regulation of ERS has attracted
increasing attention. OA is naturally found in fruits and
vegetables. Podocytes, early DKD targets, exhibit dose-dependent OA
responses: Low-dose OA mitigates injury, while high-dose OA
triggers ERS-PERK-mediated apoptosis, impairing podocyte protein
synthesis and filtration barrier integrity. Mechanistically, CHOP
upregulation inhibits Bcl-2, activating mitochondrial apoptotic
cascades, such as the caspase-3-mediated cascade (173,174). OA also exerts context-dependent
effects on tubular fibrosis: Low ERS inhibits fibrosis, while
OA-induced ERS activates ATF6/CHOP to upregulate TGF-β1/α-smooth
muscle actin (α-SMA), driving EMT (175). Excessive OA drives mesangial cell
proliferation and ECM accumulation through ERS-hyperactivated mTOR
signaling, contributing to glomerulosclerosis (176). This underscores OA concentration
modulation as a promising DKD therapeutic strategy.
In preclinical models, OlA exhibits antioxidant,
antiglycation, anti-inflammatory and bactericidal activity, notably
ameliorating DKD by reducing ERS. In diabetic rats, OlA treatment
enhances the renal protein expression of p-AMPK/AMPK and peroxisome
proliferator-activated receptor γ coactivator 1-α (PGC-1α), while
concurrently decreasing the expression of CD68, collagen-IV, TLR4,
NF-κB and TGF-β1 (177). By
regulating lipid metabolism and inflammation through the
AMPK/PGC-1α and TLR4/NF-κB pathways, OlA mitigates renal injury in
diabetic rats. In DKD model mice, OlA administration notably
reduces the ALB-creatinine ratio (ACR). The marked DKD-induced
elevation in ERS markers, such as p-PERK, p-eIF2α, ATF-6, BiP and
CHOP, was notably decreased following OlA treatment, accompanied by
marked reductions in ROS and nuclear factor erythroid 2-related
factor 2 levels in glomerular mesangial cells (177,178). Furthermore, OlA administration
reduces TGF-β/Smad2/3 signaling and α-SMA expression, repairs renal
damage, alleviates albuminuria and suppresses diabetes-induced
renal fibrosis by inhibiting ERS and apoptosis (179). In combination with
N-acetylcysteine, OlA further attenuates oxidative stress and
reduces the ERS-induced activation of TGF-β/Smad2/3 signaling
(180).
Quercetin, a widely distributed natural polyphenol,
exhibits potent antioxidant, anti-inflammatory and multi-target
regulatory effects, making it a focal point of interdisciplinary
research in disease prevention (181,182). Quercetin exhibits efficacy in
treating metabolic diseases, including diabetes and its
complications. Quercetin enhances cell-mediated immunity by
upregulating IFN-γ secretion from Th1 lymphocytes and reduces
inflammation by downregulating IL-4 secretion from Th2 lymphocytes
(183). The protective effects of
quercetin are mediated through multiple mechanisms, including
inhibition of ROS production and mitochondrial permeability
transition-pore opening, suppression of lipid peroxidation,
elevation of glutathione and superoxide dismutase levels (184), inhibition of NF-κB, TNF-α and
IL-6 signaling (185), reduction
of inflammatory mediator release, suppression of
angiotensin-converting enzyme and NF-κB activity, and enhancement
of endothelial-dependent vasodilation (184,185). These actions collectively
regulate blood pressure and reduce urinary protein excretion.
Quercetin may improve glucose homeostasis by
modulating the expression of the microRNA-29 family, thereby
increasing glucose transporters and insulin-like growth factor 1
gene expression, ultimately mitigating diabetic complications
(186). In STZ-induced diabetic
male Wistar rats, quercetin exhibited hypoglycemic effects,
restored pancreatic morphology and β-cell function, reduced ERS
markers such as CHOP and endothelin-1, inhibited lipid
peroxidation, and enhanced antioxidant enzyme activities (183,187). Consequently, it lowered blood
glucose levels, protected pancreatic tissues, alleviated ERS and
improved oxidative status. Quercetin also inhibits platelet
aggregation, hypertension and lipid peroxidation (188). In metabolic disorders, quercetin
ameliorates diabetic endothelial dysfunction by targeting ERS,
highlighting its role in metabolic-endothelial crosstalk (189).
As metabolic disease therapy, the renin-angiotensin
system (RAS) inhibitors aliskiren (193,194) and valsartan (195,196) reduce DKD proteinuria by lowering
glomerular pressure, with evidence showing that ERS pathway
regulation contributes to their renoprotective effects in
RAS-driven DKD.
ACEIs and ARBs such as aliskiren and valsartan
inhibit renal ERS in DKD, and dual therapy yields cumulative
renoprotective effects via synergistic RAS blockade and ERS control
(193). Valsartan is recommended
by several guidelines, including the Kidney Disease: Improving
Global Outcomes (KDIGO) guidelines (197), as the preferred ARB for DKD with
hypertension, and its nephroprotective effect is partly attributed
to ERS modulation. The clinical use of aliskiren is currently
limited due to early clinical trials, such as the ALTITUDE study
(72,198), showing an increased risk of
hyperkalemia and renal injury when used in combination with an ARB,
but its single-agent modulation of ERS remains worth studying
(199–201). Aliskiren and valsartan exert
anti-apoptotic, anti-inflammatory and anti-fibrotic effects in DKD
by inhibiting RAS overactivation and modulating upstream or
downstream ERS. Valsartan, as a classical ARB, has been clinically
validated as an ERS regulator, particularly in the context of DKD
treatment. However, the ERS-targeting effect of aliskiren (a direct
renin inhibitor) remains to be further evaluated and balanced in
terms of its efficacy and safety (193). Basic research has confirmed that
angiotensin II exacerbates ERS in renal tubular epithelial cells by
activating the PERK/eIF2α/CHOP pathway, while ACEIs and ARBs
downregulate stress markers such as GRP78 and CHOP in renal tissue
(196,202). However, clinical studies have yet
to incorporate assessments of ERS-related biomarkers, thus direct
clinical evidence of ACEI and ARB exerting effects via inhibition
of ERS is lacking (193,196). The 2024 KDIGO guidelines
recommend RAS inhibitors for patients with DKD with proteinuria,
emphasizing blood pressure and proteinuria management but omitting
the discussion of ERS-related mechanisms (202).
CB1R, a notable cannabinoid receptor, is widely
distributed in renal vascular endothelial, mesangial, tubular and
immune cells (203,204). In DKD, hyperglycemia, oxidative
stress and inflammation activate CB1R, inducing renal inflammation,
fibrosis and metabolic dysfunction, including insulin resistance
(205–207). CB1R antagonists, such as
rimonabant and AM251, have been shown to improve glucose-lipid
metabolism in metabolic disorders through the blockade of CB1R
signaling, and their protective mechanism against DKD has been
demonstrated to be closely associated with modulation of the ERS
pathway (208,209). The protective effect of CB1R
inhibition in DKD is linked to ERS pathway modulation, with CB1R,
abundantly expressed in diabetic rats kidneys, mediating ERS and
apoptosis in rat mesangial Cells) under high glucose. CB1R
regulates palmitic acid-induced apoptosis in human renal proximal
tubular cells through the ERS pathway (210,211). CB1R antagonists are not
recommended in DKD guidelines; rimonabant has been withdrawn
globally due to severe psychiatric side effects such as depression
and anxiety (212). Subsequent
candidate CB1R antagonists, such as AM6545, remain in preclinical
research for DKD and have not yet been incorporated into guidelines
(213,214).
Endogenous cannabinoids activating CB1R may serve a
notable role in the pathogenesis of diabetic cardiomyopathy by
promoting MAPK activation, type-1 angiotensin II receptor
expression and signaling, AGE accumulation, oxidative and
nitrosative stress, inflammation, and fibrosis (215,216). Conversely, CB1R antagonists exert
anti-diabetic effects by increasing pancreatic β-cell glucokinase
and glucose transporter (GLUT)-2 expression, improving β-cell
insulin signaling and proliferation (for example, mitigating β-cell
loss in male Zucker diabetic obese rats, independent of weight
loss) (216,217). CB1R antagonists also upregulate
skeletal muscle GLUT-4 to ameliorate intermittent hypoxia-induced
insulin resistance, whereas CB1R agonists interfere with insulin
signaling pathways (218,219). CB1R antagonists modulate the ERS
pathway by blocking high-glucose- or inflammation-induced CB1R
signaling on several levels: i) Inhibiting oxidative stress and
calcium disruption (211); and
ii) attenuating inflammation and fibrosis, thereby improving renal
cell survival and renal function (220). The use of CB1R antagonists is
subject to notable limitations. Currently, to the best of our
knowledge, no human studies using CB1R antagonists have been
conducted, and the risk of psychiatric side effects remains to be
assessed (221,222). Consequently, clinical advancement
cannot proceed at this stage (223). However, it holds potential and
may provide a basis for DKD treatment following future animal
studies and clinical trials.
Chemical chaperones, such as TUDCA and 4-PBA, are
small molecules responsible for stabilizing protein conformation,
enhancing ER folding and promoting mutant protein transport. TUDCA,
a secondary bile acid, exhibits beneficial effects in various
disorders, including diabetes, obesity and neurodegenerative
disease (224). The
cytoprotective mechanisms of chemical chaperones primarily involve
the alleviation of ERS. 4-PBA, a small-molecule chaperone employed
in urea-cycle disorders, mitigates ERS to normalize hyperglycemia
and insulin resistance (225–227). Preclinical study has demonstrated
the efficacy of 4-PBA in preventing podocyte apoptosis in type 2
diabetes (228). In addition,
three widely used ERS inducers, clindamycin, dithiothreitol and
carbobenzoxy-Leu-Leu-leucinal, have been applied to assess ERS in
animal models and cell lines. 4-PBA was shown to alleviate
drug-induced ERS (229), while
drugs targeting PERK, IRE1α, eIF2α and ATF4, such as GSK2606414,
MKC-3946, salubrinal and trazodone, offer promise for ERS-related
disorder management (230,231).
In rats fed a high-salt diet, damage to the glomeruli and proximal
tubules was exacerbated, accompanied by increased urinary protein
excretion. Following treatment with TUDCA, the levels of proximal
tubular giant cells increased and urinary protein levels were
markedly decreased (232). TUDCA
and 4-PBA are not formally included in clinical guidelines for DKD.
However, the Chinese Guidelines for the Prevention and Treatment of
Diabetic Kidney Disease (2021 Edition) mention in the section on
‘Novel Therapeutic Targets for DKD’ that ‘chemical companions may
improve renal injury by alleviating ERS’, listing TUDCA as a
potential candidate drug (233).
Sodium-glucose co-transporter 2 (SGLT2) inhibitors,
such as empagliflozin and dapagliflozin, currently represent
first-line therapies for DKD. Their regulation of ERS involves
indirect multi-pathway interventions, and the direct association
between their renal protective effects and ERS inhibition requires
validation through mechanistic clinical trials (244,245). The 2024 KDIGO guidelines classify
SGLT2 inhibitors as a first-line therapy with Class 1A
recommendation for patients with DKD with an estimated glomerular
filtration rate (eGFR) ≥20 ml/min/1.73 m2, emphasizing
their role in delaying renal progression (246,247). However, ERS-related mechanisms
are not addressed. Existing large-scale clinical trials have
primarily focused on definitive renal endpoints, such as eGFR
decline and end-stage kidney disease, without systematic assessment
of ERS biomarkers (202,248–251). Thus, attention to ERS treatment
is warranted for kidney disease.
The aforementioned drugs exhibit pleiotropic
effects, indicating their lack of specificity for ERS inhibition;
thus, development of ERS-targeted therapeutics is warranted.
Notably, ERS exhibits dual effects: Moderate ERS restores
intracellular homeostasis, while sustained ERS drives renal damage
in DKD, meaning that human-targeted ERS research is warranted
(252–254). A deeper understanding of the
mechanisms underlying this process will help improve the treatment
of DKD.
As a major microvascular complication of diabetes
mellitus, DKD is closely associated with aberrant activation of
ERS. Factors such as hyperglycemia and oxidative stress trigger an
imbalance of protein folding in renal podocytes, tubular epithelial
and renal capsule cells, which activates the three core pathways of
the UPR, namely the IRE1α/XBP1, PERK/eIF2α/ATF4 and ATF6 pathways
(50,255). Short-term ERS maintains cellular
homeostasis by promoting protein folding and degradation, whereas
sustained overactivation induces pro-apoptotic factor expression,
such as CHOP expression, exacerbating inflammatory responses
through NF-κB pathway activation and mesangial fibrosis via
TGF-β1/Smad3 pathway dysregulation, which ultimately leads to
glomerulosclerosis and renal failure. Several stimuli disrupting
cellular homeostatic function induce ERS (58,256) (Fig.
1). The present review discusses ERS-related experimental
agents, preclinical models and clinical data, alongside an analysis
of the three UPR pathways in DKD renal histopathology.
Phase I and II clinical trials are underway for
ERS-modulating compounds such as alginate and TUDCA (257). Pharmacological interventions
against ERS have focused on blocking the excessive stress response.
However, although the aforementioned RAS inhibitors, SGLT2
inhibitors, 4-PBA and CB1R antagonists may regulate ERS through
indirect pathways, existing guidelines do not provide specific
recommendations based on this mechanism (258–260). Clinical trials lack ERS-related
endpoint data, and the causal relationship between medications for
the treatment of DKD, ERS) requires further validation through
targeted research. Western drugs, such as PERK inhibitors, IRE1α
endonuclease inhibitors and CHOP antagonists, have demonstrated
potential in attenuating apoptosis and fibrosis in animal models
(203,261–263); however, challenges associated
with target specificity and long-term safety remain.
ERS inhibitors commonly used in laboratory studies,
such as the chemical chaperone 4-PBA or the pathway-specific PERK
inhibitor GSK2606414, generally exhibit good tolerability in rodent
DKD models (264,265), but their clinical safety
assessments present significant challenges. Primarily, ERS is a
fundamental physiological mechanism enabling cellular responses to
abnormal protein folding, which is important in metabolically
active tissues such as pancreatic β-cells and hepatocytes. While
4-PBA alleviates ERS in renal tissues of rat models (266–268), its prolonged clinical application
may cause hyperammonemia and gastrointestinal mucosal irritation.
Furthermore, suppressing ERS responses in pancreatic β-cells can
impair insulin synthesis, potentially exacerbating glycemic
dysregulation (225). Secondly,
the risk of kidney-specific toxicity may be underestimated. Animal
models commonly involve young, healthy rats, whereas clinical
patients with DKD typically present with comorbidities such as
hypertension, cardiovascular disease and renal insufficiency,
markedly reducing drug metabolism capacity. In addition, drug
interactions remain unclear: Patients with DKD frequently require
concomitant hypoglycemic, antihypertensive and lipid-lowering
medications (269,270). Furthermore, ERS inhibitors may
compete with SGLT2 inhibitors for renal tubular excretion pathways
or affect the renal metabolism of ACEIs, increasing the risk of
adverse reactions such as hyperkalemia (271). Such interactions are challenging
to detect in controlled, single-factor laboratory settings.
Additionally, the complex multi-pathway regulatory nature of ERS,
coordinated by PERK, IRE1α and ATF6, combined with individual
heterogeneity among patients with DKD makes ‘precision targeting’ a
translational challenge. Laboratory studies often target single
pathways, whereas clinical ERS activation patterns vary notably
among patients with DKD: Some cases of DKD primarily involve
activation of the PERK/eIF2α pathway, accompanied by extensive
tubular epithelial apoptosis, while others predominantly involve
activation of the ATF6 pathway, accompanied by inflammatory
mediator release (for example, elevated IL-6 and TNF-α levels)
(71). Furthermore, ERS in DKD
involves glomerular mesangial cells, podocytes and tubular
epithelial cells; however, existing drugs exhibit systemic
distribution, complicating precise delivery to specific renal cell
subpopulations (18,19).
Addressing these bottlenecks requires further
research. Safety evaluations should incorporate multiple animal
models with renal insufficiency to more accurately replicate drug
metabolism characteristics observed in clinical populations. For
target optimization, kidney-specific drug formulations could be
developed to enhance renal tissue accumulation. Only through these
approaches can ERS inhibition strategies transition from being
mechanistically effective in laboratory settings to being safe and
beneficial in clinical practice.
The active constituents of TCM, such as AS and
Huangkui total flavonoids, exert multi-target modulatory effects on
ERS, including the downregulation of CHOP and inhibition of IRE1α
phosphorylation (47,166). These compounds also exert
antioxidant, anti-inflammatory and podocyte-protective effects,
supporting clinical evidence of reduced proteinuria and delayed
renal function decline in DKD. Such findings underscore the
potential of TCM-Western medicine integration as a promising
research avenue for DKD management. The development and translation
of ER-specific reagents targeting protein folding, UPR signaling
and ER calcium homeostasis hold promise for novel therapeutic
strategies (7).
In future research, it is important to investigate
ERS biomarkers for early DKD diagnosis by elucidating the
underlying ERS mechanisms. BiP/GRP78, as a sentinel protein for ERS
activation, exhibits increased expression levels that signal early
ERS and are directly associated with the severity of renal
pathological damage in DKD, thereby providing a foundational
reference for assessing disease progression (112). Concurrently, p-PERK, p-eIF2α and
ATF4 from the PERK pathway, p-IRE1α and XBP1s from the IRE1α
pathway and ATF6f (a type II transmembrane protein that under ER
stress is translocated to the Golgi apparatus where it is
proteolytically processed, releasing the cytoplasmic fragment of
ATF6) (77). from the ATF6 pathway
indicate the activation status of the three principal UPR branches,
accurately reflecting the ERS-mediated pathological transition from
adaptive protection to apoptosis initiation. The comprehensive
assessment of these markers not only clarifies the molecular
mechanisms of ERS in DKD but also provides molecular evidence for
staging disease progression (16).
These biomarkers thus hold potential for translation into early
diagnostic tools and therapeutic efficacy-monitoring indicators for
DKD, enabling precise clinical interventions. Simultaneously, the
development of inhibitors targeting specific pathways, such as
IRE1α-specific inhibitors, is important, offering novel therapeutic
strategies for DKD management. Given the growing potential of
ERS-targeted therapies, focused clinical studies are required to
deepen the current understanding of DKD pathogenesis and refine
targeted interventions. Collectively, these efforts will advance
DKD treatment and provide insights that are relevant to broader
ERS-associated disorders.
Not applicable.
The present study was supported by the Jilin Province Science
and Technology Development Program (grant no. 20210101201JC).
Not applicable.
PZ contributed to writing the manuscript and
prepared the figures. YZ made substantial contributions to the
conception and design of the study, conducting a comprehensive and
systematic literature review to identify key studies and ensuring
the research was grounded in relevant and current findings in the
field of DKD. YZ also played a crucial role in the analysis and
interpretation of data, particularly in understanding the
mechanisms of ERS in DKD. Additionally, YZ contributed to drafting
significant portions of the manuscript and critically revised it
for important intellectual content. YZ approved the final version
of the manuscript and takes full responsibility for the integrity
and accuracy of the work, ensuring all aspects of the research were
thoroughly addressed. CC, YC, FL, SZ and CL revised the manuscript.
All authors read and approved the final version of the manuscript.
Data authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Zhang L, Long J, Jiang W, Shi Y, He X,
Zhou Z, Li Y, Yeung RO, Wang J, Matsushita K, et al: Trends in
chronic kidney disease in China. N Engl J Med. 375:905–906. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cybulsky AV: Endoplasmic reticulum stress,
the unfolded protein response and autophagy in kidney diseases. Nat
Rev Nephrol. 13:681–696. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
American Diabetes Association: 11
Microvascular complications and foot care: Standards of medical
care in diabetes-2020. Diabetes Care. 43 (Suppl 1):S135–S151. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pena MJ, Mischak H and Heerspink HJ:
Proteomics for prediction of disease progression and response to
therapy in diabetic kidney disease. Diabetologia. 59:1819–1831.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bustamante P, Tsering T, Coblentz J,
Mastromonaco C, Abdouh M, Fonseca C, Proença RP, Blanchard N, Dugé
CL, Andujar RAS, et al: Circulating tumor DNA tracking through
driver mutations as a liquid biopsy-based biomarker for uveal
melanoma. J Exp Clin Cancer Res. 40:1962021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tang M, Sun J and Cai Z: PCK2 inhibits
lung adenocarcinoma tumor cell immune escape through oxidative
stress-induced senescence as a potential therapeutic target. J
Thorac Dis. 15:2601–2615. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Almanza A, Carlesso A, Chintha C,
Creedican S, Doultsinos D, Leuzzi B, Luís A, McCarthy N,
Montibeller L, More S, et al: Endoplasmic reticulum stress
signalling - from basic mechanisms to clinical applications. FEBS
J. 286:241–278. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tilija Pun N, Lee N, Song SH and Jeong CH:
pitavastatin induces cancer cell apoptosis by blocking autophagy
flux. Front Pharmacol. 13:8545062022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Çiftçi YC, Yurtsever Y and Akgül B: Long
non-coding RNA-mediated modulation of endoplasmic reticulum stress
under pathological conditions. J Cell Mol Med. 28:e185612024.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kober L, Zehe C and Bode J: Development of
a novel ER stress based selection system for the isolation of
highly productive clones. Biotechnol Bioeng. 109:2599–2611. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hotamisligil GS: Endoplasmic reticulum
stress and the inflammatory basis of metabolic disease. Cell.
140:900–917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shahzad K, Ghosh S, Mathew A and Isermann
B: Methods to detect endoplasmic reticulum stress and apoptosis in
diabetic nephropathy. Methods Mol Biol. 2067:153–173. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hetz C and Papa FR: The unfolded protein
response and cell fate control. Mol Cell. 69:169–181. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guzmán Mendoza NA, Homma K, Osada H, Toda
E, Ban N, Nagai N, Negishi K, Tsubota K and Ozawa Y:
Neuroprotective effect of 4-phenylbutyric acid against photo-stress
in the Retina. Antioxidants (Basel). 10:11472021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen YY, Peng XF, Liu GY, Liu JS, Sun L,
Liu H, Xiao L and He LY: Protein arginine methyltranferase-1
induces ER stress and epithe-lial-mesenchymal transition in renal
tubular epithelial cells and contributes to diabetic nephropathy.
Biochim Biophys Acta Mol Basis Dis. 1865:2563–2575. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu H and Sun HL: LncRNA TCF7 triggered
endoplasmic reticulum stress through a sponge action with miR-200c
in patients with diabetic nephropathy. Eur Rev Med Pharmacol Sci.
23:5912–5922. 2019.PubMed/NCBI
|
|
17
|
Chen N, Song S, Yang Z, Wu M, Mu L, Zhou T
and Shi Y: ChREBP deficiency alleviates apoptosis by inhibiting
TXNIP/oxidative stress in diabetic nephropathy. J Diabetes
Complications. 35:1080502021. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tervaert TW, Mooyaart AL, Amann K, Cohen
AH, Cook HT, Drachenberg CB, Ferrario F, Fogo AB, Haas M, de Heer
E, et al: Pathologic classification of diabetic nephropathy. J Am
Soc Nephrol. 21:556–563. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Oshima M, Shimizu M, Yamanouchi M, Toyama
T, Hara A, Furuichi K and Wada T: Trajectories of kidney function
in diabetes: A clinicopathological update. Nat Rev Nephrol.
17:740–750. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Garg P: A review of podocyte biology. Am J
Nephrol. 47 (Suppl 1):S3–S13. 2018. View Article : Google Scholar
|
|
21
|
O'Toole JF: Renal manifestations of
genetic mitochondrial disease. Int J Nephrol Renovasc Dis. 7:57–67.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nath KA: Tubulointerstitial changes as a
major determinant in the progression of renal damage. Am J Kidney
Dis. 20:1–17. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tang J, Yao D, Yan H, Chen X, Wang L and
Zhan H: The role of MicroRNAs in the pathogenesis of diabetic
nephropathy. Int J Endocrinol. 2019:87190602019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bazzi C, Bakoush O and Gesualdo L:
Proteinuria: From molecular to clinical applications in
glomerulonephritis. Int J Nephrol. 2012:4249682012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Edirs S, Jiang L, Xin X and Aisa HA: Kursi
Wufarikun Ziyabit improves the physiological changes by regulating
endoplasmic reticulum stress in the type 2 diabetes db/db mice.
Evid Based Complement Alternat Med. 2021:21001282021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Guo H, Cao A, Chu S, Wang Y, Zang Y, Mao
X, Wang H, Wang Y, Liu C, Zhang X and Peng W: Astragaloside IV
attenuates podocyte apoptosis mediated by endoplasmic reticulum
stress through upregulating sarco/endoplasmic reticulum Ca2+-ATPase
2 expression in diabetic nephropathy. Front Pharmacol. 7:5002016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang D, Wang YS, Zhao HM, Lu P, Li M, Li
W, Cui HT, Zhang ZY and Lv SQ: Plantamajoside improves type 2
diabetes mellitus pancreatic β-cell damage by inhibiting
endoplasmic reticulum stress through Dnajc1 up-regulation. World J
Diabetes. 16:990532025. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang HX, Yuan J and Li RS: Thalidomide
mitigates apoptosis via endoplasmic reticulum stress in diabetic
nephropathy. Endocr Metab Immune Disord Drug Targets. 22:787–794.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Van Krieken R, Mehta N, Wang T, Zheng M,
Li R, Gao B, Ayaub E, Ask K, Paton JC, Paton AW, et al: Cell
surface expression of 78-kDa glucose-regulated protein (GRP78)
mediates diabetic nephropathy. J Biol Chem. 294:7755–7768. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang MZ, Wang Y, Paueksakon P and Harris
RC: Epidermal growth factor receptor inhibition slows progression
of diabetic nephropathy in association with a decrease in
endoplasmic reticulum stress and an increase in autophagy.
Diabetes. 63:2063–2072. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen J, Hou XF, Wang G, Zhong QX, Liu Y,
Qiu HH, Yang N, Gu JF, Wang CF, Zhang L, et al: Terpene glycoside
component from Moutan Cortex ameliorates diabetic nephropathy by
regulating endoplasmic reticulum stress related inflammatory
responses. J Ethnopharmacol. 193:433–444. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yao F, Li Z, Ehara T, Yang L, Wang D, Feng
L, Zhang Y, Wang K, Shi Y, Duan H and Zhang L: Fatty acid-binding
protein 4 mediates apoptosis via endoplasmic reticulum stress in
mesangial cells of diabetic nephropathy. Mol Cell Endocrinol.
411:232–242. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ge J, Miao JJ, Sun XY and Yu JY: Huangkui
capsule, an extract from Abelmoschus manihot (L.) medic, improves
diabetic nephropathy via activating peroxisome
proliferator-activated receptor (PPAR)-α/γ and attenuating
endoplasmic reticulum stress in rats. J Ethnopharmacol.
189:238–249. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Park MJ, Han HJ and Kim DI:
Lipotoxicity-Induced PRMT1 exacerbates mesangial cell apoptosis via
endoplasmic reticulum stress. Int J Mol Sci. 18:14212017.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Khoi CS, Xiao CQ, Hung KY, Lin TY and
Chiang CK: Oxidative stress-induced growth inhibitor (OSGIN1), a
Target of X-Box-Binding Protein 1, protects palmitic acid-induced
vascular lipotoxicity through maintaining autophagy. Biomedicines.
10:9922022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jeon HY, Moon CH, Kim EB, Sayyed ND, Lee
AJ and Ha KS: Simultaneous attenuation of hyperglycemic
memory-induced retinal, pulmonary, and glomerular dysfunctions by
proinsulin C-peptide in diabetes. BMC Med. 21:492023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jiao Y, Liu X, Shi J, An J, Yu T, Zou G,
Li W and Zhuo L: Unraveling the interplay of ferroptosis and immune
dysregulation in diabetic kidney disease: A comprehensive molecular
analysis. Diabetol Metab Syndr. 16:862024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu F, Yang Z, Li J, Wu T, Li X, Zhao L,
Wang W, Yu W, Zhang G and Xu Y: Targeting programmed cell death in
diabetic kidney disease: from molecular mechanisms to
pharmacotherapy. Mol Med. 30:2652024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Cao Y, Hao Y, Li H, Liu Q, Gao F, Liu W
and Duan H: Role of endoplasmic reticulum stress in apoptosis of
differentiated mouse podocytes induced by high glucose. Int J Mol
Med. 33:809–816. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Tian N, Gao Y, Wang X, Wu X, Zou D, Zhu Z,
Han Z, Wang T and Shi Y: Emodin mitigates podocytes apoptosis
induced by endoplasmic reticulum stress through the inhibition of
the PERK pathway in diabetic nephropathy. Drug Des Devel Ther.
12:2195–2211. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Garner KL, Betin VMS, Pinto V, Graham M,
Abgueguen E, Barnes M, Bedford DC, McArdle CA and Coward RJM:
Enhanced insulin receptor, but not PI3K,signalling protects
podocytes from ER stress. Sci Rep. 8:39022018. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang Y, Gao X, Chen S, Zhao M, Chen J,
Liu R, Cheng S, Qi M, Wang S and Liu W: Cyclin-dependent kinase 5
contributes to endoplasmic reticulum stress induced podocyte
apoptosis via promoting MEKK1 phosphorylation at Ser280 in diabetic
nephropathy. Cell Signal. 31:31–40. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cybulsky AV: The intersecting roles of
endoplasmic reticulum stress, ubiquitin-proteasome system, and
autophagy in the pathogenesis of proteinuric kidney disease. Kidney
Int. 84:25–33. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Dong Z, Liu N and Sun M: The distinct
biological role of JAML positions it as a promising target for
treating human cancers and a range of other diseases. Front
Immunol. 16:15584882025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Feng Z, Tang L, Wu L, Cui S, Hong Q, Cai
G, Wu D, Fu B, Wei R and Chen X: Na+/H+ exchanger-1 reduces
podocyte injury caused by endoplasmic reticulum stress via
autophagy activation. Lab Invest. 94:439–454. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Fang L, Li X, Luo Y, He W, Dai C and Yang
J: Autophagy inhibition induces podocyte apoptosis by activating
the proapoptotic pathway of endoplasmic reticulum stress. Exp Cell
Res. 322:290–301. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Guo H, Wang Y, Zhang X, Zang Y, Zhang Y,
Wang L, Wang H, Wang Y, Cao A and Peng W: Astragaloside IV protects
against podocyte injury via SERCA2-dependent ER stress reduction
and AMPKα-regulated autophagy induction in streptozotocin-induced
diabetic nephropathy. Sci Rep. 7:68522017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kato M: Intercellular transmission of
endoplasmic reticulum stress through gap junction targeted by
microRNAs as a keystep of diabetic kidney diseases? Ann Transl Med.
9:8272021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Liang X, Duan N, Wang Y, Shu S, Xiang X,
Guo T, Yang L, Zhang S, Tang X and Zhang J: Advanced oxidation
protein products induce endothelial-to-mesenchymal transition in
human renal glomerular endothelial cellsthrough induction of
endoplasmic reticulum stress. J Diabetes Complications. 30:573–579.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ma X, Ma J, Leng T, Yuan Z, Hu T, Liu Q
and Shen T: Advances in oxidative stress in pathogenesis of
diabetic kidney disease and efficacy of TCM intervention. Ren Fail.
45:21465122023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Cao Y, Chen Z, Hu J, Feng J, Zhu Z, Fan Y,
Lin Q and Ding G: Mfn2 regulates high glucose-induced MAMs
dysfunction and apoptosis in podocytes via PERK pathway. Front Cell
Dev Biol. 9:7692132021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mega C, Teixeira-de-Lemos E, Fernandes R
and Reis F: Renoprotective effects of the dipeptidyl peptidase-4
inhibitor sitagliptin: A review in type 2 diabetes. J Diabetes Res.
2017:51642922017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Feng A, Yin R, Xu R, Zhang B and Yang L:
An update on renal tubular injury as related to glycolipid
metabolism in diabetic kidney disease. Front Pharmacol.
16:15590262025. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bondue T, van den Heuvel L, Levtchenko E
and Brock R: The potential of RNA-based therapy for kidney
diseases. Pediatr Nephrol. 38:327–344. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhang J, Dong XJ, Ding MR, You CY, Lin X,
Wang Y, Wu MJ, Xu GF and Wang GD: Resveratrol decreases high
glucose-induced apoptosis in renal tubular cells via suppressing
endoplasmic reticulum stress. Mol Med Rep. 22:4367–4375.
2020.PubMed/NCBI
|
|
56
|
Zhang J, Cao P, Gui J, Wang X, Han J, Wang
Y and Wang G: Arc-tigenin ameliorates renal impairment and inhibits
endoplasmic reticulum stress in diabetic db/db mice. Life Sci.
223:194–201. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Han J, Pang X, Shi X, Zhang Y, Peng Z and
Xing Y: Ginkgo biloba extract EGB761 ameliorates the extracellular
matrix accumulation and mesenchymal transformation of renal tubules
in diabetic kidney disease by inhibiting endoplasmic reticulum
stress. Biomed Res Int. 2021:66572062021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Huang KH, Guan SS, Lin WH, Wu CT, Sheu ML,
Chiang CK and Liu SH: Role of calbindin-D28k in diabetes-associated
advanced glycation end-products-induced renal proximal tubule cell
injury. Cells. 8:6602019. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Sun X, Sun Y, Lin S, Xu Y and Zhao D:
Histone deacetylase inhibitor valproic acid attenuates high
glucoseinduced endoplas-mic reticulum stress and apoptosis in
NRK52E cells. Mol Med Rep. 22:4041–4047. 2020.PubMed/NCBI
|
|
60
|
Shibusawa R, Yamada E, Okada S, Nakajima
Y, Bastie CC, Maeshima A. Kaira K and Yamada M: Dapagliflozin
rescues endoplasmic reticulum stress-mediated cell death. Sci Rep.
9:98872019. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wu L, Wang Q, Guo F, Ma X, Wang J, Zhao Y,
Yan Y and Qin G: Involvement of miR-27a-3p in diabetic nephropathy
via affecting renal fibrosis, mitochondrial dysfunction, and
endoplasmic reticulum stress. J Cell Physiol. 236:1454–1468. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Fang L, Xie D, Wu X, Cao H, Su W and Yang
J: Involvement of endoplasmic reticulum stress in albuminuria
induced inflam-masome activation in renal proximal tubular cells.
PLoS One. 8:e723442013. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kang JM, Lee HS, Kim J, Yang DH, Jeong HY,
Lee YH, Kim DJ, Park SH, Sung M, Kim J, et al: Beneficial effect of
Chloroquine and Amodiaquine on type 1 Diabetic Tubulopathy by
attenuating mitochondrial Nox4 and endoplasmic reticulum stress. J
Korean Med Sci. 35:e3052020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Sun H, Yuan Y and Sun Z: Update on
mechanisms of renal tubule injury caused by advanced glycation end
products. Biomed Res Int. 2016:54751202019.PubMed/NCBI
|
|
65
|
Iwai T, Kume S, Chin-Kanasaki M, Kuwagata
S, Araki H, Takeda N, Sugaya T, Uzu T, Maegawa H and Araki SI:
Stearoyl-CoA Desaturase-1 protects cells against
lipotoxicity-mediated apoptosis in proximal tubular cells. Int J
Mol Sci. 17:18682016. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Liu J, Yang JR, Chen XM, Cai GY, Lin LR
and He YN: Impact of ER stress-regulated ATF4/p16 signaling on the
premature senescence of renal tubular epithelial cells in diabetic
nephropathy. Am J Physiol Cell Physiol. 308:C621–C630. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Barati MT, Powell DW, Kechavarzi BD,
Isaacs SM, Zheng S, Epstein PN, Cai L, Coventry S, Rane MJ and
Klein JB: Differential expression of endoplasmic reticulum
stress-response proteins in different renal tubule subtypes of
OVE26 diabetic mice. Cell Stress Chaperones. 21:155–166. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Liu Y, Chen DQ, Han JX, Zhao TT and Li SJ:
A review of traditional Chinese medicine in treating renal
interstitial fibrosis via endoplasmic reticulum stress-mediated
apoptosis. Biomed Res Int. 2021:66677912021. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Wang J, Lu L, Chen S, Xie J, Lu S, Zhou Y
and Jiang H: PERK overexpression-mediated Nrf2/HO-1 pathway
alleviates hypoxia/reoxygenation-induced injury in neonatal murine
cardiomyocytes via improving endoplasmic reticulum stress. Biomed
Res Int. 2020:64580602020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Chen Z, Feng H, Peng C, Zhang Z, Yuan Q,
Gao H, Tang S and Xie C: Renoprotective effects of tanshinone IIA:
A literature review. Molecules. 28:19902023. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Nakka VP, Prakash-Babu P and Vemuganti R:
Crosstalk between endoplasmic reticulum stress, oxidative stress,
and autophagy: Potential therapeutic targets for acute CNS
injuries. Mol Neurobiol. 53:532–544. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Qiu M, Li S, Jin L, Feng P, Kong Y, Zhao
X, Lin Y, Xu Y, Li C and Wang W: Combination of chymostatin and
aliskiren attenuates ER stress induced by lipid overload in kidney
tubular cells. Lipids Health Dis. 17:1832018. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Chai H, Yao S, Gao Y, Hu Q and Su W:
Developments in the connection between epithelial-mesenchymal
transition and endoplasmic reticulum stress (Review). Int J Mol
Med. 56:1022025. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Wang WW, Liu YL, Wang MZ, Li H, Liu BH, Tu
Y, Yuan CC, Fang QJ, Chen JX, Wang J, et al: Inhibition of renal
tubular epithelial mesenchymal transition and endoplasmic reticulum
stress-induced apoptosis with shenkang injection attenuates
diabetic tubulopathy. Front Pharmacol. 12:6627062021. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Wang W, Ke B, Wang C, Xiong X, Feng X and
Yan H: Targeting ion channel networks in diabetic kidney disease:
from molecular crosstalk to precision therapeutics and clinical
innovation. Front Med (Lausanne). 12:16077012025. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Jo HJ, Yang JW, Park JH, Choi ES, Lim CS,
Lee S and Han CY: Endoplasmic reticulum stress increases DUSP5
expression via PERK-CHOP pathway, leading to hepatocyte death. Int
J Mol Sci. 20:43692019. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Hu H, Tian M, Ding C and Yu S: The C/EBP
homologous protein (CHOP) transcription factor functions in
endoplasmic reticulum Stress-Induced apoptosis and microbial
infection. Front Immunol. 9:30832019. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Jin R, Zhao A, Han S, Zhang D, Sun H, Li
M, Su D and Liang X: The interaction of S100A16 and GRP78 actives
endoplasmic reticulum stress-mediated through the IRE1α/XBP1
pathway in renal tubulointerstitial fibrosis. Cell Death Dis.
12:9422021. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Fan Y, Zhang J, Xiao W, Lee K, Li Z, Wen
J, He L, Gui D, Xue R, Jian G, et al: Rtn1a-Mediated endoplasmic
reticulum stress in podocyte injury and diabetic nephropathy. Sci
Rep. 7:3232017. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Zhang GQ, Tao YK, Bai YP, Yan ST and Zhao
SP: Inhibitory effects of simvastatin on oxidized low-density
lipoprotein-induced endoplasmic reticulum stress and apoptosis in
vascular endothelial cells. Chin Med J (Engl). 131:950–955. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Ni L, Yang L and Lin Y: Recent progress of
endoplasmic reticulum stress in the mechanism of atherosclerosis.
Front Cardiovasc Med. 11:14134412024. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Li HY, Huang LF, Huang XR, Wu D, Chen XC,
Tang JX, An N, Liu HF and Yang C: Endoplasmic reticulum stress in
systemic lupus erythematosus and lupus nephritis: Potential
therapeutic target. J Immunol Res. 2023:76258172023. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Shu S, Wang H, Zhu J, Liu Z, Yang D, Wu W,
Cai J, Chen A, Tang C and Dong Z: Reciprocal regulation between ER
stress and autophagy in renal tubular fibrosis and apoptosis. Cell
Death Dis. 12:10162021. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Li D, Zhang J, Su X, Yang Y, Lai J, Wei X,
Chen H, Liu Y, Wang H and Sun L: Calpain1 inhibition enhances
autophagy-lysosomal pathway and ameliorates tubulointerstitial
fibrosis in Nephronophthisis. Mol Med. 31:1662025. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Laorodphun P, Cherngwelling R, Panya A and
Arjinajarn P: Curcumin protects rats against gentamicin-induced
nephrotoxicity by amelioration of oxidative stress, endoplasmic
reticulum stress and apoptosis. Pharm Biol. 60:491–500. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Ma N, Xu N, Yin D, Zheng P, Liu W, Wang G,
Hui Y, Han G, Yang C and Cheng X: Levels of circulating GRP78 and
CHOP in endoplasmic reticulum stress pathways in Chinese type 2
diabetic kidney disease patients. Medicine (Baltimore).
100:e268792021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Zhao DM, Zhong R, Wang XT and Yan ZH:
Mitochondrial dysfunction in diabetic nephropathy: Insights and
therapeutic avenues from traditional Chinese medicine. Front
Endocrinol (Lausanne). 15:14294202024. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Yan DY and Xu B: The role of autophagy in
manganese-induced neurotoxicity. Front Neurosci. 14:5747502020.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Abo-Zaid OA, Moawed FS, Taha EF, Ahmed ESA
and Kawara RS: Melissa officinalis extract suppresses endoplasmic
reticulum stress-induced apoptosis in the brain of
hypothyroidism-induced rats exposed to γ-radiation. Cell Stress
Chaperones. 28:709–720. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Kong FJ, Ma LL, Guo JJ, Xu LH, Li Y and Qu
S: Endoplasmic reticulum stress/autophagy pathway is involved in
diabetes induced neuronal apoptosis and cognitive decline in mice.
Clin Sci (Lond). 132:111–125. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Tomicic MT, Meise R, Aasland D, Berte N,
Kitzinger R, Krämer OH, Kaina B and Christmann M: Apoptosis induced
by temozolomide and nimustine in glioblastoma cells is supported by
JNK/c-Jun-mediated induction of the BH3-only protein BIM.
Oncotarget. 6:33755–33768. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Gao Z, Liu G, Hu Z, Shi W, Chen B, Zou P
and Li X: Grape seed proanthocyanidins protect against
streptozotocin-induced diabetic nephropathy by attenuating
endoplasmic reticulum stress-induced apoptosis. Mol Med Rep.
18:1447–1454. 2018.PubMed/NCBI
|
|
93
|
Sun XY, Qin HJ, Zhang Z, Xu Y, Yang XC,
Zhao DM, Li XN and Sun LK: Valproate attenuates diabetic
nephropathy through inhibition of endoplasmic reticulum
stress-induced apoptosis. Mol Med Rep. 13:661–668. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Suzuki Y, Inoue T, Murai M,
Suzuki-Karasaki M, Ochiai T and Ra C: Depolarization potentiates
TRAIL-induced apoptosis in human melanoma cells: role for
ATP-sensitive K+ channels and endoplasmic reticulum stress. Int J
Oncol. 41:465–475. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Hibi M, Lin A, Smeal T, Minden A and Karin
M: Identification of an oncoprotein- and UV-responsive protein
kinase that binds and potentiates the c-Jun activation domain.
Genes Dev. 7:2135–2148. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Shang H, Cao Z, Zhao J, Guan J, Liu J,
Peng J, Chen Y, Joseph Sferra T, Sankararaman S and Lin J: Babao
Dan induces gastric cancer cell apoptosis via regulating MAPK and
NF-κB signaling pathways. J Int Med Res. 47:5106–5119. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Liu Y, Long Y, Xing Z and Zhang D: C-Jun
recruits the NSL complex to regulate its target gene expression by
modulating H4K16 acetylation and promoting the release of the
repressive NuRD complex. Oncotarget. 6:14497–14506. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Vilas-Boas EA, Almeida DC, Roma LP, Ortis
F and Carpinelli AR: Lipotoxicity and β-Cell failure in type 2
diabetes: Oxidative stress linked to NADPH oxidase and ER stress.
Cells. 10:33282021. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Chhabra R, Dubey R and Saini N: Gene
expression profiling indicate role of ER stress in miR-23a ~ 27a ~
24 - 2 cluster induced apoptosis in HEK293T cells. RNA Biol.
8:648–664. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Rodríguez-Hernández MA, González R, de la
Rosa ÁJ, Gallego P, Ordóñez R, Navarro-Villarán E, Contreras L,
Rodríguez-Arribas M, González-Gallego J, Álamo-Martínez JM, et al:
Molecular characterisation of autophagic and apoptotic signalling
induced by sorafenib in liver cancer cells. J Cell Physiol.
234:692–708. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Zeng T, Peng L, Chao H, Xi H, Fu B, Wang
Y, Zhu Z and Wang G: IRE1α-TRAF2-ASK1 complex-mediated endoplasmic
reticulum stress and mitochondrial dysfunction contribute to
CXC195-induced apoptosis in human bladder carcinoma T24 cells.
Biochem Biophys Res Commun. 460:530–536. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Zhang J, Liang Y, Lin Y, Liu Y and YouYou
Yin W: IRE1α-TRAF2-ASK1 pathway is involved in CSTMP-induced
apoptosis and ER stress in human non-small cell lung cancer A549
cells. Biomed Pharmacother. 82:281–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Ha J, Kang E, Seo J and Cho S:
Phosphorylation dynamics of JNK signaling: Effects of
dual-specificity phosphatases (DUSPs) on the JNK pathway. Int J Mol
Sci. 20:61572019. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Li L, Chen J, Lin L, Pan G, Zhang S, Chen
H, Zhang M, Xuan Y, Wang Y and You Z: Quzhou Fructus Aurantii
Extract suppresses inflammation via regulation of MAPK, NF-κB, and
AMPK signaling pathway. Sci Rep. 10:15932020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Muraleva NA, Tikhonov DI, Zhdankina AA,
Plotnikov MB, Khlebnikov AI, Logvinov SV and Kolosova NG:
Alterations of JNK signaling pathway activity in the rat retina:
Effects of age, age-related macular degeneration-like pathology,
and a JNK inhibitor (IQ-1S). Cells. 14:8962025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Bain J, Later L, Elliot M, Shpiro N,
Hastie CJ, Mclauchlan H, Klevernic I, Arthur JS, Alessi DR and
Cohen P: The selectivity of protein kinase inhibitors: A further
update. Biochem J. 408:297–315. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Bennett BL, Sasaki DT, Murray BW, O'Leary
EC, Sakata ST, Xu W, Leisten JC, Motiwala A, Pierce S, Satoh Y, et
al: SP600125, an anthrapyrazolone inhibitor of Jun N-Terminal
kinase. Proc Natl Acad Sci USA. 98:136812021. View Article : Google Scholar
|
|
108
|
Inesta-Vaquera FA, Campbell DG, Arthur JS
and Cuenda A: ERK5 pathway regulates the phosphorylation of tumour
suppressor hDlg during mitosis. Biochem Biophy Res Commun.
399:84–90. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Zhang T, Inesta-Vaquera F, Niepel M, Zhang
J, Ficarro SB, Machleidt T, Xie T, Marto JA, Kim N, Sim T, et al:
Discovery of potent and selective covalent inhibitors of JNK. Chem
Biol. 19:140–154. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Lupachyk S, Watcho P, Stavniichuk R,
Shevalye H and Obrosova IG: Endoplasmic reticulum stress plays a
key role in the pathogenesis of diabetic peripheral neuropathy.
Diabetes. 62:944–952. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Mei Y, Thompson MD, Cohen RA and Tong X:
Endoplasmic reticulum stress and related pathological processes. J
Pharmacol Biomed Anal. 1:10001072013.PubMed/NCBI
|
|
112
|
Zhang SX, Sanders E, Fliesler SJ and Wang
JJ: Endoplasmic reticulum stress and the unfolded protein responses
in retinal degeneration. Exp Eye Res. 125:30–40. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Faria JA, Reis PA, Reis MT, Rosado GL,
Pinheiro GL, Mendes GC and Fontes EP: The NAC domain-containing
protein, GmNAC6, is a downstream component of the ER stress- and
osmotic stress-induced NRP-mediated cell-death signaling pathway.
BMC Plant Biol. 11:1292011. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Tsai TC, Lai KH, Su JH, Wu YJ and Sheu JH:
7-Acetylsinumaximol B induces apoptosis and autophagy in human
gastric carcinoma cells through Mitochondria Dysfunction and
activation of the PERK/eIF2α/ATF4/CHOP signalling pathway. Mar
Drugs. 16:1042018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Narasimhan M and Rajasekaran NS: Reductive
potential-a savior turns stressor in protein aggregation
cardiomyopathy. Biochim Biophys Acta. 1852:53–60. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Wu K, Li B, Zhang X, Fang Y, Zeng S, Hu W,
Liu X, Liu X, Lu Z, Li X, et al: Correction for Wu et al.,
‘CSFV restricts necroptosis to sustain infection by inducing
autophagy/mitophagy-targeted degradation of RIPK3’. Microbiol
Spectr. 13:e03188242025. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Koniari I, Velissaris D, Kounis NG, Koufou
E, Artopoulou E, de Gregorio C, Mplani V, Paraskevas T, Tsigkas G,
Hung MY, et al: Anti-diabetic therapy, heart failure and oxidative
stress: An update. J Clin Med. 11:46602022. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Gonen N, Sabath N, Burge CB and Shalgi R:
Widespread PERK-dependent repression of ER targets in response to
ER stress. Sci Rep. 9:43302019. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Nita M and Grzybowski A: Antioxidative
role of heterophagy, autophagy, and mitophagy in the retina and
their association with the age-related macular degeneration (AMD)
etiopathogenesis. Antioxidants (Basel). 12:13682023. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Bae D, Jones RE, Piscopo KM, Tyagi M,
Shepherd JD and Hollien J: Regulation of Blos1 by IRE1 prevents the
accumulation of Huntingtin protein aggregates. Mol Biol Cell.
33:ar1252022. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Jin B, Ishikawa T, Taniguchi M, Ninagawa
S, Okada T, Kagaya S and Mori K: Development of a rapid in vivo
assay to evaluate the efficacy of IRE1-specific inhibitors of the
unfolded protein response using medaka fish. Cell Struct Funct.
45:23–31. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Niu F, Liu W, Ren Y, Tian Y, Shi W, Li M,
Li Y, Xiong Y and Qian L: β-cell neogenesis: A rising star to
rescue diabetes mellitus. J Adv Res. 62:71–89. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Flintoaca Alexandru PR, Chiritoiu GN,
Lixandru D, Zurac S, Ionescu-Targoviste C and Petrescu SM: EDEM1
regulates the insulin mRNA level by inhibiting the endoplasmic
reticulum stress-induced IRE1/JNK/c-Jun pathway. iScience.
26:1079562023. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Flores-Santibáñez F, Medel B, Bernales JI
and Osorio F: Understanding the role of the unfolded protein
response sensor IRE1 in the biology of antigen presenting cells.
Cells. 8:15632019. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Arunagiri A, Haataja L, Cunningham CN,
Shrestha N, Tsai B, Qi L, Liu M and Arvan P: Misfolded proinsulin
in the endoplasmic reticulum during development of beta cell
failure in diabetes. Ann NY Acad Sci. 1418:5–19. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Figueroa-Juárez E, Noriega LG,
Pérez-Monter C, Alemán G, Hernández-Pando R, Correa-Rotter R,
Ramírez V, Tovar AR, Torre-Villalvazo I and Tovar-Palacio C: The
role of the unfolded protein response on renal lipogenesis in
C57BL/6 mice. Biomolecules. 11:732021. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Wu L, He S, Ye W, Shen J, Zhao K, Zhang Y,
Zhang R, Wei J, Cao S, Chen K, et al: Surf4 facilitates
reprogramming by activating the cellular response to endoplasmic
reticulum stress. Cell Prolif. 54:e131332021. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Fu YL, Han DY, Wang YJ, Di XJ, Yu HB and
Mu TW: Remodeling the endoplasmic reticulum proteostasis network
restores proteostasis of pathogenic GABAA receptors. PLoS One.
13:e02079482018. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Wang D, Qu S, Zhang Z, Tan L, Chen X,
Zhong HJ and Chong CM: Strategies targeting endoplasmic reticulum
stress to improve Parkinson's disease. Front Pharmacol.
14:12888942023. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Pérez-Martí A, Ramakrishnan S, Li J,
Dugourd A, Molenaar MR, De La Motte LR, Grand K, Mansouri A,
Parisot M, Lienkamp SS, et al: Reducing lipid bilayer stress by
monounsaturated fatty acids protects renal proximal tubules in
diabetes. Elife. 11:e743912022. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Hollien J and Weissman JS: Decay of
endoplasmic reticulum-localized mRNAs during the unfolded protein
response. Science. 313:104–107. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Huang M, Cai S and Su J: The pathogenesis
of sepsis and potential therapeutic targets. Int J Mol Sci.
20:53762019. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Wise R, Duhachek-Muggy S, Qi Y, Zolkiewski
M and Zolkiewska A: Protein disulfide isomerases in the endoplasmic
reticulum promote anchorage-independent growth of breast cancer
cells. Breast Cancer Res Treat. 157:241–252. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Schäffer DE, Iyer LM, Burroughs AM and
Aravind L: Functional innovation in the evolution of the
calcium-dependent system of the eukaryotic endoplasmic reticulum.
Front Genet. 11:342020. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
AlBashtawi J, Al-Jaber H, Ahmed S and
Al-Mansoori L: Impact of obesity-related endoplasmic reticulum
stress on cancer and associated molecular targets. Biomedicines.
12:7932024. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Park SJ, Li C and Chen YM: Endoplasmic
reticulum calcium homeostasis in kidney disease: Pathogenesis and
therapeutic targets. Am J Pathol. 191:256–265. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Krebs J, Agellon LB and Michalak M: Ca(2+)
homeostasis and endoplasmic reticulum (ER) stress: an integrated
view of calcium signaling. Biochem. Biophys Res Commun.
460:114–121. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Lim W, Yang C, Jeong M, Bazer FW and Song
G: Coumestrol induces mitochondrial dysfunction by stimulating ROS
production and calcium ion influx into mitochondria in human
placental choriocarcinoma cells. Mol Hum Reprod. 23:786–802. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Bahar E, Kim H and Yoon H: ER
stress-mediated signaling: Action potential and ca(2+) as key
players. Int J Mol Sci. 17:15582016. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Zheng Q, Chen Y, Chen D, Zhao H, Feng Y,
Meng Q, Zhao Y and Zhang H: Calcium transients on the ER surface
trigger liquid-liquid phase separation of FIP200 to specify
autophagosome initiation sites. Cell. 185:4082–4098.e22. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Høyer-Hansen M, Bastholm L, Szyniarowski
P, Campanella M, Szabadkai G, Farkas T, Bianchi K, Fehrenbacher N,
Elling F, Rizzuto R, et al: Control of macroautophagy by calcium,
calmodulin-dependent kinase kinase-beta, and Bcl-2. Mol Cell.
25:193–205. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
La Rovere RM, Roest G, Bultynck G and
Parys JB: Intracellular ca(2+) signalling and ca(2+) microdomains
in the control of cell survival, apoptosis and autophagy. Cell
Calcium. 60:74–87. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Sakaki K, Wu J and Kaufman RJ: Protein
kinase Ctheta is required for autophagy in response to stress in
the endoplasmic reticulum. J Biol Chem. 283:15370–15380. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Levin-Salomon V, Bialik S and Kimchi A:
DAP-kinase and autophagy. Apoptosis. 19:346–356. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Zhong Y, Jin C, Han J, Zhu J, Liu Q, Sun
D, Xia X, Zhang Y and Peng X: Diosgenin protects against kidney
Injury and mitochondrial apoptosis Induced by 3-MCPD through the
regulation of ER stress, Ca(2+) homeostasis, and Bcl2 expression.
Mol Nutr Food Res. 65:e20012022021. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Pu Q, Yu L, Wang X, Yan H, Xie Y, Jiang Y
and Yang Z: Immunomodulatory effect of traditional Chinese medicine
combined with systemic therapy on patients with liver cancer: A
systemic review and network meta-analysis. J Cancer. 13:3280–3296.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Wan CP, Gao LX, Hou LF, Yang XQ, He PL,
Yang YF, Tang W, Yue JM, Li J and Zuo JP: Astragaloside II triggers
T cell activation through regulation of CD45 protein tyrosine
phosphatase activity. Acta Pharmacol Sin. 34:522–530. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Shen L, Luo H, Fan L, Tian X, Tang A, Wu
X, Dong K and Su Z: Potential immunoregulatory mechanism of plant
saponins: A review. Molecules. 29:1132023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Nalbantsoy A, Nesil T, Yılmaz-Dilsiz O,
Aksu G, Khan S and Bedir E: Evaluation of the immunomodulatory
properties in mice and in vitro anti-inflammatory activity of
cycloartane type saponins from Astragalus species. J
Ethnopharmacol. 139:574–581. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Ju Y, Su Y, Chen Q, Ma K, Ji T, Wang Z and
Li W and Li W: Protective effects of astragaloside IV on
endoplasmic reticulum stress-induced renal tubular epithelial cells
apoptosis in type 2 diabetic nephropathy rats. Biomed Pharmacother.
109:84–92. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Chen Y, Gui D, Chen J, He D, Luo Y and
Wang N: Down-regulation of PERK-ATF4-CHOP pathway by astragaloside
IV is associated with the inhibition of endoplasmic reticulum
stress-induced podocyte apoptosis in diabetic rats. Cell Physiol
Biochem. 33:1975–1987. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Wang ZS, Xiong F, Xie XH, Chen D, Pan JH
and Cheng L: Astragaloside IV attenuates proteinuria in
streptozotocin-induced diabetic nephropathy via the inhibition of
endoplasmic reticulum stress. BMC Nephrol. 16:442015. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Li M, Wang W, Xue J, Gu Y and Lin S:
Meta-analysis of the clinical value of Astragalus membranaceus in
diabetic nephropathy. J Ethnopharmacol. 133:412–419. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Xue HZ, Chen Y, Wang SD, Yang YM, Cai LQ,
Zhao JX, Huang WJ and Xiao YH: Radix astragali and its
representative extracts for diabetic nephropathy: Efficacy and
molecular mechanism. J Diabetes Res. 2024:52161132024. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Li Y and Wang J: Possible mechanism for
the protective effect of active ingredients of astragalus
membranaceus on diabetes nephropathy. J Asian Nat Prod Res.
26:1276–1284. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Zhang L, Shergis JL, Yang L, Zhang AL, Guo
X, Zhang L, Zhou S, Zeng L, Mao W and Xue CC: Astragalus
membranaceus (Huang Qi) as adjunctive therapy for diabetic kidney
disease: an updated systematic review and meta-analysis. J
Ethnopharmacol. 239:1119212019. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Guo JC, Pan HC, Yeh BY, Lu YC, Chen JL,
Yang CW, Chen YC, Lin YH and Chen HY: Associations between using
Chinese herbal medicine and long-term outcome among pre-dialysis
diabetic nephropathy patients: A retrospective population-based
cohort study. Front Pharmacol. 12:6165222021. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Cao JL, Liang LY, Liu YH, Xu ZM, Wang X,
Wei WX, Wan HJ, Lyu XH, Li WX, Zhang YX, et al: Medication rules of
Astragali Radix in ancient Chinese medical books based on
‘disease-medicine-dose’ pattern. Zhongguo Zhong Yao Za Zhi.
50:798–811. 2025.(In Chinese). PubMed/NCBI
|
|
159
|
Hassanpour Fard M, Naseh G, Lotfi N,
Hosseini SM and Hosseini M: Effects of aqueous extract of turnip
leaf (Brassica rapa) in alloxan-induced diabetic rats. Avicenna J
Phytomed. 5:148–156. 2015.PubMed/NCBI
|
|
160
|
Santos M, Fortunato RH and Spotorno VG:
Analysis of flavonoid glycosides with potential medicinal
properties on Bauhinia uruguayensis and Bauhinia forficate
subspecies pruinosa. Nat Prod Res. 33:2574–2578. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Chhatre S, Nesari T, Somani G, Kanchan D
and Sathaye S: Phytopharmacological overview of Tribulus
terrestris. Pharmacogn Rev. 8:45–51. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Abdou HM and Abd Elkader HAE: The
potential therapeutic effects of Trifolium alexandrinum extract,
hesperetin and quercetin against diabetic nephropathy via
attenuation of oxidative stress, inflammation, GSK-3β and apoptosis
in male rats. Chem Biol Interact. 352:1097812022. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Alshehri AS: Kaempferol attenuates
diabetic nephropathy in streptozotocin-induced diabetic rats by a
hypoglycaemic effect and concomitant activation of the
Nrf-2/Ho-1/antioxidants axis. Arch Physiol Biochem. 129:984–997.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Luo W, Chen X, Ye L, Chen X, Jia W, Zhao
Y, Samorodov AV, Zhang Y, Hu X, Zhuang F, et al: Kaempferol
attenuates streptozotocin-induced diabetic nephropathy by
downregulating TRAF6 expression: The role of TRAF6 in diabetic
nephropathy. J Ethnopharmacol. 268:1135532021. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Li K, Wang YJ, Chen C, Wang XJ and Li W:
Targeting pyroptosis: A novel strategy of ginseng for the treatment
of diabetes and its chronic complications. Phytomedicine.
138:1564302025. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Wu W, Hu W, Han WB, Liu YL, Tu Y, Yang HM,
Fang QJ, Zhou MY, Wan ZY, Tang RM, et al: Inhibition of
Akt/mTOR/p70S6K signaling activity with Huangkui capsule alleviates
the early glomerular pathological changes in diabetic nephropathy.
Front Pharmacol. 9:4432018. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Han W, Ma Q, Liu Y, Wu W, Tu Y, Huang L,
Long Y, Wang W, Yee H, Wan Z, et al: Huangkui capsule alleviates
renal tubular epithelial-mesenchymal transition in diabetic
nephropathy via inhibiting NLRP3 inflammasome activation and
TLR4/NF-κB signaling. Phytomedicine. 57:203–214. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Zhu Z, Luan G, Peng S, Fang Y, Fang Q,
Shen S, Wu K, Qian S, Jia W, Ye J and Wei L: Huangkui capsule
attenuates diabetic kidney disease through the induction of
mitophagy mediated by STING1/ PINK1 signaling in tubular cells.
Phytomedicine. 119:1549752023. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Shi R, Tao Y, Tang H, Wu C, Fei J, Ge H,
Gu HF and Wu J: Abelmoschus Manihot ameliorates the levels of
circulating metabolites in diabetic nephropathy by modulating gut
microbiota in non-obese diabetes mice. Microb Biotechnol.
16:813–826. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Zhang L, Li P, Xing CY, Zhao JY, He YN,
Wang JQ, Wu XF, Liu ZS, Zhang AP, Lin HL, et al: Efficacy and
safety of Abelmoschus manihot for primary glomerular disease: A
prospective, multicenter randomized controlled clinical trial. Am J
Kidney Dis. 64:57–65. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Chen X, Yu Y, Xiao C, Li S, Wu T, Wu H, Li
X, Lin C, Chen X, Guo X and Liu S: Huangkui capsules for diabetic
nephropathy: Comprehensive review of efficacy and molecular
mechanisms. Phytomedicine. 147:1572072025. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Liu X, Zhang C, Fu Y, Dai J, Lu J, Liu G
and Yang X: Huangkui capsule combined with finerenone attenuates
diabetic nephropathy by regulating the JAK2/STAT3 signaling pathway
based on network pharmacology, molecular docking, and experimental
verification. Front Pharmacol. 16:16252862025. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Wang Y, He X, Xue M, Yu H, He Q and Jin J:
Integrated 16S rRNA sequencing and metabolomic analysis reveals the
potential protective mechanism of Germacrone on diabetic
nephropathy in mice. Acta Biochim Biophys Sin (Shanghai).
56:414–426. 2024.PubMed/NCBI
|
|
174
|
Lee HS, Suh JY, Kang BC and Lee E:
Lipotoxicity dysregulates the immunoproteasome in podocytes and
kidneys in type 2 diabetes. Am J Physiol Renal Physiol.
320:F548–F558. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Nemecz M, Constantin A, Dumitrescu M,
Alexandru N, Filippi A, Tanko G and Georgescu A: The distinct
effects of palmitic and oleic acid on pancreatic beta cell
function: The elucidation of associated mechanisms and effector
molecules. Front Pharmacol. 9:15542021. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Liu Y, Jia Z, Liu S, Downton M, Liu G, Du
Y and Yang T: Combined losartan and nitro-oleic acid remarkably
improves diabetic nephropathy in mice. Am J Physiol Renal Physiol.
305:F1555–F1562. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Liu Y, Hu Z, Xing H, Kang L, Chen X, Liu B
and Niu K: Renoprotective effects of oleanolic acid and its
possible mechanisms in rats with diabetic kidney disease. Biochem
Biophys Res Commun. 636((Pt 1)): 1–9. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Lee ES, Kim HM, Kang JS, Lee EY, Yadav D,
Kwon MH, Kim YM, Kim HS and Chung CH: Oleanolic acid and
N-acetylcysteine ameliorate diabetic nephropathy through reduction
of oxidative stress and endoplasmic reticulum stress in a type 2
diabetic rat model. Nephrol Dial Transplant. 31:391–400. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Gao D, Li Q, Li Y, Liu Z, Liu Z, Fan Y,
Han Z, Li J and Li K: Antidiabetic potential of oleanolic acid from
Ligustrum lucidum Ait. Can J Physiol Pharmacol. 85:1076–1083. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Sultana N and Ata A: Oleanolic acid and
related derivatives as medicinally important compounds. J Enzyme
Inhib Med Chem. 23:739–756. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Abudoureyimu A, Chen C, Hu Y, Nuermaimaiti
D and Liu T: Quercetin alleviates diabetic nephropathy by
inhibiting M1 macrophage polarization via targeting NLRC5/NLRP3
pathway. Cell Immunol. 414:1049972025. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Yan X, Li P, Liu C, Yin F, Han J, Sun H,
Zheng Y, Chen X, Guan S and Wang X: Exploring the molecular
mechanisms for renoprotective effects of Huangkui capsule on
diabetic nephropathy mice by comprehensive serum metabolomics
analysis. J Ethnopharmacol. 340:1192232025. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Brito JCM, Lima WG, Cordeiro LPB and da
Cruz Nizer WS: Effectiveness of supplementation with quercetin-type
flavonols for treatment of viral lower respiratory tract
infections: Systematic review and meta-analysis of preclinical
studies. Phytother Res. 35:4930–4942. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Hu Q, Qu C, Xiao X, Zhang W, Jiang Y, Wu
Z, Song D, Peng X, Ma X and Zhao Y: Flavonoids on diabetic
nephropathy: Advances and therapeutic opportunities. Chin Med.
16:742021. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Hu T, Yue J, Tang Q, Cheng KW, Chen F,
Peng M, Zhou Q and Wang M: The effect of quercetin on diabetic
nephropathy (DN): A systematic review and meta-analysis of animal
studies. Food Funct. 13:4789–4803. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Lai WF and Wong WT: Design and
optimization of quercetin-based functional foods. Crit Rev Food Sci
Nutr. 62:7319–7335. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Albadrani GM, Binmowyna MN, Bin-Jumah MN,
El-Akabawy G, Aldera H and Al-Farga AM: Quercetin protects against
experimentally-induced myocardial infarction in rats by an
antioxidant potential and concomitant activation of signal
transducer and activator of transcription 3. J Physiol Pharmacol.
Apri 22–2020.(Epub ahead of print). PubMed/NCBI
|
|
188
|
Dini S, Zakeri M, Ebrahimpour S,
Dehghanian F and Esmaeili A: Quercetin-conjugated superparamagnetic
iron oxide nanoparticles modulate glucose metabolism-related genes
and miR-29 family in the hippocampus of diabetic rats. Sci Rep.
11:86182021. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Suganya N, Dornadula S, Chatterjee S and
Mohanram RK: Quercetin improves endothelial function in diabetic
rats through inhibition of endoplasmic reticulum stress-mediated
oxidative stress. Eur J Pharmacol. 819:80–88. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Zhong Y, Lee K, Deng Y, Ma Y, Chen Y, Li
X, Wei C, Yang S, Wang T, Wong J, et al: Arctigenin attenuates
diabetic kidney disease through the activation of PP2A in
podocytes. Nat Commun. 10:45232019. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Li X, Wang J, Yan J, He JC, Li Y and Zhong
Y: Additive renal protective effects between arctigenin and
puerarin in diabetic kidney disease. Biomed Pharmacother.
171:1161072024. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Medras ZJH, Mostafa YM, Ahmed AAM and
El-Sayed NM: Arctigenin improves neuropathy via ameliorating
apoptosis and modulating autophagy in streptozotocin-induced
diabetic mice. CNS Neurosci Ther. 29:3068–3080. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Wang W, Qiu L, Howard A, Solis N, Li C,
Wang X, Kopp JB and Levi M: Protective effects of aliskiren and
valsartan in mice with diabetic nephropathy. J Renin Angiotensin
Aldosterone Syst. 15:384–395. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
López V, Martin M, Cobelo C, Aranda P,
Cabello M, Sola E, Gutierrez C, Burgos D, Martínez D and Hernandez
D: Renin-angiotensin system dual blockade using angiotensin
receptor plus aliskiren decreases severe proteinuria in kidney
transplant recipients. Transplant Proc. 42:2883–2885. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Sun Y, Peng PA, Ma Y, Liu XL, Yu Y, Jia S,
Xu XH, Wu SJ and Zhou YJ: Valsartan protects against
contrast-induced acute kidney injury in rats by inhibiting
endoplasmic reticulum stress-induced apoptosis. Curr Vasc
Pharmacol. 15:174–183. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Li C, Lin Y, Luo R, Chen S, Wang F, Zheng
P, Levi M, Yang T and Wang W: Intrarenal renin-angiotensin system
mediates fatty acid-induced ER stress in the kidney. Am J Physiol
Renal Physiol. 310:F351–F363. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Kidney Disease: Improving Global Outcomes
(KDIGO) Diabetes Work Group: KDIGO 2020 clinical practice guideline
for diabetes management in chronic kidney disease. Kidney Int.
98((4S)): S1–S115. 2020.
|
|
198
|
Chang TT, Wu TC, Huang PH, Lin CP, Chen
JS, Lin LY, Lin SJ and Chen JW: Direct renin inhibition with
aliskiren improves ischemia-induced neovasculogenesis in diabetic
animals via the SDF-1 related mechanism. PLoS One. 10:e01366272015.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Wang Z, Yuan A, Liu C, Liu Y, Qiao L, Xu
Z, Bi S, Tian J, Yu B, Lin Z, et al: Identification of key
antifibrotic targets FPR1, TAS2R5, and LRP2BP of valsartan in
diabetic nephropathy: A transcriptomics-driven study integrating
machine learning, molecular docking, and dynamics simulations. Int
J Biol Macromol. 297:1398422025. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Zhang H, Liu G, Zhou W, Zhang W, Wang K
and Zhang J: Neprilysin inhibitor-angiotensin II receptor blocker
combination therapy (Sacubitril/valsartan) suppresses
atherosclerotic plaque formation and inhibits inflammation in
apolipoprotein E-deficient mice. Sci Rep. 9:65092019. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Ma Y, Xie D, Liu J, Han X, Xu H and Chen
Y: Angiopoietin-like protein 3 deficiency combined with valsartan
administration protects better against podocyte damage in
streptozotocin-induced diabetic nephropathy mice. Int
Immunopharmacol. 115:1097152023. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Kidney Disease: Improving Global Outcomes
(KDIGO) CKD Work Group: KDIGO 2024 clinical practice guideline for
the evaluation and management of chronic kidney disease. Kidney
Int. 105:S117–S314. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Mori K, Togo A, Ohta K, Asahi T, Nozaki C
and Kataoka K: CB1 receptor agonist ACEA Resists ER stress-mediated
apoptosis via CB1R-independent mechanism. Biol Pharm Bull.
48:769–781. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Vasincu A, Rusu RN, Ababei DC, Neamțu M,
Arcan OD, Macadan I, Beșchea Chiriac S, Bild W and Bild V:
Exploring the therapeutic potential of cannabinoid receptor
antagonists in inflammation, diabetes mellitus, and obesity.
Biomedicines. 11:16672023. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Aguirre CA, Castillo VA and Llanos MN:
Excess of the endocannabinoid anandamide during lactation induces
overweight, fat accumulation and insulin resistance in adult mice.
Diabetol Metab Syndr. 4:352012. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Kumawat VS and Kaur G: Cannabinoid
receptor 2 (CB2) agonists and L-arginine ameliorate diabetic
nephropathy in rats by suppressing inflammation and fibrosis
through NF-κβ pathway. Naunyn Schmiedebergs Arch Pharmacol.
397:381–393. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Jourdan T, Park JK, Varga ZV, Pálóczi J,
Coffey NJ, Rosenberg AZ, Godlewski G, Cinar R, Mackie K, Pacher P
and Kunos G: Cannabinoid-1 receptor deletion in podocytes mitigates
both glomerular and tubular dysfunction in a mouse model of
diabetic nephropathy. Diabetes Obes Metab. 20:698–708. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Barutta F, Piscitelli F, Pinach S, Bruno
G, Gambino R, Rastaldi MP, Salvidio G, Di Marzo V, Cavallo Perin P
and Gruden G: Protective role of cannabinoid receptor type 2 in a
mouse model of diabetic nephropathy. Diabetes. 60:2386–2396. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Yang HM, Kim J, Kim BK, Seo HJ, Kim JY,
Lee JE, Lee J, You J, Jin S, Kwon YW, et al: Resistin regulates
inflammation and insulin resistance in humans via the
endocannabinoid system. Research (Wash D C). 7:03262024.PubMed/NCBI
|
|
210
|
Lim JC, Lim SK, Park MJ, Kim GY, Han HJ
and Park SH: Cannabinoid receptor 1 mediates high glucose induced
apoptosis via endoplasmic reticulum stress in primary cultured rat
mesangial cells. Am J Physiol Renal Physiol. 301:F179–F188. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Lim JC, Lim SK, Han HJ and Park SH:
Cannabinoid receptor 1 mediates palmitic acid-induced apoptosis via
endoplasmic reticulum stress in human renal proximal tubular cells.
J Cell Physiol. 225:654–663. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Panlilio LV, Goldberg SR and Justinova Z:
Cannabinoid abuse and addiction: Clinical and preclinical findings.
Clin Pharmacol Ther. 97:616–627. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Cinar R, Iyer MR and Kunos G: The
therapeutic potential of second and third generation
CB1R antagonists. Pharmacol Ther. 208:1074772020.
View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Mińczuk K, Baranowska-Kuczko M, Krzyżewska
A, Schlicker E and Malinowska B: Cross-Talk between the
(Endo)cannabinoid and renin-angiotensin systems: Basic evidence and
potential therapeutic significance. Int J Mol Sci. 23:63502022.
View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Jourdan T, Szanda G, Cinar R, Godlewski G,
Holovac DJ, Park JK, Nicoloro S, Shen Y, Liu J, Rosenberg AZ, et
al: Developmental role of macrophage cannabinoid-1 receptor
signaling in type 2 diabetes. Diabetes. 66:994–1007. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Jourdan T, Szanda G, Rosenberg AZ, Tam J,
Earley BJ, Godlewski G, Cinar R, Liu Z, Liu J, Ju C, et al:
Overactive cannabinoid 1 receptor in podocytes drives type 2
diabetic nephropathy. Proc Natl Acad Sci USA. 111:E5420–E5428.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Ghosh A, Peyot ML, Leung YH, Ravenelle F,
Madiraju SRM and Prentki M: A peripherally restricted cannabinoid-1
receptor inverse agonist promotes insulin secretion and protects
from cytokine toxicity in human pancreatic islets. Eur J Pharmacol.
944:1755892023. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Rajesh M, Bátkai S, Kechrid M,
Mukhopadhyay P, Lee WS, Horváth B, Holovac E, Cinar R, Liaudet L,
Mackie K, et al: Cannabinoid 1 receptor promotes cardiac
dysfunction, oxidative stress, inflammation, and fibrosis in
diabetic cardiomyopathy. Diabetes. 61:716–727. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Haspula D and Clark MA: Cannabinoid
receptors: An update on cell signaling, pathophysiological roles
and therapeutic opportunities in neurological, cardiovascular, and
inflammatory diseases. Int J Mol Sci. 21:76932020. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Pointeau O, Ba AI, Geissler A, Barbosa R,
Basu A, Muhammad A, Nivot M, Loriot M, Leemput J, Passilly-Degrace
P, et al: Blockade of cannabinoid CB receptors potentiates the
anti-fibrotic effects mediated by SGLT2 inhibition in a mouse model
of diabetic nephropathy. Br J Pharmacol. 182:5355–5377. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Dagon Y, Avraham Y, Link G, Zolotarev O,
Mechoulam R and Berry EM: The synthetic cannabinoid HU-210
attenuates neural damage in diabetic mice and hyperglycemic
pheochromocytoma PC12 cells. Neurobiol Dis. 27:174–181. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Navas-Madroñal M, Almendra-Pegueros R,
Puertas-Umbert L, Jiménez-Altayó F, Julve J, Pérez B,
Consegal-Pérez M, Kassan M, Martínez-González J, Rodriguez C and
Galán M: Targeting mitochondrial stress with szeto-schiller 31
prevents experimental abdominal aortic aneurysm: Crosstalk with
endoplasmic reticulum stress. Br J Pharmacol. 180:2230–2249. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Hinden L, Udi S, Drori A, Gammal A,
Nemirovski A, Hadar R, Baraghithy S, Permyakova A, Geron M, Cohen
M, et al: Modulation of renal GLUT2 by the cannabinoid-1 receptor:
Implications for the treatment of diabetic nephropathy. J Am Soc
Nephrol. 29:434–448. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Kusaczuk M: Tauroursodeoxycholate-bile
acid with chaperoning activity: Molecular and cellular effects and
therapeutic perspectives. Cell. 8:14712019. View Article : Google Scholar
|
|
225
|
Zheng P, Lin Y, Wang F, Luo R, Zhang T, Hu
S, Feng P, Liang X, Li C and Wang W: 4-PBA improves lithiuminduced
nephrogenic diabetes insipidus by attenuating ER stress. Am J
Physiol Renal Physiol. 311:F763–F776. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Guo Q, Xu L, Li H, Sun H, Wu S and Zhou B:
4-PBA reverses autophagic dysfunction and improves insulin
sensitivity in adipose tissue of obese mice via Akt/mTOR signaling.
Biochem Biophys Res Commun. 484:529–535. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Ji X, Yao L, Wang M, Liu X, Peng S, Li K,
Xu M, Shen N, Luo L and Sun C: Cystatin C attenuates insulin
signaling transduction by promoting endoplasmic reticulum stress in
hepatocytes. FEBS Lett. 589((24 Part B)): 3938–3944. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Cao AL, Wang L, Chen X, Wang YM, Guo HJ,
Chu S, Liu C, Zhang XM and Peng W: Ursodeoxycholic acid and
4-phenylbutyrate prevent endoplasmic reticulum stressinduced
podocyte apoptosis in diabetic nephropathy. Lab Invest. 96:610–622.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Qin L, Wang Z, Tao L and Wang Y: ER stress
negatively regulates AKT/TSC/mTOR pathway to enhance autophagy.
Autophagy. 6:239–247. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Ni L, Yuan C and Wu X: Endoplasmic
reticulum stress in diabetic nephrology: Regulation, pathological
role, and therapeutic potential. Oxid Med Cell Longev.
2021:72779662021. View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Chang YC, Hee SW, Hsieh ML, Jeng YM and
Chuang LM: The role of organelle stresses in diabetes mellitus and
obesity: Implication for treatment. Anal Cell Pathol (Amst).
2015:9728912015.PubMed/NCBI
|
|
232
|
De Miguel C, Sedaka R, Kasztan M, Lever
JM, Sonnenberger M, Abad A, Jin C, Carmines PK, Pollock DM and
Pollock JS: Tauroursodeoxycholic acid (TUDCA) abolishes chronic
high salt-induced renal injury and inflammation. Acta Physiol
(Oxf). 226:e132272019. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Microvascular Complications Group of
Chinese Diabetes Society, . Clinical guideline for the prevention
and treatment of diabetic kidney disease in China (2021 edition).
Chin J Diabetes Mellitus. 13:762–784. 2021.(In Chinese).
|
|
234
|
Li Z, Li Y, Overstreet JM, Chung S, Niu A,
Fan X, Wang S, Wang Y, Zhang MZ and Harris RC: Inhibition of
epidermal growth factor receptor activation is associated with
improved diabetic nephropathy and insulin resistance in type 2
diabetes. Diabetes. 67:1847–1857. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Rao VRA/LBV, Tan SH, Candasamy M and
Bhattamisra SK: Diabetic nephropathy: An update on pathogenesis and
drug development. Diabetes Metab Syndr. 13:754–762. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Shih JY, Lin YW, Fisch S, Cheng JT, Kang
NW, Hong CS, Chen ZC and Chang WT: Dapagliflozin suppresses ER
stress and improves subclinical myocardial function in diabetes.
From bedside to bench. Diabetes. 70:262–267. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Yu Y, Xia X, Li H, Zhang Y, Zhou X and
Jiang H: A new rhodopsin R135W mutation induces endoplasmic
reticulum stress and apoptosis in retinal pigment epithelial cells.
J Cell Physiol. 234:14100–14108. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
238
|
Fang Q, Zou C, Zhong P, Lin F, Li W, Wang
L, Zhang Y, Zheng C, Wang Y, Li X and Liang G: EGFR mediates
hyperlipidemia-induced renal injury via regulating inflammation and
oxidative stress: The detrimental role and mechanism of EGFR
activation. Oncotarget. 7:24361–24373. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
239
|
Martín-Pérez R, Palacios C, Yerbes R,
Cano-González A, Iglesias-Serret D, Gil J, Reginato MJ and
López-Rivas A: Activated ERBB2/HER2 licenses sensitivity to
apoptosis upon endoplasmic reticulum stress through a
PERK-dependent pathway. Cancer Res. 74:1766–1777. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
240
|
Zhou K, Zi X, Song J, Zhao Q, Liu J, Bao H
and Li L: Molecular mechanistic pathways targeted by natural
compounds in the prevention and treatment of diabetic kidney
disease. Molecules. 27:62212022. View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Zhang L, Li C, Fu L, Yu Z, Xu G, Zhou J,
Shen M, Feng Z, Zhu H, Xie T, et al: Protection of catalpol against
triptolide-induced hepatotoxicity by inhibiting excessive autophagy
via the PERK-ATF4-CHOP pathway. PeerJ. 10:e127592022. View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Itoh T, Hatano R, Horimoto Y, Yamada T,
Song D, Otsuka H, Shirakawa Y, Mastuoka S, Iwao N, Aune TM, et al:
IL-26 mediates epidermal growth factor receptor-tyrosine kinase
inhibitor resistance through endoplasmic reticulum stress signaling
pathway in triple-negative breast cancer cells. Cell Death Dis.
12:5202021. View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Wang L, Ding S, Hu Y, Su J, Zhu G, Hong H,
Hou B, Dong Z, Xue Z, Wang J, et al: Targeting programmed cell
death pathways: Emerging therapeutic strategies for diabetic kidney
disease. Front Endocrinol (Lausanne). 16:15138952025. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Mima A: A narrative review of diabetic
kidney disease: Previous and current evidence-based therapeutic
approaches. Adv Ther. 39:3488–3500. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
245
|
Kume S and Maegawa H: Lipotoxicity,
nutrient-sensing signals, and autophagy in diabetic nephropathy.
JMA J. 3:87–94. 2020.PubMed/NCBI
|
|
246
|
Beal B, Buizen L, Yeung EK, Heath L,
Houston L, Cherney DZI, Jardine M, Pollock C, Arnott C, Kotwal SS,
et al: Effects of SGLT2 inhibition on insulin use in CKD and type 2
diabetes: Insights from the CREDENCE trial. Nephrol Dial
Transplant. 40:1727–1735. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
247
|
Kidney Disease: Improving Global Outcomes
(KDIGO) Diabetes Work Group: KDIGO 2022 clinical practice guideline
for diabetes management in chronic kidney disease. Kidney Int.
102((5S)): S1–S127. 2022.
|
|
248
|
Wanner C, Inzucchi SE, Lachin JM, Fitchett
D, von Eynatten M, Mattheus M, Johansen OE, Woerle HJ, Broedl UC
and Zinman B; EMPA-REG OUTCOME Investigators, : Empagliflozin and
progression of kidney disease in type 2 diabetes. N Engl J Med.
375:323–334. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
249
|
Neal B, Perkovic V, Mahaffey KW, de Zeeuw
D, Fulcher G, Erondu N, Shaw W, Law G, Desai M and Matthews DR;
CANVAS Program Collaborative Group, : Canagliflozin and
cardiovascular and renal events in type 2 diabetes. N Engl J Med.
377:644–657. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Wiviott SD, Razl Bonaca MP, Mosenzon O,
Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, et
al: Dapagliflozinand cardiovascular outcomes in type 2 diabetes. N
Engl J Med. 380:347–357. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
251
|
Perkovic V, Jardine MJ, Neal B, Bompoint
S, Heerspink HJL, Charytan DM, Edwards R, Agarwal R, Bakris G, Bull
S, et al: Canagliflozin and renal outcomes in type 2 diabetes and
nephropathy. N Engl J Med. 380:2295–2306. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
252
|
Barreiro E, Salazar-Degracia A,
Sancho-Muñoz A and Gea J: Endoplasmic reticulum stress and unfolded
protein response profile in quadriceps of sarcopenic patients with
respiratory diseases. J Cell Physiol. 234:11315–11329. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
253
|
Bohnert KR, McMillan JD and Kumar A:
Emerging roles of ER stress and unfolded protein response pathways
in skeletal muscle health and disease. J Cell Physiol. 233:67–78.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
254
|
Liu CY, Hsu CC, Huang TT, Lee CH, Chen JL,
Yang SH, Jiang JK, Chen WS, Lee KD and Teng HW: ER stress-related
ATF6 upregulates CIP2A and contributes to poor prognosis of colon
cancer. Mol Oncol. 12:1706–1717. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Ryu D, Seo WY, Yoon YS, Kim YN, Kim SS,
Kim HJ, Park TS, Choi CS and Koo SH: Endoplasmic reticulum stress
promotes LIPIN2-dependent hepatic insulin resistance. Diabetes.
60:1072–1081. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Toma L, Stancu CS and Sima AV: Endothelial
dysfunction in diabetes is aggravated by glycated lipoproteins;
Novel molecular therapies. Biomedicines. 9:182020. View Article : Google Scholar : PubMed/NCBI
|
|
257
|
Marciniak SJ, Chambers JE and Ron D:
Pharmacological targeting of endoplasmic reticulum stress in
disease. Nat Rev Drug Discov. 21:115–140. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
Anumas S and Inagi R: Mitigating
lipotoxicity: A potential mechanism to delay chronic kidney disease
progression using current pharmacological therapies. Nephrology
(Carlton). 30:e700982025. View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Zheng T, Luo L, Wang X, Deng X and Xue M:
Canagliflozin ameliorates diabetic podocyte damage via enriching
mitochondria-associated endoplasmic reticulum membranes. Cell
Signal. 135:1120382025. View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Wang Y, Liu S, Zhu F, Wang X, Wang H, Long
L, Xiao J and Guo C: Improved bioavailability and
anti-nephrotoxicity efficacy of polydatin on cisplatin-induced AKI
via a dual-targeting fucoidan delivery system. Int J Pharm X.
10:1004222025.PubMed/NCBI
|
|
261
|
Fang Z, Liu R, Xie J and He JC: Molecular
mechanism of renal lipid accumulation in diabetic kidney disease. J
Cell Mol Med. 28:e183642024. View Article : Google Scholar : PubMed/NCBI
|
|
262
|
Ye S, Cheng Z, Zhuo D and Liu S: Different
types of cell death in diabetic neuropathy: A focus on mechanisms
and therapeutic strategies. Int J Mol Sci. 25:81262024. View Article : Google Scholar : PubMed/NCBI
|
|
263
|
Ghemrawi R, Battaglia-Hsu SF and Arnold C:
Endoplasmic reticulum stress in metabolic disorders. Cells.
7:632018. View Article : Google Scholar : PubMed/NCBI
|
|
264
|
Chattopadhyay A, Kwartler CS, Kaw K, Li Y,
Kaw A, Chen J, LeMaire SA, Shen YH and Milewicz DM:
Cholesterol-induced phenotypic modulation of smooth muscle cells to
macrophage/fibroblast-like cells is driven by an unfolded protein
response. Arterioscler Thromb Vasc Biol. 41:302–316. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
265
|
Dong L, Xu M, Li Y, Xu W, Wu C, Zheng H,
Xiao Z, Sun G, Ding L, Li X, et al: SMURF1 attenuates endoplasmic
reticulum stress by promoting the degradation of KEAP1 to activate
NRF2 antioxidant pathway. Cell Death Dis. 14:3612023. View Article : Google Scholar : PubMed/NCBI
|
|
266
|
Rabhi N, Denechaud PD, Gromada X, Hannou
SA, Zhang H, Rashid T, Salas E, Durand E, Sand O, Bonnefond A, et
al: KAT2B is required for pancreatic beta cell adaptation to
metabolic stress by controlling the unfolded protein response. Cell
Rep. 15:1051–1061. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Wang Y, Zhou X, Zhao D, Wang X, Gurley EC,
Liu R, Li X, Hylemon PB, Chen W and Zhou H: Berberine inhibits free
fatty acid and LPS-induced inflammation via modulating ER stress
response in macrophages and hepatocytes. PLoS One. 15:e02326302020.
View Article : Google Scholar : PubMed/NCBI
|
|
268
|
Yang M, Liu C, Jiang N, Liu Y, Luo S, Li
C, Zhao H, Han Y, Chen W, Li L, et al: Endoplasmic reticulum
homeostasis: A potential target for diabetic nephropathy. Front
Endocrinol (Lausanne). 14:11828482023. View Article : Google Scholar : PubMed/NCBI
|
|
269
|
Pennell JP: Optimizing medical management
of patients with pre-end-stage renal disease. Am J Med.
111:559–568. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Kim SH, Yoo JH, Lee WJ and Park CY:
Gemigliptin: An update of its clinical use in the management of
type 2 diabetes mellitus. Diabetes Metab J. 40:339–353. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
271
|
Frățilă VG, Lupușoru G, Sorohan BM,
Obrișcă B, Mocanu V, Lupușoru M and Ismail G: Nephrotic Syndrome:
From pathophysiology to novel therapeutic approaches. Biomedicines.
12:5692024. View Article : Google Scholar : PubMed/NCBI
|