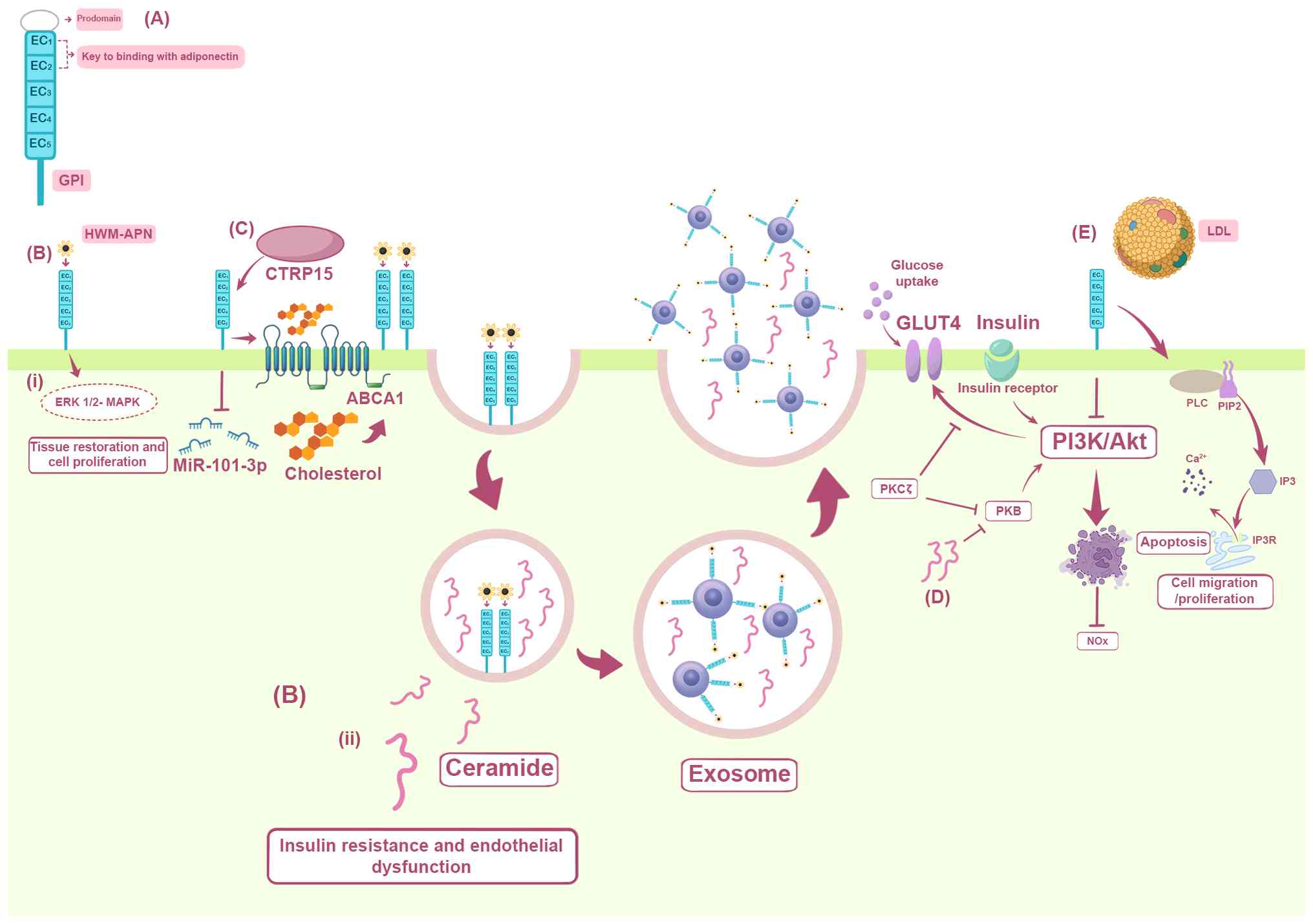
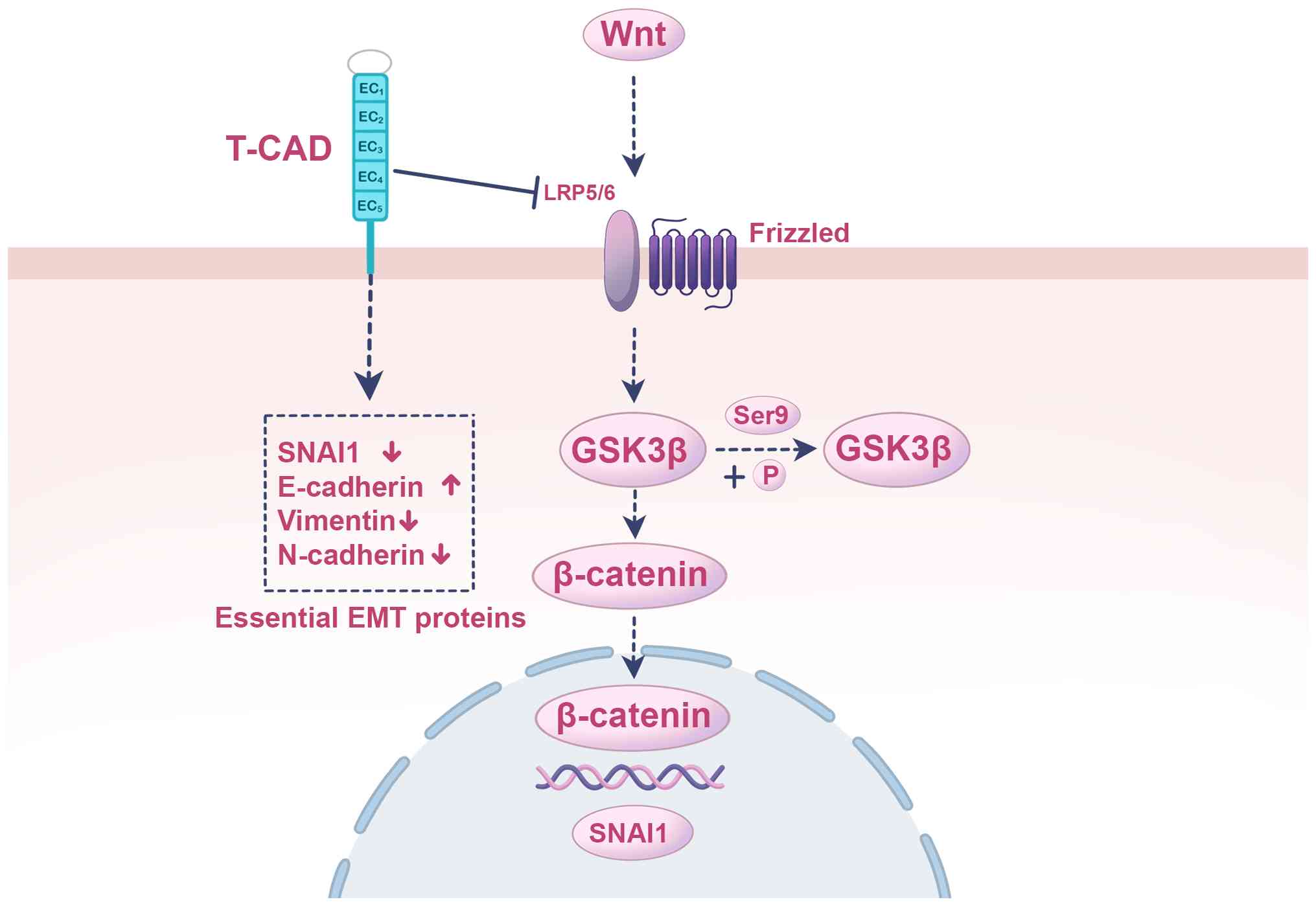
|
1
|
Roth GA, Mensah GA, Johnson CO, Addolorato
G, Ammirati E, Baddour LM, Barengo NC, Beaton AZ, Benjamin EJ,
Benziger CP, et al: Global burden of cardiovascular diseases and
risk factors, 1990–2019: Update from the GBD 2019 study. J Am Coll
Cardiol. 76:2982–3021. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wu Z, Xia F and Lin R: Global burden of
cancer and associated risk factors in 204 countries and
territories, 1980–2021: A systematic analysis for the GBD 2021. J
Hematol Oncol. 17:1192024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
GBD 2021 Diabetes Collaborators, . Global,
regional, and national burden of diabetes from 1990 to 2021, with
projections of prevalence to 2050: A systematic analysis for the
global burden of disease study 2021. Lancet. 402:203–234. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Spaander MCW, Zauber AG, Syngal S, Blaser
MJ, Sung JJ, You YN and Kuipers EJ: Young-onset colorectal cancer.
Nat Rev Dis Primers. 9:212023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Misra S, Ke C, Srinivasan S, Goyal A,
Nyriyenda MJ, Florez JC, Khunti K, Magliano DJ and Luk A: Current
insights and emerging trends in early-onset type 2 diabetes. Lancet
Diabetes Endocrinol. 11:768–782. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mousavi I, Suffredini J, Virani SS,
Ballantyne CM, Michos ED, Misra A, Saeed A and Jia X: Early-onset
atherosclerotic cardiovascular disease. Eur J Prev Cardiol.
32:100–112. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sun J, Qiao Y, Zhao M, Magnussen CG and Xi
B: Global, regional, and national burden of cardiovascular diseases
in youths and young adults aged 15–39 years in 204
countries/territories, 1990–2019: A systematic analysis of global
burden of disease study 2019. BMC Med. 21:2222023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ciumărnean L, Milaciu MV, Negrean V,
Orășan OH, Vesa SC, Sălăgean O, Iluţ S and Vlaicu SI:
Cardiovascular risk factors and physical activity for the
prevention of cardiovascular diseases in the elderly. Int J Environ
Res Public Health. 19:2072021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rouland A, Thuillier P, Al-Salameh A,
Benzerouk F, Bahougne T, Tramunt B, Berlin I, Clair C, Thomas D, Le
Faou AL, et al: Smoking and diabetes. Ann Endocrinol (Paris).
85:614–622. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ranscht B and Dours-Zimmermann MT:
T-cadherin, a novel cadherin cell adhesion molecule in the nervous
system lacks the conserved cytoplasmic region. Neuron. 7:391–402.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Philippova M, Joshi MB, Kyriakakis E,
Pfaff D, Erne P and Resink TJ: A guide and guard: The many faces of
T-cadherin. Cell Signal. 21:1035–1044. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rubina KA, Semina EV, Kalinina NI, Sysoeva
VY, Balatskiy AV and Tkachuk VA: Revisiting the multiple roles of
T-cadherin in health and disease. Eur J Cell Biol. 100:1511832021.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rubina KA and Tkachuk VA: Guidance
receptors in the nervous and cardiovascular systems. Biochemistry
(Mosc). 80:1235–1253. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Di Palo A, Siniscalchi C, Polito R, Nigro
E, Russo A, Daniele A and Potenza N: microRNA-377-3p downregulates
the oncosuppressor T-cadherin in colorectal adenocarcinoma cells.
Cell Biol Int. 45:1797–1803. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hayano Y, Zhao H, Kobayashi H, Takeuchi K,
Norioka S and Yamamoto N: The role of T-cadherin in axonal pathway
formation in neocortical circuits. Development. 141:4784–4793.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sysoeva V, Semina E, Klimovich P,
Kulebyakin K, Dzreyan V, Sotskaya E, Shchipova A, Popov V, Shilova
A, Brodsky I, et al: T-cadherin modulates adipogenic
differentiation in mesenchymal stem cells: Insights into ligand
interactions. Front Cell Dev Biol. 12:14463632024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kita S, Fukuda S, Maeda N and Shimomura I:
Native adiponectin in serum binds to mammalian cells expressing
T-cadherin, but not AdipoRs or calreticulin. Elife. 8:e486752019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamauchi T, Iwabu M, Okada-Iwabu M and
Kadowaki T: Adiponectin receptors: A review of their structure,
function and how they work. Best Pract Res Clin Endocrinol Metab.
28:15–23. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hug C, Wang J, Ahmad NS, Bogan JS, Tsao TS
and Lodish HF: T-cadherin is a receptor for hexameric and
high-molecular-weight forms of Acrp30/adiponectin. Proc Natl Acad
Sci USA. 101:10308–10313. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fukuda S, Kita S, Obata Y, Fujishima Y,
Nagao H, Masuda S, Tanaka Y, Nishizawa H, Funahashi T, Takagi J, et
al: The unique prodomain of T-cadherin plays a key role in
adiponectin binding with the essential extracellular cadherin
repeats 1 and 2. J Biol Chem. 292:7840–7849. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ciatto C, Bahna F, Zampieri N,
VanSteenhouse HC, Katsamba PS, Ahlsen G, Harrison OJ, Brasch J, Jin
X, Posy S, et al: T-cadherin structures reveal a novel adhesive
binding mechanism. Nat Struct Mol Biol. 17:339–347. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bochkov VN, Tkachuk VA, Philippova MP,
Stambolsky DV, Bühler FR and Resink TJ: Ligand selectivity of 105
and 130 kDa lipoprotein-binding proteins in
vascular-smooth-muscle-cell membranes is unique. Biochem J.
317:297–304. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Niermann T, Kern F, Erne P and Resink T:
The glycosyl phosphatidylinositol anchor of human T-cadherin binds
lipoproteins. Biochem Biophys Res Commun. 276:1240–1247. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Parker-Duffen JL, Nakamura K, Silver M,
Kikuchi R, Tigges U, Yoshida S, Denzel MS, Ranscht B and Walsh K:
T-cadherin is essential for adiponectin-mediated revascularization.
J Biol Chem. 288:24886–24897. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Balatskaya MN, Sharonov GV, Baglay AI,
Rubtsov YP and Tkachuk VA: Different spatiotemporal organization of
GPI-anchored T-cadherin in response to low-density lipoprotein and
adiponectin. Biochim Biophys Acta Gen Subj. 1863:1294142019.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rubina K, Talovskaya E, Cherenkov V,
Ivanov D, Stambolsky D, Storozhevykh T, Pinelis V, Shevelev A,
Parfyonova Y, Resink T, et al: LDL induces intracellular signalling
and cell migration via atypical LDL-binding protein T-cadherin. Mol
Cell Biochem. 273:33–41. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kipmen-Korgun D, Osibow K, Zoratti C,
Schraml E, Greilberger J, Kostner GM, Jürgens G and Graier WF:
T-cadherin mediates low-density lipoprotein-initiated cell
proliferation via the Ca(2+)-tyrosine kinase-Erk1/2 pathway. J
Cardiovasc Pharmacol. 45:418–430. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Iioka M, Fukuda S, Maeda N, Natsukawa T,
Kita S, Fujishima Y, Sawano H, Nishizawa H and Shimomura I:
Time-series change of serum soluble T-Cadherin concentrations and
its association with creatine kinase-MB levels in ST-segment
elevation myocardial infarction. J Atheroscler Thromb.
29:1823–1834. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tsugawa-Shimizu Y, Fujishima Y, Kita S,
Minami S, Sakaue TA, Nakamura Y, Okita T, Kawachi Y, Fukada S,
Namba-Hamano T, et al: Increased vascular permeability and severe
renal tubular damage after ischemia-reperfusion injury in mice
lacking adiponectin or T-cadherin. Am J Physiol Endocrinol Metab.
320:E179–E190. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kita S and Shimomura I: Stimulation of
exosome biogenesis by adiponectin, a circulating factor secreted
from adipocytes. J Biochem. 169:173–179. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lee MH, Klein RL, El-Shewy HM, Luttrell DK
and Luttrell LM: The adiponectin receptors AdipoR1 and AdipoR2
activate ERK1/2 through a Src/Ras-dependent pathway and stimulate
cell growth. Biochemistry. 47:11682–11692. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Popov VS, Brodsky IB, Balatskaya MN,
Balatskiy AV, Ozhimalov ID, Kulebyakina MA, Semina EV, Arbatskiy
MS, Isakova VS, Klimovich PS, et al: T-cadherin deficiency is
associated with increased blood pressure after physical activity.
Int J Mol Sci. 24:142042023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dasen B, Pigeot S, Born GM, Verrier S,
Rivero O, Dittrich PS, Martin I and Filippova M: T-cadherin is a
novel regulator of pericyte function during angiogenesis. Am J
Physiol Cell Physiol. 324:C821–C836. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Obata Y, Kita S, Koyama Y, Fukuda S,
Takeda H, Takahashi M, Fujishima Y, Nagao H, Masuda S, Tanaka Y, et
al: Adiponectin/T-cadherin system enhances exosome biogenesis and
decreases cellular ceramides by exosomal release. JCI Insight.
3:e996802018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Clark JL, Taylor CG and Zahradka P:
Exploring the cardio-metabolic relevance of T-cadherin: A
pleiotropic adiponectin receptor. Endocr Metab Immune Disord Drug
Targets. 17:200–206. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kita S, Maeda N and Shimomura I:
Interorgan communication by exosomes, adipose tissue, and
adiponectin in metabolic syndrome. J Clin Invest. 129:4041–4049.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Okita T, Kita S, Fukuda S, Fukuoka K,
Kawada-Horitani E, Iioka M, Nakamura Y, Fujishima Y, Nishizawa H,
Kawamori D, et al: Soluble T-cadherin promotes pancreatic β-cell
proliferation by upregulating Notch signaling. iScience.
25:1054042022. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fujishima Y, Kita S, Nishizawa H, Maeda N
and Shimomura I: Cardiovascular significance of adipose-derived
adiponectin and liver-derived xanthine oxidoreductase in metabolic
syndrome. Endocr J. 70:663–675. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tan WH, Peng ZL, You T and Sun ZL: CTRP15
promotes macrophage cholesterol efflux and attenuates
atherosclerosis by increasing the expression of ABCA1. J Physiol
Biochem. 78:653–666. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Rubina K, Maier A, Klimovich P, Sysoeva V,
Romashin D, Semina E and Tkachuk V: T-cadherin (CDH13) and
non-coding RNAs: The crosstalk between health and disease. Int J
Mol Sci. 26:61272025. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Nicolas A, Aubert R, Bellili-Muñoz N,
Balkau B, Bonnet F, Tichet J, Velho G, Marre M, Roussel R and
Fumeron F: T-cadherin gene variants are associated with type 2
diabetes and the fatty liver index in the French population.
Diabetes Metab. 43:33–39. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Tarantino G, Costantini S, Finelli C,
Capone F, Guerriero E, La Sala N, Gioia S and Castello G: Carotid
intima-media thickness is predicted by combined eotaxin levels and
severity of hepatic steatosis at ultrasonography in obese patients
with nonalcoholic fatty liver disease. PLoS One. 9:e1056102014.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Philippova M, Suter Y, Toggweiler S,
Schoenenberger AW, Joshi MB, Kyriakakis E, Erne P and Resink TJ:
T-cadherin is present on endothelial microparticles and is elevated
in plasma in early atherosclerosis. Eur Heart J. 32:760–771. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Philippova MP, Bochkov VN, Stambolsky DV,
Tkachuk VA and Resink TJ: T-cadherin and signal-transducing
molecules co-localize in caveolin-rich membrane domains of vascular
smooth muscle cells. FEBS Lett. 429:207–210. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Göddeke S, Knebel B, Fahlbusch P, Hörbelt
T, Poschmann G, van de Velde F, Benninghoff T, Al-Hasani H, Jacob
S, Van Nieuwenhove Y, et al: CDH13 abundance interferes with
adipocyte differentiation and is a novel biomarker for adipose
tissue health. Int J Obes (Lond). 42:1039–1050. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Summerhill VI, Grechko AV, Yet SF, Sobenin
IA and Orekhov AN: The atherogenic role of circulating modified
lipids in atherosclerosis. Int J Mol Sci. 20:35612019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Fujishima Y, Maeda N, Matsuda K, Masuda S,
Mori T, Fukuda S, Sekimoto R, Yamaoka M, Obata Y, Kita S, et al:
Adiponectin association with T-cadherin protects against neointima
proliferation and atherosclerosis. FASEB J. 31:1571–1583. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Philippova M, Banfi A, Ivanov D,
Gianni-Barrera R, Allenspach R, Erne P and Resink T: Atypical
GPI-anchored T-cadherin stimulates angiogenesis in vitro and in
vivo. Arterioscler Thromb Vasc Biol. 26:2222–2230. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yan F, Sun SY and Wu H: The
‘angiogenesis-plaque stability paradox’ in atherosclerosis
pathogenesis. Front Cardiovasc Med. 12:16590062025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lin A, Miano JM, Fisher EA and Misra A:
Chronic inflammation and vascular cell plasticity in
atherosclerosis. Nat Cardiovasc Res. 3:1408–1423. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Joshi MB, Kyriakakis E, Pfaff D, Rupp K,
Philippova M, Erne P and Resink TJ: Extracellular cadherin repeat
domains EC1 and EC5 of T-cadherin are essential for its ability to
stimulate angiogenic behavior of endothelial cells. FASEB J.
23:4011–4021. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Semina EV, Rubina KA, Sysoeva VY,
Rutkevich PN, Kashirina NM and Tkachuk VA: Novel mechanism
regulating endothelial permeability via T-cadherin-dependent
VE-cadherin phosphorylation and clathrin-mediated endocytosis. Mol
Cell Biochem. 387:39–53. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Semina E, Popov V, Khabibullin N,
Klimovich P, Sysoeva V, Kurilina E, Tsokolaeva Z, Tkachuk V and
Rubina K: New evidence for T-cadherin in COVID-19 pathogenesis,
endothelial dysfunction, and lung fibrosis. Front Cell Dev Biol.
13:14763292025. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Schäfer S, Gogiraju R, Rösch M, Kerstan Y,
Beck L, Garbisch J, Saliba AE, Gisterå A, Hermanns HM, Boon L, et
al: CD8+ T cells drive plaque smooth muscle cell
dedifferentiation in experimental atherosclerosis. Arterioscler
Thromb Vasc Biol. 44:1852–1872. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wang H, Tao L, Ambrosio A, Yan W, Summer
R, Lau WB, Wang Y and Ma X: T-cadherin deficiency increases
vascular vulnerability in T2DM through impaired NO bioactivity.
Cardiovasc Diabetol. 16:122017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Balatskiy A, Teterina M, Pisaryuk A,
Balabanenko I, Kadrev A, Tishuk A, Balatskaya M, Samokhodskaya L,
Boytsov S, Kalinina N and Tkachuk V: T-cadherin and the ratio of
its ligands as predictors of carotid atherosclerosis: A pilot
study. Biomedicines. 9:13982021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Denzel MS, Scimia MC, Zumstein PM, Walsh
K, Ruiz-Lozano P and Ranscht B: T-cadherin is critical for
adiponectin-mediated cardioprotection in mice. J Clin Invest.
120:4342–4352. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Vitale MR, Zöller JEM, Jansch C, Janz A,
Edenhofer F, Klopocki E, van den Hove D, Vanmierlo T, Rivero O,
Nadif Kasri N, et al: Generation of induced pluripotent stem cell
(iPSC) lines carrying a heterozygous (UKWMPi002-A-1) and null
mutant knockout (UKWMPi002-A-2) of cadherin 13 associated with
neurodevelopmental disorders using CRISPR/Cas9. Stem Cell Res.
51:1021692021. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Mills KT, Bundy JD, Kelly TN, Reed JE,
Kearney PM, Reynolds K, Chen J and He J: Global disparities of
hypertension prevalence and control: A systematic analysis of
population-based studies from 90 countries. Circulation.
134:441–450. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Kidambi S, Ghosh S, Kotchen JM, Grim CE,
Krishnaswami S, Kaldunski ML, Cowley AW Jr, Patel SB and Kotchen
TA: Non-replication study of a genome-wide association study for
hypertension and blood pressure in African Americans. BMC Med
Genet. 13:272012. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lee JH, Shin DJ, Park S, Kang SM, Jang Y
and Lee SH: Association between CDH13 variants and cardiometabolic
and vascular phenotypes in a Korean population. Yonsei Med J.
54:1305–1312. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Vargas-Alarcon G, Martinez-Rodriguez N,
Velazquez-Cruz R, Perez-Mendez O, Posadas-Sanchez R, Posadas-Romero
C, Peña-Duque MA, Martinez-Rios MA, Ramirez-Fuentes S and Fragoso
JM: The T>A (rs11646213) gene polymorphism of cadherin-13
(CDH13) gene is associated with decreased risk of developing
hypertension in Mexican population. Immunobiology. 222:973–978.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kim HJ, Seo YS, Sung J, Son HY, Yun JM,
Kwon H, Cho B, Kim JI and Park JH: Interactions of CDH13 gene
polymorphisms and ambient PM10 air pollution exposure
with blood pressure and hypertension in Korean men. Chemosphere.
218:292–298. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Spijkers LJA, van den Akker RFP, Janssen
BJA, Debets JJ, De Mey JGR, Stroes ESG, van den Born BJH,
Wijesinghe DS, Chalfant CE, MacAleese L, et al: Hypertension is
associated with marked alterations in sphingolipid biology: A
potential role for ceramide. PLoS One. 6:e218172011. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Berry C, Touyz R, Dominiczak AF, Webb RC
and Johns DG: Angiotensin receptors: Signaling, vascular
pathophysiology, and interactions with ceramide. Am J Physiol Heart
Circ Physiol. 281:H2337–H2365. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Fenger M, Linneberg A, Jørgensen T,
Madsbad S, Søbye K, Eugen-Olsen J and Jeppesen J: Genetics of the
ceramide/sphingosine-1-phosphate rheostat in blood pressure
regulation and hypertension. BMC Genet. 12:442011. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Fang G, Li Y, Yuan J, Cao W, Song S, Chen
L, Wang Y and Wang Q: Cadherin-11-interleukin-6 signaling between
cardiac fibroblast and cardiomyocyte promotes ventricular
remodeling in a mouse pressure overload-induced heart failure
model. Int J Mol Sci. 24:65492023. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Groenewegen A, Rutten FH, Mosterd A and
Hoes AW: Epidemiology of heart failure. Eur J Heart Fail.
22:1342–1356. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Tromp J, Ferreira JP, Janwanishstaporn S,
Shah M, Greenberg B, Zannad F and Lam CSP: Heart failure around the
world. Eur J Heart Fail. 21:1187–1196. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Bleumink GS, Knetsch AM, Sturkenboom MCJM,
Straus SMJM, Hofman A, Deckers JW, Witteman JCM and Stricker BHC:
Quantifying the heart failure epidemic: Prevalence, incidence rate,
lifetime risk and prognosis of heart failure the Rotterdam study.
Eur Heart J. 25:1614–1619. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Yancy CW, Jessup M, Bozkurt B, Butler J,
Casey DE Jr, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi
JL, et al: 2013 ACCF/AHA guideline for the management of heart
failure: A report of the American college of cardiology
foundation/American heart association task force on practice
guidelines. J Am Coll Cardiol. 62:e147–e239. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Yoo SGK, Lam CSP and Sweitzer NK: ‘Asian’
heart failure. Circulation. 150:177–179. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Fukuda S, Kita S, Miyashita K, Iioka M,
Murai J, Nakamura T, Nishizawa H, Fujishima Y, Morinaga J, Oike Y,
et al: Identification and clinical associations of 3 forms of
circulating T-cadherin in human serum. J Clin Endocrinol Metab.
106:1333–1344. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Nakamura Y, Kita S, Tanaka Y, Fukuda S,
Obata Y, Okita T, Nishida H, Takahashi Y, Kawachi Y,
Tsugawa-Shimizu Y, et al: Adiponectin stimulates exosome release to
enhance mesenchymal stem-cell-driven therapy of heart failure in
mice. Mol Ther. 28:2203–2219. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Chen X, Qin M, Jiang W, Zhang Y and Liu X:
Electrophysiological characteristics of pressure overload-induced
cardiac hypertrophy and its influence on ventricular arrhythmias.
PLoS One. 12:e01836712017. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Cutler MJ, Jeyaraj D and Rosenbaum DS:
Cardiac electrical remodeling in health and disease. Trends
Pharmacol Sci. 32:174–180. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Armoundas AA, Wu R, Juang G, Marbán E and
Tomaselli GF: Electrical and structural remodeling of the failing
ventricle. Pharmacol Ther. 92:213–230. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Nemeth J, Skronska-Wasek W, Keppler S,
Schundner A, Groß A, Schoenberger T, Quast K, El Kasmi KC, Ruppert
C, Günther A and Frick M: Adiponectin suppresses
stiffness-dependent, profibrotic activation of lung fibroblasts. Am
J Physiol Lung Cell Mol Physiol. 327:L487–L502. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wang L, Tsakiroglou P, Gonzales R, Cho S,
Li A, Dos Remedios C, Wright N and Kontrogianni-Konstantopoulos A:
Essential role of obscurin kinase-1 in cardiomyocyte coupling via
N-cadherin phosphorylation. JCI Insight. 9:e1621782024.PubMed/NCBI
|
|
80
|
Ma G, Li Y, Meng F, Sui C, Wang Y and
Cheng D: Hsa_circ_0000119 promoted ovarian cancer development via
enhancing the methylation of CDH13 by sponging miR-142-5p. J
Biochem Mol Toxicol. 37:e232642023. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Straub LG and Scherer PE: Metabolic
messengers: Adiponectin. Nat Metab. 1:334–339. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Okita T, Kita S, Fukuda S, Kondo Y, Sakaue
TA, Iioka M, Fukuoka K, Kawada K, Nagao H, Obata Y, et al: Soluble
T-cadherin secretion from endothelial cells is regulated via
insulin/PI3K/Akt signalling. Biochem Biophys Res Commun.
732:1504032024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Bartolome A, Zhu C, Sussel L and Pajvani
UB: Notch signaling dynamically regulates adult β cell
proliferation and maturity. J Clin Invest. 129:268–280. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Eom YS, Gwon AR, Kwak KM, Youn JY, Park H,
Kim KW and Kim BJ: Notch1 has an important role in β-cell mass
determination and development of diabetes. Diabetes Metab J.
45:86–96. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
De Groef S, Renmans D, Cai Y, Leuckx G,
Roels S, Staels W, Gradwohl G, Baeyens L, Heremans Y, Martens GA,
et al: STAT3 modulates β-cell cycling in injured mouse pancreas and
protects against DNA damage. Cell Death Dis. 7:e22722016.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Ma J, Xing B, Cao Y, He X, Bennett KE,
Tong C, An C, Hojnacki T, Feng Z, Deng S, et al: Menin-regulated
Pbk controls high fat diet-induced compensatory beta cell
proliferation. EMBO Mol Med. 13:e135242021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Rosado-Olivieri EA, Aigha II, Kenty JH and
Melton DA: Identification of a LIF-responsive,
replication-competent subpopulation of human β cells. Cell Metab.
31:327–338.e6. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Philippova M, Joshi MB, Pfaff D,
Kyriakakis E, Maslova K, Erne P and Resink TJ: T-cadherin
attenuates insulin-dependent signalling, eNOS activation, and
angiogenesis in vascular endothelial cells. Cardiovasc Res.
93:498–507. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Nicolas A, Mohammedi K, Bastard JP,
Fellahi S, Bellili-Muñoz N, Roussel R, Hadjadj S, Marre M, Velho G
and Fumeron F: T-cadherin gene variants are associated with
nephropathy in subjects with type 1 diabetes. Nephrol Dial
Transplant. 32:1987–1993. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Li Y, Yang Y, Yao Y, Li X, Shi L, Zhang Y,
Xiong Y, Yan M, Yao Y and Xiao C: Association study of ARL15 and
CDH13 with T2DM in a Han Chinese population. Int J Med Sci.
11:522–527. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lee G, Wong C, Cho A, West JJ, Crawford
AJ, Russo GC, Si BR, Kim J, Hoffner L, Jang C, et al: E-cadherin
induces serine synthesis to support progression and metastasis of
breast cancer. Cancer Res. 84:2820–2835. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Agiostratidou G, Hulit J, Phillips GR and
Hazan RB: Differential cadherin expression: Potential markers for
epithelial to mesenchymal transformation during tumor progression.
J Mammary Gland Biol Neoplasia. 12:127–133. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Paredes J, Correia AL, Ribeiro AS,
Albergaria A, Milanezi F and Schmitt FC: P-cadherin expression in
breast cancer: A review. Breast Cancer Res. 9:2142007. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Xu D, Yuan H, Meng Z, Yang C, Li Z, Li M,
Zhang Z, Gan Y and Tu H: Cadherin 13 inhibits pancreatic cancer
progression and epithelial-mesenchymal transition by Wnt/β-catenin
signaling. J Cancer. 11:2101–2112. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhao J, Yang T, Ji J, Li C, Li Z and Li L:
Garcinol exerts anti-cancer effect in human cervical cancer cells
through upregulation of T-cadherin. Biomed Pharmacother.
107:957–966. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Shao Y, Li W, Zhang L, Xue B, Chen Y,
Zhang Z, Wang D and Wu B: CDH13 is a prognostic biomarker and a
potential therapeutic target for patients with clear cell renal
cell carcinoma. Am J Cancer Res. 12:4520–4544. 2022.PubMed/NCBI
|
|
97
|
Dasen B, Vlajnic T, Mengus C, Ruiz C,
Bubendorf L, Spagnoli G, Wyler S, Erne P, Resink TJ and Philippova
M: T-cadherin in prostate cancer: Relationship with cancer
progression, differentiation and drug resistance. J Pathol Clin
Res. 3:44–57. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Wang Y, Zhang L, Yang J, Li B and Wang J:
CDH13 promoter methylation regulates cisplatin resistance of
non-small cell lung cancer cells. Oncol Lett. 16:5715–5722.
2018.PubMed/NCBI
|
|
99
|
Lin J, Chen Z, Huang Z, Chen F, Ye Z, Lin
S and Wang W: Effect of T-cadherin on the AKT/mTOR signaling
pathway, gastric cancer cell cycle, migration and invasion, and its
association with patient survival rate. Exp Ther Med. 17:3607–3613.
2019.PubMed/NCBI
|
|
100
|
Lin J, Chen Z, Huang Z, Chen F, Ye Z, Lin
S and Wang W: Upregulation of T-cadherin suppresses cell
proliferation, migration and invasion of gastric cancer in
vitro. Exp Ther Med. 14:4194–4200. 2017.PubMed/NCBI
|
|
101
|
Wei B, Shi H, Lu X, Shi A, Cheng Y and
Dong L: Association between the expression of T-cadherin and
vascular endothelial growth factor and the prognosis of patients
with gastric cancer. Mol Med Rep. 12:2075–2081. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Tang Y, Dai Y and Huo J: Decreased
expression of T-cadherin is associated with gastric cancer
prognosis. Hepatogastroenterology. 59:1294–1298. 2012.PubMed/NCBI
|
|
103
|
Wang Q, Chen Y, Chen Y, Jiang J, Song X,
Zhang L, He Q, Ye B, Wu L, Wu R, et al: Aberrant promoter
methylation of T-cadherin in sera is associated with a poor
prognosis in oral squamous cell carcinoma. Neoplasma. 68:528–534.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Wang Q, Zhang X, Song X and Zhang L:
Overexpression of T-cadherin inhibits the proliferation of oral
squamous cell carcinoma through the PI3K/AKT/mTOR intracellular
signalling pathway. Arch Oral Biol. 96:74–79. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Kong DD, Wang MH, Yang J, Li L, Wang W,
Wang SB and Zhou YZ: T-cadherin is associated with prognosis in
triple-negative breast cancer. Oncol Lett. 14:2975–2981. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Yang J, Niu H, Huang Y and Yang K: A
systematic analysis of the relationship of CDH13 promoter
methylation and breast cancer risk and prognosis. PLoS One.
11:e01491852016. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Baranová I, Samec M, Dvorská D, Šťastný I,
Janíková K, Kašubová I, Hornáková A, Lukáčová E, Kapinová A,
Biringer K, et al: Droplet digital PCR analysis of CDH13
methylation status in Slovak women with invasive ductal breast
cancer. Sci Rep. 14:147002024. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Pang JM, Deb S, Takano EA, Byrne DJ, Jene
N, Boulghourjian A, Holliday A, Millar E, Lee CS, O'Toole SA, et
al: Methylation profiling of ductal carcinoma in situ and its
relationship to histopathological features. Breast Cancer Res.
16:4232014. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Yuan XD, Wang JW, Fang Y, Qian Y, Gao S,
Fan YC and Wang K: Methylation status of the T-cadherin gene
promotor in peripheral blood mononuclear cells is associated with
HBV-related hepatocellular carcinoma progression. Pathol Res Pract.
216:1529142020. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Tuoya AD, Wang DX, Xing YS, Liu RJ, Hu YX,
Zhang MD, Bai TY, Lv XL, Li J and Chang FH: Relationship between
methylation of FHIT and CDH13 gene promoter region and liver
cancer. Curr Med Sci. 40:502–509. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wang M, Long K, Li E, Li L, Li B, Ci S, He
L, Pan F, Hu Z and Guo Z: DNA polymerase beta modulates cancer
progression via enhancing CDH13 expression by promoter
demethylation. Oncogene. 39:5507–5519. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Nieto MA, Huang RY, Jackson RA and Thiery
JP: EMT: 2016. Cell. 166:21–45. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Kalluri R and Weinberg RA: The basics of
epithelial-mesenchymal transition. J Clin Invest. 119:1420–1428.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Zajchowski LD and Robbins SM: Lipid rafts
and little caves. Compartmentalized signalling in membrane
microdomains. Eur J Biochem. 269:737–752. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Lucero HA and Robbins PW: Lipid
rafts-protein association and the regulation of protein activity.
Arch Biochem Biophys. 426:208–224. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Vouret-Craviari V, Boulter E, Grall D,
Matthews C and Van Obberghen-Schilling E: ILK is required for the
assembly of matrix-forming adhesions and capillary morphogenesis in
endothelial cells. J Cell Sci. 117:4559–4569. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Isik OA and Cizmecioglu O: Rafting on the
plasma membrane: Lipid rafts in signaling and disease. Adv Exp Med
Biol. 1436:87–108. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Sviridov D, Mukhamedova N and Miller YI:
Lipid rafts as a therapeutic target. J Lipid Res. 61:687–695. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Balatskaya MN, Balatskii AV, Sharonov GV
and Tkachuk VA: T-cadherin as a novel receptor regulating
metabolism in the blood vessel and heart cells: From structure to
function. J Evol Biochem Physiol. 52:103–118. 2016. View Article : Google Scholar
|
|
120
|
Ruzzi F, Cappello C, Semprini MS,
Scalambra L, Angelicola S, Pittino OM, Landuzzi L, Palladini A,
Nanni P and Lollini PL: Lipid rafts, caveolae, and epidermal growth
factor receptor family: Friends or foes? Cell Commun Signal.
22:4892024. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Rubina KA, Semina EA, Balatskaya MN,
Plekhanova OS and Tkachuk VA: Mechanisms of Regulation of the
Targeted Grown of Nerves and Vessels by Components of the
Fibrinolytic System and GPI-Anchored Navigation Receptors. Neurosci
Behav Physiol. 50:217–230. 2020. View Article : Google Scholar
|