Introduction
Considering incidence and mortality rates,
cardiovascular diseases, diabetes and cancer have become the most
notable diseases worldwide (1–3).
Despite notable progress in prevention approaches and treatment
modalities, the incidence rates of these diseases continue to
increase steadily and are gradually shifting toward younger
populations (4–7). Besides known risk factors, some risks
cannot be explained by traditional factors, indicating the presence
of unknown variables (8,9). This emphasizes how important it is to
find and understand novel molecular regulators at the
pathophysiological nexus of metabolic, cancer and cardiovascular
diseases.
An atypical member of the cadherin superfamily,
truncated-cadherin (T-cadherin), also known as heart-cadherin or
cadherin-13 (CDH13), was first discovered in the embryonic
brains of chicks (10). T-cadherin
contains five Ca2+-binding extracellular structural
domains, which are typical components of cadherins. However, it
lacks transmembrane and cytoplasmic structural domains and binds to
the plasma membrane via glycosylphosphatidylinositol (GPI) anchors
(11,12). In the embryonic nervous system,
T-cadherin-mediated homophilic cell adhesion regulates angiogenesis
and tumor neoangiogenesis, and negatively affects motor neuron axon
projections and neural crest cell migration (13). Owing to the unique structure of
T-cadherin, T-cadherin-mediated homophilic cell adhesion
fundamentally alters its mechanism of action, preventing it from
forming stable intercellular junctions in the same way as a
traditional adhesion molecule; specifically, its distinct modes of
action are determined by its GPI anchor, which enables it to
function as both a receptor and a ligand, thereby allowing it to
participate in regulating intracellular signaling pathways
governing proliferation, differentiation, migration and
regeneration (11). T-cadherin, as
a physiological receptor, can bind to hexameric and high molecular
weight (HMW) forms of the cardiovascular preventive factor
adiponectin, while it can also independently serve as a receptor
for atherosclerotic low-density lipoprotein (LDL); this paradoxical
ligand affinity thereby positions T-cadherin at a critical
signaling hub (11,12). The present study reviewed the
current research progress on the structure, regulation, expression,
function, signaling and role of T-cadherin in cardiovascular
diseases, metabolic disorders and cancer.
Molecular foundation of T-cadherin
T-cadherin: Structure and
function
A comprehensive understanding of the molecular
structure, ligand interactions and regulation of T-cadherin
expression is necessary to fully understand its multifaceted roles
in various diseases. This information lays the groundwork for
investigating the specific contribution of T-cadherin to
cardiovascular diseases, metabolic disorders and cancer.
The 3′ untranslated region of the
T-cadherin-encoding gene CDH13 has a microRNA (miR)-377-3p
binding site that controls T-cadherin protein expression (14). T-cadherin is a member of the
‘atypical cadherin’ subgroup of the cadherin superfamily. The five
Ca2+-binding domains present in T-cadherin are important
for homo- and hetero-dimerization and interactions with other
T-cadherin molecules in the vascular and neurological systems
(12) (Fig. 1). Similar to other traditional
cadherins, such as epithelial-cadherin (E-cadherin) and
neural-cadherin (N-cadherin), T-cadherin is linked to the cell
membrane by a GPI anchor; however, T-cadherin does not have
intracellular and transmembrane structural domains (15). Protein distribution of T-cadherin
within plasma membrane rafts is often guided by the presence of the
GPI anchor (11), a structural
component that provides dynamic modulation of cell adhesion and may
play a role in signal transduction via lipid raft microregions. The
dual ligand-binding capability of T-cadherin implies that its
extracellular domain may contain multiple binding sites, as its
extracellular region acts as a receptor for both LDL and HMW
adiponectin (16). In vitro
experiments have shown that adiponectin binds to cells expressing
T-cadherin but not adiponectin receptor (AdipoR)1 or calreticulin.
AdipoR and calreticulin may require additional co-receptors or
accessory proteins to acquire the ability to bind natural HMW
adiponectin. T-cadherin knockout notably reduces adiponectin
binding to cells of the 293 cell line, Chinese hamster ovary cells
and C2C12 myotubes. These findings indicate that T-cadherin is the
primary binding partner of native adiponectin (17,18)
Importantly, T-cadherin is a receptor for hexameric and HMW
adiponectin, but not for trimeric or globular adiponectin (19). A study by Fukuda et al
(20) showed that the binding of
adiponectin to T-cadherin required the extracellular EC1-EC2 region
of T-cadherin, and that the pro-domain of T-cadherin played a role
in binding with adiponectin. The extracellular cadherin repeat
(EC1-EC2) region overlaps with the reported homologous
transmembrane region of T-cadherin (21). Notably, compared with the mature
form comprising 100 kDa T-cadherin without the pro-domain, the 130
kDa T-cadherin form with the pro-domain shows a greater binding
affinity for adiponectin. The 130 kDa form is also more likely to
be expressed on the surface of endothelial cells both in
vitro and in vivo than the mature form, demonstrating
positive feedback regulation of T-cadherin by adiponectin (20). Adiponectin binding further
increases membrane expression and functional activity of 130 kDa
T-cadherin, continuously elevating T-cadherin abundance at the
post-translational level. A study by Bochkov et al (22) primarily showed that the atypical
lipoprotein-binding protein P105 that is present on the vascular
smooth muscle membrane of humans and rats was T-cadherin, providing
evidence that the mechanism underlying binding of LDL to T-cadherin
may be similar to that mediating homophilic cadherin-cadherin
interactions. A subsequent study revealed that T-cadherin is
expressed in Escherichia coli with a GPI anchor signal
sequence but without GPI modification, and that recombinant human
T-cadherin expressed in 293 cells without a GPI anchor signal
sequence cannot bind to lipoproteins, providing evidence that the
GPI anchor of T-cadherin is important for lipoprotein binding
(23). Previous studies have
provided evidence that T-cadherin is a physiological receptor for
LDL (24–26). T-cadherin binds to LDL on the cell
surface, triggering intracellular calcium ion mobilization,
increasing tyrosine phosphorylation and stimulating ERK1/2 and
NF-κB to promote cell proliferation, thereby converting
extracellular LDL into mitogenic signals (25,27).
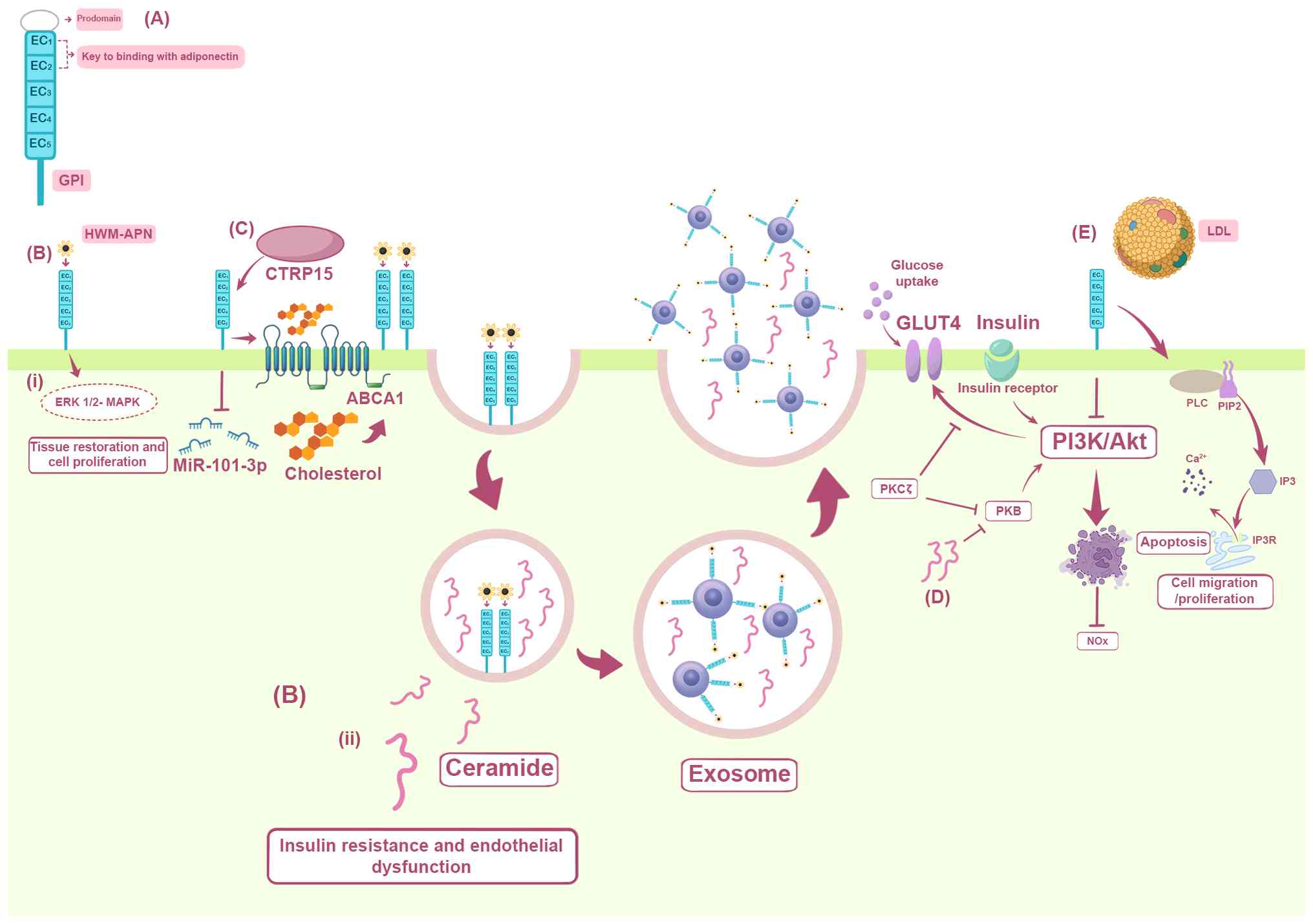 | Figure 1.T-cadherin modulates atherosclerosis
through adiponectin signaling, insulin sensitivity and cholesterol
transport. The figure summarizes the structure and pleiotropic
functions of T-cadherin. (A) Molecular architecture of T-cadherin.
The preproprotein consists of a signal pre-peptide, five
extracellular cadherin repeats, EC1-EC5, and a C-terminal GPI
anchor. The EC1 and EC2 domains are necessary for adiponectin
binding and the GPI anchor targets the protein to membrane lipid
rafts. (B) Adiponectin signaling and ceramide export. The binding
of HMW-APN to T-cadherin triggers two important events: i)
Activation of the ERK1/2/MAPK pathway, which promotes tissue repair
and cell proliferation; and ii) adiponectin sequestration into
multivesicular bodies, enhancing exosome biogenesis and secretion.
This process concurrently exports intracellular ceramide, a lipid
that inhibits insulin signaling. (C) Promotion of macrophage
cholesterol efflux. The CTRP15/T-cadherin axis downregulates
miR-101-3p, which directly represses the cholesterol transporter
ABCA1. This enhances ABCA1-mediated cholesterol efflux, reducing
lipid accumulation in macrophages and slowing the progression of
atherosclerosis. (D) Inhibition of insulin signaling by ceramide.
High intracellular ceramide levels block the PI3K/Akt pathway
either by inhibiting the PKB/Akt pathway or by activating PKCζ,
ultimately preventing GLUT4 translocation to the membrane and
reducing cellular glucose uptake, which fosters insulin resistance.
(E) Consequences of T-cadherin loss. T-cadherin deficiency: i)
Promotes apoptosis and reduces bioavailable nitric oxide via
diminished Akt phosphorylation and endothelial nitric oxide
synthase activity; and ii) ablates T-cadherin binding to LDL,
preventing the activation of PLC/PIP2/IP3 signaling and subsequent
calcium release, which are important for normal endothelial
function and cell motility. LDL, low-density lipoprotein; miR,
microRNA; ABCA1, ATP-binding cassette transporter A1; Akt, Protein
kinase B; CTRP15, C1q/TNF-related protein 15; EC1-EC5,
extracellular cadherin repeats 1–5; eNOS, endothelial nitric oxide
synthase; ERK1/2, extracellular signal-regulated kinases 1 and 2;
GLUT4, glucose transporter type 4; GPI,
glycosylphosphatidylinositol; HMW-APN, high-molecular-weight
adiponectin; IP3, inositol trisphosphate; MAPK, mitogen-activated
protein kinase; miR-101-3p, microRNA-101-3p; NO, nitric oxide,
PI3K, phosphoinositide 3-kinase; PIP, phosphatidylinositol
4,5-bisphosphate; PKB, protein kinase B; PKCζ, protein kinase C ζ;
PLC, phospholipase C. |
T-cadherin accumulation in cardiovascular tissues,
such as the aorta, heart and skeletal muscles, allows adiponectin
to exert cardioprotective effects (28). Clinical research has shown a
notable association between serum HMW adiponectin levels and
T-cadherin expression. The binding between T-cadherin in HMW
adiponectin is specific and independent of other known adiponectin
receptors, such as AdipoR1 and AdipoR2 (17,29).
In addition to its role as a receptor for adiponectin, T-cadherin
contributes to the pleiotropic organ-protective effects of
adiponectin. T-cadherin-dependent secretion of adiponectin via
exosomes in the cardiovascular system improves vascular homeostasis
and myocardial regeneration, and is the predominant mechanism
underlying therapeutic effects of mesenchymal stem cells (MSCs)
(30). In muscle and endothelial
cells, T-cadherin binds to adiponectin to activate exosome
secretion. Additionally, inhibition of the AdipoR1/R2 signaling
pathway, potentially mediated by competitive binding of adiponectin
to T-cadherin, activates the ERK1/2/MAPK pathway, promoting tissue
repair and cell proliferation (Fig.
1) (30,31).
T-cadherin expression and control in
the cardiovascular system
Vascular endothelial cells, vascular smooth muscle
cells and pericytes are the primary cells of the cardiovascular
system that express T-cadherin (32,33).
Immunofluorescence results in a previous study showed that
T-cadherin is markedly expressed in the basal layer of healthy
arteries and is closely linked to the functional control of
pericytes (33). Furthermore, its
presence in cardiac tissues, particularly in the coronary
endothelium and cardiomyocytes, raises the possibility that
T-cadherin plays a cardioprotective role (20,34).
As a receptor for LDL and HMW adiponectin,
T-cadherin possesses dual ligand-binding capabilities (12,16,25).
These two ligands exert opposing effects on the cardiovascular
system. When LDL binds to T-cadherin, it triggers the calcium
signaling cascade, accelerating the formation of atherosclerotic
plaques by promoting endothelial cell migration, proliferation and
inflammatory responses (25,35).
HMW adiponectin accumulation in cardiovascular tissues via
T-cadherin improves insulin resistance and inhibits vascular
inflammation by increasing exosome production and release while
decreasing cellular ceramide accumulation (20,34,36)
(Fig. 1). This process activates
Notch signaling (37), a pathway
closely associated with adiponectin-dependent vascular regeneration
and myocardial protection (38,39).
Although T-cadherin is generally associated with the cardiovascular
protective effects of adiponectin, it also displays an association
with non-alcoholic fatty liver disease (NAFLD) due to shared
metabolic dysfunction. Reduced adiponectin signals via its
receptors, including T-cadherin, cause insulin resistance and
hepatic fat accumulation in NAFLD (40). Additionally, a study has shown that
genetic variations, known as polymorphisms, in CDH13 are
associated with NAFLD-related metabolic diseases, such as type 2
diabetes (T2D) and metabolic syndrome (40). A previous study has shown that a
high fatty liver index score can predict the risk of T2D. It is
plausible that CDH13 alleles are associated with T2D through
liver indicators (41).
Atherosclerosis, assessed using carotid intima-media
thickness (IMT), is closely associated with NAFLD (42). The dual role of T-cadherin as a
receptor for both cardioprotective adiponectin and atherogenic LDL
suggests that it is an important metabolic sensor that may
influence NAFLD and its associated cardiovascular risk. T-cadherin
is uniquely expressed on the plasma membranes of vascular
endothelial cells, smooth muscle cells and cardiomyocytes. Its
distribution is not uniform but rather concentrated at signaling
hubs such as membrane microdomains rich in caveolin proteins. This
‘strategic localization’ enables efficient binding to ligands in
the bloodstream (such as adiponectin) and initiates signaling
pathways that regulate cell growth, survival and function. Due to
the strategic placement and dual ligand-binding capabilities of
T-cadherin within the cardiovascular system, its dysregulation or
altered expression is expected to contribute towards the
development of major cardiovascular disorders (19,43,44).
The following sections discuss the evidence linking T-cadherin to
atherosclerosis, hypertension, heart failure and diabetes.
T-cadherin and cardiovascular diseases
T-cadherin and atherosclerosis
T-cadherin regulates lipid metabolism, which
directly affects the development of atherosclerotic plaques
(45). A study by Göddeke et
al (45), while studying the
role of T-cadherin in 3T3-L1 adipocyte differentiation, observed
that T-cadherin expression was highest in undifferentiated
adipocytes and almost undetectable in mature adipocytes. During
adipogenesis, T-cadherin knockdown has been shown to reduce the
expression of adipogenic genes, including peroxisome
proliferator activated receptor γ and CCAAT enhancer binding
protein α. Additionally, the authors of the study noted that
T-cadherin knockdown resulted in a reduction in both fatty acid
uptake and lipid content in developing adipocytes (45). Another study by Sysoeva et
al (16) subsequently reported
that LDL stimulated adipogenic differentiation, whereas T-cadherin
expression appears to mitigate the promotional effect of LDL on
lipid droplet accumulation. When LDL was added to adipogenic and
standard media, the proportion of lipid droplet-positive cells in
both T-cadherin−/− MSCs and wild-type MSCs markedly
increased, suggesting that LDL promotes lipid droplet accumulation.
However, the proportion of lipid droplet-positive cells in
wild-type MSCs was lower compared with that in
T-cadherin−/− MSCs, indicating that T-cadherin plays a
specific role in mitigating the impact of LDL on adipogenic
differentiation (16).
Atherosclerosis is characterized by excessive
accumulation of lipids in the intima of the cardiovascular system
(46). By inhibiting the
expression of miR-101-3p, erythroferrone can increase
phospholipid-transporting ATPase ABCA1 expression and cholesterol
efflux, thereby increasing T-cadherin expression and high-density
lipoprotein cholesterol levels in the circulation, as well as the
efficiency of reverse cholesterol transport, thereby inhibiting the
development of atherosclerosis (Fig.
1) (39). Furthermore, a study
on T-cadherin/apolipoprotein E dual-knockout model mice
demonstrated markedly elevated plasma lipocalin levels in
T-cadherin-deficient mice reared on a high-cholesterol diet
compared with T-cadherin-expressing mice, alongside a notable
increase in atherosclerotic plaque area and neointimal thickness,
indicating that T-cadherin is an important receptor for the
protective function of adiponectin (47).
Atherosclerosis is often accompanied by
neovascularization, a process induced by inflammation and tissue
ischemia. Neovascularization within the plaque extends to the
intima at the site of vascular injury, which may lead to
intra-plaque hemorrhage and rupture, thereby promoting the
progression and acute onset of atherosclerosis (48–50).
Notably, T-cadherin promotes angiogenesis by facilitating vascular
cell cycle progression and inducing cell de-adhesion, thereby
promoting the migration of smooth muscle and endothelial cells
(33,48,51).
In human umbilical vein endothelial cells, T-cadherin
overexpression leads to impaired endothelial barrier function
through chloride-dependent endocytosis and lysosomal degradation of
vascular endothelial-cadherin (VE-cadherin), activation of Rho
GTPase and increased actin stress fiber assembly (52). It has been further demonstrated
that increased T-cadherin expression negatively affects endothelial
barrier functions. A study on the human endothelial cell line
Ea.hy926 showed that overexpression of T-cadherin is linked to
lower VE-cadherin and β-catenin mRNA levels, resulting in the loss
of endothelial barrier function (53). The present review speculates that
under pathological conditions, overexpression of T-cadherin further
suppresses endothelial function and increases endothelial
permeability. Additionally, a previous study has shown that the
extracellular cadherin repeats EC1 and EC5 of T-cadherin are
necessary for its ability to stimulate endothelial cell angiogenic
behavior (51). These findings
suggest that T-cadherin promotes the progression of
atherosclerosis.
The severity of atherosclerotic lesions is closely
associated with immune cell infiltration (54). It is hypothesized that T-cadherin
may influence plaque progression by modulating CD8+ T
cell functions. In advanced plaques, depletion of CD8+ T
cells reduces smooth muscle apoptosis and improves plaque stability
(54). Furthermore, animal
experiments have demonstrated that T-cadherin deficiency causes
endothelial cell death by inhibiting the PI3K/Akt pathway, which
leads to decreased Akt phosphorylation, rather than by inhibiting
endothelial nitric oxide synthase, which leads to decreased
production of endothelial cell nitrogen oxides and a marked
reduction in vasodilatory function. These changes in endothelial
cells (55) (Fig. 1) exacerbate oxidative stress and
the release of inflammatory factors, such as IL-1β and IL-6, which
further encourage macrophage infiltration into the plaque (53). Adiponectin reduces the intra-plaque
inflammatory response by blocking inflammatory signaling pathways,
such as the NF-κB signaling pathway, via mechanisms dependent on
T-cadherin (56). A clinical study
has shown that patients with particular genotypes, for example the
thymine allele, carrying the CDH13 rs12444338
single-nucleotide polymorphism (SNP), have lower plasma T-cadherin
levels, although the carotid IMT of these patients was lower than
patients without these SNPs. These findings suggest that this
particular genotype may have a protective effect against
atherosclerosis (56).
Furthermore, T-cadherin plays dual roles in diabetes-related
vasculopathy; increases in T-cadherin expression increase insulin
secretion, while the excessive upregulation of T-cadherin may
worsen endothelial insulin resistance (35,37).
Several studies have proposed treatment options for
atherosclerosis based on the regulatory mechanisms of T-cadherin.
Supplementation with recombinant adiponectin or increased
T-cadherin expression can prevent lipid accumulation and
inflammatory responses in plaques, protecting the heart from
stress-induced pathological remodeling (39,47,57).
Generating specific antibodies or small molecule inhibitors to
prevent LDL from binding to T-cadherin can reduce its
pro-atherosclerotic effects (25,56).
Furthermore, CRISPR/Cas9 technology can be used to alter
CDH13 polymorphisms (58),
while miRs, such as miR-101-3p, can be used to modify T-cadherin
expression for improving the efficacy of reverse cholesterol
transporters (16,39).
Hypertension and T-cadherin
In total, ~30% of adults have hypertension, one of
the most prevalent cardiovascular conditions worldwide.
Hypertension is closely associated with metabolic irregularities
and the risk of cardiovascular events (59). SNPs in CDH13 have been
linked to hypertension in genome-wide association studies (GWASs).
Reduced circulating T-cadherin levels and carotid IMT are linked to
the CDH13 SNP rs12444338, indicating that this genotype may
exert antihypertensive effects by altering the vascular structure
(56). CDH13-specific SNPs,
such as rs12444338 and rs1048612, have been linked to variation in
systolic blood pressure levels in hypertensive patients, accounting
for ~1% of the phenotypic variance in blood pressure (60). Notably, these associations vary by
population: In the South Korean population, carriers of the
rs6565105 allele display elevated levels of LDL cholesterol
compared with other genotypes (61), which may indirectly increase the
risk of elevated blood pressure, whereas in African Americans, the
effect of CDH13 SNPs on systolic blood pressure has been
identified in independent samples (60). These findings imply that
CDH13 polymorphism may regulate blood pressure differently
in different ethnicities, and metabolic pathways may modulate its
effects. A previous study showed that the CDH13 rs11646213
polymorphism is associated with a lower risk of hypertension
compared with other SNPs (62).
These results suggest that certain CDH13 polymorphisms may
protect against hypertension. Notably, the greatest signal
associated with blood pressure features, such as systolic and
diastolic blood pressure, as well as hypertension, was the
rs7500599 intron SNP of CDH13, which also interacts with
particulate matter exposure (63).
These findings suggest that variations in CDH13 may affect
the susceptibility of an individual to environmental stimuli,
thereby affecting the likelihood of developing hypertension.
Animal models offer new perspectives on the
pathophysiology of hypertension. Based on comparative studies of
T-cadherin-knockout and wild-type mice, it has been reported that
T-cadherin plays an important role in controlling blood pressure
and endurance. Furthermore, T-cadherin knockout mice demonstrate a
marked increase in systolic blood pressure and reduced endurance
following exercise compared with wild-type mice, indicating that
T-cadherin plays a notable role in regulating vascular reactivity
and that T-cadherin deficiency reduces the compensatory capacity
for cardiovascular functions (32). Furthermore, in a model of
angiotensin II-mediated endothelial dysfunction,
T-cadherin-deficient mice show markedly lower reactive oxygen
species generation and NADPH oxidase 2 (Nox2) expression than their
wild-type counterparts (53).
These findings suggest that T-cadherin plays a role in controlling
angiotensin II-induced vascular reactivity, which in turn
influences blood pressure regulation.
Compared with rats with normal blood pressure, the
levels of ceramide in the arterial tissue of spontaneously
hypertensive rats have been demonstrated to be markedly elevated
(64). A further study showed that
ceramide is associated with thromboxane A2, a protein that may
cause endothelial dysfunction in hypertension (64). Similar associations between
ceramide levels and blood pressure have been reported in patients
with hypertension. In patients with primary hypertension, plasma
ceramide levels increase stepwise with the severity of
hypertension. Ceramide levels in patients with stage 1 hypertension
are between those in individuals with healthy blood pressure and
stage 2–3 hypertension (65). A
previous study has shown that ceramide/sphingosine-1-phosphate
regulators play a role in blood pressure regulation in patients
with hypertension (66). The
adiponectin/T-cadherin system reduces intracellular ceramide levels
via exosome secretion (34).
The interaction between T-cadherin and VE-cadherin
was further supported by the observation that overexpression of
T-cadherin in cultured human endothelial cells decreases
VE-cadherin mRNA expression. This interaction may disrupt
endothelial barrier function and affect vascular tone, which may
have a stabilizing effect on vascularity and blood pressure
regulation (53). According to
previous studies, T-cadherin regulates blood pressure via several
mechanisms, including oxidative stress, vascular remodeling and
endothelial function modification (32,53,57).
Although T-cadherin has been linked to hypertension in several
studies, the generalizability of these results may be affected by
confounding variables and population variations. A study conducted
in Amish and European populations have validated statistically
significant effects of CDH13 SNPs on blood pressure (odds
ratio, 1.2–2.0); however, phenotypic evaluations may be complicated
due to the use of antihypertensive drugs (60). The study further showed that after
controlling for confounders, the effect of certain CDH13
SNPs on hypertension among African Americans was weakly significant
(P=0.04) (60), indicating that
contextual or epigenetic variables may alter the impact of genotype
on hypertensive susceptibility. Cross-ethnic large-sample cohort
studies and the integration of multi-omics data are needed to
elucidate the precise role of CDH13 polymorphisms in
hypertension and thoroughly examine its molecular network to
identify novel targets for the precise treatment of
hypertension.
T-cadherin and heart failure
Heart failure, a common outcome in several
cardiovascular disorders, is becoming a notable public health
issue. Patients with heart failure typically have a low quality of
life in addition to high hospitalization and mortality rates
(67–69). According to estimates, 1–2% of all
individuals in industrialized cultures have heart failure, while in
those >70 years of age, the prevalence of heart failure is
>10%. In Europe, the lifetime risk of the disease is ~33% for
males and 28% for females aged >55 years (70), whereas in the US, the lifetime risk
is 20% for those aged >40 years (71). Compared with Western populations,
heart failure is more severe and develops at a younger age in
Asians and in low- and middle-income nations (69,72).
When MSCs are used to treat pressure-overload heart failure, the
adiponectin/T-cadherin system plays a key role in promoting
myocardial regeneration and reducing oxidative stress by increasing
exosome production and secretion. Experiments have demonstrated
that mice lacking T-cadherin or adiponectin have much lower exosome
counts and do not benefit from MSC therapy (30,73,74).
Heart failure frequently coexists with cardiac
remodeling, which is defined as changes in the structure and
function of the heart, and includes perivascular and interstitial
fibrosis, ventricular arrhythmias (75) and structural remodeling. These
changes alter the composition and structure of the heart, which in
turn causes ventricular hypertrophy, cardiac fibrosis and heart
failure (76,77). T-cadherin plays a key role in heart
failure by regulating oxidative stress and inflammatory signaling
implicated in cardiac fibrosis. The production of reactive oxygen
species and Nox2 mRNA expression are markedly downregulated in
T-cadherin-deficient mouse models of angiotensin II-induced
endothelial dysfunction, indicating that T-cadherin may affect
cardiac remodeling by regulating redox homeostasis. Furthermore,
the absence of T-cadherin reduces bleomycin-induced lung fibrosis;
however, further research is required to determine the precise
mechanism by which T-cadherin affects myocardial fibrosis (53). Adiponectin may suppress fibrosis
via the CDH13/p38 MAPKγ signaling axis, and reduced
CDH13 expression is linked to increased fibroblast
activation in pulmonary fibrosis (78). Since cardiac fibrosis is a
fundamental pathophysiological alteration in heart failure,
CDH13 may be a novel target for myocardial remodeling
interventions.
Other calcineurin proteins, including N-cadherin and
VE-cadherin, interact functionally with T-cadherin. Abnormal
cardiomyocyte coupling in patients with end-stage heart failure may
result from deregulation of the obscurin-kinase 1/N-cadherin axis
(79). Further research is needed
to determine whether a relationship exists between T-cadherin
expression and the obscurin-kinase 1/N-cadherin axis in heart
failure models. A previous study reported by Fukuda et al
(73) identified three novel
soluble isoforms of T-cadherin and reported that the 30 kDa isoform
was associated with clinically important indicators of heart
failure, such as B-type natriuretic peptide. Further studies are
needed to determine whether other signaling pathways or molecular
mechanisms are involved in heart failure pathology. T-cadherin
overexpression can damage endothelial intercellular junction
integrity and decrease VE-cadherin mRNA levels, affecting heart
function and vascular permeability (53). An ovarian cancer study revealed a
regulatory relationship between CDH13 and DNA
methyltransferase 1 (DNMT1) (80).
Since DNMT1 is important in myocardial hypertrophy and fibrosis,
CDH13 may indirectly affect the development of heart failure
through epigenetic networks. Future studies using data from various
sources, including studies at the cellular level, animal models and
clinical investigations, are needed to corroborate the association
between T-cadherin and heart failure.
T-cadherin and diabetes
Although the underlying molecular mechanisms are
unknown, several studies have linked the plasma levels of
adiponectin and T-cadherin to an increased risk of diabetes
(37,81,82).
In obese individuals and patients with type 2 diabetes, plasma
adiponectin levels are often reduced, a finding linked to insulin
resistance and β-cell dysfunction (81). This suggests that low adiponectin
levels are closely associated with diabetes. An animal study
demonstrated that T-cadherin deficiency increases the risk of
diabetes (37). Elevated levels of
T-cadherin do not directly increase the risk of diabetes; rather,
increased soluble T-cadherin levels serve primarily as a
biochemical marker of insulin deficiency or impaired insulin
signaling (82). Vascular
endothelial cells, cardiac muscles, MSCs and skeletal muscles are
the predominant cells that express T-cadherin, although pancreatic
β-cells also rarely express T-cadherin (37,57,74,82).
Although not expressed in β-cells, the endogenous humoral factor
soluble T-cadherin notably promotes β-cell proliferation.
Additionally, the Notch and IL-6/STAT3 pathways in pancreatic
islets are regulated by T-cadherin (37). The Notch pathway contributes to the
differentiation and proliferation of β-cells (83,84),
which are promoted by soluble T-cadherin regulation. However, the
absence of soluble T-cadherin suppresses the IL-6/STAT3 pathway and
the activity of protein kinases involved in β-cell proliferation
and apoptosis (37,85–87).
Notably, the proliferative effects of T-cadherin are tissue
specific; knocking out T-cadherin in the heart or skeletal muscles
does not impact β-cell function (82). However, in mice with
streptozotocin-induced diabetes, endothelial cell-specific
T-cadherin knockout is associated with a 26% decrease in plasma
soluble T-cadherin levels, which is accompanied by a marked
increase in blood glucose (82).
These findings imply that T-cadherin produced by endothelial cells
may modulate β-cell activity or systemic insulin sensitivity,
thereby regulating glucose metabolism.
A previous study demonstrated that diabetic mouse
models, such as leptin receptor-deficient db/db model mice,
have increased levels of plasma soluble T-cadherin (82), which could be an adaptive reaction
to the compensatory requirement for β-cell activity. Furthermore,
insulin can positively regulate the production of soluble
T-cadherin from endothelial cells through the insulin receptor/Akt
signaling pathway, and inadequate insulin secretion or insulin
resistance may interfere with this regulatory mechanism (35,37).
T-cadherin regulates insulin sensitivity via two different
mechanisms: i) The effects of adiponectin on ceramide turnover are
mediated via the T-cadherin/adiponectin signaling pathway, which
improves endothelial function and suppresses inflammatory
responses, resulting in increased insulin sensitivity (Fig. 1) (12,16,81);
and ii) T-cadherin upregulation in vascular cells may interfere
with insulin receptor signaling, resulting in endothelial insulin
resistance (12,88).
GWASs have shown that the risk of diabetes is
closely associated with CDH13 polymorphism. The minor allele
at the rs3865188 locus may have a protective effect against
diabetes (OR, 0.71; 95% CI, 0.58–0.88; P=0.002), whereas the
T-allele at the rs11646213 locus is associated with an elevated
risk of T2D (OR, 1.11; 95% CI, 1.04–1.18; P=0.001) (41,89).
Notably, the rs11646213 locus is also associated with elevated body
mass index (P=0.03), indicating that CDH13 polymorphisms may
influence metabolic pathways associated with obesity, thereby
indirectly increasing the risk of T2D (41). Although CDH13 polymorphisms
are markedly associated with T2D in white populations (42), in Han Chinese populations the
rs4783244 locus is not significantly associated with T2D (P=0.56),
whereas the guanine-allele of rs4311394 may be associated with an
increased risk of T2D (OR, 1.20; 95% CI, 1.01–1.41) (90). These differences may stem from the
modifying effects of the genetic background or environmental
factors. Similarly, the rs11646213 and rs3865188 variants are
associated with the prevalence, incidence and progression of type 1
diabetic nephropathy and this association may depend on plasma
adiponectin levels (89). The
absence of T-cadherin in the vascular segments of T2D models leads
to endothelial dysfunction, providing evidence that T-cadherin is
involved in the pathogenesis of T2D complications (55).
There has been limited research directly linking
T-cadherin to diabetes (12);
additional research is needed to determine its precise role in the
vascular endothelium, β-cells and insulin signaling. Future
research should also concentrate on the molecular mechanism of
T-cadherin/adiponectin interactions, variations in CDH13
polymorphisms and the possibility of targeting this receptor for
the treatment of diabetes. Furthermore, a bidirectional research
strategy of validating mechanistic targets derived from model
systems in well-phenotyped clinical cohorts, and conversely, using
genetic or phenotypic associations discovered in cohorts to inform
new mechanistic studies, will be essential to de-risk and
accelerate the development of T-cadherin-targeted therapies.
Although T-cadherin has a notable impact on
cardiovascular and metabolic health, it also plays a prominent role
in cancer biology. Unlike conventional cadherins, which are
typically associated with cancer progression, T-cadherin frequently
functions as a tumor suppressor. The loss of T-cadherin expression,
primarily due to CDH13 promoter hypermethylation, represents
a prevalent characteristic in several epithelial malignancies
(91–94).
T-cadherin and tumors
Previously, classical cadherins, including E-,
placental- and N-cadherins, have been the focus of the majority of
studies on cancer. It has been speculated that tumor progression is
linked to abnormal cadherin expression (92,95).
The role of T-cadherin in signaling pathways implicated in tumor
cell proliferation is discussed in the present section.
Tumorigenesis may activate various signaling
pathways, and T-cadherin regulates tumors via several signaling
mechanisms. CDH13 overexpression prevents activation of the
PI3K/Akt pathway, leading to decreased tumor cell survival and
metastasis (95). Additionally,
CDH13 inhibits β-catenin nuclear translocation and decreases
the expression of downstream genes such as c-Myc and
cyclin D1 to stop tumor cell growth (94). The degree of tumor immune
infiltration in clear-cell renal cell carcinoma (ccRCC) is
associated with CDH13 expression, and patient prognosis is
affected by CDH13 methylation status and N6-methyladenosine
alterations (96). Research has
demonstrated that CDH13 expression is positively associated
with prognosis in patients with ccRCC. Specifically, high
CDH13 expression is notably associated with improved overall
survival and progression-free survival, and associated with lower
tumor stage and grade. Conversely, high DNA methylation in the
CDH13 promoter region is negatively associated with its mRNA
expression and leads to poor patient prognosis. Furthermore,
CDH13 expression is regulated by N6-methyladenosine (m6A)
RNA modifications, and its associations with multiple m6A
regulatory factors collectively constitute a key epigenetic
mechanism influencing patient prognosis (96).
Numerous tumors exhibit markedly reduced T-cadherin
expression compared with healthy tissues (14,94,97,98).
In gastric cancer, lower T-cadherin protein levels have been
observed compared with normal tissues both in vivo and in
cell lines, and these reduced levels have been notably associated
with tumor aggressiveness and poor prognosis (99–102). T-cadherin expression is
associated with the overall survival rate of patients with gastric
cancer (99). Furthermore, in oral
squamous cell carcinoma, the methylation status of CDH13 is
markedly associated with tumor and lymph node stages, lymph node
metastasis and the risk of recurrence. Furthermore, patients with
methylated CHD13 have markedly lower overall and
progression-free survival than patients without methylated
CHD13 (103,104). Reduced expression of T-cadherin
in patients with triple-negative breast cancer is associated with
lower postoperative survival, indicating its potential as an
independent prognostic marker (105). Unlike other cancers, the
expression of T-cadherin is increased in ccRCC, suggesting a unique
expression pattern of this receptor. T-cadherin levels are markedly
higher in low-grade than high-grade ccRCC, and this expression
pattern can assist clinicians in diagnosis as well as tumor staging
and grading. Further survival analysis has revealed that increased
levels of T-cadherin are associated with improved overall and
progression-free survival in patients with ccRCC, indicating that
T-cadherin is a novel prognostic biomarker for ccRCC (96). However, the role of T-cadherin in
specific tumor types remains controversial. For example, in
prostate cancer, the expression of mRNA and protein levels of
T-cadherin are not perfectly associated, with only the protein
level being elevated. In prostate cancer, T-cadherin undergoes
dynamic regulation and may exhibit stage-dependent effects,
promoting differentiation and chemotherapy sensitivity in early
stages but gradually diminishing during disease progression
(97). This indicates that the
relationship between the expression pattern and function of
T-cadherin is highly context-dependent, rather than simply acting
as a tumor suppressor or promoter. To further investigate the
potential mechanism by which T-cadherin inhibits gastric cancer
cell proliferation, a study by Lin et al (100) reported that the overexpression of
T-cadherin downregulates the expression of CDK4 and cyclin D1,
inducing G0/G1 phase cell cycle arrest.
Additionally, T-cadherin overexpression upregulates E-cadherin and
downregulates vimentin and MMP-2 expression, preventing the
migration and invasion of gastric cancer cells (100). A molecular study has supported
that T-cadherin inhibits gastric tumorigenesis by suppressing the
Akt/mTOR signaling pathway (99).
These findings suggest that T-cadherin may serve as a potential
target for the treatment of gastric cancer and for predicting the
prognosis of patients with gastric cancer.
Animal experiments have provided evidence that
T-cadherin overexpression in pancreatic cancer reduces the volume
of in situ-grafted tumors and the incidence of hepatic and
mesenteric metastases. It also markedly inhibits the proliferation,
migration and invasiveness of tumor cells (94). Furthermore, the carcinogenic
effects of T-cadherin are dependent on the tumor microenvironment,
which might impact tumor growth by blocking pathways linked to
angiogenesis, such as the PI3K/Akt and Wnt/β-catenin pathways
(94,96). The primary mechanism for inhibiting
the expression of T-cadherin is hypermethylation of its promoter
region, which is common in ovarian, breast, pancreatic and
hepatocellular carcinomas. Studies have demonstrated that breast
cancer molecular subtypes can be differentiated based on T-cadherin
promoter methylation, with human epidermal growth factor receptor
2-positive and progesterone receptor-negative tumors exhibiting
noticeably greater levels of promoter methylation than other types
(106–108). miR-142-5p and the cyclic RNA
hsa_circ_0000119 regulate CDH13 promoter methylation in
ovarian cancer, with notably greater levels observed in malignant
tissues compared with those in adjacent normal tissues. By
increasing the expression of DNMT1, hsa_circ_0000119 promotes
hypermethylation of CDH13, thereby stimulating the
production of T-cadherin, which in turn accelerates the development
of ovarian cancer (80).
Furthermore, patients with hepatocellular carcinoma
exhibit a much greater frequency of CDH13 methylation than
the general population, and this methylation is linked to
environmental variables such as alcohol consumption and smoking
(109,110). These findings suggest that
CDH13 promoter methylation levels may serve as a potential
biomarker for predicting outcomes in patients with hepatocellular
carcinoma. Notably, by controlling the demethylation of the
CDH13 promoter, the DNA repair enzyme DNA polymerase β can
restore T-cadherin expression and prevent tumor spreading (111). Thus, restoring T-cadherin
expression or focusing on its regulatory mechanisms represent
possible treatment approaches. Demethylating medications such as
5-aza-2′-deoxycytidine have the potential to reverse cisplatin
resistance in lung cancer cells (112) and to restore T-cadherin
expression, which prevents tumor spreading (14,103). By upregulating T-cadherin
expression and blocking the PI3K/Akt pathway, the natural substance
garcinol markedly inhibits the development of transplanted tumors
and the proliferation of cervical cancer cells (95).
T-cadherin exerts tumor-suppressive effects in a
range of epithelial-derived malignancies (112,113). Epithelial-mesenchymal transition
(EMT) is an important step in morphogenesis that gives epithelial
cells a mesenchymal appearance. EMT occurs during various stages of
embryonic development, including placental creation, neural crest
development and gastrulation. Researchers have speculated that
epithelial malignancies must have mesenchymal characteristics to
penetrate and spread, since EMT is an important signature of
embryogenesis (112,113). A previous study demonstrated that
T-cadherin overexpression suppresses the Wnt/β-catenin signaling
pathway and prevents its activation by controlling the expression
of markers linked to EMT (Fig. 2)
(90).
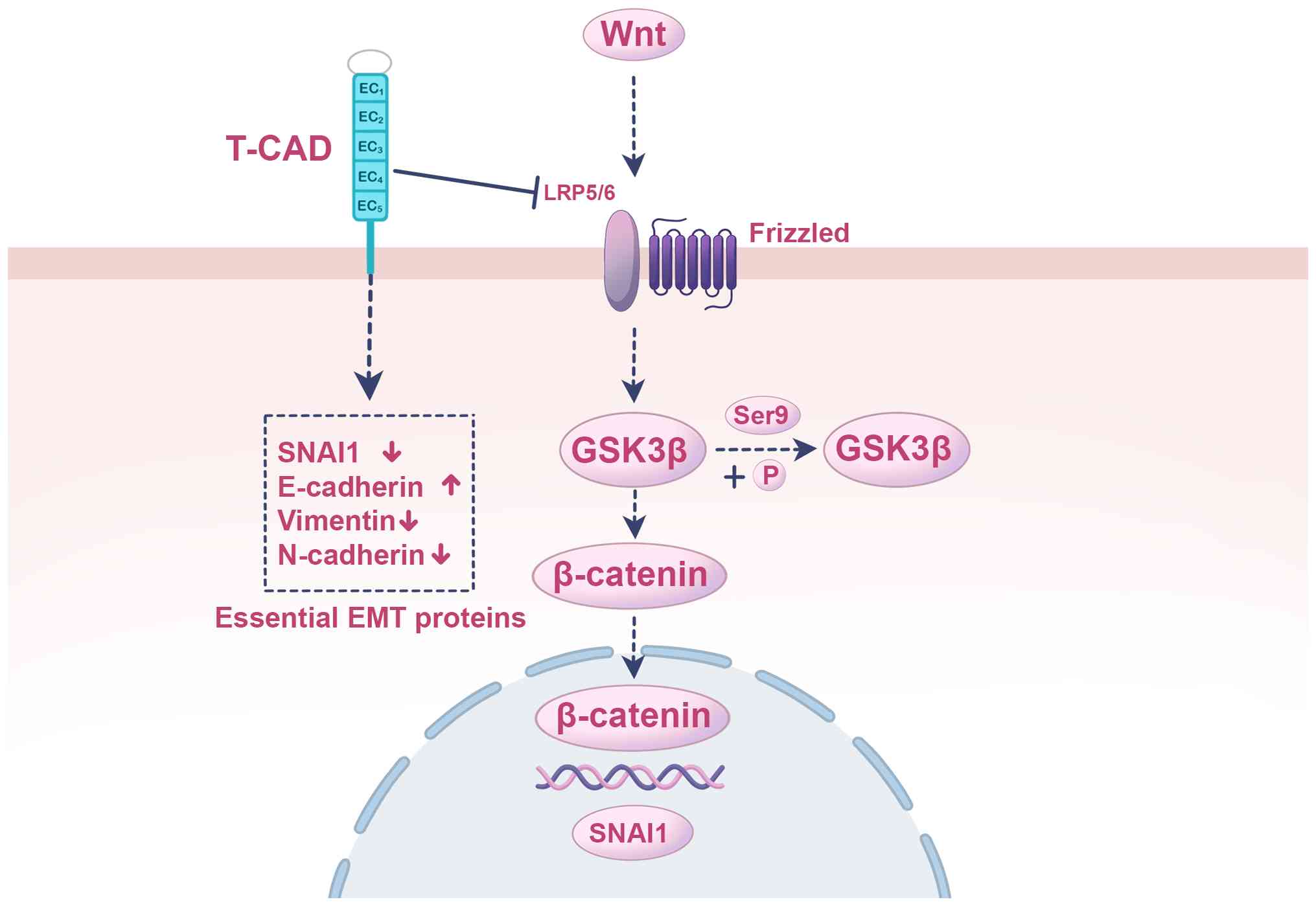 | Figure 2.T-cadherin upregulation inhibits EMT
by inactivating the Wnt/β-catenin pathway. The illustration depicts
the mechanism through which T-cadherin suppresses core signaling
that drives the EMT. Initiation of this suppression at the membrane
requires T-cadherin upregulation that downregulates the Wnt
co-receptor LRP5/6, impairing the canonical Wnt signaling pathway
initiated by Wnt ligands binding to Frizzled receptors. The
cytoplasmic signaling cascade is as follows: In the absence of the
Wnt signal, GSK3β remains active, involving a lack of
phosphorylation at Ser9. Active GSK3β then phosphorylates
β-catenin, marking it for proteasomal degradation. This prevents
β-catenin accumulation and its translocation into the nucleus.
Nuclear transcriptional regulation is mediated by the loss of
nuclear β-catenin, leading to reduced transcription of its target
genes, including the key EMT transcription factor SNAI1. Reduced
SNAI1 levels cause a reversal of EMT markers, manifested by the
upregulation of the epithelial marker E-cadherin and the
downregulation of the mesenchymal markers vimentin and N-cadherin,
thereby counteracting the invasive and metastatic phenotype that
accompanies EMT. T-CAD, T-cadherin; LRP5/6, low-density lipoprotein
receptor-related protein 5/6; GSK3β, glycogen synthase kinase 3β;
Ser9, serine 9; P, phosphate group; SNAI1, zinc finger protein
SNAI1; EMT, epithelial-mesenchymal transition; E-cadherin,
epithelial-cadherin; N-cadherin, neural-cadherin. |
The tumor-suppressive role of T-cadherin is
supported by multiple studies. Future research should combine
single-cell sequencing and spatial transcriptome technologies to
fully understand the function of T-cadherin in regulating tumor
heterogeneity and microenvironmental remodeling, as well as to
explore its potential as a combination therapeutic target. The
complex, context-dependent actions of T-cadherin in various
disorders, including cardiovascular diseases, metabolic diseases
and cancer, emphasize its unique status as a GPI-anchored atypical
cadherin that mediates important extracellular signals. Thus,
unraveling the complexities of relevant signaling processes is
necessary.
Conclusions
Despite belonging to the cadherin superfamily,
T-cadherin does not function as a classical cadherin due to the
lack of transmembrane and cytoplasmic structural domains. This
unique structure makes it challenging to understand how T-cadherin
mediates intracellular signaling. Signal transduction by
GPI-anchored proteins require interactions with bridging molecules
within the lipid rafts of the plasma membrane (114,115). Both T-cadherin and
integrin-linked kinase (ILK) are localized in plasma membrane lipid
rafts (44,116). Studies have shown that ILK plays
a role in strain-induced signal transduction (117,118), and it has been shown that ILK is
a signaling regulator of GPI-anchored proteins (119). Another hypothesis for
T-cadherin-mediated signal transduction is that T-cadherin
co-localizes with caveolin, a marker of caveolae, within a smaller
detergent-insoluble low-density membrane domain. This domain is
rich in other GPI-anchored proteins, such as CD-59 and
urokinase-type plasminogen activator receptors, and signaling
molecules, such as the G protein αs subunit and Src family kinases;
therefore, T-cadherin may interact with caveolae to mediate
membrane signaling (44). Notably,
caveolae and cell membrane lipid rafts are considered centers of
signal transduction (117,118,120).
Further research is needed to identify co-adaptor molecules for
GPI-anchored proteins or specific signal transduction mediators of
T-cadherin.
As a receptor for LDL and HMW adiponectin,
T-cadherin often exerts opposing effects upon binding to these
ligands. LDL and HMW adiponectin induce the formation of distinct
T-cadherin clusters, leading to different cellular responses
(25), which may explain their
distinct physiological effects. A competitive relationship exists
between LDL and HMW adiponectin for T-cadherin binding, as both are
large complexes of similar diameters of ~25 nm (120). Adiponectin inhibits
LDL-stimulated Ca2+ signaling in
T-cadherin-overexpressing 293 cells (120,121). Furthermore, the expression of
adiponectin is decreased in obesity, whereas LDL levels are
increased. Changes in adiponectin-LDL ratio may drive different
pathological responses (56).
Numerous studies have shown that T-cadherin
primarily functions as a signaling molecule involved in cellular
regulation, proliferation, apoptosis and migration. Serving as a
receptor for both LDL and adiponectin, with genetic variations in
the CDH13 gene, T-cadherin plays notable roles in
cardiovascular diseases, diabetes and cancer. Nevertheless, key
questions regarding tissue specificity, signaling crosstalk and the
precise mechanisms of T-cadherin activity in major intracellular
signaling pathways remain to be fully elucidated.
Acknowledgements
Not applicable.
Funding
The present review was funded by the National Natural Science
Foundation of China (grant no. 82070472).
Availability of data and materials
Not applicable.
Authors' contributions
YS designed the review; YS and XW retrieved the
relevant literature. YS and YB wrote and reviewed the article. XW
wrote the figure legends and created the figures. Data
authentication is not applicable. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Roth GA, Mensah GA, Johnson CO, Addolorato
G, Ammirati E, Baddour LM, Barengo NC, Beaton AZ, Benjamin EJ,
Benziger CP, et al: Global burden of cardiovascular diseases and
risk factors, 1990–2019: Update from the GBD 2019 study. J Am Coll
Cardiol. 76:2982–3021. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wu Z, Xia F and Lin R: Global burden of
cancer and associated risk factors in 204 countries and
territories, 1980–2021: A systematic analysis for the GBD 2021. J
Hematol Oncol. 17:1192024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
GBD 2021 Diabetes Collaborators, . Global,
regional, and national burden of diabetes from 1990 to 2021, with
projections of prevalence to 2050: A systematic analysis for the
global burden of disease study 2021. Lancet. 402:203–234. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Spaander MCW, Zauber AG, Syngal S, Blaser
MJ, Sung JJ, You YN and Kuipers EJ: Young-onset colorectal cancer.
Nat Rev Dis Primers. 9:212023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Misra S, Ke C, Srinivasan S, Goyal A,
Nyriyenda MJ, Florez JC, Khunti K, Magliano DJ and Luk A: Current
insights and emerging trends in early-onset type 2 diabetes. Lancet
Diabetes Endocrinol. 11:768–782. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mousavi I, Suffredini J, Virani SS,
Ballantyne CM, Michos ED, Misra A, Saeed A and Jia X: Early-onset
atherosclerotic cardiovascular disease. Eur J Prev Cardiol.
32:100–112. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sun J, Qiao Y, Zhao M, Magnussen CG and Xi
B: Global, regional, and national burden of cardiovascular diseases
in youths and young adults aged 15–39 years in 204
countries/territories, 1990–2019: A systematic analysis of global
burden of disease study 2019. BMC Med. 21:2222023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ciumărnean L, Milaciu MV, Negrean V,
Orășan OH, Vesa SC, Sălăgean O, Iluţ S and Vlaicu SI:
Cardiovascular risk factors and physical activity for the
prevention of cardiovascular diseases in the elderly. Int J Environ
Res Public Health. 19:2072021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rouland A, Thuillier P, Al-Salameh A,
Benzerouk F, Bahougne T, Tramunt B, Berlin I, Clair C, Thomas D, Le
Faou AL, et al: Smoking and diabetes. Ann Endocrinol (Paris).
85:614–622. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ranscht B and Dours-Zimmermann MT:
T-cadherin, a novel cadherin cell adhesion molecule in the nervous
system lacks the conserved cytoplasmic region. Neuron. 7:391–402.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Philippova M, Joshi MB, Kyriakakis E,
Pfaff D, Erne P and Resink TJ: A guide and guard: The many faces of
T-cadherin. Cell Signal. 21:1035–1044. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rubina KA, Semina EV, Kalinina NI, Sysoeva
VY, Balatskiy AV and Tkachuk VA: Revisiting the multiple roles of
T-cadherin in health and disease. Eur J Cell Biol. 100:1511832021.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rubina KA and Tkachuk VA: Guidance
receptors in the nervous and cardiovascular systems. Biochemistry
(Mosc). 80:1235–1253. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Di Palo A, Siniscalchi C, Polito R, Nigro
E, Russo A, Daniele A and Potenza N: microRNA-377-3p downregulates
the oncosuppressor T-cadherin in colorectal adenocarcinoma cells.
Cell Biol Int. 45:1797–1803. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hayano Y, Zhao H, Kobayashi H, Takeuchi K,
Norioka S and Yamamoto N: The role of T-cadherin in axonal pathway
formation in neocortical circuits. Development. 141:4784–4793.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sysoeva V, Semina E, Klimovich P,
Kulebyakin K, Dzreyan V, Sotskaya E, Shchipova A, Popov V, Shilova
A, Brodsky I, et al: T-cadherin modulates adipogenic
differentiation in mesenchymal stem cells: Insights into ligand
interactions. Front Cell Dev Biol. 12:14463632024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kita S, Fukuda S, Maeda N and Shimomura I:
Native adiponectin in serum binds to mammalian cells expressing
T-cadherin, but not AdipoRs or calreticulin. Elife. 8:e486752019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yamauchi T, Iwabu M, Okada-Iwabu M and
Kadowaki T: Adiponectin receptors: A review of their structure,
function and how they work. Best Pract Res Clin Endocrinol Metab.
28:15–23. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hug C, Wang J, Ahmad NS, Bogan JS, Tsao TS
and Lodish HF: T-cadherin is a receptor for hexameric and
high-molecular-weight forms of Acrp30/adiponectin. Proc Natl Acad
Sci USA. 101:10308–10313. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fukuda S, Kita S, Obata Y, Fujishima Y,
Nagao H, Masuda S, Tanaka Y, Nishizawa H, Funahashi T, Takagi J, et
al: The unique prodomain of T-cadherin plays a key role in
adiponectin binding with the essential extracellular cadherin
repeats 1 and 2. J Biol Chem. 292:7840–7849. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ciatto C, Bahna F, Zampieri N,
VanSteenhouse HC, Katsamba PS, Ahlsen G, Harrison OJ, Brasch J, Jin
X, Posy S, et al: T-cadherin structures reveal a novel adhesive
binding mechanism. Nat Struct Mol Biol. 17:339–347. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bochkov VN, Tkachuk VA, Philippova MP,
Stambolsky DV, Bühler FR and Resink TJ: Ligand selectivity of 105
and 130 kDa lipoprotein-binding proteins in
vascular-smooth-muscle-cell membranes is unique. Biochem J.
317:297–304. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Niermann T, Kern F, Erne P and Resink T:
The glycosyl phosphatidylinositol anchor of human T-cadherin binds
lipoproteins. Biochem Biophys Res Commun. 276:1240–1247. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Parker-Duffen JL, Nakamura K, Silver M,
Kikuchi R, Tigges U, Yoshida S, Denzel MS, Ranscht B and Walsh K:
T-cadherin is essential for adiponectin-mediated revascularization.
J Biol Chem. 288:24886–24897. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Balatskaya MN, Sharonov GV, Baglay AI,
Rubtsov YP and Tkachuk VA: Different spatiotemporal organization of
GPI-anchored T-cadherin in response to low-density lipoprotein and
adiponectin. Biochim Biophys Acta Gen Subj. 1863:1294142019.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rubina K, Talovskaya E, Cherenkov V,
Ivanov D, Stambolsky D, Storozhevykh T, Pinelis V, Shevelev A,
Parfyonova Y, Resink T, et al: LDL induces intracellular signalling
and cell migration via atypical LDL-binding protein T-cadherin. Mol
Cell Biochem. 273:33–41. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kipmen-Korgun D, Osibow K, Zoratti C,
Schraml E, Greilberger J, Kostner GM, Jürgens G and Graier WF:
T-cadherin mediates low-density lipoprotein-initiated cell
proliferation via the Ca(2+)-tyrosine kinase-Erk1/2 pathway. J
Cardiovasc Pharmacol. 45:418–430. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Iioka M, Fukuda S, Maeda N, Natsukawa T,
Kita S, Fujishima Y, Sawano H, Nishizawa H and Shimomura I:
Time-series change of serum soluble T-Cadherin concentrations and
its association with creatine kinase-MB levels in ST-segment
elevation myocardial infarction. J Atheroscler Thromb.
29:1823–1834. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tsugawa-Shimizu Y, Fujishima Y, Kita S,
Minami S, Sakaue TA, Nakamura Y, Okita T, Kawachi Y, Fukada S,
Namba-Hamano T, et al: Increased vascular permeability and severe
renal tubular damage after ischemia-reperfusion injury in mice
lacking adiponectin or T-cadherin. Am J Physiol Endocrinol Metab.
320:E179–E190. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kita S and Shimomura I: Stimulation of
exosome biogenesis by adiponectin, a circulating factor secreted
from adipocytes. J Biochem. 169:173–179. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lee MH, Klein RL, El-Shewy HM, Luttrell DK
and Luttrell LM: The adiponectin receptors AdipoR1 and AdipoR2
activate ERK1/2 through a Src/Ras-dependent pathway and stimulate
cell growth. Biochemistry. 47:11682–11692. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Popov VS, Brodsky IB, Balatskaya MN,
Balatskiy AV, Ozhimalov ID, Kulebyakina MA, Semina EV, Arbatskiy
MS, Isakova VS, Klimovich PS, et al: T-cadherin deficiency is
associated with increased blood pressure after physical activity.
Int J Mol Sci. 24:142042023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dasen B, Pigeot S, Born GM, Verrier S,
Rivero O, Dittrich PS, Martin I and Filippova M: T-cadherin is a
novel regulator of pericyte function during angiogenesis. Am J
Physiol Cell Physiol. 324:C821–C836. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Obata Y, Kita S, Koyama Y, Fukuda S,
Takeda H, Takahashi M, Fujishima Y, Nagao H, Masuda S, Tanaka Y, et
al: Adiponectin/T-cadherin system enhances exosome biogenesis and
decreases cellular ceramides by exosomal release. JCI Insight.
3:e996802018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Clark JL, Taylor CG and Zahradka P:
Exploring the cardio-metabolic relevance of T-cadherin: A
pleiotropic adiponectin receptor. Endocr Metab Immune Disord Drug
Targets. 17:200–206. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kita S, Maeda N and Shimomura I:
Interorgan communication by exosomes, adipose tissue, and
adiponectin in metabolic syndrome. J Clin Invest. 129:4041–4049.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Okita T, Kita S, Fukuda S, Fukuoka K,
Kawada-Horitani E, Iioka M, Nakamura Y, Fujishima Y, Nishizawa H,
Kawamori D, et al: Soluble T-cadherin promotes pancreatic β-cell
proliferation by upregulating Notch signaling. iScience.
25:1054042022. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fujishima Y, Kita S, Nishizawa H, Maeda N
and Shimomura I: Cardiovascular significance of adipose-derived
adiponectin and liver-derived xanthine oxidoreductase in metabolic
syndrome. Endocr J. 70:663–675. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tan WH, Peng ZL, You T and Sun ZL: CTRP15
promotes macrophage cholesterol efflux and attenuates
atherosclerosis by increasing the expression of ABCA1. J Physiol
Biochem. 78:653–666. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Rubina K, Maier A, Klimovich P, Sysoeva V,
Romashin D, Semina E and Tkachuk V: T-cadherin (CDH13) and
non-coding RNAs: The crosstalk between health and disease. Int J
Mol Sci. 26:61272025. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Nicolas A, Aubert R, Bellili-Muñoz N,
Balkau B, Bonnet F, Tichet J, Velho G, Marre M, Roussel R and
Fumeron F: T-cadherin gene variants are associated with type 2
diabetes and the fatty liver index in the French population.
Diabetes Metab. 43:33–39. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Tarantino G, Costantini S, Finelli C,
Capone F, Guerriero E, La Sala N, Gioia S and Castello G: Carotid
intima-media thickness is predicted by combined eotaxin levels and
severity of hepatic steatosis at ultrasonography in obese patients
with nonalcoholic fatty liver disease. PLoS One. 9:e1056102014.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Philippova M, Suter Y, Toggweiler S,
Schoenenberger AW, Joshi MB, Kyriakakis E, Erne P and Resink TJ:
T-cadherin is present on endothelial microparticles and is elevated
in plasma in early atherosclerosis. Eur Heart J. 32:760–771. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Philippova MP, Bochkov VN, Stambolsky DV,
Tkachuk VA and Resink TJ: T-cadherin and signal-transducing
molecules co-localize in caveolin-rich membrane domains of vascular
smooth muscle cells. FEBS Lett. 429:207–210. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Göddeke S, Knebel B, Fahlbusch P, Hörbelt
T, Poschmann G, van de Velde F, Benninghoff T, Al-Hasani H, Jacob
S, Van Nieuwenhove Y, et al: CDH13 abundance interferes with
adipocyte differentiation and is a novel biomarker for adipose
tissue health. Int J Obes (Lond). 42:1039–1050. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Summerhill VI, Grechko AV, Yet SF, Sobenin
IA and Orekhov AN: The atherogenic role of circulating modified
lipids in atherosclerosis. Int J Mol Sci. 20:35612019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Fujishima Y, Maeda N, Matsuda K, Masuda S,
Mori T, Fukuda S, Sekimoto R, Yamaoka M, Obata Y, Kita S, et al:
Adiponectin association with T-cadherin protects against neointima
proliferation and atherosclerosis. FASEB J. 31:1571–1583. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Philippova M, Banfi A, Ivanov D,
Gianni-Barrera R, Allenspach R, Erne P and Resink T: Atypical
GPI-anchored T-cadherin stimulates angiogenesis in vitro and in
vivo. Arterioscler Thromb Vasc Biol. 26:2222–2230. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Yan F, Sun SY and Wu H: The
‘angiogenesis-plaque stability paradox’ in atherosclerosis
pathogenesis. Front Cardiovasc Med. 12:16590062025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lin A, Miano JM, Fisher EA and Misra A:
Chronic inflammation and vascular cell plasticity in
atherosclerosis. Nat Cardiovasc Res. 3:1408–1423. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Joshi MB, Kyriakakis E, Pfaff D, Rupp K,
Philippova M, Erne P and Resink TJ: Extracellular cadherin repeat
domains EC1 and EC5 of T-cadherin are essential for its ability to
stimulate angiogenic behavior of endothelial cells. FASEB J.
23:4011–4021. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Semina EV, Rubina KA, Sysoeva VY,
Rutkevich PN, Kashirina NM and Tkachuk VA: Novel mechanism
regulating endothelial permeability via T-cadherin-dependent
VE-cadherin phosphorylation and clathrin-mediated endocytosis. Mol
Cell Biochem. 387:39–53. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Semina E, Popov V, Khabibullin N,
Klimovich P, Sysoeva V, Kurilina E, Tsokolaeva Z, Tkachuk V and
Rubina K: New evidence for T-cadherin in COVID-19 pathogenesis,
endothelial dysfunction, and lung fibrosis. Front Cell Dev Biol.
13:14763292025. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Schäfer S, Gogiraju R, Rösch M, Kerstan Y,
Beck L, Garbisch J, Saliba AE, Gisterå A, Hermanns HM, Boon L, et
al: CD8+ T cells drive plaque smooth muscle cell
dedifferentiation in experimental atherosclerosis. Arterioscler
Thromb Vasc Biol. 44:1852–1872. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wang H, Tao L, Ambrosio A, Yan W, Summer
R, Lau WB, Wang Y and Ma X: T-cadherin deficiency increases
vascular vulnerability in T2DM through impaired NO bioactivity.
Cardiovasc Diabetol. 16:122017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Balatskiy A, Teterina M, Pisaryuk A,
Balabanenko I, Kadrev A, Tishuk A, Balatskaya M, Samokhodskaya L,
Boytsov S, Kalinina N and Tkachuk V: T-cadherin and the ratio of
its ligands as predictors of carotid atherosclerosis: A pilot
study. Biomedicines. 9:13982021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Denzel MS, Scimia MC, Zumstein PM, Walsh
K, Ruiz-Lozano P and Ranscht B: T-cadherin is critical for
adiponectin-mediated cardioprotection in mice. J Clin Invest.
120:4342–4352. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Vitale MR, Zöller JEM, Jansch C, Janz A,
Edenhofer F, Klopocki E, van den Hove D, Vanmierlo T, Rivero O,
Nadif Kasri N, et al: Generation of induced pluripotent stem cell
(iPSC) lines carrying a heterozygous (UKWMPi002-A-1) and null
mutant knockout (UKWMPi002-A-2) of cadherin 13 associated with
neurodevelopmental disorders using CRISPR/Cas9. Stem Cell Res.
51:1021692021. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Mills KT, Bundy JD, Kelly TN, Reed JE,
Kearney PM, Reynolds K, Chen J and He J: Global disparities of
hypertension prevalence and control: A systematic analysis of
population-based studies from 90 countries. Circulation.
134:441–450. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Kidambi S, Ghosh S, Kotchen JM, Grim CE,
Krishnaswami S, Kaldunski ML, Cowley AW Jr, Patel SB and Kotchen
TA: Non-replication study of a genome-wide association study for
hypertension and blood pressure in African Americans. BMC Med
Genet. 13:272012. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lee JH, Shin DJ, Park S, Kang SM, Jang Y
and Lee SH: Association between CDH13 variants and cardiometabolic
and vascular phenotypes in a Korean population. Yonsei Med J.
54:1305–1312. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Vargas-Alarcon G, Martinez-Rodriguez N,
Velazquez-Cruz R, Perez-Mendez O, Posadas-Sanchez R, Posadas-Romero
C, Peña-Duque MA, Martinez-Rios MA, Ramirez-Fuentes S and Fragoso
JM: The T>A (rs11646213) gene polymorphism of cadherin-13
(CDH13) gene is associated with decreased risk of developing
hypertension in Mexican population. Immunobiology. 222:973–978.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kim HJ, Seo YS, Sung J, Son HY, Yun JM,
Kwon H, Cho B, Kim JI and Park JH: Interactions of CDH13 gene
polymorphisms and ambient PM10 air pollution exposure
with blood pressure and hypertension in Korean men. Chemosphere.
218:292–298. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Spijkers LJA, van den Akker RFP, Janssen
BJA, Debets JJ, De Mey JGR, Stroes ESG, van den Born BJH,
Wijesinghe DS, Chalfant CE, MacAleese L, et al: Hypertension is
associated with marked alterations in sphingolipid biology: A
potential role for ceramide. PLoS One. 6:e218172011. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Berry C, Touyz R, Dominiczak AF, Webb RC
and Johns DG: Angiotensin receptors: Signaling, vascular
pathophysiology, and interactions with ceramide. Am J Physiol Heart
Circ Physiol. 281:H2337–H2365. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Fenger M, Linneberg A, Jørgensen T,
Madsbad S, Søbye K, Eugen-Olsen J and Jeppesen J: Genetics of the
ceramide/sphingosine-1-phosphate rheostat in blood pressure
regulation and hypertension. BMC Genet. 12:442011. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Fang G, Li Y, Yuan J, Cao W, Song S, Chen
L, Wang Y and Wang Q: Cadherin-11-interleukin-6 signaling between
cardiac fibroblast and cardiomyocyte promotes ventricular
remodeling in a mouse pressure overload-induced heart failure
model. Int J Mol Sci. 24:65492023. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Groenewegen A, Rutten FH, Mosterd A and
Hoes AW: Epidemiology of heart failure. Eur J Heart Fail.
22:1342–1356. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Tromp J, Ferreira JP, Janwanishstaporn S,
Shah M, Greenberg B, Zannad F and Lam CSP: Heart failure around the
world. Eur J Heart Fail. 21:1187–1196. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Bleumink GS, Knetsch AM, Sturkenboom MCJM,
Straus SMJM, Hofman A, Deckers JW, Witteman JCM and Stricker BHC:
Quantifying the heart failure epidemic: Prevalence, incidence rate,
lifetime risk and prognosis of heart failure the Rotterdam study.
Eur Heart J. 25:1614–1619. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Yancy CW, Jessup M, Bozkurt B, Butler J,
Casey DE Jr, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi
JL, et al: 2013 ACCF/AHA guideline for the management of heart
failure: A report of the American college of cardiology
foundation/American heart association task force on practice
guidelines. J Am Coll Cardiol. 62:e147–e239. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Yoo SGK, Lam CSP and Sweitzer NK: ‘Asian’
heart failure. Circulation. 150:177–179. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Fukuda S, Kita S, Miyashita K, Iioka M,
Murai J, Nakamura T, Nishizawa H, Fujishima Y, Morinaga J, Oike Y,
et al: Identification and clinical associations of 3 forms of
circulating T-cadherin in human serum. J Clin Endocrinol Metab.
106:1333–1344. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Nakamura Y, Kita S, Tanaka Y, Fukuda S,
Obata Y, Okita T, Nishida H, Takahashi Y, Kawachi Y,
Tsugawa-Shimizu Y, et al: Adiponectin stimulates exosome release to
enhance mesenchymal stem-cell-driven therapy of heart failure in
mice. Mol Ther. 28:2203–2219. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Chen X, Qin M, Jiang W, Zhang Y and Liu X:
Electrophysiological characteristics of pressure overload-induced
cardiac hypertrophy and its influence on ventricular arrhythmias.
PLoS One. 12:e01836712017. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Cutler MJ, Jeyaraj D and Rosenbaum DS:
Cardiac electrical remodeling in health and disease. Trends
Pharmacol Sci. 32:174–180. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Armoundas AA, Wu R, Juang G, Marbán E and
Tomaselli GF: Electrical and structural remodeling of the failing
ventricle. Pharmacol Ther. 92:213–230. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Nemeth J, Skronska-Wasek W, Keppler S,
Schundner A, Groß A, Schoenberger T, Quast K, El Kasmi KC, Ruppert
C, Günther A and Frick M: Adiponectin suppresses
stiffness-dependent, profibrotic activation of lung fibroblasts. Am
J Physiol Lung Cell Mol Physiol. 327:L487–L502. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wang L, Tsakiroglou P, Gonzales R, Cho S,
Li A, Dos Remedios C, Wright N and Kontrogianni-Konstantopoulos A:
Essential role of obscurin kinase-1 in cardiomyocyte coupling via
N-cadherin phosphorylation. JCI Insight. 9:e1621782024.PubMed/NCBI
|
|
80
|
Ma G, Li Y, Meng F, Sui C, Wang Y and
Cheng D: Hsa_circ_0000119 promoted ovarian cancer development via
enhancing the methylation of CDH13 by sponging miR-142-5p. J
Biochem Mol Toxicol. 37:e232642023. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Straub LG and Scherer PE: Metabolic
messengers: Adiponectin. Nat Metab. 1:334–339. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Okita T, Kita S, Fukuda S, Kondo Y, Sakaue
TA, Iioka M, Fukuoka K, Kawada K, Nagao H, Obata Y, et al: Soluble
T-cadherin secretion from endothelial cells is regulated via
insulin/PI3K/Akt signalling. Biochem Biophys Res Commun.
732:1504032024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Bartolome A, Zhu C, Sussel L and Pajvani
UB: Notch signaling dynamically regulates adult β cell
proliferation and maturity. J Clin Invest. 129:268–280. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Eom YS, Gwon AR, Kwak KM, Youn JY, Park H,
Kim KW and Kim BJ: Notch1 has an important role in β-cell mass
determination and development of diabetes. Diabetes Metab J.
45:86–96. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
De Groef S, Renmans D, Cai Y, Leuckx G,
Roels S, Staels W, Gradwohl G, Baeyens L, Heremans Y, Martens GA,
et al: STAT3 modulates β-cell cycling in injured mouse pancreas and
protects against DNA damage. Cell Death Dis. 7:e22722016.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Ma J, Xing B, Cao Y, He X, Bennett KE,
Tong C, An C, Hojnacki T, Feng Z, Deng S, et al: Menin-regulated
Pbk controls high fat diet-induced compensatory beta cell
proliferation. EMBO Mol Med. 13:e135242021. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Rosado-Olivieri EA, Aigha II, Kenty JH and
Melton DA: Identification of a LIF-responsive,
replication-competent subpopulation of human β cells. Cell Metab.
31:327–338.e6. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Philippova M, Joshi MB, Pfaff D,
Kyriakakis E, Maslova K, Erne P and Resink TJ: T-cadherin
attenuates insulin-dependent signalling, eNOS activation, and
angiogenesis in vascular endothelial cells. Cardiovasc Res.
93:498–507. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Nicolas A, Mohammedi K, Bastard JP,
Fellahi S, Bellili-Muñoz N, Roussel R, Hadjadj S, Marre M, Velho G
and Fumeron F: T-cadherin gene variants are associated with
nephropathy in subjects with type 1 diabetes. Nephrol Dial
Transplant. 32:1987–1993. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Li Y, Yang Y, Yao Y, Li X, Shi L, Zhang Y,
Xiong Y, Yan M, Yao Y and Xiao C: Association study of ARL15 and
CDH13 with T2DM in a Han Chinese population. Int J Med Sci.
11:522–527. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Lee G, Wong C, Cho A, West JJ, Crawford
AJ, Russo GC, Si BR, Kim J, Hoffner L, Jang C, et al: E-cadherin
induces serine synthesis to support progression and metastasis of
breast cancer. Cancer Res. 84:2820–2835. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Agiostratidou G, Hulit J, Phillips GR and
Hazan RB: Differential cadherin expression: Potential markers for
epithelial to mesenchymal transformation during tumor progression.
J Mammary Gland Biol Neoplasia. 12:127–133. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Paredes J, Correia AL, Ribeiro AS,
Albergaria A, Milanezi F and Schmitt FC: P-cadherin expression in
breast cancer: A review. Breast Cancer Res. 9:2142007. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Xu D, Yuan H, Meng Z, Yang C, Li Z, Li M,
Zhang Z, Gan Y and Tu H: Cadherin 13 inhibits pancreatic cancer
progression and epithelial-mesenchymal transition by Wnt/β-catenin
signaling. J Cancer. 11:2101–2112. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhao J, Yang T, Ji J, Li C, Li Z and Li L:
Garcinol exerts anti-cancer effect in human cervical cancer cells
through upregulation of T-cadherin. Biomed Pharmacother.
107:957–966. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Shao Y, Li W, Zhang L, Xue B, Chen Y,
Zhang Z, Wang D and Wu B: CDH13 is a prognostic biomarker and a
potential therapeutic target for patients with clear cell renal
cell carcinoma. Am J Cancer Res. 12:4520–4544. 2022.PubMed/NCBI
|
|
97
|
Dasen B, Vlajnic T, Mengus C, Ruiz C,
Bubendorf L, Spagnoli G, Wyler S, Erne P, Resink TJ and Philippova
M: T-cadherin in prostate cancer: Relationship with cancer
progression, differentiation and drug resistance. J Pathol Clin
Res. 3:44–57. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Wang Y, Zhang L, Yang J, Li B and Wang J:
CDH13 promoter methylation regulates cisplatin resistance of
non-small cell lung cancer cells. Oncol Lett. 16:5715–5722.
2018.PubMed/NCBI
|
|
99
|
Lin J, Chen Z, Huang Z, Chen F, Ye Z, Lin
S and Wang W: Effect of T-cadherin on the AKT/mTOR signaling
pathway, gastric cancer cell cycle, migration and invasion, and its
association with patient survival rate. Exp Ther Med. 17:3607–3613.
2019.PubMed/NCBI
|
|
100
|
Lin J, Chen Z, Huang Z, Chen F, Ye Z, Lin
S and Wang W: Upregulation of T-cadherin suppresses cell
proliferation, migration and invasion of gastric cancer in
vitro. Exp Ther Med. 14:4194–4200. 2017.PubMed/NCBI
|
|
101
|
Wei B, Shi H, Lu X, Shi A, Cheng Y and
Dong L: Association between the expression of T-cadherin and
vascular endothelial growth factor and the prognosis of patients
with gastric cancer. Mol Med Rep. 12:2075–2081. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Tang Y, Dai Y and Huo J: Decreased
expression of T-cadherin is associated with gastric cancer
prognosis. Hepatogastroenterology. 59:1294–1298. 2012.PubMed/NCBI
|
|
103
|
Wang Q, Chen Y, Chen Y, Jiang J, Song X,
Zhang L, He Q, Ye B, Wu L, Wu R, et al: Aberrant promoter
methylation of T-cadherin in sera is associated with a poor
prognosis in oral squamous cell carcinoma. Neoplasma. 68:528–534.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Wang Q, Zhang X, Song X and Zhang L:
Overexpression of T-cadherin inhibits the proliferation of oral
squamous cell carcinoma through the PI3K/AKT/mTOR intracellular
signalling pathway. Arch Oral Biol. 96:74–79. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Kong DD, Wang MH, Yang J, Li L, Wang W,
Wang SB and Zhou YZ: T-cadherin is associated with prognosis in
triple-negative breast cancer. Oncol Lett. 14:2975–2981. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Yang J, Niu H, Huang Y and Yang K: A
systematic analysis of the relationship of CDH13 promoter
methylation and breast cancer risk and prognosis. PLoS One.
11:e01491852016. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Baranová I, Samec M, Dvorská D, Šťastný I,
Janíková K, Kašubová I, Hornáková A, Lukáčová E, Kapinová A,
Biringer K, et al: Droplet digital PCR analysis of CDH13
methylation status in Slovak women with invasive ductal breast
cancer. Sci Rep. 14:147002024. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Pang JM, Deb S, Takano EA, Byrne DJ, Jene
N, Boulghourjian A, Holliday A, Millar E, Lee CS, O'Toole SA, et
al: Methylation profiling of ductal carcinoma in situ and its
relationship to histopathological features. Breast Cancer Res.
16:4232014. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Yuan XD, Wang JW, Fang Y, Qian Y, Gao S,
Fan YC and Wang K: Methylation status of the T-cadherin gene
promotor in peripheral blood mononuclear cells is associated with
HBV-related hepatocellular carcinoma progression. Pathol Res Pract.
216:1529142020. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Tuoya AD, Wang DX, Xing YS, Liu RJ, Hu YX,
Zhang MD, Bai TY, Lv XL, Li J and Chang FH: Relationship between
methylation of FHIT and CDH13 gene promoter region and liver
cancer. Curr Med Sci. 40:502–509. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wang M, Long K, Li E, Li L, Li B, Ci S, He
L, Pan F, Hu Z and Guo Z: DNA polymerase beta modulates cancer
progression via enhancing CDH13 expression by promoter
demethylation. Oncogene. 39:5507–5519. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Nieto MA, Huang RY, Jackson RA and Thiery
JP: EMT: 2016. Cell. 166:21–45. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Kalluri R and Weinberg RA: The basics of
epithelial-mesenchymal transition. J Clin Invest. 119:1420–1428.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Zajchowski LD and Robbins SM: Lipid rafts
and little caves. Compartmentalized signalling in membrane
microdomains. Eur J Biochem. 269:737–752. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Lucero HA and Robbins PW: Lipid
rafts-protein association and the regulation of protein activity.
Arch Biochem Biophys. 426:208–224. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Vouret-Craviari V, Boulter E, Grall D,
Matthews C and Van Obberghen-Schilling E: ILK is required for the
assembly of matrix-forming adhesions and capillary morphogenesis in
endothelial cells. J Cell Sci. 117:4559–4569. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Isik OA and Cizmecioglu O: Rafting on the
plasma membrane: Lipid rafts in signaling and disease. Adv Exp Med
Biol. 1436:87–108. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Sviridov D, Mukhamedova N and Miller YI:
Lipid rafts as a therapeutic target. J Lipid Res. 61:687–695. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Balatskaya MN, Balatskii AV, Sharonov GV
and Tkachuk VA: T-cadherin as a novel receptor regulating
metabolism in the blood vessel and heart cells: From structure to
function. J Evol Biochem Physiol. 52:103–118. 2016. View Article : Google Scholar
|
|
120
|
Ruzzi F, Cappello C, Semprini MS,
Scalambra L, Angelicola S, Pittino OM, Landuzzi L, Palladini A,
Nanni P and Lollini PL: Lipid rafts, caveolae, and epidermal growth
factor receptor family: Friends or foes? Cell Commun Signal.
22:4892024. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Rubina KA, Semina EA, Balatskaya MN,
Plekhanova OS and Tkachuk VA: Mechanisms of Regulation of the
Targeted Grown of Nerves and Vessels by Components of the
Fibrinolytic System and GPI-Anchored Navigation Receptors. Neurosci
Behav Physiol. 50:217–230. 2020. View Article : Google Scholar
|
















