Introduction
Vitiligo is an acquired pigmentation disorder with a
complex pathogenesis and a number of etiological factors. It is
mainly manifested by limited or generalized depigmentation spots
(1,2). Epidemiological investigations show
that its incidence accounts for 1–2% of the total population and
the total number of vitiligo patients in China is estimated to be
10–20 million. The incidence rate is increasing year on year and
the average age of onset is decreasing (3). The etiology of vitiligo is complex
and hypotheses have been proposed from various perspectives
(4,5), including genetics, autoimmunity,
oxidative stress, cytotoxicity and microenvironmental disorders.
Currently, no single hypothesis fully explains the pathogenesis of
vitiligo. However, it is clear that the final link in these
hypotheses is the apoptosis or dysfunction of melanocytes (6).
The main treatments for vitiligo are
non-pharmacological and pharmacological. Non-pharmacological
treatments include melanocyte transplantation, iontophoresis and
laser therapy, which have great limitations and are not a feasible
treatment option for patients in the developmental stage and with
large skin lesions (7).
Pharmacological treatments for vitiligo mostly use high-dose
glucocorticoids or immunosuppressants, which have significant side
effects and produce unsatisfactory results. By contrast, in
traditional Chinese medicine (TCM) the use of Psoralea
corylifolia (P. corylifolia) is believed to tonify the kidneys,
strengthen yang, and invigorate the spleen and stomach and it is
widely used to treat psoriasis, vitiligo and a number of other
dermatological conditions. P. corylifolia tincture is
commonly used in the clinical treatment of vitiligo (8). However, further research is needed to
elucidate its active ingredients, molecular targets and mechanisms
of action.
Microarray analysis is a high-throughput method
commonly used to explore the pathogenesis of human diseases.
Bioinformatics are employed to analyze online microarray databases
and identify key genes involved in disease development (9). The present study used bioinformatics
to analyze the GSE75819 vitiligo-related microarray dataset in the
Gene Expression Omnibus (GEO) database. The aim was to explore the
core genes involved in vitiligo occurrence and development and to
provide a reference index for evaluating the therapeutic efficacy
of P. corylifolia in vitiligo treatment. Network
pharmacology is a new approach that can systematically and
comprehensively analyze the mechanisms of drug action on disease by
examining the interactions between active ingredients in TCM and
disease targets (10), which can
systematically and comprehensively explore the mechanisms of drug
action on disease and coincides with the holistic and systematic
view of TCM in treating diseases (11,12).
Molecular docking technology, which is based on the principles of
molecular mechanics and quantum mechanics, is used to predict the
binding affinity of drugs and targets by calculating the energy
changes between drug molecules and target proteins. In the present
study, bioinformatics, network pharmacology and molecular docking
were used to screen the main active ingredients of P.
corylifolia, analyze the action targets and mechanisms of
active ingredients and verify them through zebrafish and cell
experiments. It not only provided theoretical foundation for the
clinical application of P. corylifolia in the therapy of
vitiligo, but also served as references for its further scientific
research.
Materials and methods
Screening of medicinal chemical
components and their targets
The components of P. corylifolia that enter
the bloodstream were obtained as potential active ingredients using
the SymMap database (http://www.symmap.org/). The non-hemolytic
constituents of psoralen were meticulously investigated within the
TCMSP database (https://old.tcmsp-e.com/tcmsp.php) using the criteria
of druglikeness ≥0.18 and oral bioavailability ≥30% as the
selection parameters. The active ingredients obtained were searched
for in the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) to determine their
chemical formulae. The Swiss Target Prediction tool (http://swisstargetprediction.ch/) was then used
to determine the target of the active ingredient.
GEO database and vitiligo target
acquisition
The GSE75819 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=gse75819)
transcriptome data (13),
downloaded from the GEO database (http://www.ncbi.nlm.nih.gov/geo/), contains data from
15 vitiligo patients and 15 healthy individuals. Differential
analysis was conducted using the ‘limma’ package (https://bioconductor.org/packages/release/bioc/html/limma.html)
in the R programming language. Differential genes were identified
using a significance level of P<0.05 and |log2FC|>1 as the
criteria. The findings were then depicted via volcano plots and
heat maps to provide a comprehensive visualization. The
differential genes and vitiligo genes from the GeneCards
(https://www.genecards.org/) and DisGeNET
databases (https://disgenet.com/) were brought
together as vitiligo target genes using a Venn diagram (http://www.bioinformatics.com.cn). With the help
of the online tool Venny 2.1.0 (http://liuxiaoyuyuan.cn/gongju/weientu.html), the
vitiligo targets were intersected with the targets of the P.
corylifolia active ingredient and the intersecting targets were
identified as potential vitiligo interventions.
Construction of the active
ingredient-target-disease network
Network and tape files consisting of P.
corylifolia and its common targets with vitiligo were produced.
These files were then be imported into Cytoscape 3.10.1 (https://apps.cytoscape.org/) application to construct
the active ingredient-target-disease network.
Construction of protein-protein
interaction (PPI) networks and screening of core targets
The possible targets of P. corylifolia for
vitiligo were searched for in the STRING database (https://cn.string-db.org). Free nodes were eliminated
and the resulting TSV files were downloaded, visualized and
analyzed using Cytoscape 3.10.1.
Gene Ontology (GO) and Kyoto
Encyclopedia of Genes and Genomes (KEGG) pathway enrichment
analysis
GO and KEGG pathway data obtained from the
intersecting genes of P. corylifolia and vitiligo were
retrieved and downloaded with the help of the DAVID database
(https://david.ncifcrf.gov). GO and KEGG
enrichment analyses were conducted to investigate the biological
pathways and potential functions of the intersecting genes, with
the aim of analyzing their biological processes and metabolic
pathways. Finally, the data were visualized using the
MicroBioInformatics online tool (https://www.bioinformatics.com.cn).
Molecular docking validation
The 3D crystalline structure of the target protein
was retrieved from the Protein Data Bank (PDB; http://www.rcsb.org/). It was then downloaded and
processed for dehydration and hydrogenation using AutoDockTools
(https://vina.scripps.edu/). This
structure was ultimately selected as the receptor and preserved in
the PDBqt file format. Active constituents were retrieved from the
TCMSP database, hydrogenated using AutoDockTools and selected as
ligands before being exported in the PDBQT file format. Molecular
docking simulations were then performed using AutoDock Vina to
determine the binding affinity of each compound to the target
protein. Subsequent analysis of these affinities was conducted
using the ‘pheatmap’ (https://cran.r-project.org/web/packages/pheatmap/index.html)
and ‘circlize’ R packages (https://cran.r-project.org/web/packages/circlize/index.html),
complemented by visualization in PyMOL (http://www.pymol.org/pymol).
Preparation of aqueous and ethanol
extracts
P. corylifolia (10 g) was crushed and added
to 100 ml of distilled water/75% ethanol. The mixture was left to
soak for 1 h and then subjected to water bath heating extraction
for a further 1 h. The extract was filtered and collected. Add a
further 100 ml of distilled water/75% ethanol and perform a water
bath heating extraction for a further 30 min. Filter and collect
the extract, then combine the two extracts and concentrate
them.
Main materials and instruments
AB wild-type zebrafish (Shanghai FishBio Co., Ltd.),
B16F10 cells (Wuhan Pricella Biotechnology Co., Ltd.; Elabscience
Bionovation Inc.),
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; MTT;
tetrazolium salt), N-phenylthiourea (PTU), levodopa (L-DOPA),
laboratory biochemical incubator (Tianjin Laboratory
Instrumentation Co., Ltd.), K-400L stereo microscope (Motic
Incorporation, Ltd.), SpectraMax iD3 multifunctional enzyme labeler
(Molecular Devices, LLC.).
Principal component analysis extract
using ultra-high-performance liquid
chromatography-triple/time-of-flight mass spectrometry
(UPLC-Q-TOF/MS)
The aqueous and ethanol extracts of P.
corylifolia were subjected to UPLC-Q-TOF/MS analysis to
ascertain the total ion mobility map, in compliance with the
chromatographic and mass spectrometric parameters as detailed in
Tables SI and SII. Drawing upon the multistage mass
spectrometric data derived from the samples, subsequent processing
and comprehensive analysis were conducted in synergy with the
high-resolution mass spectrometry database for natural products and
pertinent scholarly literature.
Establishment of a zebrafish model for
pigment loss and drug experiments
Preliminary experiments determined that 200 µM PTU
would be used to generate a pigment-deficient zebrafish model.
Zebrafish embryos were cultured in a constant-temperature incubator
at 28°C for 24 h, designated as 24hpf/1dpf. PTU-induced modeling
was then performed for 48 h, at which point the embryos were
designated as 72hpf/3dpf. Drug administration commenced immediately
afterward for 72 h, with the final time point designated as
144hpf/6dpf. Experimental groups and procedures are detailed below:
At 1 dpf, healthy zebrafish embryos were selected and placed into
6-well plates at 15 zebrafish embryos per well. Groups included:
Blank control group cultured in fresh culture water; modelling
group cultured in fresh culture water containing 200 µM PTU; and
groups with different concentrations of aqueous and ethanol
extracts (1, 2.5 and 5 µg/ml). Following drug administration,
anesthesia was induced using 0.016% MS-222 until zebrafish ceased
swimming or exhibited ventral positioning (flipping onto their
backs), indicating full anesthesia (14). Anesthetized zebrafish were observed
under a microscope within 5 min to assess pigment development, with
images captured for documentation. After the experiment, zebrafish
were immersed in a high-concentration 0.08% MS-222 solution
(typically 5X) until gill movement ceased (~5 min). Anesthesia was
maintained for an additional 10–15 min to ensure the mortality of
experimental fish (15). Finally,
they were placed in a −20°C freezer for centralized disposal of all
experimental animals (16). All
zebrafish experiments were reviewed and approved by the Animal
Ethics Committee of Bozhou University, with ethics approval number
DFDW/BZUU-2025-ZF-031.
Measurement of melanocyte
viability
B16F10 melanocytes in the exponential growth phase
were carefully selected and inoculated into 96-well plates at a
concentration of 5×104 cells/ml in 180 µl of volume per
well. Following inoculation, the cells were placed in a 5%
CO2 incubator at 37°C and allowed to proliferate for 24
h. Subsequently, the cells were treated with aqueous and ethanol
extracts of P. corylifolia at concentrations of 0.01, 0.1,
1, 10 and 100 µg/ml, followed by an incubation at 37°C period of 48
h. The solution was then evacuated and discarded. Then, 30 µl of
MTT reagent was added to each well and the reaction was carried out
at 37°C for 4 h. DMSO (150 µl) was added to each well and the
solution was vigorously agitated for 10 min. After a 48-h
incubation period, 30 µl of MTT reagent was added to each well. The
reaction was then conducted at 37°C for 4 h. Then, 150 µl of DMSO
was added to each well and shaken for 10 min to facilitate the
reaction. Absorbance readings for each well were taken at a
wavelength of 570 nm to evaluate the effect of aqueous and
alcoholic extracts derived from tonic acid on melanocyte viability
and functional integrity.
Determination of melanin content and
tyrosinase activity of cells
B16F10 melanocytes were cultured according to the
procedure in the aforementioned Measurement of melanocyte
viability. After 24 h, 0.01, 0.1, 1 and 10 µg/ml concentrations
of the aqueous and ethanol extracts of P. corylifolia were
added. After 72 h of incubation at 37°C, the cells were washed
twice with PBS, following which 100 µl of a non-denaturing lysis
buffer containing 1 nM PMSF were added. The cells were lysed at 4°C
for a duration of 20 min, following which they were collected. Upon
reaching 72 h of culture at 37°C, the cells underwent a washing
process with PBS, repeated twice. Thereafter, a volume of 100 µl of
non-denaturing lysate, which contained 1 nM PMSF, was introduced.
The cells were subsequently lysed once again at 4°C for 20 min and
then collected. The collected cells were centrifuged at 4°C for 10
min at 21,756 × g. The resulting supernatant was used to determine
the protein content via the BCA assay and the protein concentration
was subsequently calculated. The volume of the supernatant
containing 10 µg of total protein was measured and transferred to a
96-well plate. The volume was then brought up to 100 µl using PBS
(0.1 M, pH 6.8). Then, 100 µl of a 0.01% L-DOPA solution was added
and the plate was incubated at 37°C in the dark for 3 h. OD475 was
used to evaluate the catalytic activity of tyrosinase. The melanin
precipitate pellet was then suspended in a 100 µl NaOH solution
containing 10% DMSO to ensure full solubilization of the melanin
within each experimental group. The melanin precipitate with the
lower concentration was supplemented with 100 µl of a NaOH solution
containing 10% DMSO. Then, 100 µl of the melanin solution was
pipetted into each well of a 96-well plate. OD405 was determined to
assess melanocyte content.
Statistical analysis
Data were analyzed exclusively using GraphPad Prism
5 software (Dotmatics), employing one-way analysis of variance
(followed by Tukey's post hoc test) and two-tailed paired t-tests.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Screening of the chemical constituents
of P. corylifolia
Searching the SymMap and TCMSP databases, the
present study obtained nine constituents of P. corylifolia,
including isobavachin, bavachin, stigmasterol, bavachalcone,
bakuchiol, psoralidin and angelicin (Table I).
 | Table I.Main active components of Psoralea
corylifolia. |
Table I.
Main active components of Psoralea
corylifolia.
| CAS number | Active
ingredient | Basis of
selection |
|---|
| 31524-62-6 | Isobavachin | OB≥30%;
DL≥0.18% |
| 19879-32-4 | Bavachin | Blood-entry
component |
| 83-48-7 | Stigmasterol | OB≥30%;
DL≥0.18% |
| 28448-85-3 | Bavachalcone | Blood-entry
component |
| 10309-37-2 | Bakuchiol | Blood-entry
component |
| 20784-50-3 |
Isobavachalcone | Blood-entry
component |
| 523-50-2 | Angelicin | Blood-entry
component |
| 41743-38-8 | Bavachromene | Blood-entry
component |
| 18642-23-4 | Psoralidin | Blood-entry
component |
Intersection of drug active
ingredients and disease targets
A total of 259 effective targets of the nine active
components of P. corylifolia were obtained through the Swiss
Target Prediction database. The GeneCards and DisGeNET databases
screened 1,357 and 395 potential vitiligo targets, respectively.
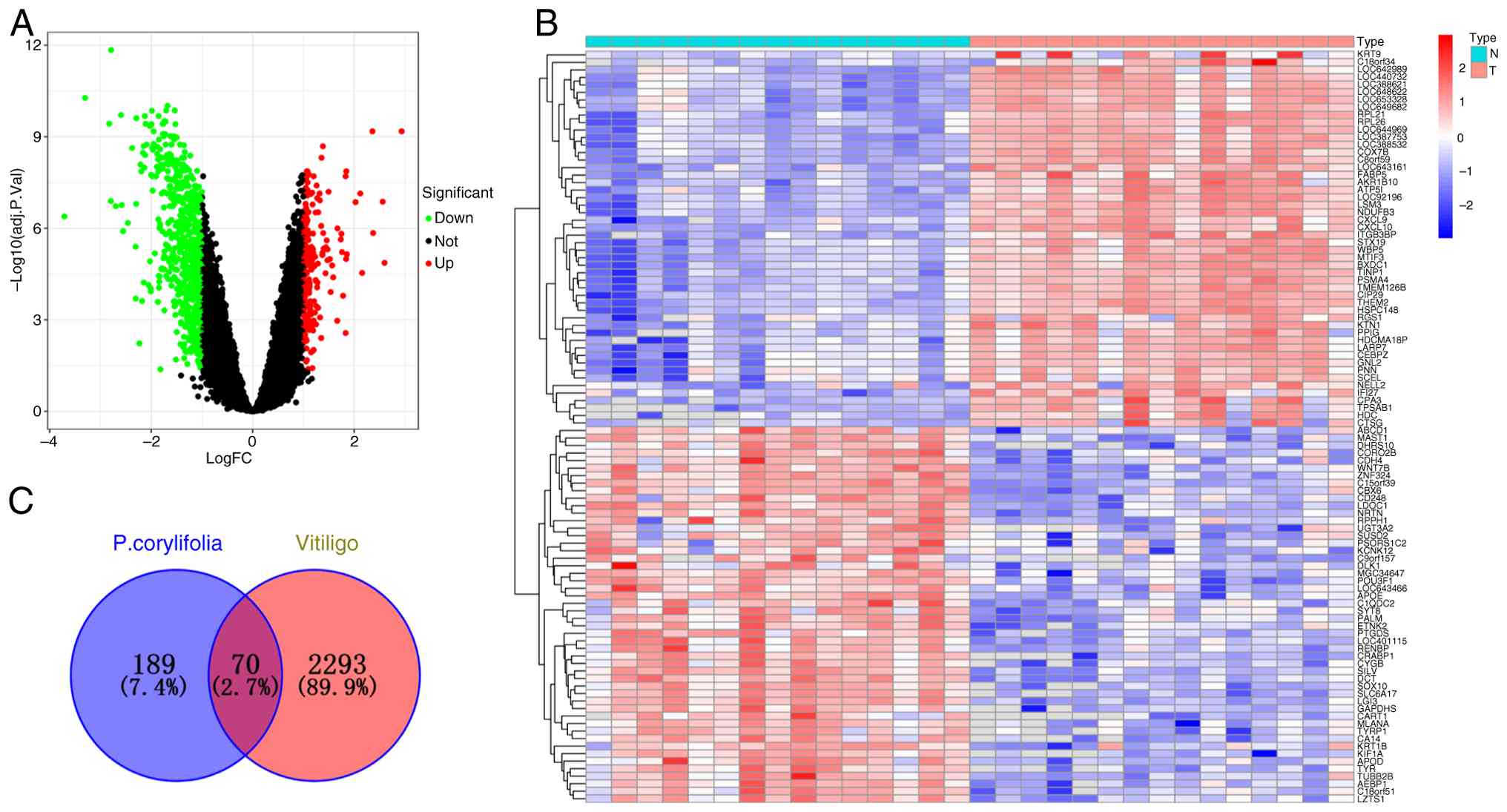
Microarray data from GSE75819 detected a total of 25,402 genes,
including 691 upregulated and 220 downregulated genes (Fig. 1A and B). Using Venny, the
intersection between the microarray differential genes related to
vitiligo and the active ingredient targets of P.
corylifolia, were identified obtaining a total of 70
intersecting genes (Fig. 1C).
These included TNF, AKT1, PPARG, PTGS2, HSP90AA1, ESR1, BCL2,
NFKB1, MTOR, GSK3B, PARP1, MDM2, MAPK1 and PIK3CA.
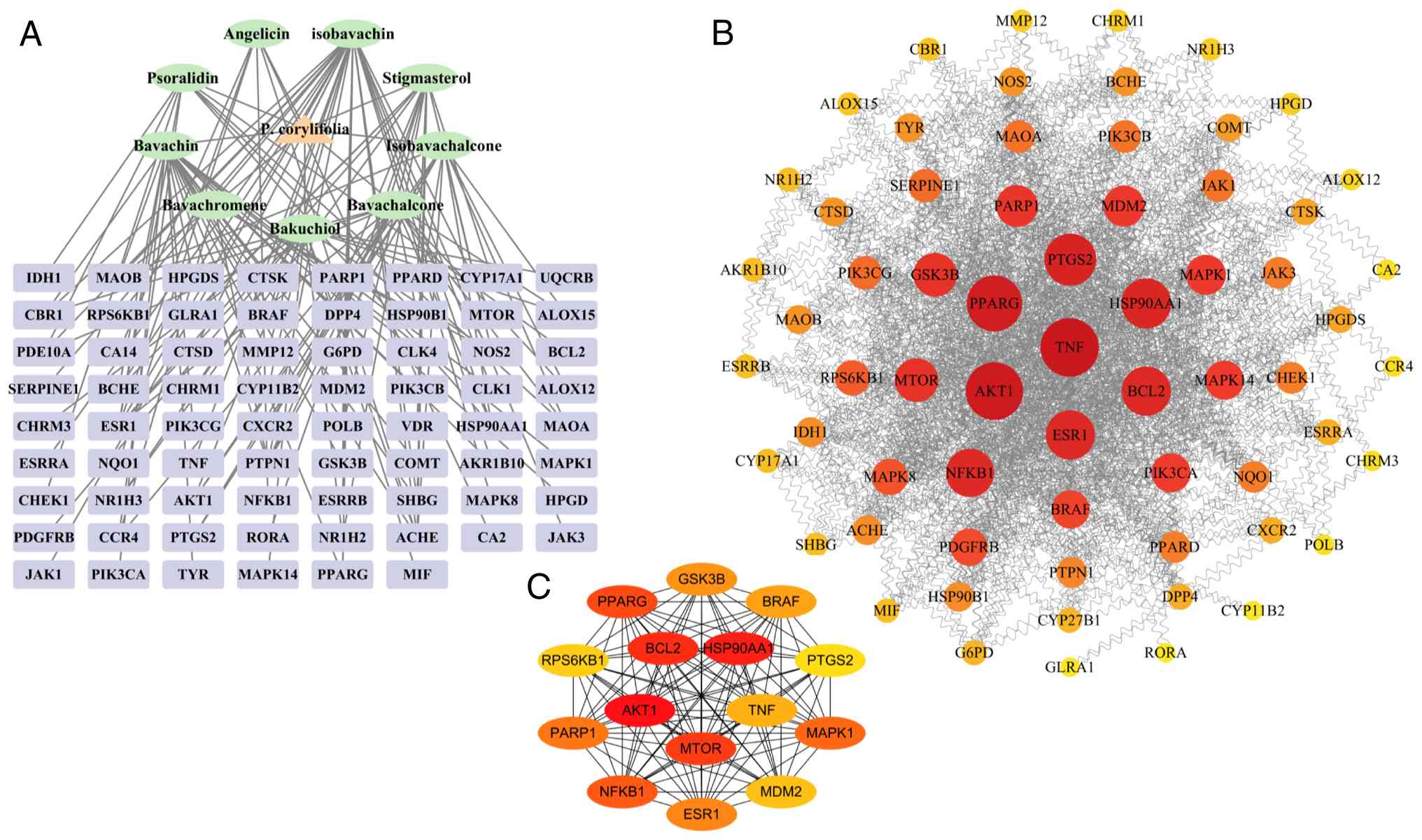
The Cytoscape 3.10.1 application was used to create
the ‘active ingredient-target-disease’ network illustrated in
Fig. 2A, comprising 80 nodes and
140 edges. The Network Analyzer, an integral component of the
software, was used to calculate the degree value for each node. A
higher degree value indicates a stronger association between the
active components of P. corylifolia and vitiligo-related
targets. The active ingredients of P. corylifolia based on
degree value were isobavachin, bavachin, stigmasterol,
bavachalcone, bakuchiol, isobavachalcone, psoralidin and angelicin,
with degree values of 29, 26, 16, 16, 16, 13, 10 and 8,
respectively. It suggested that these constituents may be crucial
in the psoralen treatment of vitiligo.
Construction of PPI network and
screening of core targets
The curated list of 70 intersecting genes was
seamlessly integrated into the STRING database, giving rise to a
sophisticated PPI network, specifically curated under the criterion
of ‘Homo sapiens’. This intricate network encompasses a total of 70
nodes interconnected by 461 edges, exhibiting an average node
degree of 13.2, as illustrated in Fig.
2B. The derived protein interaction data were meticulously
integrated into the Cytoscape 3.10.1 software suite, wherein
subsequent manipulation via the Analyze Network module facilitated
the generation of an exhaustive network visualization. This
visualization underscored the pivotal targets of P.
corylifolia in the context of vitiligo therapeutic
intervention, as depicted in Fig.
2B. In the figure the size of the circular node indicates the
degree value, the larger node degree value, the redder node
indicates that the target is more critical, the closer the
relationship between the active ingredient of P. corylifolia
and the target of vitiligo. The top 15 target proteins are TNF,
AKT1, PPARG, PTGS2, HSP90AA1, ESR1, BCL2, NFKB1, MTOR, GSK3B,
PARP1, MDM2, MAPK1, PIK3CA and MAPK14, with degree values of 86,
84, 80, 72, 68, 66, 66, 64, 54, 54, 50, 48, 46, 44, 44,
respectively. The top 15 targets were further calculated by
applying the MCC algorithm using the cytoHubba plug-in in Cytoscape
software (Fig. 2C) and the
intersection of the targets calculated by the MCC algorithm and
Degree was taken to obtain a total of AKT1, HSP90AA1, BCL2, MTOR,
NFKB1, MAPK1, ESR1, GSK3B, TNF, PTGS2 ten key targets. The
identified targets may represent the principal therapeutic targets
of P. corylifolia in the context of vitiligo treatment and
they are concurrently the focus of molecular docking validation as
presented in the present study.
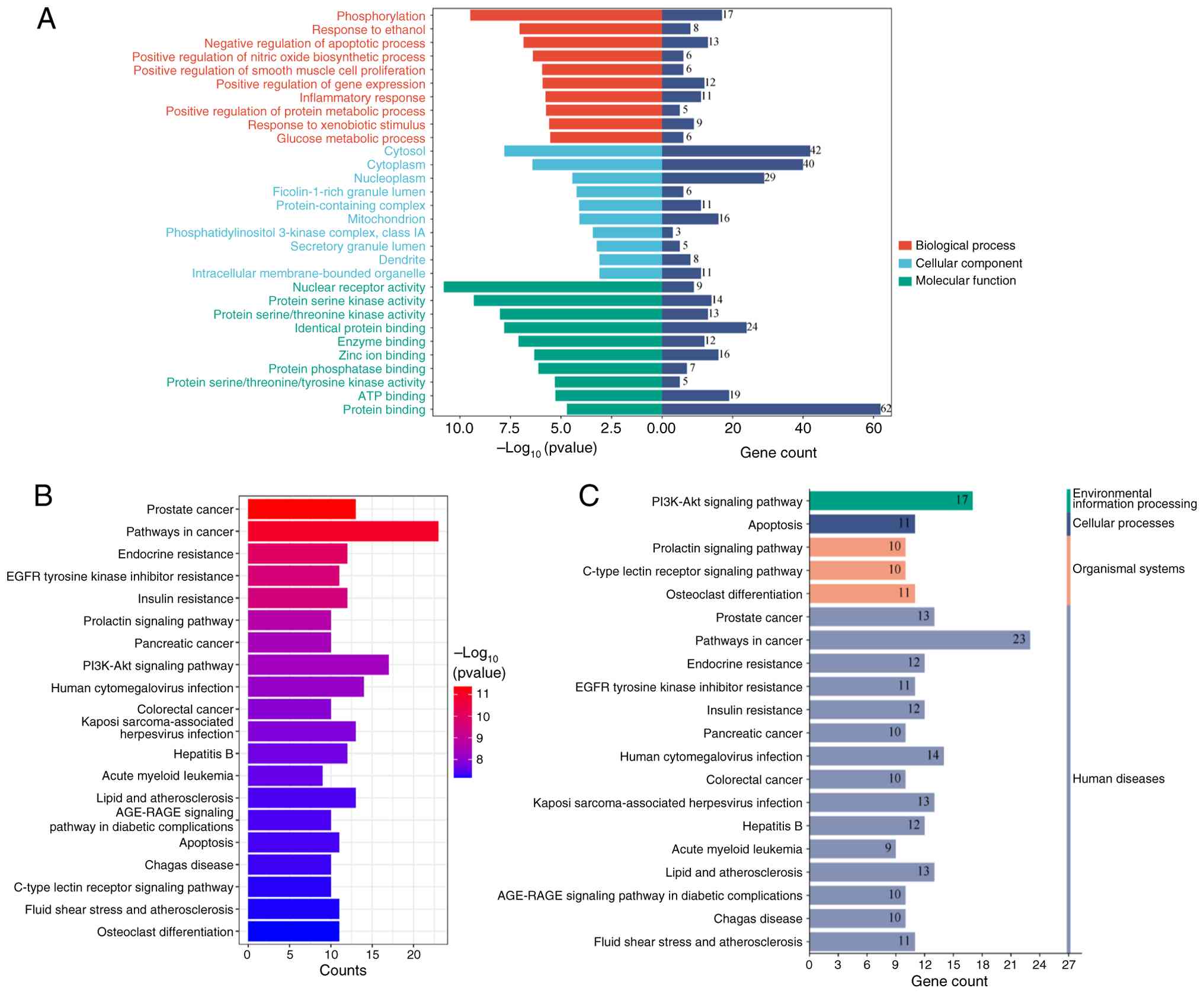
GO functional enrichment analysis
The 70 overlapping genes were uploaded to the DAVID
database for enrichment analysis, yielding results for 272
biological processes (BP), 41 cellular components (CC) and 66
molecular functions (MF). Following rigorous screening, the top ten
candidates in each category were selected based on their P-values:
BP, CC and MF. These selections were then depicted graphically via
bar charts to facilitate a comprehensive visual comparison. The
results showed that BP was mainly enriched in the regulation of
apoptosis, protein phosphorylation and the inflammatory response;
CC was mainly enriched in the mitochondria, the cytoplasm and the
phosphatidylinositol 3-kinase complex; and MF was mainly enriched
in homodimeric protein binding, enzyme binding and protein
serine/threonine kinase activity, as shown in Fig. 3.
KEGG pathway enrichment analysis
The 70 intersecting genes obtained were imported
into the DAVID database for enrichment analysis. The results of the
enrichment analysis revealed 140 KEGG pathways and a KEGG
functional enrichment bar graph was drawn according to the results
of the top 20 P-values (Fig. 3B).
The KEGG pathway enrichment results were categorized (Fig. 3C). The enrichment outcomes indicate
that the genes are predominantly associated with multiple facets of
the PI3K-Akt signaling pathway and oncogenic pathways, as well as
pathways relevant to hepatitis.
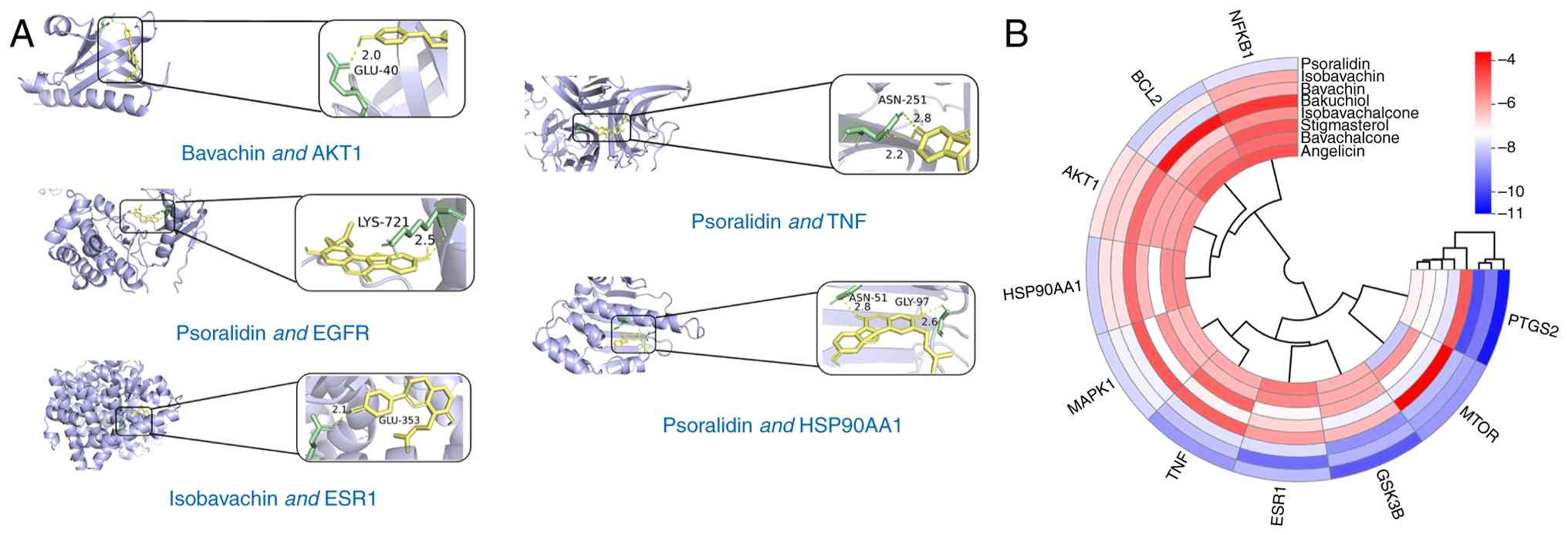
Results of molecular docking of core
components and key targets
The eight active ingredients of P.
corylifolia obtained (Isobavachin, Bavachin, Stigmasterol,
Bavachalcone, Bakuchiol, Isobavachalcone, Psoralidin and Angelicin)
and the 10 key targets (AKT1, HSP90AA1, BCL2, MTOR, NFKB1, MAPK1,
ESR1, GSK3B, TNF and PTGS2) were molecularly docked (Table II and Fig. 4A). The binding energy, measured in
kilocalories per mole (kcal/mol), was found to be <-5.0
kcal/mol, indicating a strong and stable interaction between the
drug components and the key target proteins. It should be noted
that a more negative binding energy denotes a stronger and more
stable binding effect. The binding energy was found to be <-5.0
kcal/mol for all components, signifying robust and stable
interactions with the key target proteins. The resultant binding
energies for each constituent with respect to the target were
visualized using a clustered heatmap generated via the ‘pheatmap’ R
package (Fig. 4B).
 | Table II.Molecular docking results of core
components and key targets. |
Table II.
Molecular docking results of core
components and key targets.
| Targets | PDB-ID | Components | Binding energy,
kcal/mol | RMSD (A) |
|---|
| AKT1 | 6NJS | Isobavachin | −6.5 | 4.329 |
|
|
| Bavachin | −6.7 | 1.989 |
|
|
| Stigmasterol | −6.0 | 2.927 |
|
|
| Bavachalcone | −6.0 | 2.029 |
|
|
| Bakuchiol | −5.2 | 1.395 |
|
|
|
Isobavachalcone | −6.0 | 2.376 |
|
|
| Angelicin | −5.7 | 1.471 |
|
|
| Psoralidin | −6.9 | 1.293 |
| ESR1 | 2BJ4 | Isobavachin | −9.4 | 2.653 |
|
|
| Bavachin | −7.8 | 2.698 |
|
|
| Stigmasterol | −7.2 | 2.188 |
|
|
| Bavachalcone | −5.6 | 1.603 |
|
|
| Bakuchiol | −5.9 | 1.990 |
|
|
|
Isobavachalcone | −6.7 | 2.369 |
|
|
| Angelicin | −5.4 | 3.204 |
|
|
| Psoralidin | −8.3 | 2.997 |
| GSK3B | 1H8F | Isobavachin | −8.4 | 2.412 |
|
|
| Bavachin | −8.9 | 2.995 |
|
|
| Stigmasterol | −6.1 | 4.803 |
|
|
| Bavachalcone | −6.1 | 3.931 |
|
|
| Bakuchiol | −6.3 | 2.064 |
|
|
|
Isobavachalcone | −7.5 | 2.339 |
|
|
| Angelicin | −6.2 | 1.872 |
|
|
| Psoralidin | −9.7 | 2.737 |
| HSP90AA1 | 1AUE | Isobavachin | −6.9 | 1.491 |
|
|
| Bavachin | −6.9 | 1.860 |
|
|
| Stigmasterol | −7.3 | 1.534 |
|
|
| Bavachalcone | −5.7 | 1.506 |
|
|
| Bakuchiol | −5.2 | 1.905 |
|
|
|
Isobavachalcone | −6.3 | 3.442 |
|
|
| Angelicin | −5.6 | 1.475 |
|
|
| Psoralidin | −8.0 | 1.910 |
| MAPK1 | 6D5Y | Isobavachin | −7.5 | 3.485 |
|
|
| Bavachin | −7.5 | 2.012 |
|
|
| Stigmasterol | −5.8 | 2.726 |
|
|
| Bavachalcone | −6.4 | 3.042 |
|
|
| Bakuchiol | −4.9 | 4.641 |
|
|
|
Isobavachalcone | −7.3 | 3.702 |
|
|
| Angelicin | −6.2 | 2.500 |
|
|
| Psoralidin | −7.9 | 1.597 |
| BCL2 | 1G5M | Isobavachin | −7.0 | 2.448 |
|
|
| Bavachin | −7.9 | 1.519 |
|
|
| Stigmasterol | −5.9 | 4.630 |
|
|
| Bavachalcone | −5.5 | 4.418 |
|
|
| Bakuchiol | −3.9 | 1.915 |
|
|
|
Isobavachalcone | −6.6 | 4.701 |
|
|
| Angelicin | −4.9 | 1.368 |
|
|
| Psoralidin | −8.0 | 1.939 |
| MTOR | 1AUE | Isobavachin | −8.6 | 2.072 |
|
|
| Bavachin | −8.3 | 2.156 |
|
|
| Stigmasterol | −7.3 | 1.819 |
|
|
| Bavachalcone | −5.8 | 3.613 |
|
|
| Bakuchiol | −3.6 | 2.333 |
|
|
|
Isobavachalcone | −7.6 | 2.454 |
|
|
| Angelicin | −8.1 | 1.548 |
|
|
| Psoralidin | −8.8 | 1.722 |
| NFKB1 | 2DBF | Isobavachin | −6.1 | 2.756 |
|
|
| Bavachin | −6.2 | 2.643 |
|
|
| Stigmasterol | −4.9 | 3.402 |
|
|
| Bavachalcone | −5.2 | 3.792 |
|
|
| Bakuchiol | −4.4 | 2.338 |
|
|
|
Isobavachalcone | −5.7 | 4.932 |
|
|
| Angelicin | −4.8 | 1.457 |
|
|
| Psoralidin | −7.7 | 1.939 |
| TNF | 4ZCH | Isobavachin | −8.2 | 2.607 |
|
|
| Bavachin | −7.7 | 1.316 |
|
|
| Stigmasterol | −5.1 | 3.696 |
|
|
| Bavachalcone | −6.3 | 1.544 |
|
|
| Bakuchiol | −5.0 | 5.426 |
|
|
|
Isobavachalcone | −7.1 | 5.465 |
|
|
| Angelicin | −6.1 | 2.198 |
|
|
| Psoralidin | −8.8 | 1.990 |
| PTGS2 | 5F19 | Isobavachin | −9.2 | 3.501 |
|
|
| Bavachin | −9.9 | 3.245 |
|
|
| Stigmasterol | −7.4 | 2.953 |
|
|
| Bavachalcone | −7.2 | 2.932 |
|
|
| Bakuchiol | −4.9 | 3.905 |
|
|
|
Isobavachalcone | −7.7 | 2.991 |
|
|
| Angelicin | −7.1 | 1.405 |
|
|
| Psoralidin | −10.7 | 1.006 |
UPLC-Q-TOF/MS analysis of the main
components of the aqueous and ethanol extracts of P.
corylifolia
UPLC-Q-TOF/MS analysis was employed to detect and
comprehensively analyze the main components of the aqueous and
ethanol extracts of P. corylifolia. UPLC-Q-TOF/MS technology
was employed to detect and analyze the extracts and the results
indicated that the main constituents in the aqueous and ethanolic
extracts of P. corylifolia were angelicin, psoralidin,
stigmasterol, bavachin and bakuchiol, among others (Fig. S1; Table III).
 | Table III.Identification of the main components
of Psoralea corylifolia extract samples. |
Table III.
Identification of the main components
of Psoralea corylifolia extract samples.
|
|
|
|
| Aqueous
extract | Ethanol
extract |
|---|
|
|
|
|
|
|
|
|---|
| Serial no. | m/z | Molecular
formula | Main
components | Time/min | Peak area | Time/min | Peak area |
|---|
| 1 | 187.0386 |
C11H6O3 | Isopsoralen | 58.946 | 1,51,577 | - | - |
| 2 | 273.1835 |
C18H24O2 | Bakuchiol | 65.861 | 8,76,676 | 65.857 | 881.293 |
| 3 | 893.7206 |
C57H96O7 |
2-Glc-Stigmasterol | 66.618 | 1,55,60,238 | 10.187 | 15.560.238 |
| 4 | 187.0395 |
C11H6O3 | Psoralen | 1.402 | 5,77,113 | 63.182 | 880.119 |
| 5 | 325.1428 |
C20H20O4 | Bavachin | - | - | 0.9 | 48.755 |
| 6 | 339.1586 |
C20H20O4 | Bavachinin | 2.106 | 2,90,604 | - | - |
Effect of the hydroalcoholic extract
of P. corylifolia on pigment synthesis in normal and depigmented
zebrafish
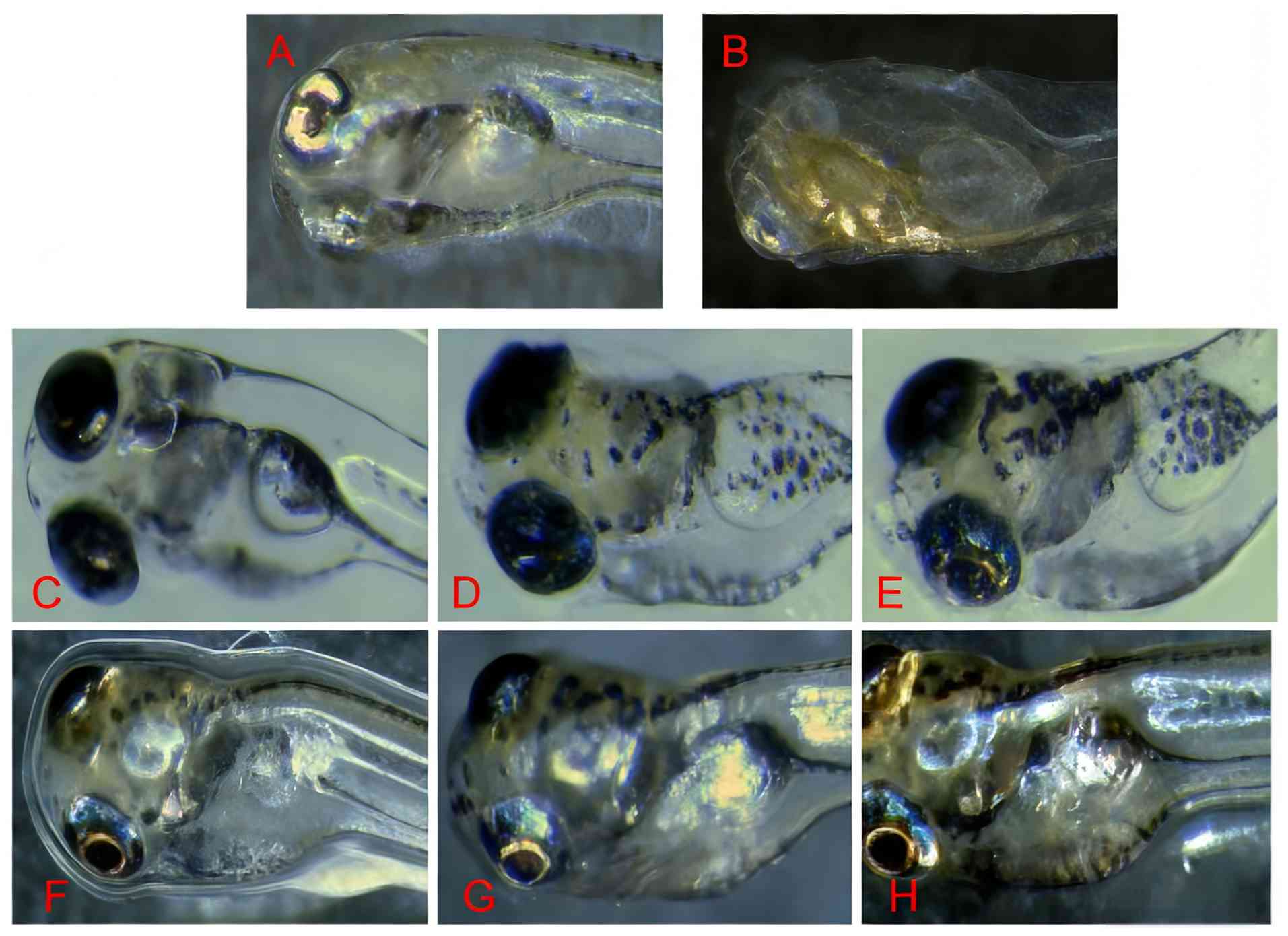
Compared with the control group, the aqueous and
ethanol extracts of P. corylifolia markedly affected pigment
synthesis in depigmented zebrafish, with increased melanin
synthesis as the administered drug concentration increased
(Fig. 5).
Effect of P. corylifolia
hydroalcoholic extract on melanin synthesis
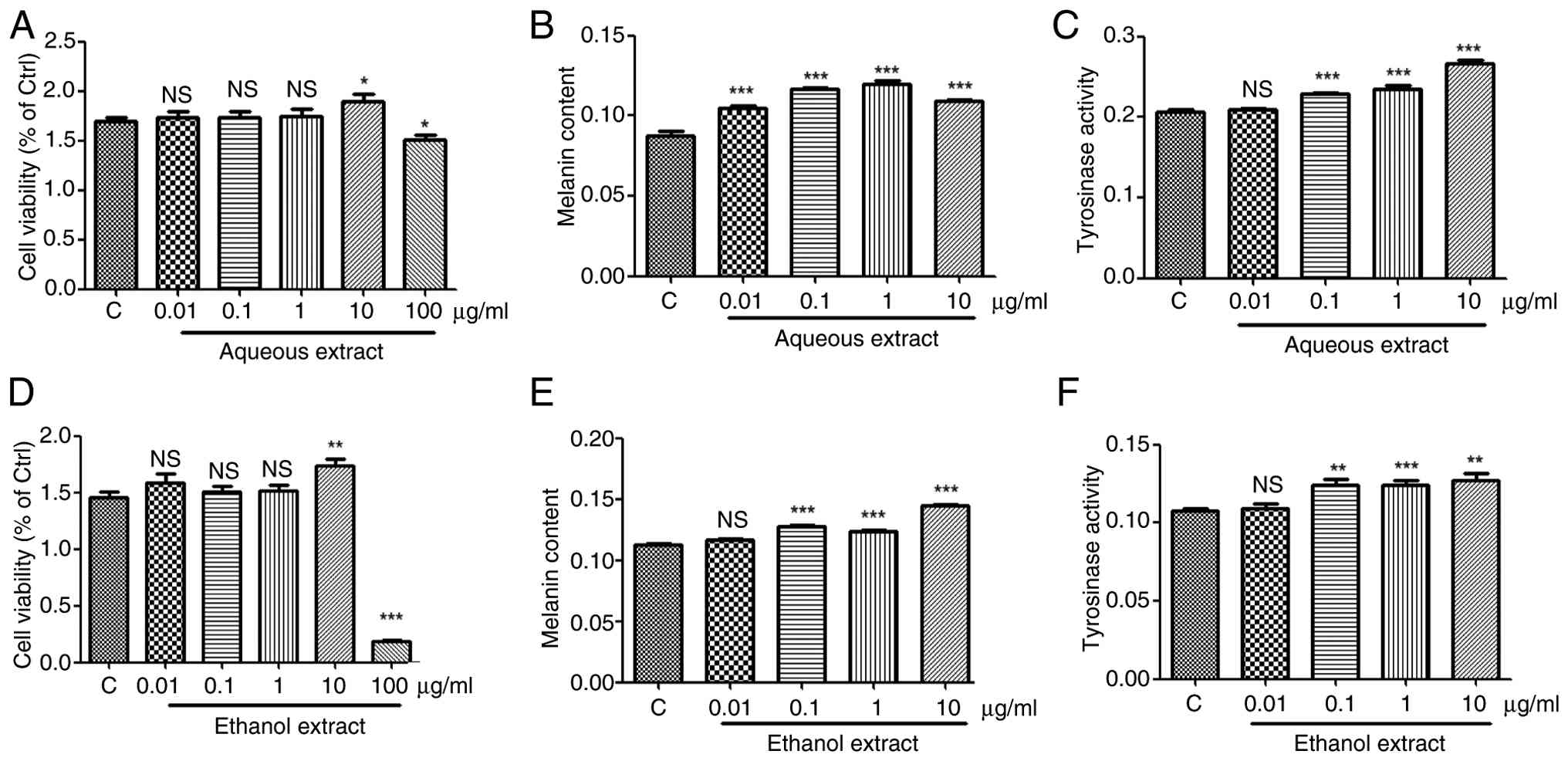
Compared with the control group, the results
revealed that 0.01–1 µg/ml of the aqueous and ethanol extracts of
P. corylifolia had no effect on cell viability, 10 µg/ml
promoted cell viability and 100 µg/ml markedly inhibited cell
viability (Fig. 6A and D).
Therefore, 0.01–1 µg/ml of the aqueous and ethanol extracts of
P. corylifolia were selected for subsequent experiments on
melanin synthesis and tyrosinase activity. Compared with the
control group, 0.01–10 µg/ml of the aqueous extracts of P.
corylifolia markedly promoted melanin synthesis, with the
effect becoming more pronounced at concentrations of 0.01–1 µg/ml
(Fig. 6B). Compared with the
control group, 0.01 µg/ml of the ethanol extract of P.
corylifolia had no significant effect on melanocyte synthesis,
0.1–10 µg/ml markedly promoted melanocyte melanin synthesis
(Fig. 6E). Compared with the
control group, 0.1–10 µg/ml of the aqueous extract markedly
increased tyrosinase activity, with a more pronounced effect at
higher concentrations (Fig. 6C and
F).
Discussion
A thorough search of the SymMap and TCMSP databases
in the current investigation revealed that the primary active
constituents of P. corylifolia that are effective in the
treatment of vitiligo potentially include isobavachin, bavachin,
stigmasterol, bavachalcone, bakuchiol, psoralidin and angelicin.
Principal component analysis using UPLC-Q-TOF/MS showed that the
aqueous and ethanol extracts of P. corylifolia contain these
active ingredients. The phytochemicals isobavachin, bavachin,
angelicin and stigmasterol have pharmacological properties
including anti-inflammatory and antioxidant effects (17,18).
Bavachalcone has the ability to modulate the NF-κB signaling
pathway, effectively suppressing the synthesis of pro-inflammatory
cytokines and demonstrating its anti-inflammatory properties
(19). Melanocyte damage caused by
free radicals induced by ultraviolet light and sunlight exposure is
one of the main causes of vitiligo exacerbation, so antioxidant and
free radical scavenging properties are important for curing
vitiligo. Bakuchiol can scavenge oxidized free radicals and inhibit
the production of interleukin-6 in various cell lines, exhibiting
antioxidant and anti-inflammatory properties (20). It has been demonstrated that
psoralidin activates tyrosinase, the rate-limiting enzyme in
melanogenesis, conferring therapeutic efficacy against vitiligo
(21). This suggests that the
treatment of vitiligo with P. corylifolia is due to the
combined effect of its active ingredients.
The establishment of the PPI network and the
outcomes of the topological analysis conducted in the present study
revealed that the key targets of P. corylifolia in its
anti-vitiligo effects include AKT1, HSP90AA1, BCL2, MTOR, NFKB1,
MAPK1, ESR1, GSK3B, TNF and PTGS2. AKT serves as a critical
junction within the signaling cascade of protein kinase B and also
plays a pivotal role in the organism's protein metabolic processes
(22). At the same time, activated
AKT1 mainly induces downstream signaling cascades by
phosphorylating substrate proteins and participates in regulating
various cellular activities (23,24),
thereby adjusting the decline in autoimmune function due to
vitiligo disease. TNF is a large group of cytokines that can
directly cause tumor cell death. The present study identifies
TNF-α, primarily produced by monocyte macrophages, as a key
pro-inflammatory and immunomodulatory factor. Not only does it
suppress the proliferation and differentiation of hematopoietic
cells, inducing apoptosis, it also mitigates skin inflammation
associated with vitiligo and enhances the immune response of the
body (25). PTGS2, also known as
cyclooxygenase, is a key prostaglandin endoperoxide synthase. It is
a key enzyme in the metabolism of arachidonic acid to generate
prostaglandins and is widely distributed in the body. When cells
are stimulated, the expression of PTGS2 is increased and it
catalyzes the production of various prostaglandins from arachidonic
acid. These prostaglandins produce therapeutic effects such as pain
relief, an anti-inflammatory response and anti-fibrosis.
Prostaglandins are produced by keratinocyte-forming cells under
ultraviolet irradiation and can promote melanocyte proliferation,
dendrite growth and melanin production. This can treat skin melanin
loss caused by vitiligo (26) and
suggests that treating vitiligo with P. corylifolia is
effective.
The findings of the present study suggested that the
therapeutic mechanisms of P. corylifolia in the treatment of
vitiligo involved the PI3K/AKT signaling pathway, oncological
pathways and hepatitis-associated pathways. Vitiligo, cancer and
hepatitis exhibit significant commonalities at the molecular
pathway level, with their core mechanisms converging in three
areas: Immune dysregulation, abnormal activation of key signaling
pathways and oxidative stress damage. Research indicates that all
three conditions rely on CD8+ T cell-mediated cytotoxic responses,
whereby perforin and granzyme are released to disrupt the membrane
integrity of target cells (melanocytes, hepatocytes or tumor
cells), thereby inducing cellular dysfunction or death (27–29).
Concurrently, sustained activation of the
IFN-γ/JAK-STAT pathway serves as a central pro-inflammatory driver.
By binding to IFNGR1/2 receptors, it induces STAT1 phosphorylation
and nuclear translocation, thereby regulating hundreds of
pro-inflammatory genes and exacerbating tissue damage. This pathway
promotes melanocyte clearance in vitiligo, amplifies antiviral
inflammatory responses in hepatitis and plays dual roles in
antitumor immune responses and immune editing in cancer (30–32).
Shared signaling networks (such as NF-κB, PI3K-AKT and MAPK)
further link the pathologies of these three conditions. NF-κB forms
a positive feedback inflammatory loop via TLR/IL-1R activation,
driving the release of proinflammatory factors (TNF-α and IL-6);
the PI3K-AKT pathway suppresses melanocyte survival signaling in
vitiligo, but promotes tumor metabolic reprogramming in cancer
(33); and MAPK mediates
neuropeptide (CGRP)-induced immune activation in vitiligo and
proinflammatory signaling from viral proteins (such as HBx) in
hepatitis (34). The synergistic
effects of oxidative stress create a vicious
‘inflammation-oxidative stress’ cycle through the excessive
accumulation of reactive oxygen species. Sources include melanin
synthesis by-products (vitiligo), mitochondrial dysfunction
(cancer) and viral protein-induced endoplasmic reticulum stress
(hepatitis), ultimately leading to cellular dysfunction or
malignant transformation (35).
Zebrafish have a number of advantages in
melanin-related studies (36,37):
i) Their transparent embryos enable real-time visualization of
melanocyte development and migration; ii) their melanogenesis
pathways are highly conserved with those of mammals; iii) they
enable high-throughput screening for genetic and pharmacological
interventions; and iv) they provide a more cost-effective
alternative to mammalian models while maintaining biological
relevance. In addition, the B16F10 cell line is a melanoma cell
line that is also widely used and recognized in research related to
pigmentation, including melanin formation, melanocyte biology and
studies on potential interventions for pigmentary disorders such as
vitiligo (38,39). The experimental results of the
present study show that the aqueous and ethanol extracts of P.
corylifolia can increase melanin production in zebrafish and
B16 melanocytes. Within a certain range, the higher the
concentration, the greater the melanin synthesis, which plays a
role in curing vitiligo. This is because P. corylifolia can
effectively enhance the proliferation rate of melanocytes and
keratinocytes, promote melanocytes to enter the S and G2
phases of the cell cycle and increase their division and
proliferation. It can also enhance the activity of tyrosinase, a
key enzyme in vitiligo treatment, ultimately leading to a
significant increase in melanin synthesis (40). Further investigations have revealed
that isobavachin acts as an anti-inflammatory modulator that
effectively inhibits inflammatory responses in vitro and
in vivo by modulating the MAPK and NF-κB signaling pathways.
This suggests that isobavachin may exert therapeutic efficacy
against vitiligo via its anti-inflammatory properties (16). Stigmasterol is a novel inhibitor of
Nrf2, a ‘master regulator’ of the antioxidant response which
reduces Nrf2 protein levels (41).
The compromised Nrf2 pathway in melanocytes from vitiligo patients
reduces the activation of the antioxidant enzyme system, resulting
in dysregulated cellular autophagy. This, in turn, increases the
sensitivity of vitiligo melanocytes to oxidative stress, thereby
facilitating the onset and progression of vitiligo (42). Therefore, it was hypothesized that
P. corylifolia exerts its therapeutic effects on vitiligo
through multiple active ingredients acting on key targets along
multiple pathways. Further refinement of quantitative assays for
melanin production in zebrafish remains necessary. Additionally,
validation of the targets and pathways implicated in the present
study should be conducted in primary melanocytes and vitiligo
animal models using approaches such as target fishing and gene
silencing (short interfering/small hairpin RNA).
P. corylifolia may exert its effects through
active ingredients such as isobavachin, bavachin and stigmasterol,
which act on core targets such as AKT1, BCL2 and MAPK1 and affect
signaling pathways such as PI3K/AKT, thereby treating vitiligo. The
current study provided a conceptual basis for the use of P.
corylifolia in the prophylaxis and therapy of vitiligo,
although the intricacies of its pharmacodynamic mechanism require
further investigation.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by 2023 Anhui Province Higher
Education Science Research Project (grant nos. 2023AH052279 and
2023AH052272) and 2024 Bozhou College Horizontal Project (grant
nos. BYH202485, BYH2025016 and BYH2025098).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
MZ and CZ conceived and supervised the project and
designed the experiments. CZ, MZ, XZ, MW, ZW, ZC and YY collected
the data. CZ, ZW and XZ analyzed the data. CZ drafted the
manuscript. XZ and CZ confirm the authenticity of all the raw data.
All authors contributed to manuscript revision. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
All zebrafish experiments were reviewed and approved
by the Animal Ethics Committee of Bozhou University, with ethics
approval number DFDW/BZUU-2025-ZF-031.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Frisoli ML, Essien K and Harris JE:
Vitiligo: Mechanisms of pathogenesis and treatment. Annu Rev
Immunol. 38:621–648. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Araj S, Kemp EH and Gawkrodger DJ:
Pathoimmunological mechanisms of vitiligo: The role of the innate
and adaptive immunities and environmental stress factors. Clin Exp
Immunol. 207:27–43. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tong XY, Wang Y, Duan C, Jia H, Wei MY and
Chang XT: Clinical epidemiologic analysis of 112 cases of vitiligo
in children. Dermatol Venereol. 42:875–876. 2020.
|
|
4
|
Faraj S, Kemp EH and Gawkrodger DJ:
Patho-immunological mechanisms of vitiligo: the role of the innate
and adaptive immunities and environmental stress factors. Clin Exp
Immunol. 207:27–43. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Marchioro HZ, Silva de Castro CC, Fava VM,
Sakiyama PH, Dellatorre G and Miot HA: Update on the pathogenesis
of vitiligo. An Bras Dermatol. 97:478–490. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen J, Li S and Li C: Mechanisms of
melanocyte death in vitiligo. Med Res Rev. 41:1138–1166. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wade-Irimada M, Tsuchiyama K, SasakiI R,
Hatchome N, Watabe A, Kimura Y, Yamasaki K and Aiba S: Efficacy and
safety of i.v. methylprednisolone pulse therapy for vitiligo: A
retrospective study of 58 therapy experiences for 33 vitiligo
patients. J Dermatol. 48:1090–1093. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shi N, Chen Y, Wang J and Ni H: Clinical
observation on the effect of Zengse Pill in treating patients with
vitiligo of qi-stagnancy and blood-stasis syndrome type. Chin J
Integr Med. 14:303–306. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang K, Guan C, Shang X, Ying X, Mei S,
Zhu H, Xia L and Chai Z: A bioinformatic analysis: The
overexpression and clinical significance of FCGBP in ovarian
cancer. Aging (Albany NY). 13:7416–7429. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
He D, Huang JH, Zhang ZY, Du Q, Peng WJ,
Yu R, Zhang SF, Zhang SH and Qin YH: A network pharmacology-based
strategy for predicting active ingredients and potential targets of
LiuWei DiHuang pill in treating type 2 diabetes mellitus. Drug Des
Devel Ther. 13:3989–4005. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li Y, Yang Q and Yu Y: A network
pharmacological approach to investigate the mechanism of action of
active ingredients of Epimedii Herba and their potential
targets in treatment of Alzheimer's disease. Med Sci Monit.
26:e9262952020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen S, Jiang H, Cao Y, Wang Y, Hu Z, Zhu
Z and Chai Y: Drug target identification using network analysis:
Taking active components in Sini decoction as an example. Sci Rep.
6:242452016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Singh A, Gotherwal V, Junni P, Vijayan V,
Tiwari M, Ganju P, Kumar A, Sharma P, Fatima T, Gupta A, et al:
Mapping architectural and transcriptional alterations in
non-lesional and lesional epidermis in vitiligo. Sci Rep.
7:98602017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Katz EM, Chu DK, Casey KM, Jampachaisri K,
Felt SA and Pacharinsak C: The stability and efficacy of tricaine
methanesulfonate (MS222) solution after long-term storage. J Am
Assoc Lab Anim Sci. 59:393–400. 2020.PubMed/NCBI
|
|
15
|
Matthews M and Varga ZM: Anesthesia and
euthanasia in zebrafish. ILAR J. 53:192–204. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wallace CK, Bright LA, Marx JO, Andersen
RP, Mullins MC and Carty AJ: Effectiveness of rapid cooling as a
method of euthanasia for young zebrafish (Danio rerio). J Am Assoc
Lab Anim Sci. 57:58–63. 2018.PubMed/NCBI
|
|
17
|
Chung YC, Song SJ, Lee A, Jang CH, Kim CS
and Hwang YH: Isobavachin, a main bioavailable compound in
psoraleae fructus, alleviates lipopolysaccharide-induced
inflammatory responses in macrophages and zebrafish by suppressing
the MAPK and NF-κB signaling pathways. J Ethnopharmacol.
321:1175012024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Park J, Seo E and Jun HS: Bavachin
alleviates diabetic nephropathy in db/db mice by inhibition of
oxidative stress and improvement of mitochondria function. Biomed
Pharmacother. 161:1144792023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wu X, Zhang Z, Zhang X, Guo YP, Liu F,
Gong JW, Li L, Chen XY and Li ZP: Upregulation of A20 and TAX1BP1
contributes to the anti-neuroinflammatory and antidepressant
effects of bavachalcone. Int Immunopharmacol. 122:1105522023.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nizam NN, Mahmud S, Ark SA, Kamruzzaman M
and Hasan M: Bakuchiol, a natural constituent and its
pharmacological benefits. F1000Res. 12:292023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shi M, Zhang Y, Song M, Sun Y, Li C and
Kang W: Screening the marker components in Psoralea
corylifolia L. with the aids of Spectrum-effect relationship
and component Knock-Out by UPLC-MS2. Int J Mol Sci.
19:34392018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Basnet R and Basnet BB: Overview of
protein kinase B enzyme: A potential target for breast and prostate
cancer. Curr Mol Pharmacol. 14:527–536. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Balasuriya N, McKenna MS, Liu XG, Li S and
O'donoghue P: Phosphorylation-dependent inhibition of Akt1. Genes
(Basel). 9:4502018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim MY, Park JY and Park HS: Akt1-mediated
phosphorylation of RBP-Jk controls notch1 signaling. Biochemistry
(Moscow). 84:1537–1546. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li W, Liu Q, Shi J, Xu X and Xu J: The
role of TNF-α in the fate regulation and functional reprogramming
of mesenchymal stem cells in an inflammatory microenvironment.
Front Immunol. 14:10748632023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fang P, Han Y, Qu Y, Wang X, Zhang Y,
Zhang W, Zhang N, Li G and Ma W: EIF3B stabilizes PTGS2 expression
by counteracting MDM2-mediated ubiquitination to promote the
development and progression of malignant melanoma. Cancer Sci.
113:4181–4192. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chang Y, Kang P, Cui T, Guo W, Zhang W, Du
P, Yi X, Guo S, Gao T, Li C and Li S: Pharmacological inhibition of
demethylzeylasteral on JAK-STAT signaling ameliorates vitiligo. J
Transl Med. 21:4342023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zimmerer JM, Ringwald BA, Chaudhari SR,
Han J, Peterson CM, Warren RT, Hart MM, Abdel-Rasoul M and
Bumgardner GL: Invariant NKT cells promote the development of
highly cytotoxic multipotent CXCR3+CCR4+CD8+ T cells that mediate
rapid hepatocyte allograft Rejection. J Immunol. 207:3107–3121.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Deo AS, Sruthika SU, Karun S, Bisaria K
and Sarkar K: Participation of T cells in generating immune
protection against cancers. Pathol Res Pract. 262:1555342024.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li C, Wang W, Shao J, Zhou S, Ji X, Xi Y,
Xu Q, Huang Y, Wang J, Wan Y and Li Z: Biomimetic polydopamine
loaded with Janus kinase inhibitor for synergistic vitiligo therapy
via hydrogel microneedles. J Nanobiotechnology. 23:632025.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ezeonwumelu IJ, Garcia-Vidal E and Ballana
E: JAK-STAT pathway: A novel target to tackle viral infections.
Viruses. 13:23792021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jia JJ, Zhou X and Chu Q: Mechanisms and
therapeutic prospect of the JAK-STAT signaling pathway in liver
cancer. Mol Cell Biochem. 480:1–17. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Teng Y, Fan Y, Ma J, Lu W, Liu N, Chen Y,
Pan W and Tao X: The PI3K/Akt pathway: Emerging roles in skin
homeostasis and a group of non-malignant skin disorders. Cells.
10:12192021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu T, Xi T, Dong X and Xu D: Research
progress on pathogenesis of skin pigmentation in chronic liver
disease. Biomol Biomed. 25:12182024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chaudhari N, Talwar P, Parimisetty A,
Hellencourt CL and Ravanan P: A molecular web: Endoplasmic
reticulum stress, inflammation, and oxidative stress. Front Cell
Neurosci. 8:2132014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Suo LH, Hou FY, Wang ZY, Wu CH, Xie J,
Miao WG, Fan YM and Zhang J: Spirodiclofen inhibited melanin
synthesis in zebrafish embryos. Pestic Biochem Physiol.
210:1063972025. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ferreira AM, de Souza AA, Koga RCR, Sena
IDS, Matos MJS, Tomazi R, Ferreira IM and Carvalho JCT:
Anti-melanogenic potential of natural and synthetic substances:
Application in zebrafish model. Molecules. 28:10532023. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Khongkarat P, Kitipaspallop W, Puthong S,
Pimtong W, Phuwapraisirisan P and Chanchao C: The in cellular and
in vivo melanogenesis inhibitory activity of safflospermidines from
Helianthus annuus L. bee pollen in B16F10 murine melanoma
cells and zebrafish embryos. PLoS One. 20:e03252642025. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wusiman Z, Zhang AM, Zhang SS, Zhao PP,
Kang YT, Zhang Y, Li ZJ and Huo SX: Galangin ameliorates
PTU-induced vitiligo in zebrafish and B16F10 cells by increasing
melanogenesis through activation of the p38/JNK MAPK pathway. Front
Pharmacol. 16:15210972025. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chen L, Chen S, Li P, Zhao X, Sun P, Liu
X, Wei H, Jiang X, Zhan Z and Wang J: Exploration of the mechanism
of Qinglongyi-Buguzhi drug pair in treating vitiligo based on
network pharmacology, molecular docking and experimental
verification. J Ethnopharmacol. 334:1185952024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Liao H, Zhu D, Bai M, Chen H, Yan S, Yu J,
Zhu H, Zheng W and Fan G: Stigmasterol sensitizes endometrial
cancer cells to chemotherapy by repressing Nrf2 signal pathway.
Cancer Cell Int. 20:4802020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lin X, Meng X, Song Z and Lin J: Nuclear
factor erythroid 2-related factor 2 (Nrf2) as a potential
therapeutic target for vitiligo. Arch Biochem Biophys.
696:1086702020. View Article : Google Scholar : PubMed/NCBI
|