Introduction
Physical exercise is considered to play an essential
role in maintaining a healthy lifestyle and exercise is essential
for maintaining health and preventing disease (1). Exercise regulates metabolism,
supports immune function and enhances cardiovascular and nervous
system activity, making it a multifaceted intervention with broad
public health benefits (2).
Traditionally, the bodily adaptations induced by exercise have been
understood in terms of improved organ function, increased endurance
and enhanced strength (3). Recent
study, however, has shown a more complex understanding of the
relationship between exercise and health, especially the notable
role of extracellular vesicles (EVs), including exosomes, played in
this relationship (4,5). The interaction between exercise and
exosomes is a complex and intriguing research direction. Delving
into this relationship holds notable academic and clinical
importance for revealing the impact of exercise on physiological
functions.
Exosomes, a specific subtype of EVs, are small,
single-membrane, secreted organelles of 30–200 nm in diameter that
have the same topology as the cell and are enriched in selected
proteins, lipids, nucleic acids and glycoconjugates (6). These nanoscale vesicles transmit
diverse signaling molecules between cells and tissues, such as
microRNA (miRNA), messenger RNA and mitochondrial DNA, modulating
physiological responses through autocrine and paracrine methods
(7,8).
Exosomes offer several notable advantages over other
delivery mediums: They can transmit diverse signals simultaneously,
facilitate the delivery of multiple miRNAs, exhibit marked
targeting specificity, shield signaling molecules from enzymatic
degradation, enhance synergistic signal transduction and
participate in the post-transcriptional regulation of gene
expression (9–11). EVs also play marked roles in
fundamental cellular processes, including survival,
differentiation, proliferation and metabolism (12–14).
Nearly all cell types secrete exosomes, which are nanoparticles
capable of interacting with proximate cells or entering distant
organs and tissues (11,15). Upon entering a recipient cell,
exosomes release bioactive substances that modulate protein
expression and activate various signaling pathways (16).
Exosomes also carry an array of surface molecules,
including adhesion molecules, integrins, and tetraspanins (CD9,
CD63, CD81 and CD151), which facilitate interactions with target
cells by binding to transmembrane receptors and ligands (17–19).
Exosomes can also enter recipient cells through endocytosis or
directly traverse the cell membrane, incorporating proteins into
their exosomal membranes (20).
The multifunctionality and widespread distribution of exosomes
underscore their marked potential and utility in the fields of
medicine and biology.
Exercise-induced exosomal signaling molecules
influence both adjacent and distant cells during the adaptive
processes initiated by physical activity, directly contributing to
the regulation of intercellular signaling pathways (20). The release of exosomes during
exercise involves intricate cellular signaling pathways,
representing dynamic adjustments within the cellular
microenvironment. Understanding how different forms of exercise
regulate exosome release, as well as how these exosomal signaling
molecules modulate cellular functions, is needed to unravel the
broader physiological impacts of exercise (21–23).
The present review aimed to comprehensively examine
the relationship between exercise-induced alterations in exosomal
miRNAs and their implications for disease pathophysiology. By
elucidating these connections, it aimed to advance the
understanding of the intricate interplay between exercise and
health, thereby identifying potential therapeutic targets for the
prevention and management of exercise-related diseases.
Impact of exercise on the production and
release of exosomes
Advances in cellular biology have established
skeletal muscle as a dynamic endocrine organ capable of systemic
communication through EVs, particularly exosomes (24,25).
These vesicles play a notable role in regulating physiological
responses to physical activity by delivering molecular signals to
distant tissues.
Research has shown that skeletal muscle releases
exosomes into the circulation, with evidence of their presence in
distant and contralateral muscles, supporting their paracrine-like
effects (26). The biogenesis of
exosomes begins with endocytosis, where the cell membrane
invaginates to form early endosomes (27). These early endosomes undergo
maturation into late endosomes, which involves marked biochemical
remodeling, including the reorganization of membrane proteins and
lipids; this process ultimately leads to the formation of
multivesicular bodies (MVBs) (28). Through complex regulatory
mechanisms, such as the activity of endosomal sorting complexes
required for transport proteins, MVBs fuse with the cell membrane
to release exosomes into the extracellular environment (28).
Exosomes circulate via the bloodstream or lymphatic
system, delivering proteins, RNA and signaling molecules to
recipient cells, thereby regulating their functions (27). Exercise markedly influences the
production and composition of exosomes. In healthy individuals,
exercise can acutely increase circulating exosome levels by
~1.5–3-fold, particularly through physiological stressors such as
mechanical strain and oxidative stress, which activate signaling
pathways during muscle contraction (29). For example, increased intracellular
Ca2+ levels trigger key pathways such as mTOR and
AM-activated protein kinase, which regulate cellular metabolism and
stress responses, directly impacting the quantity and molecular
composition of exosomes (30,31).
The illustrated process of exosome formation, highlighting the
molecules and information transfer pathways involved is illustrated
in Fig. 1. This depiction provides
a representation of the formation and fusion of intracellular
vesicles, the loading mechanisms of miRNAs and other molecules, and
the pathways of exosome-mediated intercellular communication.
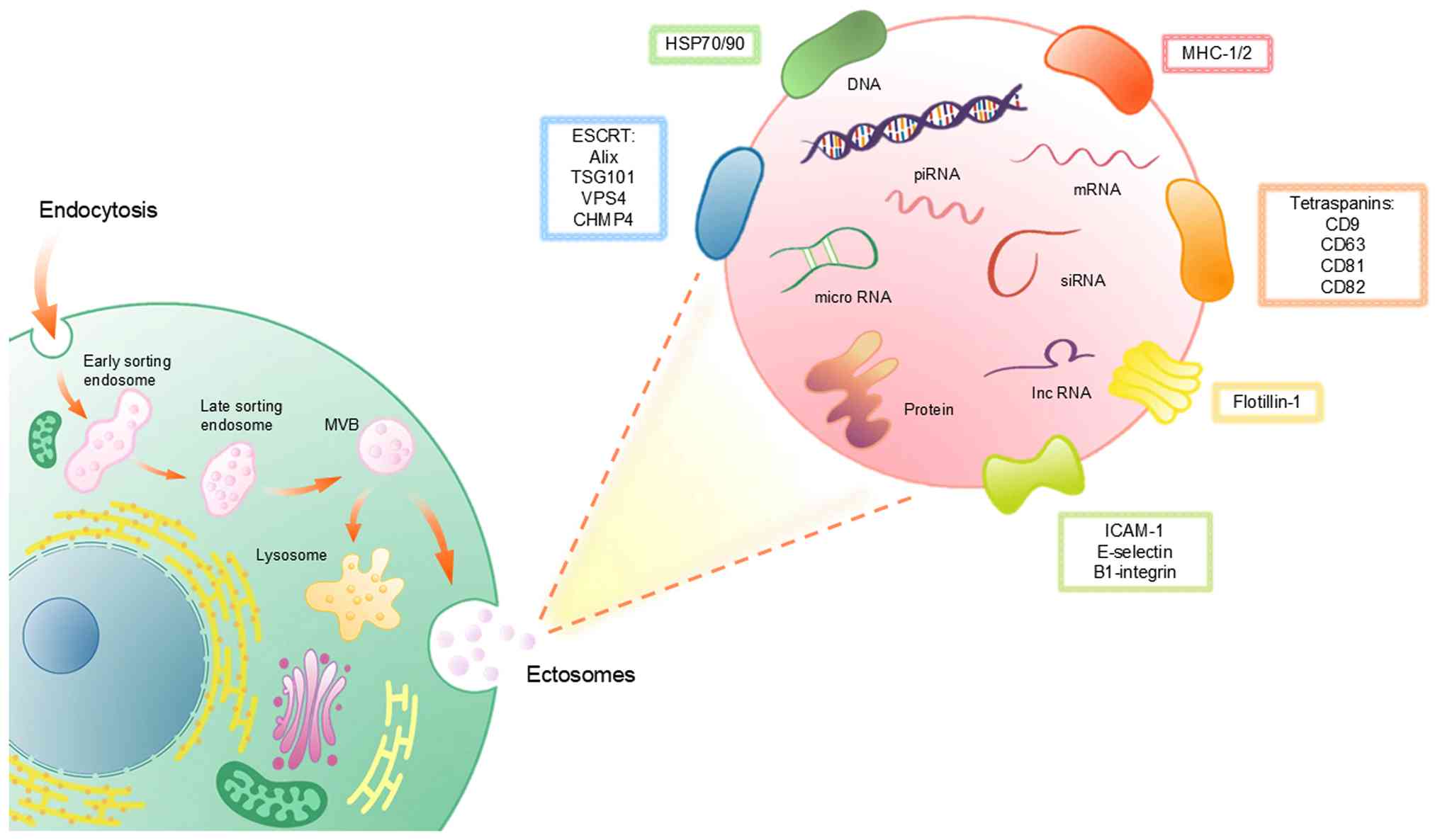 | Figure 1.Biogenesis, composition and molecular
cargo of EVs. EVs are generated through the endocytic pathway,
beginning with early sorting endosomes that mature into late
sorting endosomes and subsequently form MVBs. Upon fusion of MVBs
with the plasma membrane, intraluminal vesicles are released as
exosomes, whereas fusion with lysosomes leads to cargo degradation.
The schematic highlights the diverse molecular cargo carried by
EVs, including nucleic acids (DNA, mRNA, miRNA, siRNA, piRNA and
lncRNA), proteins and membrane-associated molecules. EV membranes
are enriched with tetraspanins (CD9, CD63, CD81 and CD82), HSPs
(HSP70/90), flotillin-1, MHC-I/II and adhesion molecules such as
ICAM-1, E-selectin and β1-integrin. The ESCRT, including Alix,
TSG101, VPS4 and CHMP4, are involved in EV biogenesis and cargo
sorting. EVs, extracellular vesicles; MVBs, multivesicular bodies;
miRNA, microRNA; siRNA, small interfering RNA; piRNA,
Piwi-interacting RNA; lncRNA; HSP, heat shock protein; MHC-I/II,
major histocompatibility complex class I and II; ICAM-1,
intercellular adhesion molecule-1; ESCRT, endosomal sorting
complexes required for transport; Alix, ALG-2-interacting protein
X; TSG101, tumor susceptibility gene 101 protein; VSP4, vacuolar
protein sorting-associated protein 4; CHMP4, charged multivesicular
body protein 4. |
Exercise modulates the dynamics of exosomes in a
pattern dependent on intensity, type and duration, leading to
variations in both their abundance and molecular composition within
circulation. High-intensity exercise, characterized by pronounced
oxidative stress, tends to provoke robust changes in exosome
secretion and cargo loading, particularly enhancing the release of
vesicles carrying stress-related or inflammatory molecules such as
specific miRNAs (e.g., miR-133a, miR-133b and miR-206) and proteins
(e.g., HSP70 and IL-6) (32,33).
By contrast, moderate- or low-intensity exercise (typically 45–65%
VO2 max) induces only mild or inconsistent increases in
exosome number but still reshapes miRNA signatures and other
molecular cargos, particularly those related to oxidative stress
regulation, endothelial protection and inflammatory modulation
(34,35).
Different exercise modalities lead to distinct EV
profiles, with variations in abundance, surface markers and
molecular cargo depending on exercise type and intensity. For
example, endurance training markedly increases exosomal miRNAs such
as miR-136-3p and miR-342-5p, both of which are involved in tissue
adaptation and cardioprotection. Interval training, including
high-intensity bouts, produces robust changes in specific exosomal
markers and a rapid increase in proteins linked to oxidative stress
response and cell signaling (29,36–38).
Trained individuals typically demonstrate more
efficient vesicle clearance, meaning that their exosomal profiles
return to baseline faster after exercise, and they exhibit distinct
miRNA responses compared with untrained subjects. For instance,
well-trained older men show altered miRNA expression in
exosome-enriched EVs after exercise, indicating durable changes in
tissue communication and adaptation pathways. Sedentary
individuals, by contrast, may have less pronounced or less
efficient exosomal responses (26,39).
These findings collectively highlight that both the
qualitative and quantitative features of exercise-induced exosome
release, such as miRNA and protein signatures, are determined by
the specific modality and intensity of exercise, as well as the
training status of the individual.
In conclusion, exercise serves as a potent
biological modulator, shaping the production, molecular cargo, and
functions of exosomes in an activity-dependent manner.
Investigating these mechanisms not only provides valuable insights
into the physiological benefits of exercise but also offers
promising therapeutic avenues for the treatment of metabolic and
inflammatory diseases.
Impact of exercise-induced exosomal miRNAs
on different organs
Exercise-induced exosomal miRNAs regulate and
protect various body systems by influencing the muscular,
metabolic, cardiovascular, nervous and immune systems (Fig. 2).
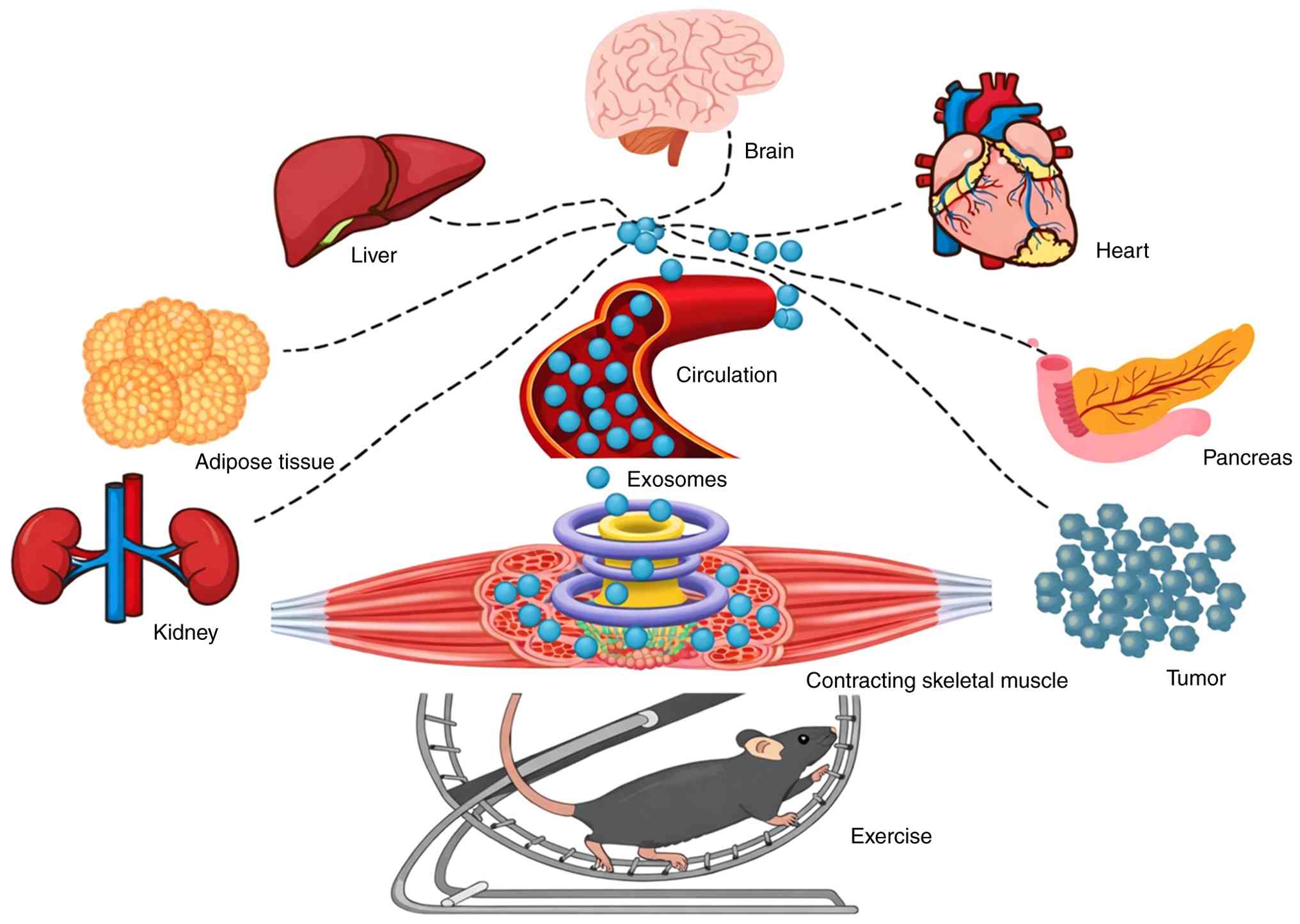 | Figure 2.Interorgan communication mediated by
exercise-induced exosomes. During exercise, contracting skeletal
muscle releases exosomes into the circulation, which act as key
mediators of interorgan communication. These circulating exosomes
can be transported via the bloodstream to multiple peripheral
organs, including the brain, heart, liver, adipose tissue, kidney
and pancreas, as well as to pathological tissues such as tumors.
Exercise-induced exosomes carry diverse bioactive cargoes,
including microRNAs, proteins and other signaling molecules, which
modulate metabolic regulation, cardiovascular function, neural
activity, immune responses and tissue remodeling in target organs.
Conversely, non-muscle organs can also release exosomes into the
circulation that act on skeletal muscle, thereby influencing muscle
metabolism, mitochondrial function, and adaptive responses under
physiological and pathological conditions. |
Multilevel metabolic regulation
involving exercise-induced exosomal miRNA release
Exercise is not only a source of physical vitality
but also a key driver of micro-metabolic regulation. In this
process, exosomes play a pivotal role in serving as nano-messengers
of intercellular communication, carrying regulatory information
related to energy metabolism, lipid metabolism, glucose metabolism,
immune regulation and metabolic communication between organs,
thereby constructing a detailed and complex metabolic balance
network (40). Muscle-derived
exosomes produced during exercise may contain molecules that
enhance insulin sensitivity or promote fat oxidation (33). These finely tuned molecular
mechanisms make exosomes play a marked role in regulating the
body's response to exercise, particularly in energy metabolism and
cellular signal transduction.
Studies have shown that muscle-derived exosomes play
a notable role in enhancing insulin sensitivity and promoting fat
oxidation; for instance, one study demonstrated that exosomes from
skeletal muscles can enhance insulin sensitivity in mice by
downregulating hepatic FoxO1 (33). Additionally, it was found that
exosomes from muscles of mice on a high-fat diet had enlarged
isolated islets in vitro and induced proliferation of
insulin-secreting cells, suggesting a potential role of exosomes in
influencing insulin secretion (41). In addition, exosomes are
hypothesized to regulate insulin sensitivity by modulating
inflammation or through direct interactions with insulin-responsive
organs, such as the phosphatidylinositol 3-kinase/Akt signaling
pathway (42). These findings
demonstrate the potential of muscle-derived exosomes in impacting
insulin sensitivity and fat oxidation, revealing their involvement
in exercise-regulated energy metabolism and cellular signal
transduction. Another study found that in patients with type 2
diabetes, the concentrations of EVs produced by endothelial cells,
platelets and monocytes were notably elevated (43), suggesting that the higher
concentrations of EVs in diabetics compared with healthy subjects
might be related to insulin resistance exacerbating vesicle release
(44). Exercise can directly
enhance insulin sensitivity by increasing skeletal muscle glucose
uptake, thereby helping to lower blood glucose levels in patients
with type 2 diabetes (45).
Exercise can reduce serum exosomal miR-27a levels, which is
considered a key regulator of PPAR-γ, thereby affecting adipose
tissue browning and insulin sensitivity (46). In addition, exercise training can
effectively improve adipocyte dysfunction caused by intermittent
hypoxia (reducing insulin resistance) by adjusting gut microbiota
balance, reducing intestinal permeability and optimizing plasma
exosomal cargo (47). Furthermore,
exercise has also been shown to reduce elevated plasma EV
concentrations in obese individuals (48). A study by Kawanishi et al
(49) showed that long-term
exercise can regulate EV expression, inhibit macrophage
infiltration in adipocytes and reduce Toll-like receptor 4
expression, aiding in the transition of macrophages from the M1
pro-inflammatory phenotype to the M2 anti-inflammatory phenotype,
thereby alleviating inflammation and enhancing insulin sensitivity.
Glucose tolerance can also be improved by exercise training, and
plasma triglyceride levels can be reduced by altering the miRNA
spectrum in circulating exosomes (such as marked increases in
miR-133a and miR-133b). Exercise-induced exosomal miRNA changes,
such as marked increases in miR-133a and miR-133b, can also reduce
FoxO1 expression in the liver, thereby impacting systemic metabolic
characteristics. These numerous studies prove the notable role of
muscle-released exosomes in improving the metabolic system through
exercise (33).
Further research in this direction may guide
clinical researchers in developing targeted interventions to
improve insulin sensitivity and metabolic health. By regulating
metabolism through exercise-induced exosomes, insight is gained
into a multilevel, synergistic network of energy and metabolic
balance. This provides a valuable perspective for understanding the
comprehensive impact of exercise on body metabolism and offers
strategic targets for future research and treatment of
metabolic-related diseases.
Impact of exercise-induced exosomal
miRNAs release on the cardiovascular system
Exercise, as a crucial factor in maintaining
cardiovascular health, especially in the modulation of exosomes and
their miRNA expression profiles. Exercise plays a crucial role in
maintaining cardiovascular health in part by modulating exosome
release and their miRNA expression profiles. Accumulating evidence
indicates that exercise-induced exosomes exert cardioprotective
effects through anti-inflammatory actions, regulation of lipid
metabolism and oxidative stress, and modulation of immune cell
function, thereby contributing to the attenuation of
atherosclerosis and the protection, survival and repair of
cardiomyocytes (50). For example,
Ruan et al (51) found that
Suxiao Jiuxin Pill, a commonly used medication for angina pectoris
and coronary heart disease, could promote the secretion of exosomes
derived from mouse myocardial mesenchymal stem cells, thereby
protecting against myocardial ischemia/reperfusion injury; however,
the specific molecular cargo and underlying mechanisms of these
exosomes were not further characterized. Long-term exercise
training plays a key role in cardiac protection through circulating
exosomes, effectively preventing damage caused by myocardial
ischemia and reperfusion. Notably, miRNAs in these exosomes,
especially miR-342-5p, are markedly increased in both human and
animal models undergoing exercise training. miR-342-5p targets
cysteine-aspartic protease-9 (caspase-9) and Jnk2 to reduce
myocardial apoptosis induced by hypoxia/reoxygenation, while
promoting Akt phosphorylation via regulation of the phosphatase
gene Ppm1f. In animal models and endothelial cell systems,
exercise training and laminar shear stress have been shown to
upregulate miR-342-5p expression, supporting its role in
exercise-associated cardioprotective mechanisms (37). Exercise-induced exosomes also
positively impact cardiovascular microcirculation by improving
tissue blood supply and oxygenation through growth factors and
miRNAs, such as the upregulation of miR-126 levels in endothelial
progenitor cell-derived exosomes in response to moderate-intensity
aerobic treadmill exercise, alleviating apoptosis in endothelial
cells (ECs) induced by high glucose and promoting EC migration and
tube formation (52). Exercise, by
modulating exosome components, improves the angiogenic potential in
patients with type 2 diabetes, increasing the expression of SOD3
and ATP7A proteins in plasma exosomes, thus promoting angiogenic
responses. Experiments have shown that exercise training restores
the impact of exosomes from type 2 diabetic mice on endothelial
cell angiogenesis, revealing the key role of SOD3 in
exercise-induced angiogenesis. Therefore, exosomal SOD3 can serve
as a type of exercise-mimetic therapy, supporting angiogenesis and
wound repair in cardiovascular diseases (53).
In summary, exercise-induced exosomes play a notable
role in regulating endothelial cell functions, anti-inflammatory
actions, slowing down atherosclerosis, protecting myocardial cells
and improving cardiovascular microcirculation.
Impact of exercise-induced exosomal
miRNAs release on the immune system
miRNA are notable regulatory factors in the immune
system, being small non-coding RNA molecules responsible for
inhibiting the expression of specific genes, thereby affecting the
behavior of immune cells (54).
These small molecules play a marked role in the differentiation,
proliferation, activation and cytokine production of immune cells
such as T cells, B cells, macrophages and dendritic cells (55–58).
Research on immune-related miRNAs is summarized in Table I, which details the miRNAs closely
related to immune functions (59–80).
 | Table I.MiRNAs closely related to immune
function. |
Table I.
MiRNAs closely related to immune
function.
| miRNA | Functions | (Refs.) |
|---|
| miR-155 | Modulates
inflammatory responses, regulating the activation and response of
various immune cells. | (59) |
| miR-146a/b | Promotes the
resolution of inflammation by producing pro-inflammatory cytokines
within inflammatory pathways, exerting regulatory functions. | (60–62) |
| miR-21 | Regulates T cell
functions, proliferation and immune suppression, and is involved in
promoting cell proliferation, invasion and the survival of
activated T cells. | (63–66) |
| miR-181a/b | Regulates T cell
development and function, playing a role in immune tolerance. | (67,68) |
| miR-223 | Acts as a potent
regulator of granulocyte, macrophage and dendritic cell
differentiation and proliferation during inflammation. | (69) |
| miR-124 | Influences the
immune response of the nervous system, regulating microglia and
immune modulation within brain tissue. | (70,71) |
| miR-17-92 | Regulates the
proliferation and survival of T and B lymphocytes, affecting the
balance of immune responses. | (72,73) |
| miR-125b | Has a notable
regulatory role in the proliferation and differentiation of immune
cells, potentially playing a role in autoimmune diseases. | (74) |
| miR-150 | Regulates the
development and function of T and B lymphocytes, particularly
playing a notable role in the differentiation of lymphocyte
subsets. | (75) |
| miR-132 | May affect
inflammation and immune responses, involved in modulating the
functions of T cells and dendritic cells. | (76,77) |
| miR-29 | Controls the
activation state of T cells and macrophages, participating in the
regulation of inflammatory responses and immune tolerance. | (78,79) |
| miR-30 | Affects the
maturation, proliferation, differentiation, and activation of
various immune cells, associated with autoimmune diseases and also
involved in the regulation of antibody production. | (80) |
Studies have shown that exercise can affect the
release of exosomes and their miRNA components, which can be
absorbed by immune cells and influence intracellular gene
expression, thus modulating the immune response (81). Specifically, exercise-associated
EV-miRNAs, such as miR-21, miR-146a and miR-126, have been reported
to regulate inflammatory signaling by promoting anti-inflammatory
cytokines including interleukin-10 (IL-10) while suppressing
pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α)
and IL-6, thereby contributing to immune homeostasis (81). After intense exercise or long-term
training, this regulation helps to reduce chronic inflammation and
excessive immune responses, enhancing the defense of the immune
system against pathogens (22).
Beyond enhancing muscle function and endurance,
exercise also modulates immune and inflammatory responses, as
exercise-induced tissue damage and metabolic changes can trigger
inflammation, which is subsequently regulated by exercise-induced
exosomes transferring specific miRNAs such as miR-146a to promote
tissue repair and recovery (81).
This alteration in miRNA helps control post-exercise inflammatory
responses, reducing tissue damage and accelerating recovery. By
regulating inflammatory responses, these miRNAs help maintain the
balance of the immune system, prevent overreactions, and protect
the body from the effects of chronic inflammation, which is
especially important for athletes engaged in high-intensity
exercise over the long term.
The changes in miRNAs within exercise-induced
exosomes are not only notable for regulating normal immune
responses and controlling inflammatory responses but may also play
a role in preventing and treating various diseases. For example,
specific miRNAs may reduce the risk or severity of autoimmune
diseases by modulating immune cell functions (22,82).
These miRNAs have potential therapeutic value in regulating
autoimmune responses and preventing excessive immune activities.
Since these miRNAs can be transported long distances within the
body via exosomes, they have the potential to be non-invasive
biomarkers for monitoring immune status, inflammatory responses and
response to exercise interventions (83).
Overall, exercise-induced exosomes have a marked and
extensive effect on the immune system. These exosomes play a role
in regulating the activity of immune cells, balancing inflammatory
responses, enhancing immune surveillance and memory formation
(81). Although research has
provided important insights, further exploration is needed to
understand precisely how exercise regulates the specific mechanisms
of exosomes. Future research should focus on the specific
mechanisms of action of exercise-induced exosomes in different
types of immune cells and how these exosomes exhibit variations in
different exercise modalities and among different populations.
Additionally, exploring the potential clinical applications of
exercise-induced exosomes, especially in the prevention and
treatment of immune diseases, will be a valuable research
direction.
Impact of exercise-induced exosomal
miRNAs release on the nervous system
The interaction between exosomes and neural cells
plays a key role in the nervous system. Neurons and glial cells
communicate and regulate the functions of each other by releasing
exosomes. First, exosomes facilitate direct information exchange
between neurons and glial cells; neurons release exosomes carrying
specific miRNAs, proteins and other signaling molecules, which can
be taken up by surrounding glial cells, affecting their functions
and states (84). Similarly, glial
cells also release exosomes, exerting regulatory effects on
surrounding neurons (85).
Exercise-stimulated exosomes markedly impact neural cell repair and
regeneration; long-term exercise improves the blood-brain barrier
function in Alzheimer's disease mouse models and facilitates the
clearance of amyloid-β (86).
Exosomes isolated from the brains of exercising mice can improve
pericyte and endothelial cell functions, upregulating
platelet-derived growth factor receptor β, zonula occludens-1 and
claudin-5 expression. Exercise also regulates the expression of
miR-532-5p in exosomes, thus affecting brain cell functions
(87). Another study investigating
EV characteristics and EV-related miRNAs post-rest and aerobic
exercise in cerebral palsy and typically developing individuals
found reduced EV concentration and increased circulating miR-486 in
patients with cerebral palsy. These findings suggest an association
between exercise-related alterations in circulating EV-miRNA
profiles and skeletal muscle-related transcriptional regulation.
Functional analyses indicated that miR-486 may influence myogenic
gene expression; however, its direct role in cerebral palsy
pathology and effects on neural cell types were not directly
examined (88). In summary,
exosomes may regulate nervous system functions and information
transmission by directly interacting with neurons and glial cells
and potentially interacting with the blood-brain barrier. Although
the mechanisms by which exosomes cross the blood-brain barrier
still need further research, hypotheses regarding these mechanisms
have been proposed, including transcytosis, paracellular and
transcellular pathways (89)
jumping from one cell to another through MVB compartments (90) and passive diffusion (91).
The effect of signal molecules carried by
exercise-induced exosomes on neural cells is a subject of interest.
Exercise-induced exosomes have been shown to promote synaptic
growth, regulate neural plasticity and enhance neural maturity
(92). Further research indicates
that exosomes from exercise can alleviate anxiety, improve
neurogenesis, regulate neuroinflammation and promote neurological
localization, demonstrating their potential in treating certain
neurological diseases (93,94).
Exosomes are involved in complex biological processes of neural
regeneration, neuroprotection and neural plasticity; accumulating
evidence indicates that miRNAs carried by exosomes can regulate
neural stem cell differentiation and promote neural regeneration,
providing a mechanistic basis for the neuroregulatory effects of
exercise-induced exosomes. For example, a study found that exosomes
from neural stem cells (NSCs) drove the differentiation of NSCs and
promoted the maturation of neurons and glial cells, with miR-9
being most abundantly expressed in NSC-derived exosomes (95). Additionally, another study
demonstrated that exosomal miR-21a derived from neural progenitor
cells regulates neurogenesis by promoting neuronal differentiation
in in vitro neural progenitor cell models (96). Research has also focused on the
role of stem cell-derived exosomes and miRNAs in spinal cord
injuries; results showed that specific miRNAs carried by bone
marrow stem cell-derived exosomes, such as miR-126, miR-124-3p and
miR-181c, can inhibit neuronal cell death and promote neuronal
differentiation in spinal cord injury models (97). Overall, these findings demonstrate
the potential of miRNAs carried by exosomes in promoting the
proliferation and differentiation of neural stem cells, which is
notable for the development of new methods for studying neural
regeneration and stimulating endogenous neurogenesis. In terms of
neurotransmitter release, neural signal transduction and
neuroinflammation, exosomes influence the synthesis and release of
neurotransmitters in neurons by modulating miRNA transport methods
(84). Exercise also affects
exosome components, regulating neural transmission and neural
plasticity. In the field of neuroscience research, early exercise
intervention has shown notable effects in neuroprotection after a
stroke. Through the release of exosomes, early exercise
intervention after stroke inhibits excessive microglial activation
and enhances synaptic plasticity, thereby improving neurological
function, promoting body weight recovery and reducing cerebral
infarct volume in rats, suggesting a potential exosome-mediated
therapeutic mechanism applicable to other neurological disorders
such as Alzheimer's disease and cerebral palsy (98). Additionally, exercise intervention
helps reduce the number of microglia, enhance dendritic complexity
and increase the expression of synaptophysin and postsynaptic
density protein 95 (98). Exercise
interventions increase the levels of exosomes in the serum of
stroke rats, thereby promoting brain synaptic growth and the
integrity of the corticospinal tract; this process aids in reducing
infarction volume, improving neural function and rat gait. Exosomes
play a key role during exercise, enhancing serum and brain exosome
levels, beneficial for neural plasticity and neuroprotection
(92). Exercise training, in
conjunction with bone marrow MSC-derived exosomes (MSC-exos), and
activation of the JNK1/c-Jun signaling pathway markedly reduce
neuronal apoptosis and cerebral infarction volume, promoting
synapse formation and axonal regeneration, thus effectively
restoring neural function, showing improved effects compared with
treadmill exercise or MSC-exos treatment alone (99). In the regulation of
neuroinflammation, exercise-induced exosomes also play a notable
role. Regarding neuroinflammatory responses, exosomes may modulate
the inflammatory state of neural cells by carrying
anti-inflammatory or pro-inflammatory molecules. A study
demonstrated that the process of neuroinflammation can be promoted
by miRNAs such as miR-155 or inhibited by miRNAs including
miR-146a, miR-124 and miR-21. miR-124 influences a wide range of
pathological processes, including mental illnesses and central
nervous system injuries; and other miRNAs, such as the let-7
family, can promote or inhibit the induction of inflammatory
responses (100).
Exercise-released exosomes carrying anti-inflammatory molecules,
such as miR-146a, miR-21 and miR-126, can attenuate
neuroinflammation by suppressing pro-inflammatory signaling
pathways (e.g., NF-κB) and modulating immune cell activation,
thereby contributing to the prevention and treatment of
neurodegenerative diseases (101). In summary, these exosomes, by
carrying specific signaling molecules, participate in regulating
physiological processes such as neurotransmitter release, neural
signal transduction and neuroinflammatory responses. The molecules
they carry may directly or indirectly affect the activity of
neurons, neural signal transduction and inflammation. These
findings underscore the notable role of exercise through exosomes
in neuroprotection and stroke treatment strategies.
The role of exosomes in emotional regulation and
stress response is also of note. They help alleviate stress and
anxiety by regulating the release and balance of neurotransmitters,
which is vital for maintaining mental health. A study showed that
in patients with depression, there was an upregulation of
has-miR-335-5p and a downregulation of has-miR-1292-3p in plasma
exosomes, affecting signaling pathways related to synaptic density,
axonogenesis and cell growth (102). These molecules may regulate the
efficiency of information transmission between neurons, affecting
neural signal transmission.
To study the role of exercise-induced exosomal
miRNAs in these physiological processes, this information was
collected in Table II to
elucidate the effect of miRNAs on the nervous system (103–112). In summary, it has been
demonstrated that exercise-induced exosomes play a notable role in
the health and functionality of the nervous system. From promoting
the repair and regeneration of neural cells, modulating neural
transmission and plasticity, to controlling neuroinflammation and
affecting mood regulation, exercise-induced exosomes offer new
research perspectives and potential therapeutic options in the
field of neuroscience.
 | Table II.Effects of exosomal miRNAs on the
nervous system. |
Table II.
Effects of exosomal miRNAs on the
nervous system.
| miRNA | Functions | (Refs.) |
|---|
| miR-9 | Regulates neural
development and synaptic plasticity. | (103) |
| miR-124 | Promotes neuronal
differentiation and post-synaptic gene expression. | (104) |
| miR-132 | Participates in the
development of neurons and synaptic plasticity. | (105) |
| miR-29 | Modulates the
functions of neurons and glial cells. | (106) |
| miR-125 | Plays a role in
synaptic formation and neuronal survival. | (107,108) |
| miR-134 | Regulates synaptic
plasticity and post-synaptic gene expression. | (109) |
| miR-137 | Regulates neural
development, synaptic formation and neurodegenerative
diseases. | (110) |
| miR-206 | Affects neuronal
growth and synaptic function. | (111) |
| miR-335 | Potentially
regulates neural system development and neuronal apoptosis. | (112) |
| miR-431 | Influences neuronal
development and synaptic plasticity. | (112) |
| miR-708 | Potentially
involved in neuronal growth and synaptic development. | (112) |
Future studies need to further explore the specific
mechanisms of action of exercise-induced exosomes in different
types of neurological diseases. Particularly for age-related
neurodegenerative diseases, such as Alzheimer's and Parkinson's,
exercise-induced exosomes may play a key role in therapy and
prevention. Additionally, research should investigate how different
exercise modalities affect the characteristics and efficacy of
exosomes and how these exosomes function in diverse populations,
thus providing more personalized exercise prescriptions. Moreover,
exploring the potential clinical applications of exercise-induced
exosomes is also a notable direction for future research. For
instance, using exosomes as biomarkers to assess the effects of
exercise interventions or as potential carriers for treating
neurological diseases. Overall, the role of exercise-induced
exosomes in the nervous system underscores the importance of
exercise as an effective neuroprotective strategy. As the
understanding of this field improves, it can be expected that more
discoveries about the interactions between exercise, exosomes and
neural health will be made, providing new strategies for improving
neural health and treating neurological diseases.
Exosome-mediated inter-organ communication
targeting skeletal muscle
Exosomes derived from non-muscle organs have emerged
as notable regulators of skeletal muscle physiology. While skeletal
muscle functions as a secretory organ, it is also a recipient of
exosomal signals originating from the brain, liver, adipose tissue
and kidney (113). These
organ-derived exosomes, enriched with tissue-specific miRNAs and
other bioactive molecules, modulate muscle metabolism,
mitochondrial function and adaptive responses under both
physiological and pathological conditions (113).
Brain-derived exosomes constitute a notable pathway
for neuro-muscular communication. Neurons and glial cells release
exosomes containing miR-124, miR-9 and miR-132, which can enter the
bloodstream and reach peripheral skeletal muscles, where they
contribute to the maintenance and regeneration of neuromuscular
junctions (114). However, under
conditions of neurodegeneration or inflammation, brain-derived
exosomes may carry pro-inflammatory miRNAs, such as miR-155, miR-21
and miR-34a, which can exacerbate muscle atrophy and fatigue and
ultimately impair neuromuscular signaling (114).
Similarly, the liver communicates with skeletal
muscle via hepatocyte-derived exosomes carrying miRNAs such as
miR-122, miR-192 and miR-21 (32).
These vesicles influence muscle insulin sensitivity and lipid
metabolism, and in metabolic disorders such as obesity and type 2
diabetes, increased levels of hepatic exosomal miR-122 have been
shown to impair glucose uptake and mitochondrial function in
skeletal muscle (115,116). Thus, exosomal miR-122 acts as a
negative regulator in skeletal muscle, linking liver pathology to
impaired muscle insulin sensitivity and mitochondrial health in
metabolic diseases.
Adipose tissue also plays an integral role in this
inter-organ communication network. Exosomes secreted by adipocytes
are rich in miR-27a and miR-29a, which target the PGC-1α signaling
pathway in skeletal muscle, suppressing mitochondrial biogenesis
and promoting insulin resistance (117). Lifestyle modifications,
particularly regular physical activity and caloric restriction, can
reshape the miRNA profile of adipose-derived exosomes to promote
metabolic health. Exercise and dietary interventions reduce the
abundance of adipose-derived exosomal miRNAs such as miR-27a and
miR-29a, which are associated with metabolic dysfunction, while
concomitantly increasing exercise-responsive exosomal miRNAs
involved in mitochondrial biogenesis, glucose metabolism and
insulin signaling. Although the specific miRNAs upregulated vary
across studies and intervention types, this overall remodeling of
exosomal cargo facilitates healthier inter-organ communication and
improves systemic metabolic balance (117–120).
The kidney also exerts regulatory effects on
skeletal muscle physiology via exosomal signaling. Renal exosomes,
detectable in both blood circulation and urine, carry uremic
toxin-responsive miRNAs, including miR-223 and miR-26a. In chronic
kidney disease, these vesicles may reach skeletal muscle and elicit
inflammatory and oxidative stress responses, contributing to muscle
wasting (41,121).
Collectively, these findings highlight a complex and
dynamic exosome-mediated inter-organ communication network, wherein
skeletal muscle acts not only as an exosome-secreting tissue but
also as a responsive target of exosomal signals from distant
organs. This bidirectional communication is modulated by metabolic
status, pathological conditions and exercise interventions. A
deeper understanding of how physical activity reshapes these
exosome-mediated interactions will offer valuable insights into
systemic adaptive mechanisms and may inform the development of
novel therapeutic strategies for metabolic and neuromuscular
diseases.
Future research directions and
prospects
Exercise, as a critical lifestyle intervention,
induces extensive physiological adaptations across multiple organ
systems. Mounting evidence demonstrates that exercise regulates the
biogenesis, molecular composition and functional dynamics of EVs,
which exhibit considerable therapeutic potential in cardiovascular,
neurodegenerative, metabolic, oncological and musculoskeletal
disorders. Exercise-induced EVs exert multifaceted regulatory
effects on recipient cells by modulating their secretion into
circulation, remodeling their protein, lipid and nucleic acid
cargo, and enhancing immune regulation and intercellular
communication. Collectively, these processes facilitate tissue
regeneration, attenuate inflammation and sustain systemic
homeostasis. Consequently, exercise-derived EVs have emerged as a
rapidly expanding field in biomedical research, offering new
mechanistic insights and therapeutic avenues for health maintenance
and disease management.
In cardiovascular diseases, EVs promote cardiac
repair by stimulating angiogenesis and suppressing inflammation
(35). In neurodegenerative
disorders, they support neuronal survival and preserve cognitive
function through the transfer of neurotrophic factors (84). In oncology, EVs enriched with
tumor-suppressive molecules impede malignant progression and
trigger apoptotic pathways (122). Furthermore, their capacity to
serve as biocompatible, targeted drug-delivery vehicles enhances
therapeutic specificity while minimizing systemic toxicity
(122).
Despite these promising advances, the clinical
translation of exercise-induced EVs remains in its infancy. Major
challenges include the standardization of isolation and
purification protocols, validation of biosafety and reproducibility
and identification of optimal sources, dosages and delivery routes.
Bridging the translational gap will require rigorous mechanistic
studies and well-designed clinical trials. Interdisciplinary
collaboration among scientists in biology, medicine and exercise
science will be essential to elucidate the molecular underpinnings
of EV-mediated effects and to refine their therapeutic
applications.
While most studies emphasize the beneficial roles of
exercise-induced EVs, the potential for adverse or maladaptive
effects cannot be entirely ruled out. To date, no direct adverse
outcomes have been associated with exosome release during exercise;
however, under extreme or pathological conditions, such as
overtraining, acute oxidative stress or underlying metabolic
disorders, the molecular composition of EV cargo may shift toward a
pro-inflammatory or stress-related profile (26,123). These alterations may transiently
influence endothelial or immune cell function, reflecting
physiological stress responses rather than overt toxicity. Such
findings highlight the need to investigate dose-response
relationships, thresholds of exercise intensity and the temporal
patterns of EV secretion, in order to better differentiate between
adaptive and maladaptive physiological outcomes.
In summary, exercise-induced EVs represent a dynamic
and rapidly evolving frontier in translational medicine, with
marked potential as next-generation therapeutic agents for health
optimization and disease intervention. Continued investigation of
their biological roles, together with advances in EV engineering
and application technologies, will accelerate their incorporation
into precision medicine and foster transformative innovations in
healthcare.
Acknowledgements
The figures were created using BioRender
(BioRender.com) and Microsoft PowerPoint 2021 (Microsoft
Corporation).
Funding
The present review was supported by grants from the Hebei
Central Guiding Science and Technology Development Fund (grant no.
246Z7731G), the Science and Technology Plan Program of Baoding
(grant no. 2472P011) and the Hebei Provincial Natural Science
Foundation Biomedicine Joint Fund Cultivation Project (grant no.
H2022201050).
Availability of data and materials
Not applicable.
Authors' contributions
ZLW conducted comprehensive literature collection
and organization, designed the structure of the review, drafted the
original manuscript, integrated the literature and completed all
revisions of the manuscript. WJG participated in drafting and
organizing the original manuscript and contributed to subsequent
revisions. HJH contributed to literature collection and content
organization, participated in drafting and integrating key
mechanistic sections of the initial manuscript, prepared schematic
figures and data visualizations, and contributed to subsequent
revisions of the manuscript. YZ contributed to methodology
development and participated in drafting and revising the original
manuscript. NL supervised the study, contributed to
conceptualization and project administration, participated in
refinement of the initial manuscript, and revised the manuscript
critically for important intellectual content. All authors have
read and approved the final version of the manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ribeiro MM, Andrade A and Nunes I:
Physical exercise in pregnancy: Benefits, risks and prescription. J
Perinat Med. 50:4–17. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Meyer-Lindemann U, Moggio A, Dutsch A,
Kessler T and Sager HB: The impact of exercise on immunity,
metabolism, and atherosclerosis. Int J Mol Sci. 24:33942023.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hughes DC, Ellefsen S and Baar K:
Adaptations to endurance and strength training. Cold Spring Harb
Perspect Med. 8:a0297692018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Huang M, Cheng S, Li Z, Chen J, Wang C, Li
J and Zheng H: Preconditioning exercise inhibits neuron ferroptosis
and ameliorates brain ischemia damage by skeletal muscle-derived
exosomes via regulating miR-484/ACSL4 axis. Antioxid Redox Signal.
41:769–792. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang Y, Yang Y and Song Y:
Cardioprotective effects of exercise: The role of Irisin and
exosome. Curr Vasc Pharmacol. 22:316–334. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pegtel DM and Gould SJ: Exosomes. Annu Rev
Biochem. 88:487–514. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
El Safadi D, Mokhtari A, Krejbich M,
Lagrave A, Hirigoyen U, Lebeau G, Viranaicken W and Krejbich-Trotot
P: Exosome-mediated antigen delivery: Unveiling novel strategies in
viral infection control and vaccine design. Vaccines (Basel).
12:2802024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kumari M and Anji A: Small but
Mighty-exosomes, novel intercellular messengers in
neurodegeneration. Biology (Basel). 11:4132022.PubMed/NCBI
|
|
9
|
Malgundkar SH and Tamimi Y: Exosomes as
crucial emerging tools for intercellular communication with
therapeutic potential in ovarian cancer. Future Sci OA.
9:FSO8332023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jella KK, Nasti TH, Li Z, Malla SR,
Buchwald ZS and Khan MK: Exosomes, their biogenesis and role in
Inter-cellular communication, tumor microenvironment and cancer
immunotherapy. Vaccines (Basel). 6:4212018.
|
|
11
|
He J, Ren W, Wang W, Han W, Jiang L, Zhang
D and Guo M: Exosomal targeting and its potential clinical
application. Drug Deliv Transl Res. 12:2385–2402. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mrowczynski OD, Madhankumar AB, Sundstrom
JM, Zhao Y, Kawasawa YI, Slagle-Webb B, Mau C, Payne RA, Rizk EB,
Zacharia BE and Connor JR: Exosomes impact survival to radiation
exposure in cell line models of nervous system cancer. Oncotarget.
9:36083–36101. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhong Y, Li X, Wang F, Wang S, Wang X,
Tian X, Bai S, Miao D and Fan J: Emerging potential of exosomes on
adipogenic differentiation of mesenchymal stem cells. Front Cell
Dev Biol. 9:6495522021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang E, Wang X, Gong Z, Yu M, Wu H and
Zhang D: Exosome-mediated metabolic reprogramming: The emerging
role in tumor microenvironment remodeling and its influence on
cancer progression. Signal Transduct Target Ther. 5:2422020.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Feng X, Peng Z, Yuan L, Jin M, Hu H, Peng
X, Wang Y, Zhang C, Luo Z and Liao H: Research progress of exosomes
in pathogenesis, diagnosis, and treatment of ocular diseases. Front
Bioeng Biotechnol. 11:11003102023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rajput A, Varshney A, Bajaj R and
Pokharkar V: Exosomes as new generation vehicles for drug delivery:
Biomedical applications and future perspectives. Molecules.
27:72892022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jankovicova J, Secova P, Michalkova K and
Antalikova J: Tetraspanins, more than markers of extracellular
vesicles in reproduction. Int J Mol Sci. 21:75682020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Buzas EI, Toth EA, Sodar BW and
Szabo-Taylor KE: Molecular interactions at the surface of
extracellular vesicles. Semin Immunopathol. 40:453–464. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hu Q, Su H, Li J, Lyon C, Tang W, Wan M
and Hu TY: Clinical applications of exosome membrane proteins.
Precis Clin Med. 3:54–66. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gonda A, Kabagwira J, Senthil GN and Wall
NR: Internalization of exosomes through receptor-mediated
endocytosis. Mol Cancer Res. 17:337–347. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Catalanotto C, Cogoni C and Zardo G:
MicroRNA in control of gene expression: An overview of nuclear
functions. Int J Mol Sci. 17:17122016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Balchin C, Tan AL, Wilson OJ, McKenna J
and Stavropoulos-Kalinoglou A: The role of microRNAs in regulating
inflammation and exercise-induced adaptations in rheumatoid
arthritis. Rheumatol Adv Pract. 7:rkac1102023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yin X, Zhao Y, Zheng YL, Wang JZ, Li W, Lu
QJ, Huang QN, Zhang CY, Chen X and Ma JZ: Time-course responses of
muscle-specific MicroRNAs following acute uphill or downhill
exercise in sprague-dawley rats. Front Physiol. 10:12752019.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mytidou C, Koutsoulidou A, Katsioloudi A,
Prokopi M, Kapnisis K, Michailidou K, Anayiotos A and Phylactou LA:
Muscle-derived exosomes encapsulate myomiRs and are involved in
local skeletal muscle tissue communication. FASEB J. 35:e212792021.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Luo J, Pu Q and Wu X: Recent advances of
exosomes derived from skeletal muscle and crosstalk with other
tissues. Int J Mol Sci. 25:108772024. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nederveen JP, Warnier G, Di Carlo A,
Nilsson MI and Tarnopolsky MA: Extracellular vesicles and exosomes:
Insights from exercise science. Front Physiol. 11:6042742020.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Krylova SV and Feng D: The machinery of
exosomes: Biogenesis, release, and uptake. Int J Mol Sci.
24:13372023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Han QF, Li WJ, Hu KS, Gao J, Zhai WL, Yang
JH and Zhang SJ: Exosome biogenesis: Machinery, regulation, and
therapeutic implications in cancer. Mol Cancer. 21:2072022.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Estebanez B, Jimenez-Pavon D, Huang CJ,
Cuevas MJ and Gonzalez-Gallego J: Effects of exercise on exosome
release and cargo in in vivo and ex vivo models: A systematic
review. J Cell Physiol. 236:3336–3353. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Taylor J, Azimi I, Monteith G and Bebawy
M: Ca2+ mediates extracellular vesicle biogenesis
through alternate pathways in malignancy. J Extracell Vesicles.
9:17343262020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Messenger SW, Woo SS, Sun Z and Martin
TFJ: A Ca2+-stimulated exosome release pathway in cancer
cells is regulated by Munc13-4. J Cell Biol. 217:2877–2890. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Slusher AL, Visavadiya NP, Fico BG,
Estebanez B, Acevedo EO and Huang CJ: Impact of BMI and
cardiorespiratory fitness on oxidative stress in plasma and
circulating exosomes following acute exercise. Biology (Basel).
13:5992024.PubMed/NCBI
|
|
33
|
Castano C, Mirasierra M, Vallejo M,
Novials A and Parrizas M: Delivery of muscle-derived exosomal
miRNAs induced by HIIT improves insulin sensitivity through
down-regulation of hepatic FoxO1 in mice. Proc Natl Acad Sci USA.
117:30335–30343. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sigdel S, Chen S, Udoh G and Wang J:
Exercise-intervened circulating extracellular vesicles alleviate
oxidative stress in cerebral microvascular endothelial cells under
hypertensive plus hypoxic conditions. Antioxidants (Basel).
14:772025. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shen P, Qiu Y, Sun YY, Jiang YY, Guan XM,
Cheng M and Wang YX: The role of exercise in regulating the
generation of extracellular vesicles in cardiovascular diseases.
Rev Cardiovasc Med. 25:3922024. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Katayama M, Caria E, Van Simaeys D, Sanz
AY, Barrès R, Caidahl K, Wiklander OPB, El Andaloussi S, Berggren
PO, Zierath JR and Krook A: Exercise training-induced extracellular
miR-136-3p modulates glucose uptake and myogenesis through
targeting of NRDC in human skeletal muscle. J Sport Health Sci.
15:1010912025. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hou Z, Qin X, Hu Y, Zhang X, Li G, Wu J,
Li J, Sha J, Chen J, Xia J, et al: Longterm Exercise-derived
exosomal miR-342-5p: A novel exerkine for cardioprotection. Circ
Res. 124:1386–1400. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li H, Liu G, Wang B and Momeni MR:
Exosomes and microRNAs as mediators of the exercise. Eur J Med Res.
30:382025. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nair VD, Ge Y, Li S, Pincas H, Jain N,
Seenarine N, Amper MAS, Goodpaster BH, Walsh MJ, Coen PM, et al:
Sedentary and trained older men have distinct circulating exosomal
microRNA profiles at baseline and in response to acute exercise.
Front Physiol. 11:6052020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hu S, Hu Y and Yan W: Extracellular
vesicle-mediated interorgan communication in metabolic diseases.
Trends Endocrinol Metab. 34:571–582. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Aoi W and Tanimura Y: Roles of skeletal
Muscle-derived exosomes in organ metabolic and immunological
communication. Front Endocrinol (Lausanne). 12:6972042021.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kumar A, Sundaram K, Mu J, Dryden GW,
Sriwastva MK, Lei C, Zhang L, Qiu X, Xu F, Yan J, et al: High-fat
diet-induced upregulation of exosomal phosphatidylcholine
contributes to insulin resistance. Nat Commun. 12:2132021.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Li S, Wei J, Zhang C, Li X, Meng W, Mo X,
Zhang Q, Liu Q, Ren K, Du R, et al: Cell-derived microparticles in
patients with type 2 diabetes mellitus: A systematic review and
meta-analysis. Cell Physiol Biochem. 39:2439–2450. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Freeman DW, Noren Hooten N, Eitan E, Green
J, Mode NA, Bodogai M, Zhang Y, Lehrmann E, Zonderman AB, Biragyn
A, et al: Altered extracellular vesicle concentration, cargo, and
function in diabetes. Diabetes. 67:2377–2388. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gilbertson NM, Eichner NZM, Francois M,
Gaitán JM, Heiston EM, Weltman A and Malin SK: Glucose tolerance is
linked to postprandial fuel use independent of exercise dose. Med
Sci Sports Exerc. 50:2058–2066. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wang D, Zhang X, Li Y, Jia L, Zhai L, Wei
W, Zhang L, Jiang H and Bai Y: Exercise-induced browning of white
adipose tissue and improving skeletal muscle insulin sensitivity in
Obese/Non-obese growing mice: Do not neglect exosomal miR-27a.
Front Nutr. 9:9406732022. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Khalyfa A, Ericsson A, Qiao Z, Almendros
I, Farré R and Gozal D: Circulating exosomes and gut microbiome
induced insulin resistance in mice exposed to intermittent hypoxia:
Effects of physical activity. EBioMedicine. 64:1032082021.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lei LM, Lin X, Xu F, Shan SK, Guo B, Li
FX, Zheng MH, Wang Y, Xu QS and Yuan LQ: Exosomes and
Obesity-related insulin resistance. Front Cell Dev Biol.
9:6519962021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kawanishi N, Yano H, Yokogawa Y and Suzuki
K: Exercise training inhibits inflammation in adipose tissue via
both suppression of macrophage infiltration and acceleration of
phenotypic switching from M1 to M2 macrophages in
high-fat-diet-induced obese mice. Exerc Immunol Rev. 16:105–118.
2010.PubMed/NCBI
|
|
50
|
Guo W, Wang J, Chang Z, Lin S, Sha G, Wang
S, Huang J, Hu M and Xia J: Treadmill exercise ameliorates
atherogenesis and vascular inflammation in ApoE−/− mice
via circulating exosome-derived let-7c-5p. Sci Rep. 16:5852025.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ruan XF, Ju CW, Shen Y, Liu YT, Kim IM, Yu
H, Weintraub N, Wang XL and Tang Y: Suxiao Jiuxin pill promotes
exosome secretion from mouse cardiac mesenchymal stem cells in
vitro. Acta Pharmacol Sin. 39:569–578. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ma C, Wang J, Liu H, Chen Y, Ma X, Chen S,
Chen Y, Bihl JI and Yang YI: Moderate exercise enhances endothelial
progenitor cell exosomes release and function. Med Sci Sports
Exerc. 50:2024–2032. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Abdelsaid K, Sudhahar V, Harris RA, Das A,
Youn SW, Liu Y, McMenamin M, Hou Y, Fulton D, Hamrick MW, et al:
Exercise improves angiogenic function of circulating exosomes in
type 2 diabetes: Role of exosomal SOD3. FASEB J. 36:e221772022.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Raisch J, Darfeuille-Michaud A and Nguyen
HT: Role of microRNAs in the immune system, inflammation and
cancer. World J Gastroenterol. 19:2985–2996. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Jeker LT and Bluestone JA: MicroRNA
regulation of T-cell differentiation and function. Immunol Rev.
253:65–81. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Scalavino V, Liso M and Serino G: Role of
microRNAs in the regulation of dendritic cell generation and
function. Int J Mol Sci. 21:13192020. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Schell SL and Rahman ZSM: miRNA-mediated
control of B cell responses in immunity and SLE. Front Immunol.
12:6837102021. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Essandoh K, Li Y, Huo J and Fan GC:
MiRNA-mediated macrophage polarization and its potential role in
the regulation of inflammatory response. Shock. 46:122–131. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Pashangzadeh S, Motallebnezhad M,
Vafashoar F, Khalvandi A and Mojtabavi N: Implications the role of
miR-155 in the pathogenesis of autoimmune diseases. Front Immunol.
12:6693822021. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Fan W, Liang C, Ou M, Zou T, Sun F, Zhou H
and Cui L: MicroRNA-146a is a Wide-reaching neuroinflammatory
regulator and potential treatment target in neurological diseases.
Front Mol Neurosci. 13:902020. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhou C, Zhao L, Wang K, Qi Q, Wang M, Yang
L, Sun P and Mu H: MicroRNA-146a inhibits NF-κB activation and
pro-inflammatory cytokine production by regulating IRAK1 expression
in THP-1 cells. Exp Ther Med. 18:3078–3084. 2019.PubMed/NCBI
|
|
62
|
Luly FR, Leveque M, Licursi V, Cimino G,
Martin-Chouly C, Théret N, Negri R, Cavinato L, Ascenzioni F and
Del Porto P: MiR-146a is over-expressed and controls IL-6
production in cystic fibrosis macrophages. Sci Rep. 9:162592019.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ruan Q, Wang P, Wang T, Qi J, Wei M, Wang
S, Fan T, Johnson D, Wan X, Shi W, et al: MicroRNA-21 regulates
T-cell apoptosis by directly targeting the tumor suppressor gene
Tipe2. Cell Death Dis. 5:e10952014. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Wang S, Wan X and Ruan Q: The MicroRNA-21
in autoimmune diseases. Int J Mol Sci. 17:8642016. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Buscaglia LE and Li Y: Apoptosis and the
target genes of microRNA-21. Chin J Cancer. 30:371–380. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Sun J, Liu R, He X, Bian J, Zhao W, Shi W
and Ruan Q: MicroRNA-21 regulates diametrically opposed biological
functions of regulatory T cells. Front Immunol. 12:7667572021.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Kroesen BJ, Teteloshvili N,
Smigielska-Czepiel K, Brouwer E, Boots AM, van den Berg A and
Kluiver J: Immuno-miRs: Critical regulators of T-cell development,
function and ageing. Immunology. 144:1–10. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Ye Z, Li G, Kim C, Hu B, Jadhav RR, Weyand
CM and Goronzy JJ: Regulation of miR-181a expression in T cell
aging. Nat Commun. 9:30602018. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Shi M, Lu Q, Zhao Y, Ding Z, Yu S, Li J,
Ji M, Fan H and Hou S: miR-223: A key regulator of pulmonary
inflammation. Front Med (Lausanne). 10:11875572023. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zhao J, He Z and Wang J: MicroRNA-124: A
key player in microglia-mediated inflammation in neurological
diseases. Front Cell Neurosci. 15:7718982021. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Han D, Dong X, Zheng D and Nao J: MiR-124
and the underlying therapeutic promise of neurodegenerative
disorders. Front Pharmacol. 10:15552019. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Khan AA, Penny LA, Yuzefpolskiy Y, Sarkar
S and Kalia V: MicroRNA-17~92 regulates effector and memory CD8
T-cell fates by modulating proliferation in response to infections.
Blood. 121:4473–4483. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Mihailovich M, Bremang M, Spadotto V,
Musiani D, Vitale E, Varano G, Zambelli F, Mancuso FM, Cairns DA,
Pavesi G, et al: miR-17-92 fine-tunes MYC expression and function
to ensure optimal B cell lymphoma growth. Nat Commun. 6:87252015.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Lee HM, Kim TS and Jo EK: MiR-146 and
miR-125 in the regulation of innate immunity and inflammation. BMB
Rep. 49:311–318. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Hu YZ, Li Q, Wang PF, Li XP and Hu ZL:
Multiple functions and regulatory network of miR-150 in B
lymphocyte-related diseases. Front Oncol. 13:11408132023.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Li D, Wang A, Liu X, Meisgen F, Grünler J,
Botusan IR, Narayanan S, Erikci E, Li X, Blomqvist L, et al:
MicroRNA-132 enhances transition from inflammation to proliferation
during wound healing. J Clin Invest. 125:3008–3026. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Cui J, Zheng W, Sun Y and Xu T: Inducible
MicroRNA-132 inhibits the production of inflammatory cytokines by
targeting TRAF6, TAK1, and TAB1 in teleost fish. Infect Immun.
90:e00120222022. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Stelekati E, Cai Z, Manne S, Chen Z,
Beltra JC, Buchness LA, Leng X, Ristin S, Nzingha K, Ekshyyan V, et
al: MicroRNA-29a attenuates CD8 T cell exhaustion and induces
memory-like CD8 T cells during chronic infection. Proc Natl Acad
Sci USA. 119:e21060831192022. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Dey S, Udari LM, RiveraHernandez P, Kwon
JJ, Willis B, Easler JJ, Fogel EL, Pandol S and Kota J: Loss of
miR-29a/b1 promotes inflammation and fibrosis in acute
pancreatitis. JCI Insight. 6:e1495392021. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Baulina NM, Kulakova OG and Favorova OO:
MicroRNAs: The role in autoimmune inflammation. Acta Naturae.
8:21–33. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Maggio S, Canonico B, Ceccaroli P,
Polidori E, Cioccoloni A, Giacomelli L, Ferri Marini C, Annibalini
G, Gervasi M, Benelli P, et al: Modulation of the circulating
extracellular vesicles in response to different exercise regimens
and study of their inflammatory effects. Int J Mol Sci.
24:30392023. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Orlandella FM, De Stefano AE, Iervolino
PLC, Buono P, Soricelli A and Salvatore G: Dissecting the molecular
pathways involved in the effects of physical activity on breast
cancers cells: A narrative review. Life Sci. 265:1187902021.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Garai K, Adam Z, Herczeg R, Banfai K,
Gyebrovszki A, Gyenesei A, Pongracz JE, Wilhelm M and Kvell K:
Physical activity as a preventive lifestyle intervention acts
through specific exosomal miRNA Species-evidence from human Short-
and Long-term pilot studies. Front Physiol. 12:6582182021.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Huo L, Du X, Li X, Liu S and Xu Y: The
emerging role of neural Cell-derived exosomes in intercellular
communication in health and neurodegenerative diseases. Front
Neurosci. 15:7384422021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Gayen M, Bhomia M, Balakathiresan N and
Knollmann-Ritschel B: Exosomal MicroRNAs released by activated
astrocytes as potential neuroinflammatory biomarkers. Int J Mol
Sci. 21:23122020. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
He XF, Liu DX, Zhang Q, Liang FY, Dai GY,
Zeng JS, Pei Z, Xu GQ and Lan Y: Voluntary exercise promotes
glymphatic clearance of amyloid beta and reduces the activation of
astrocytes and microglia in aged mice. Front Mol Neurosci.
10:1442017. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Liang X, Fa W, Wang N, Peng Y, Liu C, Zhu
M, Tian N, Wang Y, Han X, Qiu C, et al: Exosomal miR-532-5p induced
by long-term exercise rescues blood-brain barrier function in 5XFAD
mice via downregulation of EPHA4. Aging Cell. 22:e137482023.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Vechetti IJ, Norrbom J, Alkner B,
Hjalmarsson E, Palmcrantz A, Pontén E, Pingel J, von Walden F and
Fernandez-Gonzalo R: Extracellular vesicle characteristics and
microRNA content in cerebral palsy and typically developed
individuals at rest and in response to aerobic exercise. Front
Physiol. 13:10720402022. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Heidarzadeh M, Gürsoy-Özdemir Y, Kaya M,
Eslami Abriz A, Zarebkohan A, Rahbarghazi R and Sokullu E: Exosomal
delivery of therapeutic modulators through the blood-brain barrier;
promise and pitfalls. Cell Biosci. 11:1422021. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Ramos-Zaldívar HM, Polakovicova I,
Salas-Huenuleo E, Corvalán AH, Kogan MJ, Yefi CP and Andia ME:
Extracellular vesicles through the blood-brain barrier: A review.
Fluids Barriers CNS. 19:602022. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li C, Qin S, Wen Y, Zhao W, Huang Y and
Liu J: Overcoming the blood-brain barrier: Exosomes as theranostic
nanocarriers for precision neuroimaging. J Control Release.
349:902–916. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Li C, Ke C, Su Y and Wan C: Exercise
intervention promotes the growth of synapses and regulates
neuroplasticity in rats with ischemic stroke through exosomes.
Front Neurol. 12:7525952021. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Yoon KJ, Park S, Kwak SH and Moon HY:
Effects of voluntary running wheel Exercise-induced extracellular
vesicles on anxiety. Front Mol Neurosci. 14:6658002021. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Liu W, Bai X, Zhang A, Huang J, Xu S and
Zhang J: Role of exosomes in central nervous system diseases. Front
Mol Neurosci. 12:2402019. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Yuan P, Ding L, Chen H, Wang Y, Li C, Zhao
S, Yang X, Ma Y, Zhu J, Qi X, et al: Neural stem cell-derived
exosomes regulate neural stem cell differentiation through
miR-9-Hes1 axis. Front Cell Dev Biol. 9:6016002021. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Ma Y, Li C, Huang Y, Wang Y, Xia X and
Zheng JC: Exosomes released from neural progenitor cells and
induced neural progenitor cells regulate neurogenesis through
miR-21a. Cell Commun Signal. 17:962019. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Hwang J, Jang S, Kim C, Lee S and Jeong
HS: Role of stem Cell-derived exosomes and microRNAs in spinal cord
injury. Int J Mol Sci. 24:138492023. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Li C, Hu J, Liu W, Ke C, Huang C, Bai Y,
Pan B, Wang J and Wan C: Exercise intervention modulates synaptic
plasticity by inhibiting excessive microglial activation via
exosomes. Front Cell Neurosci. 16:9536402022. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Jiang XH, Li HF, Chen ML, Zhang YX, Chen
HB, Chen RH, Xiao YC and Liu N: Treadmill exercise exerts a
synergistic effect with bone marrow mesenchymal stem cell-derived
exosomes on neuronal apoptosis and synaptic-axonal remodeling.
Neural Regen Res. 18:1293–1299. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Slota JA and Booth SA: MicroRNAs in
neuroinflammation: Implications in disease pathogenesis, biomarker
discovery and therapeutic applications. Noncoding RNA.
5:352019.PubMed/NCBI
|
|
101
|
Catitti G, De Bellis D, Vespa S, Simeone
P, Canonico B and Lanuti P: Extracellular vesicles as players in
the Anti-inflammatory inter-cellular crosstalk induced by exercise
training. Int J Mol Sci. 23:140982022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Li LD, Naveed M, Du ZW, Ding H, Gu K, Wei
LL, Zhou YP, Meng F, Wang C, Han F, et al: Abnormal expression
profile of plasma-derived exosomal microRNAs in patients with
treatment-resistant depression. Hum Genomics. 15:552021. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Sim SE, Lim CS, Kim JI, Seo D, Chun H, Yu
NK, Lee J, Kang SJ, Ko HG, Choi JH, et al: The Brain-enriched
MicroRNA miR-9-3p regulates synaptic plasticity and memory. J
Neurosci. 36:8641–8652. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Makeyev EV, Zhang J, Carrasco MA and
Maniatis T: The MicroRNA miR-124 promotes neuronal differentiation
by triggering brain-specific alternative pre-mRNA splicing. Mol
Cell. 27:435–448. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Qian Y, Song J, Ouyang Y, Han Q, Chen W,
Zhao X, Xie Y, Chen Y, Yuan W and Fan C: Advances in roles of
miR-132 in the nervous system. Front Pharmacol. 8:7702017.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Xia X, Teotia P and Ahmad I: miR-29c
regulates neurogliogenesis in the mammalian retina through REST.
Dev Biol. 450:90–100. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Rashidi SK, Kalirad A, Rafie S, Behzad E
and Dezfouli MA: The role of microRNAs in neurobiology and
pathophysiology of the hippocampus. Front Mol Neurosci.
16:12264132023. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Elramah S, Landry M and Favereaux A:
MicroRNAs regulate neuronal plasticity and are involved in pain
mechanisms. Front Cell Neurosci. 8:312014. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Li Y, Lu X, Nie J, Hu P, Ge F, Yuan TF and
Guan X: MicroRNA134 of ventral hippocampus is involved in cocaine
extinction-induced Anxiety-like and Depression-like behaviors in
mice. Mol Ther Nucleic Acids. 19:937–950. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Abuelezz NZ, Nasr FE, AbdulKader MA,
Bassiouny AR and Zaky A: MicroRNAs as Potential orchestrators of
Alzheimer's disease-related pathologies: Insights on current status
and future possibilities. Front Aging Neurosci. 13:7435732021.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Bartel DP: Metazoan MicroRNAs. Cell.
173:20–51. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Hawley ZCE, Campos-Melo D, Droppelmann CA
and Strong MJ: MotomiRs: miRNAs in Motor neuron function and
disease. Front Mol Neurosci. 10:1272017. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Jia J, Wang L, Zhou Y, Zhang P and Chen X:
Muscle-derived extracellular vesicles mediate crosstalk between
skeletal muscle and other organs. Front Physiol. 15:15019572024.
View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Wang Z, Yang J, Sun X, Sun X, Yang G and
Shi X: Exosome-mediated regulatory mechanisms in skeletal muscle: A
narrative review. J Zhejiang Univ Sci B. 24:1–14. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Castano C, Kalko S, Novials A and Parrizas
M: Obesity-associated exosomal miRNAs modulate glucose and lipid
metabolism in mice. Proc Natl Acad Sci USA. 115:12158–12163. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Liu J, Zhang Y, Tian Y, Huang W, Tong N
and Fu X: Integrative biology of extracellular vesicles in diabetes
mellitus and diabetic complications. Theranostics. 12:1342–1372.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Zhou Y and Tan C: miRNAs in
Adipocyte-derived extracellular vesicles: Multiple roles in
development of obesity-associated disease. Front Mol Biosci.
7:1712020. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Hernandez-Gomez KG, Avila-Nava A,
Gonzalez-Salazar LE, Noriega LG, Serralde-Zúñiga AE, Guizar-Heredia
R, Medina-Vera I, Gutiérrez-Solis AL, Torres N, Tovar AR and
Guevara-Cruz M: Modulation of MicroRNAs and exosomal MicroRNAs
after dietary interventions for obesity and insulin resistance: A
narrative review. Metabolites. 13:11902023. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Ke S, Cao X, Lu X, Xu J, Zhang CY, Xu L
and Jiang X: Atheroprotective roles of exercise-regulated
microRNAs. Atherosclerosis. 405:1192292025. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Lark DS: The rise and fall of circulating
miRNAs in response to exercise. Acta Physiol (Oxf). 236:e138972022.
View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Zhou S, Cheing GLY and Cheung AKK: Role of
exosomes and exosomal microRNA in muscle-Kidney crosstalk in
chronic kidney disease. Front Cell Dev Biol. 10:9518372022.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Lyu C, Sun H, Sun Z, Liu Y and Wang Q:
Roles of exosomes in immunotherapy for solid cancers. Cell Death
Dis. 15:1062024. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Darragh IAJ, O'Driscoll L and Egan B:
Exercise training and circulating small extracellular vesicles:
Appraisal of methodological approaches and current knowledge. Front
Physiol. 12:7383332021. View Article : Google Scholar : PubMed/NCBI
|
















