Introduction
Natural products have historically served as a
notable reservoir for drug discovery, with plant-derived alkaloids
representing one of the most successful classes of therapeutic
agents (1). Nelumbo
nucifera Gaertn., also known as sacred lotus, has served as a
fundamental component in traditional Chinese medicine for >2,000
years to treat various disorders, including insomnia, hematuria and
chronic dyspepsia (2). The
medicinal properties of this plant are largely due to its diverse
array of bioactive alkaloids, with nuciferine [NF;
C19H21NO2; Chemical Abstracts
Service (CAS) no. 475-83-2] being a particularly notable
aporphine-type compound that has shown promise in recent research
(3).
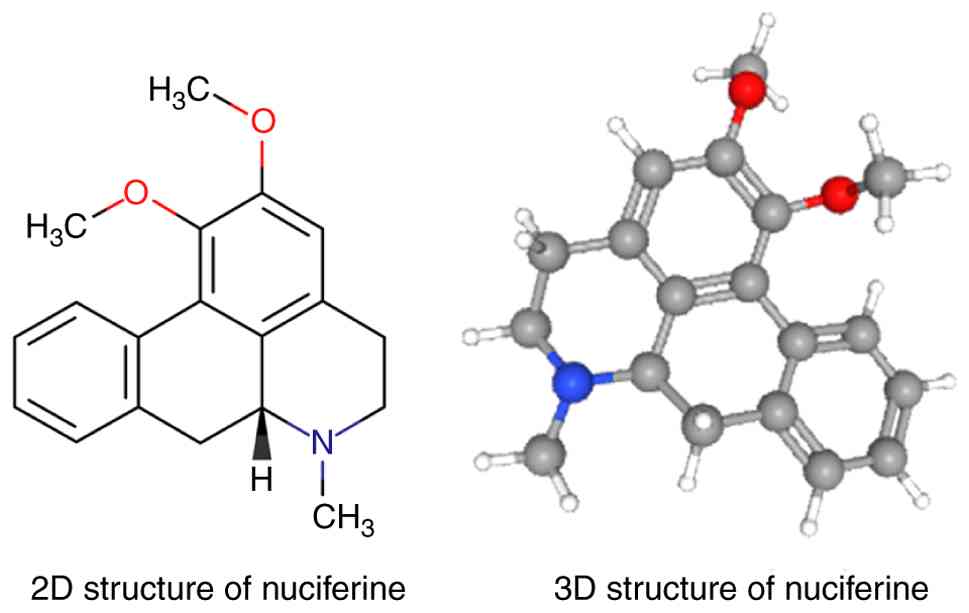
NF (Fig. 1), also
known as Sanjoinine E, VLT049 and (−)-NF, is a natural aporphine
alkaloid widely used in traditional Chinese medicine. While
Nelumbo nucifera Gaertn. remains the most extensively
studied source of NF, this bioactive compound has also been
detected in multiple other plant species, including Annona
crassiflora Mart (4),
Michelia champaca L (5),
Onychopetalum amazonicum R.E.Fr (6) and Jujubae Fructus (https://www.tcmsp-e.com/molecule.php?qn=7213).
Initially recognized for its sedative and anxiolytic properties,
which align with its traditional medicinal uses, NF has recently
garnered notable attention due to its diverse pharmacological
effects (7). Over the past decade,
preclinical research has uncovered its ability to modulate multiple
critical pathways involved in various diseases (3,8).
Accumulating evidence has demonstrated that NF modulates key
pathophysiological pathways implicated in inflammatory diseases
[including the Toll-like receptor 4 (TLR4)/NF-κB and NOD-like
receptor family pyrin domain containing 3 (NLRP3) pathways]
(9,10), neurodegenerative conditions (such
as the JAK2/STAT3 pathway) (11),
musculoskeletal degeneration (such as the MAPK/NF-κB pathway)
(12), cardiovascular dysfunction
[including the PI3K/Akt and nitric oxide (NO)/cGMP pathway]
(13), metabolic disorders [such
as the AMP-activated protein kinase (AMPK) and peroxisome
proliferator-activated receptor (PPAR)α pathways] (14,15)
and oncogenesis (for example, the PI3K/Akt, Wnt/β-catenin and STAT3
pathways) (16–18). Furthermore, NF may combat obesity
by modulating metabolic pathways and reducing fat accumulation
(15). NF also effectively reduces
lipid concentrations by inhibiting cholesterol synthesis and
cholesterol esterase activity (19). Additionally, NF exhibits
antidiabetic potential, regulating blood glucose levels and
enhancing insulin sensitivity (20). The anti-inflammatory effects of NF
are primarily mediated through its ability to regulate oxidative
stress, metabolic signaling and gut microbiota (21). The neuroprotective properties of NF
stem from its interactions with neurotransmitter receptors such as
5-HT2A, 5-HT2C, 5-HT2B and
D2, which in turn modulate neurosignaling transduction
(22). Furthermore, NF has been
shown to suppress the proliferation and migration of melanoma
cells, suggesting its potential as an antitumor agent (16). This expanding body of research
highlights the multifaceted therapeutic potential of NF across a
wide range of health conditions.
Despite this expanding body of evidence, the
literature on NF remains fragmented and lacks synthesis, presenting
several major challenges. First, the interactions of NF with
multiple receptors, such as dopaminergic, serotonergic and
adrenergic systems, and signaling pathways are well-documented;
however, the relative importance of these targets in different
disease contexts remains unclear. A number of studies have focused
on isolated mechanisms without exploring how these pathways
interact or integrate. Second, the clinical potential of NF is
limited due to its low water solubility and modest oral
bioavailability (~58% in rodents), as shown by Ye et al
(23). Although researchers have
tested new delivery systems (24–26),
there is no thorough evaluation of their effectiveness or
scalability for broader use. Third, safety concerns arise from
NF-mediated inhibition of key cytochrome P450 (CYP) enzymes,
including CYP2D6 (27) and CYP1A2
(28), which could lead to drug
interactions, yet comprehensive toxicity studies and human
pharmacokinetic data are still missing. Finally, while
computational models have predicted numerous potential protein
targets for NF (29–31), experimental validation is limited,
leaving uncertainties about its true binding affinities and
functional importance in biological systems.
The present review therefore offers a comprehensive
analysis of NF, exploring its diverse biological activities and
underlying molecular mechanisms across seven key therapeutic areas.
The safety profile of NF is assessed by examining toxicity data and
pharmacokinetic challenges while also evaluating innovative drug
delivery approaches such as liposome formulations, poly
lactic-co-glycolic acid (PLGA) nanoparticles and hydrogel scaffold
systems. The discussion integrates computational docking
predictions with existing experimental evidence, identifying areas
where further validation is needed. Synthesizing these multifaceted
aspects reveals the promising potential of NF as a novel
therapeutic candidate for various medical conditions, facilitating
the integration of traditional knowledge with modern pharmaceutical
development.
Chemistry of NF
NF, also known as Sanjoinine E, is a natural
alkaloid metabolite predominantly isolated from the leaves of the
sacred lotus plant, Nelumbo nucifera Gaertn (3,8).
This metabolite is characterized by the molecular formula
C19H21NO2 and possesses a molar
mass of 295.4 g/mol (3). It is a
colorless liquid with a pungent odor, and features a tetracyclic
core, one nitrogen atom and a chiral center. The CAS registry
number of NF is 475-83-2 (http://www.badd-cao.net:2345/browse/compound/C0586).
The International Union of Pure and Applied Chemistry name of NF is
(6aR)-1,2-dimethoxy-6-methyl-5,6,6a,7-tetrahydro-4H-dibenzo(de,g)quinoline
(http://47.92.70.12/Detail/?v=HBIN037568&label=Ingredient).
NF is notable for its high solubility in low-polarity organic
solvents, such as benzene, ether, chloroform and halogenated
alkanes. It also dissolves well in hydrophilic organic solvents,
such as acetone and ethanol. However, its solubility in water is
low or almost negligible, which can limit its bioavailability. NF
exhibits a characteristic melting point of 168°C. Fig. 1 presents both the 2D and 3D
structures of NF, which can be accessed via the PubChem database
(https://pubchem.ncbi.nlm.nih.gov/compound/10146).
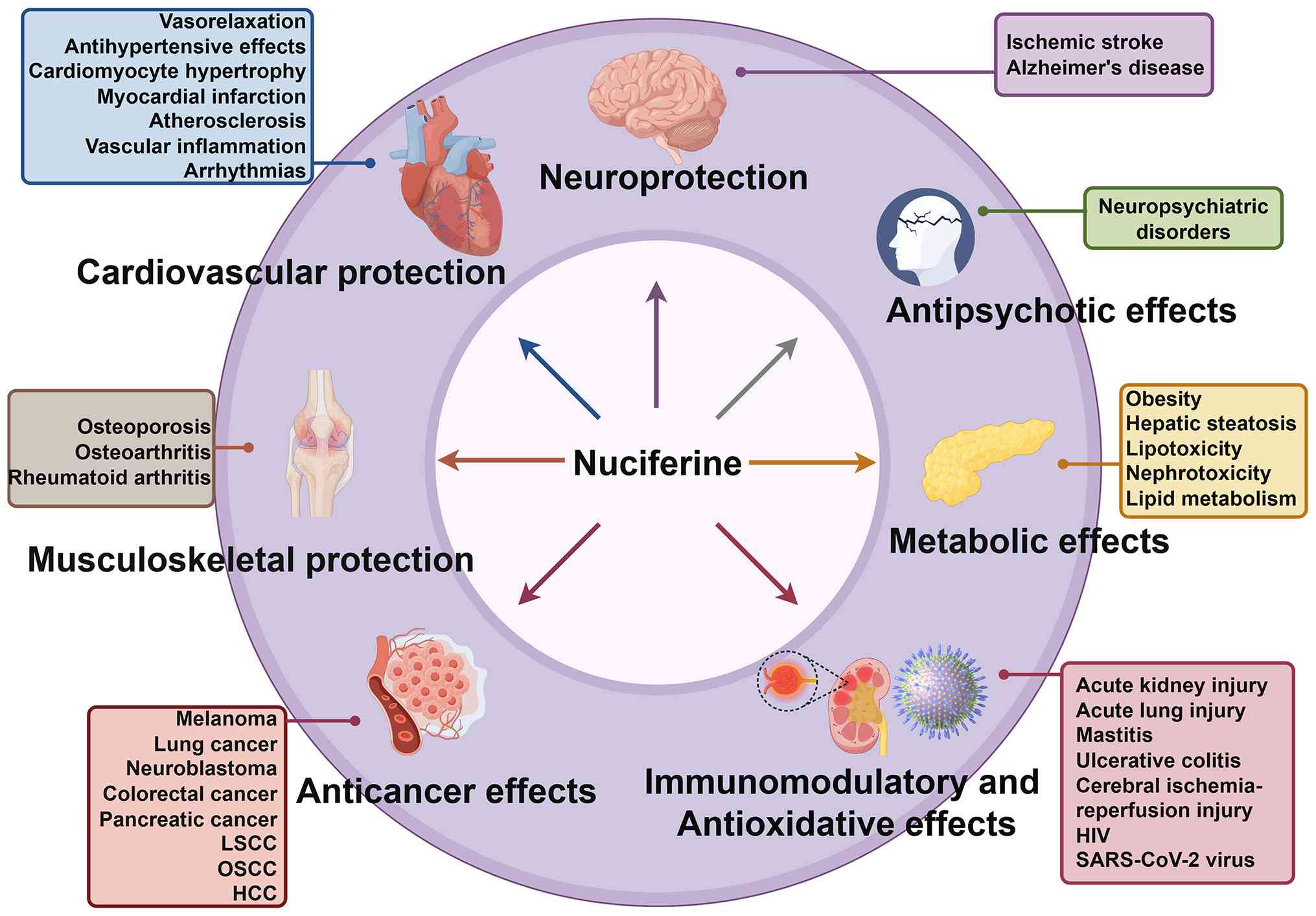
Multifunctional biological activities of
NF
As a natural alkaloid metabolite, NF has garnered
considerable attention for its diverse pharmacological effects,
which encompass immunomodulatory and antioxidative effects,
neuroprotection, osteogenic activity, cardiovascular protection,
metabolic regulation, antipsychotic and anticancer activities. The
diverse biological activities of NF are comprehensively summarized
in Table SI and illustrated in
Fig. 2.
Immunomodulatory and antioxidative
effects
Inflammation represents a fundamental protective
response that defends the human body from various infections agents
(32). Nevertheless, chronic
inflammation processes are frequently implicated in the
pathogenesis of several inflammatory disorders, including acute
kidney injury (33,34), acute lung injury (10), mastitis (35), ulcerative colitis (9,36)
and cerebral ischemia-reperfusion injury (37). Given the key role of inflammation
in disease pathology, NF has demonstrated considerable potential in
therapeutics as an anti-inflammatory agent. In fructose-fed rats,
NF mitigates renal injury by alleviating inflammatory responses via
suppressing the TLR4/PI3K/NF-κB pathway and activation of the NLRP3
inflammasome (33). Similarly, in
lipopolysaccharide (LPS)-induced acute lung injury, NF demonstrates
anti-inflammatory effects by suppressing the TLR4-mediated pathway,
thereby reducing inflammation and tissue damage (10). NF has also been shown to exhibit
potential in mitigating LPS-induced mastitis in mice through
suppression of the TLR4/NF-κB signaling pathway (35). In addition, in acute kidney injury
models, it protects against folic acid-induced damage by inhibiting
ferroptosis (34). Furthermore, NF
attenuates inflammatory responses in LPS-stimulated macrophages and
microglial cells via activation of PPARγ (38) and inhibition of p38 MAPK/activating
transcription factor 2 signaling pathways (39). In an ulcerative colitis model, NF
has been shown to modulate immune function and intestinal microbial
composition, restoring the balance of T-cell subsets and improving
colonic health (9). NF also
suppresses MAPK/NF-κB and NLRP3/caspase 1 pathways, reducing
cytokines and improving the disease activity index (DAI) and
histological alternations in colitic mice (36). In cerebral ischemia-reperfusion
injury, NF exerts its protective effects through modulation of
actions via the PI3K/Akt/NF-κB pathway, reducing inflammation and
improving cognitive and motor functions (37). These findings underscore the
potential of NF as a therapeutic agent for various inflammatory
diseases by modulating key signaling pathways and immune
responses.
In addition to its immunomodulatory effects, NF has
been reported to exhibit antiviral activity (40,41).
For example, in a comprehensive structure-activity relationship
investigation, Kashiwada et al (40) reported that NF exhibited potent
anti-HIV efficacy, with an EC50 of 0.8 µg/ml and a
favorable therapeutic window (therapeutic index, 36.3). NF has also
been demonstrated to possess both direct virucidal and antiviral
activities against the SARS-CoV-2 virus, with a virucidal
EC50 of 13.24 µM, and an antiviral EC50 for
inhibiting viral infection of 30.59 µM (41). These findings highlight the
potential of NF in combating viral infections. Oxidative stress,
defined by an overabundance of reactive oxygen species (ROS) and
reactive nitrogen species, serves a substantial role in the aging
process and the development of various diseases (42–44).
The antioxidant properties of NF have also been extensively
studied. NF has been shown to mitigate hydrogen peroxide
(H2O2)-induced oxidative stress in
cardiomyocytes and fibroblasts, demonstrating cardioprotective and
anti-aging properties by downregulating pro-inflammatory cytokines
and maintaining cellular homeostasis (45,46).
In a mouse model, NF has been shown to improve the survival rate of
random skin flaps through multiple functions (including promoting
angiogenesis, reducing oxidative stress, inflammation and
apoptosis), which effectively promotes vascular formation,
mitigates oxidative damage and stimulates autophagy by mediating
transcription factor EB (TFEB) nuclear translocation via the
AMPK/mTOR signaling pathway (47).
Nuciferine has also been reported to have applications in
nutraceuticals as a gummy bear supplement with antioxidant and
antiproliferative potential, by reducing endogenous ROS levels
under oxidative stress (48). In
summary, NF exhibits both immunomodulatory and antioxidant
properties, making it a potential therapeutic agent for managing
diseases associated with inflammation and oxidative stress.
Regulation of metabolism by NF
Emerging evidence has highlighted NF as a potential
therapeutic agent for the management of diverse metabolic
disorders, including obesity, hepatic steatosis, lipotoxicity,
nephrotoxicity and hyperuricemia. Its diverse mechanisms of action
involve the activation of key signaling pathways such as AMPK
(15), PPARα (49) and TFEB (50) pathways, as well as the regulation
of intestinal microbial composition and bile acid (BA) metabolism.
These effects collectively contribute to its benefits in metabolic
health.
NF has demonstrated a range of pharmacological
properties, including the ability to stimulate insulin secretion in
pancreatic β cells, which may offer a new therapeutic option for
diabetes management (51).
Additionally, NF has shown the anti-hyperuricemic and
anti-inflammatory effects in reducing serum urate levels and
interleukin-1β (IL-1β) secretion in mice, suggesting its potential
use in treating hyperuricemia with kidney inflammation (52). Metabolism studies have elucidated
various phase I and II metabolites of NF in rats and mice,
revealing key metabolic pathways such as oxidation, demethylation,
glucuronidation and sulfation (53–55).
Furthermore, the interaction of NF with metformin, a common
antidiabetic drug, has been explored, indicating that NF may
attenuate hepatic metformin accumulation, potentially affecting the
glucose-lowering effect of metformin (56). Metabolomics and gut microbiota
analyses have also elucidated the glycine, serine and threonine
metabolism regulatory mechanisms of NF in hyperuricemia,
highlighting its impact on metabolic pathways and gut microbiota
(57).
NF has shown promising effects in managing obesity
and hepatic steatosis. In high-fat diet (HFD)-fed mice, NF has
demonstrated notable efficacy in attenuating body weight, fat
accumulation and improving glycolipid profiles by activating the
AMPK-mediated fatty acid synthase/hormone sensitive lipase pathway,
promoting fatty acid oxidation in the liver and thermogenesis in
white adipose tissue (58).
Another study has demonstrated that NF ameliorates glucose
intolerance, diminishes lipid accumulation and enhances glycogen
storage in hepatocytes of C57BL/6J mice with HFD-induced
gestational diabetes mellitus. Additionally, it may restore
intestinal microbial balance by promoting the abundance of
beneficial genera, including Akkermansia, Lactobacillus and
Bifidobacterium, and suppressing pathogenic bacteria, such
as Staphylococcus and Escherichia-Shigella (59).
NF may also protect against non-alcoholic fatty
liver disease (NAFLD) by activating the TFEB-mediated
autophagy-lysosomal pathway through direct interaction with the
regulatory subunit hepatitis B virus X-interacting protein (HBXIP)
and inhibiting mTORC1 activity, thereby reducing hepatic lipid
accumulation and insulin resistance (60). In HFD-fed rats, NF has been
reported to modulate BA metabolism and gut microbiota, altering the
composition of colonic BA, promoting BA synthesis and excretion,
and inhibiting BA reabsorption. It also reduces the abundance of
bile salt hydrolase-producing and 7α-dehydroxylation bacteria,
leading to increased conjugated BAs, which contributes to its
protective effects against HFD-induced hepatic steatosis (61).
NF has been shown to mitigate non-esterified fatty
acid-induced lipotoxicity in bovine mammary epithelial cells by
activating the LKB1/AMPK signaling pathway, which is a key pathway
for lipid metabolism and energy homeostasis (62). In another study, NF has been
reported to block MIB2-mediated caspase recruitment domain protein
family member 6 polyubiquitination and degradation, thereby
reducing high fructose-induced lipid accumulation in hepatocytes,
highlighting its potential in preventing fatty liver disease
(63). Furthermore, NF alleviates
hepatic steatosis and ferroptosis in NAFLD mice by modulating the
PPARα signaling pathway, suggesting its therapeutic potential for
NAFLD (49).
NF protects against obesity-induced nephrotoxicity
by reducing lipid accumulation, inflammation and oxidative stress
in kidneys and renal cells through the AMPK pathway (15). Additionally, NF may prevent obesity
by activating brown adipose tissue through the TFEB/PPARλ
coactivator 1-α (PGC-1α) pathway in excessive free fatty
acid-induced oxidative injury model, enhancing energy expenditure
and improving metabolic health (50,64).
NF has been extensively studied for its potential
health benefits, particularly in the context of lipid metabolism
and liver health (19–21,65–70).
NF can markedly reduce body weight gain, liver and visceral fat
weight, and improve serum and hepatic lipid profiles in HFD-induced
hamster models (65). In
vitro studies using L02 and HepG2 cells have demonstrated that
NF reduces lipid accumulation and oxidative stress by activating
the AMPK and PPARα pathways, suggesting its potential therapeutic
applications for NAFLD (58,66).
Further mechanistic studies have revealed that NF downregulates
Per-Arnt-Sim kinase expression, which is associated with reduced
lipogenesis and inflammation in HepG2 cells (67). Additionally, NF has been shown to
inhibit lipogenesis and promote glucose uptake in insulin-resistant
3T3-L1 adipocytes by activating the AMPK signaling pathway
(20). In a murine model of type 2
diabetes mellitus induced by HFD and streptozocin (STZ), NF has
been shown to improve glucose tolerance, insulin resistance and
hepatic steatosis through the PPARα/PGC-1α pathway (68). Similar protective effects have been
observed in STZ-induced diabetic rats, where NF down-regulated
lipogenesis associated genes (e.g. ACC2, PPAR-α and CPT-1α) and
up-regulated lipolysis and fatty acid β-oxidation associated genes
(e.g. SREBP-1c, LXR-a, ACC1, SCD-1, FAS, and DGAT-2) to reduce
steatosis and liver injury (69).
In broiler chickens, dietary NF reduces fat deposition by
controlling triglyceride and cholesterol concentrations,
highlighting its potential in animal nutrition (19). Furthermore, the anti-hyperlipidemic
effects of NF have been associated with the liver
steatosis-alleviating and anti-hyperglycemic abilities to modulate
gut microbiota, particularly by enriching Akkermansia
muciniphila (21). Finally, in
3T3-L1 preadipocytes, NF can inhibit cell proliferation and lipid
accumulation by downregulating key lipogenic genes and promoting
the expression of adipokines, such as FGF21 and ZAG, suggesting its
potential as a therapeutic agent for obesity (70). In summary, the benefits of
modulating lipid metabolism, and potential to improve liver health
and enhance metabolic health, highlight the potential of NF as a
novel therapeutic agent for the management of various metabolic
disorders.
Neuroprotective activity of NF
The neuroprotective effects of NF are displayed in
its potential to combat various neurological disorders, including
ischemic stroke (71,72) and Alzheimer's disease (73). Its considerable brain penetration
and potential as a neuropharmacological agent highlight its
therapeutic potential.
Wu et al (72) elucidated the anti-ischemic stroke
mechanism of NF in a rat model of middle cerebral artery occlusion
(MCAO) by utilizing serum metabolomics. NF markedly improved
neurological deficit scores (according to the Longa scoring
system), reduced cerebral infarction and edema and modulated the
metabolic pathways involved in energy metabolism, inflammation and
lipid metabolism (72). Metabolic
changes were analyzed using 1H nuclear magnetic
resonance spectroscopy, revealing 19 metabolites and nine key
metabolic pathways involved in the therapeutic effects of NF,
including amino acid metabolism, fatty acid metabolism and
antioxidant pathways (11). Chen
et al (74) further
revealed that NF markedly alleviated neurological deficits, reduced
infarct volume and regulated lipid metabolism pathways such as
arachidonic acid metabolism, sphingolipid metabolism and the PPAR
signaling pathway on MCAO-induced brain injury in rats through
transcriptomic analysis. Li et al (71) demonstrated that NF maintained
blood-brain barrier (BBB) integrity in ischemic stroke through
suppression of the JAK2/STAT3 signaling pathway, thereby exhibiting
neuroprotective properties. Mechanistically, NF was shown to
protect the BBB structure via dual mechanisms: Attenuating
endothelial cell apoptosis and mitochondrial impairment while
simultaneously restoring tight junction protein balance (71). The findings of this previous study
provide experimental evidence supporting the therapeutic efficacy
of NF in improving energy metabolism and protecting neurons in
ischemic stroke, thereby suggesting a pharmacological foundation
for potential application in ischemic neurological disorders.
Khan et al (73) explored the anti-Alzheimer's disease
effect of NF in alloxan-induced diabetic rats. NF markedly reduced
blood glucose levels, improved body weight and enhanced antioxidant
enzyme activities. It also inhibited lipid peroxidation and
restored acetylcholinesterase activity in the brain, indicating
potential therapeutic applications for diabetes and Alzheimer's
disease. Ye et al (23)
investigated the pharmacokinetics of NF and its associated
metabolite N-nornuciferine in rats, revealing their rapid
absorption and notable brain penetration. NF exhibited an oral
bioavailability of 58.13%, whereas N-nornuciferine had an oral
bioavailability of 79.91%. Both metabolites were found to have a
relatively wide volume of distribution and slow elimination
half-lives, suggesting their potential as neuropharmacological
agents. In summary, the neuroprotective effects of NF are
attributed to its ability to modulate multiple pathways and
cellular processes. However, further research is required to
elucidate its detailed mechanisms of action and to translate these
findings into clinical applications.
Musculoskeletal protective effects of
NF
Musculoskeletal diseases impose a considerable
economic burden on individuals and social-care systems,
encompassing conditions such as osteoporosis, osteoarthritis (OA),
rheumatoid arthritis (RA), gout, lower back pain and psoriatic
arthritis (75). Previous studies
have extensively explored the therapeutic potential of NF in
osteoporosis (76), OA (77,78)
and RA (79,80), revealing its multifaceted benefits
in these conditions.
NF has demonstrated notable promise in preventing
bone loss in osteoporosis. In ovariectomized mice, NF has been
reported to disrupt the maturation of multinucleated osteoclasts
and enhance type H vessel formation through the inhibition of MAPK
and NF-κB signaling pathways, both in in vitro and in
vivo models (76).
Additionally, NF suppresses glycolysis and ROS production by
inhibiting the expression of glycolysis-related genes (such as
hexokinase 2, pyruvate kinase M2 and lactate dehydrogenase A) and
NADPH oxidase 1, while enhancing antioxidant enzyme expression
during osteoclastogenesis (12).
These mechanisms collectively contribute to the prevention of bone
loss and the maintenance of bone health.
NF alleviates OA progression by inhibiting the
production of inflammatory cytokines and targeting the
PI3K/Akt/NF-κB signaling pathway. This results in reduced
degradation of the extracellular matrix, decreased joint swelling
and cartilage protection (77).
Peng et al (78) further
revealed that NF markedly reduced the production of inflammatory
mediators, such as inducible NO synthase, prostaglandin E2 and IL-6
in IL-1β-induced chondrocytes through the PTEN/Akt/NF-κB signaling
pathway. By modulating these pathways, NF effectively mitigated
cartilage degradation and joint inflammation in OA.
In RA, NF has shown efficacy in reducing joint
swelling, synovial hyperplasia, cartilage damage and inflammatory
infiltration in rats by inhibiting the proliferation and invasion
of fibroblast-like synoviocytes and rectifying the T-helper 17/T
regulatory imbalance in collagen-induced arthritis (79). Kulhari et al (80) also demonstrated that NF treatment
markedly reduced joint swelling, inflammatory cytokines (TNF-α and
IL-1β) and oxidative stress markers (including malondialdehyde,
superoxide dismutase and glutathione) in the paw tissue by
inhibiting the TLR4/NF-κB/MAPK signaling axis in complete Freund's
adjuvant (CFA)-induced arthritic rats. These findings highlight the
ability of NF to modulate key inflammatory and immune pathways
involved in RA. In conclusion, the advantages of NF in
osteoporosis, OA and RA suggest its potential as a novel
therapeutic agent for inflammation and bone related diseases.
Cardiovascular protective effects of
NF
NF has emerged as a promising therapeutic agent for
a wide range of cardiovascular conditions, demonstrating
multifaceted benefits such as inducing vasorelaxation (81), antihypertensive effects (82), reducing cardiomyocyte hypertrophy
(83), mitigating cardiotoxicity
(84), protecting against
myocardial infarction (13,85,86),
regulating atherosclerosis (87),
alleviating vascular inflammation (88) and arrhythmias (89). The present review summarizes the
key findings and mechanisms underlying the cardiovascular effects
of NF.
NF has been shown to induce vasorelaxation in rat
mesenteric arteries through both endothelium-dependent and
independent mechanisms. Specifically, it activates NO production
and regulates calcium levels in vascular smooth muscle cells
(VSMCs) (81). Similarly, NF
exhibits vasodilatory effects on the rat thoracic aorta, mediated
through the NO/cGMP signaling pathway and calcium regulation,
indicating its potential as an antihypertensive agent (82).
In vitro and in vivo studies have
revealed that NF reduces angiotensin II-induced cardiomyocyte
hypertrophy by attenuating oxidative stress and apoptosis (45,83,90).
Additionally, NF mitigates doxorubicin-induced cardiotoxicity by
enhancing endogenous antioxidants and inhibiting caspase 3/7
activity (84). NF protects
against isoproterenol-induced myocardial infarction in Wistar rats,
normalizing heart rate and preventing pathological changes in heart
and liver tissues (85). It also
reduces vascular leakage and improves cardiac function in acute
myocardial infarction by regulating the PI3K/Akt pathway, thereby
preserving endothelial barrier function (13). Furthermore, NF enhances cardiac
function in myocardial ischemia-reperfusion injury through the
upregulation of PPARγ and the suppression of cardiomyocyte
apoptosis (86).
NF shows potential in treating atherosclerosis. In
apolipoprotein E (−/-) mice fed a HFD, NF has been shown to
regulate the proliferation and migration of VSMCs by targeting the
Calm4/MMP12/Akt pathway, thereby reducing atherosclerotic plaque
formation (87). In the context of
vascular inflammation, NF induces autophagy to alleviate vascular
cell adhesion molecule 1 activation via the Akt/mTOR/AP1 pathway,
thus protecting against cardiovascular events (88). NF analogs have demonstrated
efficacy in treating arrhythmias by blocking calcium, voltage-gated
sodium and potassium channels, thereby regulating the action
potential (89). In conclusion,
the broad-spectrum cardiovascular benefits of NF are attributed to
its ability to regulate multiple pathways and cellular processes,
making it a potential new therapeutic agent for the treatment of
various cardiovascular diseases.
Antipsychotic effects of NF
The pharmacological profile of NF was
comprehensively characterized by Farrell et al (22) through both in vitro and
in vivo assays. The findings revealed that NF exhibited a
complex pharmacological profile (e.g. blocking head-twitch
responses and discriminative stimulus effects of a 5-HT2A agonist)
similar to certain antipsychotic drugs, demonstrating potential
antipsychotic-like effects in animal models. The aforementioned
study emphasized the interactions of NF with multiple receptors and
its impact on locomotor activity and prepulse inhibition,
indicating potential therapeutic application for neuropsychiatric
disorders. The receptor profile of NF shares similarities with
aripiprazole-like antipsychotic drugs (22). Specifically, NF functions as an
antagonist at 5-HT2A, 5-HT2B and
5-HT2C receptors, an inverse agonist at 5-HT7
receptors, a partial agonist at D5, D2 and
5-HT6 receptors, and an agonist at D4 and
5-HT1A receptors, while also inhibiting the dopamine
transporter (VMAT2) (22). In
rodent models relevant to antipsychotic pharmacology, NF has been
reported to suppress head-twitch responses and discriminative
stimulus effects elicited by a 5-HT2A agonist, produce a
clozapine discriminative stimulus, potentiate amphetamine-induced
locomotor activity, attenuate phencyclidine (PCP)-induced locomotor
activity and reverse PCP-induced disruption of prepulse inhibition,
all without causing catalepsy. The receptor interaction profile of
NF resembles, but is not identical to, that of several established
antipsychotic agents, indicating that NF exhibits several
properties consistent with an atypical antipsychotic profile
(22). The pharmacological profile
of NF suggests that it possesses atypical antipsychotic-like
actions, making it a promising candidate for the treatment of
neuropsychiatric disorders. Its ability to modulate multiple
neurotransmitter systems while avoiding catalepsy is particularly
noteworthy, as it may offer therapeutic benefits with fewer side
effects compared with existing antipsychotic drugs.
Anticancer effects of NF
Increasing research has demonstrated that NF may
exert considerable influence over various aspects of cancer
biology, including cell proliferation, apoptosis, autophagy,
metastasis, invasion, transcription and drug-resistance. These
effects have been observed across multiple types of cancer, such as
melanoma (16,91,92),
lung cancer (18,93,94),
neuroblastoma (95), colorectal
cancer (96,97), breast cancer (98–100), pancreatic cancer (PC) (101), laryngeal squamous cell carcinoma
(102), oral squamous cell
carcinoma (OSCC) (17) and
hepatocellular carcinoma (HCC) (103). In these types of cancer, NF
modulates several signaling pathways, such as the PI3K/Akt pathway
in lung cancer and neuroblastoma (95,104), the NF-κB pathway in melanoma
(16), the AMPK pathway in PC
(101), the TGF-β1 signaling
pathway in estrogen receptor-negative (ER−) breast
cancer (99), the Wnt/β-catenin
signaling in non-small cell lung cancer (NSCLC) (18) and colon cancer (96), and the STAT3 signaling pathway in
OSCC (17) (Fig. 3). NF also regulates the expression
of different genes associated with factors, such as tumor cell
proliferation, cell cycle progression, programmed cell death,
autophagic pathways, metastasis and transcription (Fig. 4). Collectively, these above studies
provide substantial evidence that NF is a promising multi-targeting
agent for the prevention and treatment of various types of cancer.
The mechanisms underlying the anticancer effects of NF are briefly
summarized in Table SII.
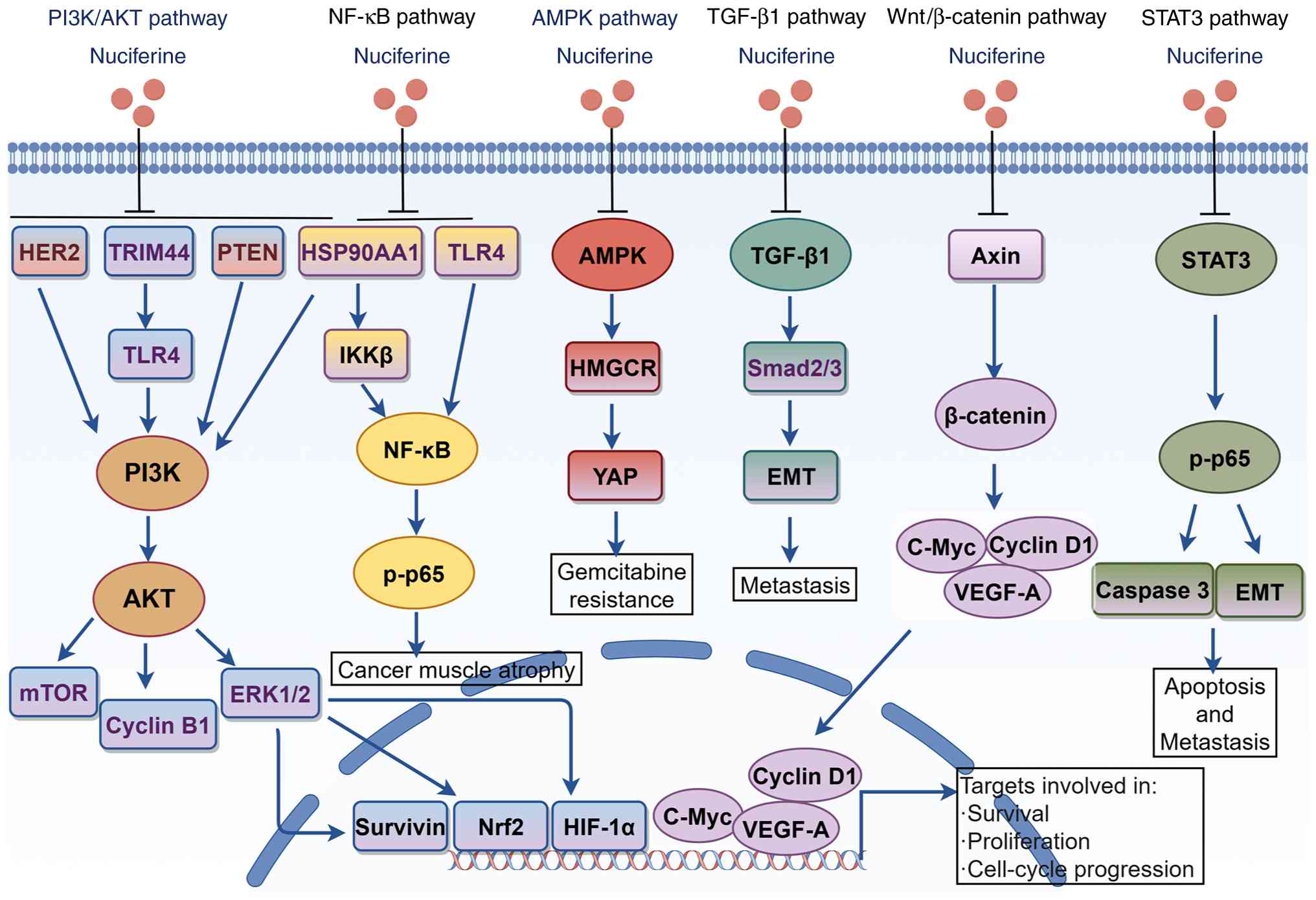 | Figure 3.Anticancer effects of nuciferine in
various types of cancer through different molecular signaling
pathways. This figure was drawn by Figdraw (https://www.figdraw.com). TRIM44, tripartite motif
containing 44; TLR4, Toll-like receptor 4; HSP90AA1, heat shock
protein 90 α family class A member; p-, phosphorylated; AMPK,
AMP-activated protein kinase; HMGCR, 3-hydroxy-3-methylglutaryl-CoA
reductase; HIF-1α, hypoxia inducible factor-1α; YAP, yes-associated
protein; Nrf2, nuclear factor erythroid 2-related factor 2; VEGF-A,
vascular endothelial growth factor A; EMT, epithelial-mesenchymal
transition. |
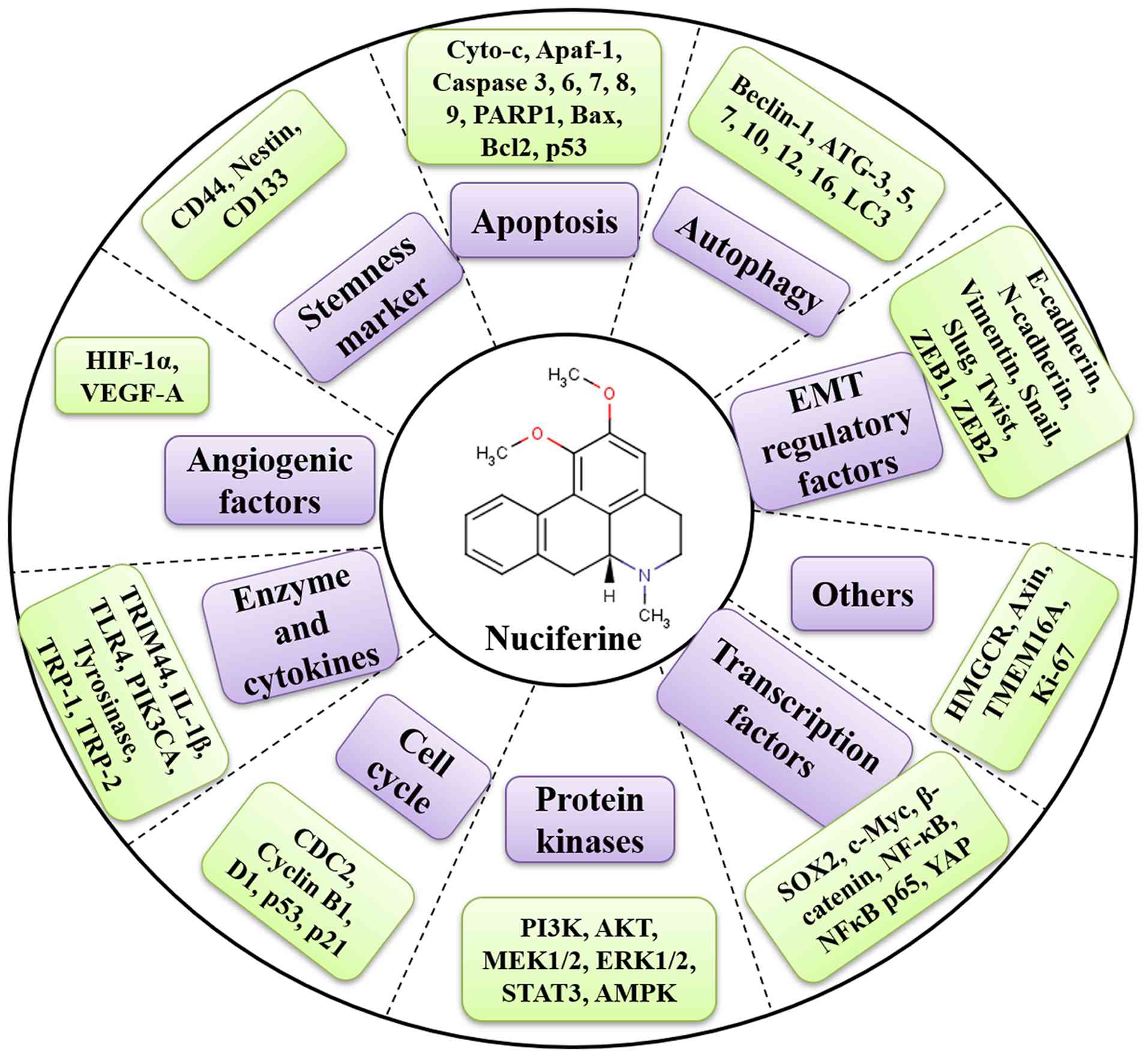 | Figure 4.Various molecular targets modulated
by nuciferine. HIF-1α, hypoxia inducible factor-1α; VEGF-A,
vascular endothelial growth factor A; TRIM44, tripartite motif
containing 44; TLR4, Toll-like receptor 4; TRP, tyrosinase-related
protein; YAP, yes-associated protein; HMGCR,
3-hydroxy-3-methylglutaryl-CoA reductase; ZEB, zinc finger E-box
binding homeobox; ATG, autophagy related; Cyto-c, cytochrome
c; EMT, epithelial-mesenchymal transition. |
PI3K/Akt signaling pathway
The PI3K/Akt signaling pathway serves as a
fundamental regulatory axis governing a diverse array of cellular
functions, including cell proliferation, survival, growth and
metabolism (104). Its
dysregulation is a hallmark of several types of cancer,
contributing notably to tumorigenesis. The pathway is often
activated by mutations or amplifications in its metabolites,
leading to enhanced oncogenic signaling. For example, Liu et
al (97) showed that NF
combined with paclitaxel considerably inhibited lung cancer growth
without causing notable toxicity, positioning NF as a promising
multidrug resistance (including paclitaxel, doxorubicin, docetaxel
and daunorubicin) sensitizer by suppressing the PI3K/Akt/ERK
pathway, reducing the activation of hypoxia inducible factor-1α and
nuclear factor erythroid 2-related factor 2 (Nrf2) in an A549/T
xenograft mouse model.
An et al (105) revealed that NF exhibited
protective effects against muscle wasting associated with lung
cancer cachexia by modulating the Akt/mTOR signaling cascade
through direct interaction with heat shock protein 90 α family
class A (HSP90AA1). Qi et al (95) revealed that NF exerted its effects
by suppressing the PI3K/Akt signaling pathway and lowering IL-1
concentrations in in vitro and in vivo models of
neuroblastoma. Li et al (102) reported that NF reduced the
expression of tripartite motif containing 44 and TLR4 proteins in a
dose-dependent manner, inhibiting activation of the Akt signaling
pathway. Li et al (103)
demonstrated that NF inhibited HCC cell proliferation in a
concentration-dependent manner through the induction of
G2/M cell-cycle arrest and the promotion of apoptosis,
and suppressed tumor growth and angiogenesis by downregulating the
HER2/PI3K/Akt/ERK signaling pathway. Collectively, the PI3K/Akt
signaling pathway serves as a pivotal oncogenic pathway implicated
in the pathogenesis of multiple malignancies. Its activation is
associated with enhanced tumor growth, invasion and resistance to
therapy, making it a key target for cancer treatment. The
development of targeted therapies against this pathway,
particularly in combination with other treatments, holds promise
for improving outcomes for patients with cancer.
NF-κB signaling pathway
The NF-κB family comprises transcription factors
that serve pivotal roles in modulating gene expression associated
with immune responses, inflammation, cell proliferation and
survival (106). Its
dysregulation contributes to inflammatory disorders, metabolic
disturbances and autoimmune diseases (107). The role of NF-κB in promoting
oncogenesis, tumor cell proliferation, survival, metastasis,
angiogenesis and therapy resistance highlights its potential as a
therapeutic target for both inflammatory diseases and cancer
(108). Xu et al (16) demonstrated that NF suppressed
melanoma cell proliferation and tumor size by inhibiting the
TLR4/NF-κB signaling pathway, Whereas An et al (105) showed that NF protected against
muscle wasting in lung cancer cachexia by modulating the NF-κB
signaling pathway via direct interaction with HSP90AA1.
AMPK signaling pathway
Gemcitabine, the current standard chemotherapeutic
agent for advanced PC, frequently encounters notable drug
resistance (109). NF has been
identified as a potential therapeutic agent that may improve
gemcitabine responsiveness in PC cells through modulation of the
AMPK pathway. This activation leads to the downregulation of
3-hydroxy-3-methylglutaryl-CoA reductase, a key regulator of the
mevalonate pathway essential for yes-associated protein activity
(101). In a PANC-1 ×enograft
mouse model, the combination of NF and gemcitabine considerably
reduced tumor volume and weight without notable toxicity (101). Additionally, NF lowered the
IC50 of gemcitabine in PC cell lines and exhibited
synergistic cytotoxic effects when used in combination (101). These findings underscore the
potential of NF as an effective adjuvant therapy for PC by
leveraging the AMPK pathway to overcome gemcitabine resistance.
TGF-β1 signaling pathway
TGF-β1 serves as the archetypal cytokine within an
extensive family of multifunctional secreted proteins. These
molecules are pivotal in governing a wide array of cellular
activities, including cell proliferation, differentiation and
apoptosis via activation of the TGF-β1/SMAD signaling pathway
(110). Given its modulation by
various elements at different levels, this pathway is also closely
involved in cancer development and progression (111). Gautam et al (94) revealed that NF exhibited potent
therapeutic effects against pulmonary fibrosis induced by both
TGF-β1 and bleomycin in both cellular and animal models, involving
modulation of the TGF-β1/Smad2/3 signaling cascade and suppression
of epithelial-mesenchymal transition (EMT) processes. Tong et
al (99) indicated that NF
markedly inhibited cell migration and metastasis of ER−
breast cancer cells by involving the TGF-β1 signaling pathway, the
ERK1/2 and SMAD3 pathways and reducing intracellular
H2O2 levels.
Wnt/β-catenin signaling pathway
The Wnt/β-catenin signaling pathway serves an
essential role in development, tissue homeostasis and cell
proliferation. However, its aberrant activation drives tumor
initiation and progression, making it a promising target for cancer
therapies (112). Liu et
al (18) demonstrated that NF
markedly reduced the proliferation of NSCLC cells, enhanced the
stabilization of Axin and induced apoptosis by decreasing the
Bcl-2/Bax ratio, and further demonstrated its efficacy in
inhibiting tumor growth and alleviating nicotine-induced liver
damage by the inhibition of Wnt/β-catenin signaling in in
vivo experiments. Okayama et al (96) evaluated the anti-proliferative
activities of NF against HT-29 human colon cancer cells and their
cancer stem cells. Mechanistically, NF inhibited the expression of
Wnt/β-catenin pathway target proteins, such as survivin, suggesting
that its cytotoxic effects are mediated through inhibition of this
pathway (96).
STAT3 signaling pathway
STAT3 is a central regulator of the antitumor
immune response and is often hyperactivated in the tumor ecosystem,
inhibiting immune activation and promoting immunosuppression
(113). NF has been reported to
inhibit the proliferation, migration and invasion of OSCC cells,
while promoting apoptosis through suppressing the STAT3 signaling
pathway (17). At the molecular
level, NF increases the expression of pro-apoptotic markers
cleaved-caspase 3 and Bax, enhances E-cadherin expression, and
reduces Bcl-2 and vimentin levels, thereby promoting apoptosis and
reversing the EMT in OSCC cells. However, colivelin, a potent STAT3
activator, can attenuate these effects by downregulating
cleaved-caspase 3 and Bax, upregulating Bcl-2, and reversing
changes in E-cadherin and vimentin expression, thereby reactivating
the STAT3 signaling pathway and counteracting the antitumor effects
of NF (17). In conclusion, the
ability of NF to modulate multiple signaling pathways and molecular
targets involved in cancer progression underscores its potential as
a multi-targeting agent for cancer therapy.
Toxicity or undesirable effects of NF
While the pharmacological activities of NF have
been extensively studied, it is essential to provide a more
comprehensive understanding of its potential toxicity and
undesirable effects (Table SIII).
In addition to the aforementioned interactions with CYP enzymes and
receptor antagonism, studies have begun to explore the broader
toxicological profile of NF.
Previous in vitro studies have investigated
the cytotoxic effects of NF on various cell lines. For example, a
study by Li et al (103)
evaluated the cytotoxicity of NF in human HCC (HepG2) cells using
the MTT assay. The results indicated that NF exhibited time- and
dose-dependent cytotoxicity, with an IC50 value of
101.88, 87.51 and 67.97 µM after 24, 48 and 72 h of treatment. Xie
et al (17) investigated
the impact of various concentrations of NF (0–120 µM) on the
proliferation and viability of OSCC cells using the Cell Counting
Kit-8 assay. Their findings demonstrated that NF effectively
inhibited the proliferation of OSCC cells in a dose-dependent
manner.
NF has been shown to have various interactions and
effects with notable implications in the medical field. As reported
by Ye et al (27) and Hu
et al (28), the inhibition
of CYP enzymes by NF suggests a potential for drug-drug
interactions. NF potently inhibits CYP2D6, with an IC50
value of 3.78 µM and also inhibits CYP2C9 and CYP2C19, with
IC50 values of 94.88 and 29.0 µM, respectively. Given
the key role of CYP2D6 in the metabolism of several clinically
important drugs, this interaction is particularly concerning. This
could lead to increased plasma levels of co-administered drugs,
potentially resulting in adverse effects or therapeutic failure;
thus, careful consideration of these interactions is necessary in
clinical settings.
Beyond these CYP-related interactions, NF has
demonstrated specific antagonistic activity against the
β2-adrenergic receptor (β2-AR), which holds
potential therapeutic benefits for conditions such as hypertension,
heart failure, glaucoma and infantile hemangioma. In human
epidermoid carcinoma A431 cells, NF was reported to exhibit an
IC50 value of 15.8±2.6 µM and a Ki value of 0.091
µM, highlighting its efficacy as a β2-AR antagonist
(114).
Heng et al (115) further explored the inhibitory
effects of NF. Using a TGF-α shedding assay, this previous study
assessed the impact of NF on human 5-HT2 and adrenergic
α1 receptor subtypes. The results showed that NF effectively
inhibited 5-HT2A, 5-HT2B and
5-HT2C receptors, with IC50 values of
7.18±0.03, 7.51±0.21 and 7.44±0.01 nM, respectively. Additionally,
it exhibited strong inhibition against α1A,
α1B and α1D receptors, with IC50
values of 7.42±0.07, 7.22±0.01 and 6.78±0.01 nM, respectively.
Liu et al (29) investigated the inhibitory effects
of NF on Mucor miehei lipase, revealing an IC50
value of 0.194 mg/ml. NF acts as a non-competitive inhibitor with a
Ki value of 0.16 mg/ml. Moreover, Guan et al
(116) demonstrated that NF can
act as a potent inhibitor of TNF-α with an IC50 value of
61.19 µM. These multiple inhibitory effects of NF across different
targets suggest its complex pharmacological profile and diverse
potential applications in medicine.
These findings highlight the potential for NF to
interact with various enzymes and receptors, which may have both
therapeutic and adverse effects. The inhibition of CYP enzymes by
NF suggests the possibility of drug-drug interactions, which need
to be carefully considered in clinical settings. Additionally, the
antagonistic activity against β2-AR and other receptors
indicates potential therapeutic applications, but also underscores
the need for further investigation into the safety profile of NF.
Future research should focus on further elucidating the
toxicological profile of NF. Comprehensive in vitro and
in vivo studies are needed to fully understand its potential
adverse effects, particularly at higher doses and with long-term
exposure. Additionally, clinical trials should be designed to
evaluate the safety and efficacy of NF in humans, with a focus on
identifying potential drug-drug interactions and mitigating
strategies.
Molecular docking simulation of NF through
an in silico approach
Molecular docking simulations have emerged as a
powerful tool to predict the binding modes and affinities between
NF and its potential targets such as PI3K (30), prostaglandin-endoperoxide synthase
2 (PTGS2) (31), taste 2 receptor
member 46 (TAS2R46) (117), JAK2
(71), Mucor miehei lipase
(29), CYPs (55), ARs (85), α-glycosidase and α-amylase
(73), HBXIP (60), cyclooxygenase-2 (COX-2) (118), URAT1 (119) and TNF-α (116), providing valuable insights into
their interactions. Various computational methods, such as
AutoDockTools software and SYBYL-X 2.0 software have been employed
to achieve this (120–124). Protein structures were sourced
from the Protein Data Bank (https://www.rcsb.org/), while the chemical structure
of NF was obtained from the PubChem database (PubChem CID: 10146).
Docking parameters were set with a grid box size of 40 Å centered
on the active site of each protein and binding affinities were
calculated using the default scoring function of AutoDockTools
software (Table SIV).
PI3K
The binding affinity of NF to PI3K was calculated
to be −6.33 kcal/mol, indicating strong interactions with key
residues such as Ser594 and Asn634 (30).
PTGS2
Similarly, NF exhibited a binding affinity of −9.33
kcal/mol with PTGS2. Its key amino acids, including Gln203, His207
and His388, serve a key role in forming interactions and
contributing to the non-competitive binding site (31).
TAS2R46
Ding et al (117) confirmed the specific target
binding ability of NF to TAS2R46 through molecular docking. The
results indicated that NF effectively interacted with the Ser248
residue of TAS2R46, with a binding affinity of −8.20±0.28 kcal/mol,
suggesting a stable binding interaction.
JAK2
Li et al (71) demonstrated a strong binding
affinity between NF and JAK2, with a binding energy of
−7.5903±0.7926 kcal/mol. NF was effectively accommodated within the
kinase active pocket of JAK2, forming a hydrogen bond with the
Gly993 residue.
Mucor miehei lipase
Liu et al (29) reported that NF can bind to the
catalytic pocket of Mucor miehei lipase, forming two
hydrogen bonds (O2-Arg169 and N-Asp238), and interacting
with Tyr195 and Pro194 through hydrophobic and stacking actions by
molecular docking analysis. The binding energy was −6.13 kcal/mol,
primarily contributed by van der Waals forces and hydrogen bonds.
The methyl and benzene ring structures of NF are key for binding,
and its inhibitory mechanism involves both active center insertion
and external region binding, inducing conformational changes in the
lipase and reducing its activity.
CYPs
Gao et al (55) investigated the interaction between
NF and CYPs using ADMET Predictor 9.0 and SYBYL-X 2.0 software. NF
was predicted to be a substrate of nine CYP family isoenzymes
(CYP1A2, 2C9, 2B6, 2A6, 2C8, 2C19, 2E1, 2D6 and 3A4), with
potential sites of metabolism located at various carbon and
nitrogen atoms. Molecular docking revealed hydrogen bonds and
hydrophobic interactions between NF and these enzymes, with
specific binding modes varying among different enzymes. For
example, NF can form one hydrogen bond (with Arg106) and seven
hydrophobic contacts when binding to CYP1A1 and one hydrogen bond
(with Thr321) and 10 hydrophobic contacts when binding to CYP1A2
(55).
ARs
Harishkumar and Selvaraj (85) reported that NF exhibited binding
energies of −8.46 and −5.02 kcal/mol with the β1- and
β2-ARs, respectively, which were higher than those of
isoproterenol (−5.27 and −4.3 kcal/mol). NF can bind to the
β1-AR at residues Val122, Thr126 and Ser211, and to the
β2-AR at residues Lys140, Gln229 and Glu1005 (85). These differences in binding
residues may account for its antagonistic activity against
isoproterenol-mediated interactions.
α-glycosidase and α-amylase
Khan et al (73) demonstrated that NF exhibits
considerable inhibitory activity against α-glycosidase and
α-amylase. NF was successfully docked into the active sites of both
enzymes. Specifically, it forms a π-π interaction with Trp484 and a
metal-ligand interaction with a calcium ion at position 701, in
α-glycosidase, interacting with key residues such as Glu173,
Glu474, Glu456 and Glu480 (73).
In α-amylase, the oxygen atom at position 39 of NF forms a bond
with Lys35, and its six-membered ring participates in a π-π
interaction with Trp396 (73).
HBXIP
Du et al (60) confirmed that NF directly interacts
with HBXIP, binding to a conserved potential pocket that includes
residues such as His87, Ile74, Gly72, His41, Ile45, Glu40, Val44
and Thr36. This interaction effectively disrupts the assembly of
the Rag GTPase-Ragulator complex, leading to the inhibition of
mTORC1 activity and the subsequent activation of TFEB-mediated
autophagy.
COX-2 and URAT1
Zhang et al (118) reported a binding energy of −5.78
kcal/mol between NF and COX-2, mediated through hydrogen bonds with
Lys328. Additionally, NF exhibited a binding energy of −7.44
kcal/mol with URAT1, forming a hydrogen bond with Lys393 and a π-π
interaction with Phe364 (119).
TNF-α
Guan et al (116) investigated the binding
interactions between NF and TNF-α, revealing a binding energy of
−8.4 kcal/mol. NF formed a carbon-hydrogen bond with Ser60 and
engaging in two π-π interactions with Tyr119. Surface plasmon
resonance (SPR) assays confirmed a strong affinity for TNF-α, with
a dissociation constant of 61.19 µM (116).
Summary of the molecular docking
simulation of NF
The molecular docking simulations of NF detailed in
the present review reveal its interactions with multiple biological
targets, providing information on potential pharmacological
mechanisms and a basis for developing more effective derivatives.
These simulations identify key binding affinities and interactions,
demonstrating the versatility of NF as a promising natural
therapeutic compound. However, these computational findings are
preliminary and require experimental validation to confirm their
biological relevance. Future studies should combine computational
predictions with laboratory techniques, such as site-directed
mutagenesis, ligand-protein interaction assays and cell-based
functional tests, to verify the proposed mechanisms.
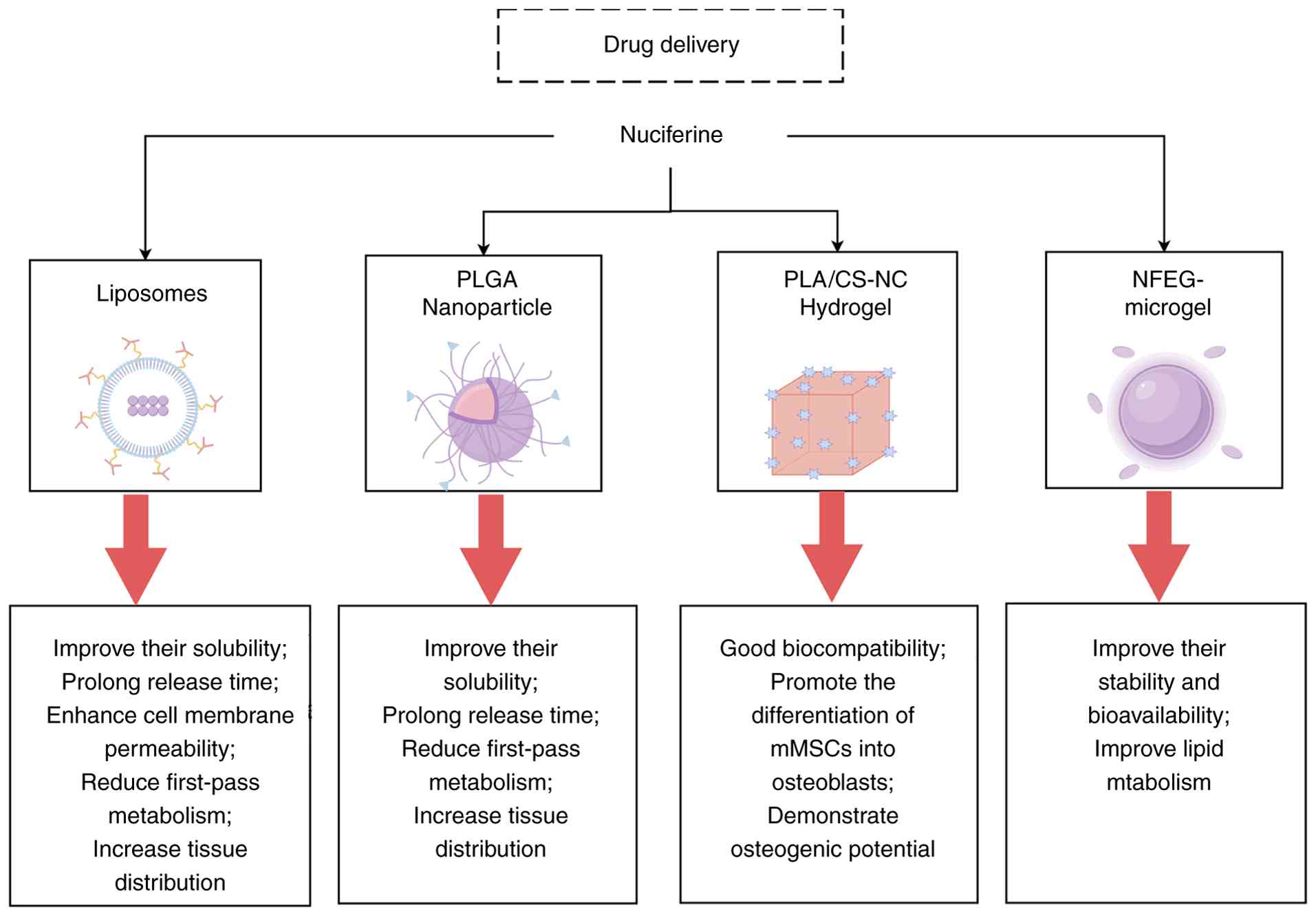
Bioavailability and drug delivery of NF
NF, a hydrophobic metabolite, exhibits poor
solubility in water, which considerably limits its bioavailability.
To address this challenge, various advanced drug delivery systems
capable of encapsulating NF have been developed. These include
liposomes (125), PLGA
nanoparticles (24), 3D-printed
polylactic acid (PLA) scaffolds incorporating chitosan (CS)-NF
hydrogel (25) and
NF-epigallocatechin-3-gallate (EGCG) double-encapsulated microgel
(NFEG-microgel) (26) (Table SV; Fig. 5). These innovative carriers have
demonstrably enhanced the aqueous solubility and bioavailability of
NF.
Liposomes notably enhance the bioavailability of NF
through multiple mechanisms, including improving its solubility,
prolonging release time, enhancing cell membrane permeability,
reducing first-pass metabolism and increasing tissue distribution.
Specifically, NF liposomes exhibit smaller and more uniform
particle sizes, slower release rates conforming to a first-order
release model and notably increased pharmacokinetic parameters,
such as half-life (T1/2), mean residence time and area
under the curve (AUC), while reducing clearance and peak
concentration (125). For
example, a pharmacokinetic study of NF liposomes demonstrated a
notable increase in bioavailability compared with free NF. The
T1/2 of NF liposomes was extended to 6.46 h, compared
with 3.15 h for free NF. The AUC increased by 2-fold, indicating
enhanced absorption and distribution (125). These characteristics enable NF
liposomes to function more effectively in the body, thereby
enhancing their therapeutic efficacy.
PLGA nanoparticles notably enhance the
bioavailability of NF by improving its solubility, prolonging
release time, reducing first-pass metabolism and increasing tissue
distribution. Specifically, the nanoparticles have small and
uniform particle sizes, slower release rates with sustained release
characteristics and higher release efficiency in simulated
intestinal fluid compared with simulated gastric fluid.
Pharmacokinetic parameters show extended T1/2, markedly increased
AUC and a 3.3-fold increase in relative bioavailability, with
sustained release characteristics over 24 h (24). These features enable NF-PLGA
nanoparticles to function more effectively in the body, thereby
enhancing therapeutic efficacy.
Bharathi et al (25) developed a NF-loaded PLA scaffold
using 3D printing technology and chitosan hydrogel for bone tissue
engineering. The results of the study showed that this composite
scaffold has good biocompatibility and can promote the
differentiation of mouse mesenchymal stem cells into osteoblasts,
demonstrating osteogenic potential at both cellular and molecular
levels. Specifically, NF is released in a sustained manner, with
~70% released over 21 days. Moreover, the NF-treated scaffolds
considerably enhance alkaline phosphatase (ALP) activity, increase
calcium deposition and upregulate the expression of
osteogenesis-related genes (such as RUNX family transcription
factor 2, ALP, COL-1 and OC). These findings suggest that the
NF-loaded PLA/chitosan scaffold is a promising material for bone
tissue engineering.
Zhu et al (26) successfully encapsulated NF and EGCG
into a microgel system, improving their stability and
bioavailability. In HFD-induced rats, NFEG-microgel intervention
effectively reduced body weight and serum lipid levels by
modulating key lipid metabolism genes and specific microRNAs
(miRs), such as miR-30b-5p and miR-126a-5p, in serum extracellular
vesicles (26). It also enhanced
gut microbiota diversity, enriching beneficial short-chain fatty
acid-producing bacteria while reducing harmful ones, thereby
improving lipid metabolism through intestinal flora regulation.
These results highlight NFEG-microgel as a promising delivery
system and therapeutic strategy for lipid metabolism disorders.
To further enhance the bioavailability of NF,
future research should prioritize optimizing nanoparticle
formulations through strategies such as surface modification with
polyethylene glycol, which can improve stability and reduce
immunogenicity. Additionally, exploring targeted delivery systems
such as antibody-drug conjugates could enable more precise delivery
of NF to diseased tissues, minimizing off-target effects. These
advanced drug delivery approaches show notable potential for
overcoming the solubility and bioavailability challenges of NF,
ultimately improving its therapeutic efficacy.
Comparative analysis and integrative
discussion
NF demonstrates a marked capacity to regulate the
PI3K/Akt pathway, which is evident in both its metabolic regulation
and anticancer activities. In the context of metabolic disorders,
NF activates the AMPK pathway, thereby enhancing lipid metabolism
and reducing inflammation (58).
In cancer models, the inhibition of the PI3K/Akt pathway by NF has
been shown to suppress tumor growth and enhance chemotherapy
sensitivity (97). These
overlapping mechanisms suggest that NF could have broad therapeutic
applications by targeting common signaling pathways involved in
both metabolic and oncogenic processes.
NF has also demonstrated clinical potential. For
example, in immunomodulation and antioxidant effects, the ability
of NF to regulate inflammatory pathways (such as TLR4/NF-κB) and
reduce oxidative stress is associated with the management of
chronic inflammatory diseases and age-related diseases. NF shows
potential in the treatment of acute kidney injury, cerebral
ischemia-reperfusion injury and ulcerative colitis. In metabolic
regulation, its ability to regulate lipid metabolism and improve
metabolic health by activating AMPK and PPARα pathways has
attracted attention, indicating that it may become a valuable drug
for the treatment of obesity, liver steatosis, type 2 diabetes and
other metabolic diseases. In musculoskeletal protection, its
ability to inhibit osteoclastogenesis and reduce cartilage
degradation in osteoporosis and OA is notable, with the potential
to address musculoskeletal diseases through a single drug. In
anticancer effects, its ability to regulate key pathways involved
in cancer cell proliferation, apoptosis and metastasis suggests it
could be a valuable addition to cancer treatment.
Despite the encouraging preclinical research
results, the application of NF in clinical practice remains a
considerable challenge. In preclinical studies, there is a notable
difference in the concentrations of NF. In in vitro studies,
its concentration typically ranges from low micromolar (1–10 µM) to
higher levels (30–200 µM), while in in vivo studies, its
concentration is typically between 1 and 50 mg/kg. Although these
concentrations have shown biological effects in vitro and in
animal models, achieving such levels in humans may face notable
challenges. For example, to achieve effective concentrations in
target tissues without causing toxicity or adverse reactions,
careful consideration of pharmacokinetics, bioavailability and
potential drug interactions is required. At the same time, detailed
mechanistic studies are required to comprehensively elucidate the
downstream effects of NF on various signaling pathways.
In addition, the studies included in the present
review collectively demonstrate the diverse interactions of NF with
a range of biological targets, including enzymes, receptors and
protein complexes. These interactions are not only indicative of
the potential therapeutic applications of NF but also provide a
detailed understanding of its molecular mechanisms. For example,
the strong binding affinities observed between NF and key targets,
such as PI3K, PTGS2 and TNF-α, suggest potential roles in
modulating signaling pathways and inflammatory responses.
Similarly, interactions with enzymes such as α-glycosidase and
α-amylase highlight its potential as an antidiabetic agent. The
docking results also reveal specific binding modes and key residues
involved in these interactions, which can guide the design of more
potent NF derivatives. However, the present review appreciates that
these in silico findings are a key first step and must be
complemented by experimental validation. Future research should
focus on integrating computational predictions with wet-lab
techniques, such as site-directed mutagenesis, ligand-protein
interaction assays and cell-based functional studies, to confirm
the mechanistic relevance of these interactions.
Although there have been several reviews on NF in
the past 5 years, these have primarily focused on summarizing
specific aspects of NF activity. For example, Huang et al
(126) comprehensively summarized
extensive knowledge on chemistry and biology of NF including
separation technology, structural modification, total synthesis,
structure-activity relationship, as well as in vivo
metabolism, biological activity and biosynthesis. Zhao et al
(3) critically summarized the
information regarding the structure-activity relationship of NF,
its biological activity and clinical application in
inflammation-related diseases (such as diabetes, obesity,
cardiovascular diseases, liver diseases and cancer), as well as its
potential mechanisms. Ren et al (8) provided a comprehensive overview of
the chemical properties, pharmacological activities (such as
improving hyperlipidemia, relaxing smooth muscles, vasodilatation,
stimulating insulin secretion, antiarrhythmic activity, inducing
hypotension, as well as anti-HIV and antibacterial activities) of
NF and their potential regulatory mechanisms. Zhao et al
(127) summarized studies on the
neuroprotective mechanisms of alkaloids derived from Nelumbo
nucifera, which encompass anti-inflammatory and antioxidant
effects, modulation of ion channels and calcium signaling,
promotion of neurogenesis and regulation of key neurotransmitter
systems. However, the present review consolidates and synthesizes a
broader range of data, including previous studies on
immunomodulation, metabolism regulation, neuroprotection,
musculoskeletal protection, cardiovascular benefits, antipsychotic
effects and anticancer activities. Secondly, the present review
gives a detailed analysis of the potential applications of NF in
fields such as molecular docking simulation and drug delivery. The
latest research results on the molecular mechanism of NF in
vivo and in vitro in recent studies have also been
included, and put forward novel views on the challenges and future
directions of NF research, distinguishing the present work from
prior reviews.
Conclusions, current challenges and future
research directions
Conclusions and challenges
NF, a natural alkaloid derived from the lotus plant
(Nelumbo nucifera Gaertn.), exhibits a wide range of
biological activities, positioning it as a promising candidate for
therapeutic applications. Its ability to modulate multiple
signaling pathways and interact with various molecular targets
underscores its potential in treating diverse diseases, including
inflammatory conditions, metabolic disorders, neurodegenerative
diseases and several types of cancer. The present review has
elucidated the multifaceted biological activities of NF,
highlighting its potential as a novel therapeutic agent.
To advance NF from bench to bedside, several key
challenges must be addressed. Firstly, the lack of standardized
extraction and purification protocols has led to notable
variability in the yield, purity and composition of NF across
studies. Factors such as solvent type, extraction time and plant
source all influence the final product, which may affect biological
activity and reproducibility (3,26).
Future studies should adopt standardized, reproducible extraction
methods and include rigorous quality control measures, such as
high-performance liquid chromatography and mass spectrometry
fingerprinting, to ensure consistency and comparability of
results.
Secondly, species differences in metabolism,
receptor expression and pharmacokinetics limit the direct
translational relevance of findings from animal models. For
example, the oral bioavailability and tissue distribution of NF may
differ markedly between rodents and humans (24–26,125). To bridge this gap, cross-species
pharmacokinetic studies, human-relevant in vitro models and
organoid or organ-on-chip systems should be employed to improve the
ability to predict human responses.
Thirdly, inconsistent findings across studies, such
as variable efficacy in inflammation or cancer models, highlight
the need for greater standardization in experimental design. This
includes dose selection, treatment duration and endpoint
assessment. Publication bias and underreporting of negative results
may also skew the literature. Meta-analyses, systematic reviews and
transparent data reporting are essential to reconcile
contradictions and identify true effect sizes.
Fourthly, while molecular docking provides useful
hypotheses about the binding of NF to target proteins, these
predictions remain speculative without experimental validation.
Several docking studies lack biophysical confirmation (29,60,73),
such as SPR, isothermal titration calorimetry and crystallography,
or functional assays to verify binding affinity and downstream
effects. Future research should integrate computational predictions
with wet-lab validation, using techniques such as site-directed
mutagenesis, ligand-protein interaction assays and cell-based
functional readouts to confirm mechanistic relevance.
Comprehensive toxicology studies are also lacking,
making it difficult to assess the safety profile of NF. Long-term
studies are needed to evaluate potential chronic effects, and
identify biomarkers for monitoring treatment efficacy and safety.
Conducting comprehensive toxicology studies, including acute and
chronic toxicity assays, will be key to establish a safety profile.
Developing biomarkers to monitor treatment efficacy and safety in
clinical trials is also essential.
The poor solubility and bioavailability of NF limit
its therapeutic potential. Advanced drug delivery systems are
needed to enhance their solubility and targeted delivery.
Developing advanced drug delivery systems, such as liposomes, PLGA
nanoparticles and prodrug strategies, can enhance the solubility
and bioavailability of NF. Optimizing nanoparticle formulations to
improve cellular uptake and reduce off-target effects will further
improve its therapeutic efficacy. Comparative studies with standard
drugs are limited, making it difficult to assess the relative
efficacy and potential advantages of NF. Conducting comparative
studies with standard drugs to evaluate the efficacy and safety of
NF is essential. Exploring synergistic effects of NF with existing
drugs, such as metformin or chemotherapeutics, could enhance
therapeutic outcomes.
Lastly, as the current research on NF mainly
focuses on cellular and animal models, or molecular docking
techniques, comprehensive pharmacokinetics, toxicology and phase I
trials are necessary steps before considering any clinical
translation. Designing human trials that accurately reflect the
therapeutic potential of NF requires careful consideration of
dosing, patient selection and endpoint assessment. Phase I trials
should be designed to assess safety, tolerability and
pharmacokinetics in healthy volunteers. Using biomarker-guided
trials to stratify patient populations and monitor therapeutic
response will be key. Considering adaptive trial designs to
optimize dosing and treatment duration based on interim results
will also enhance the efficiency and success of clinical trials.
Addressing these challenges will be essential to unlock the full
therapeutic potential of NF and advance its clinical
application.
Therapeutic prospects and future
research directions
To fully realize the therapeutic potential of NF,
several key priorities should guide future research. Firstly, the
anti-inflammatory and antioxidant properties of NF position it as a
promising therapeutic agent for various conditions, including acute
kidney injury, acute lung injury and ulcerative colitis. The
ability of NF to modulate key signaling pathways, such as
TLR4/NF-κB and Nrf2, further suggests its potential for managing
chronic inflammatory diseases. Additionally, the effects of NF on
lipid metabolism and glucose regulation indicate its therapeutic
potential in metabolic disorders, such as obesity, type 2 diabetes
and NAFLD. By activating AMPK and PPARα pathways, NF shows promise
in addressing metabolic syndrome.
Moreover, the neuroprotective effects of NF
demonstrated in ischemic stroke and Alzheimer's disease models
highlight its potential in neurodegenerative and psychiatric
disorders. Its ability to penetrate the BBB and modulate
neurotransmitter signaling underscores its promise in central
nervous system disorders. In the field of oncology, the anticancer
effects of NF, mediated through the modulation of signaling
pathways such as PI3K/Akt, NF-κB and Wnt/β-catenin, suggest its
potential as a multi-targeting agent for various types of cancer.
Its ability to inhibit tumor cell proliferation, induce apoptosis
and overcome drug resistance further highlights its therapeutic
potential in cancer therapy.
To advance the clinical application of NF, it is
essential to conduct detailed mechanistic studies to elucidate the
molecular mechanisms underlying its therapeutic effects. This
includes investigating its interactions with specific receptors and
signaling pathways, as well as identifying downstream targets.
Comprehensive toxicity studies are also required to fully
understand the safety profile of NF, including long-term studies to
assess potential chronic effects and the development of biomarkers
to monitor treatment efficacy and safety.
The development of advanced drug delivery systems,
such as liposomes and nanoparticles, should be prioritized to
enhance the bioavailability of NF and reduce its hydrophobicity.
These systems should be designed to improve targeted delivery and
minimize off-target effects. Well-designed clinical trials are
necessary to translate the therapeutic potential of NF into
clinical practice, focusing on specific disease indications such as
metabolic disorders, neurodegenerative diseases and different types
of cancer to establish its efficacy and safety in humans.
Lastly, investigating the potential of NF in
combination with existing therapies could enhance its therapeutic
effects. For example, combining NF with chemotherapeutic agents or
metabolic regulators could provide synergistic benefits in cancer
and metabolic diseases. Overall, addressing these priorities will
be important in identifying the full therapeutic of potential of NF
and advancing its clinical application.
In conclusion, NF demonstrates considerable
potential as a versatile natural compound with broad therapeutic
applications, but its transition to clinical use demands thorough
standardization, mechanistic understanding and proven delivery
methods. Overcoming these obstacles through collaborative,
interdisciplinary efforts and translational research will be
essential to fully realize the benefits of NF in modern medicine.
Future research should focus on comprehensive toxicology studies,
advanced drug delivery approaches, comparisons with existing
treatments, and carefully designed human trials to ensure the
safety and efficacy of NF in clinical practice.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This study was financially supported by the National Natural
Science Foundation of China (grant. no. 81902802), the Guangzhou
Municipal Science and Technology Project (grant. nos. 2025A03J3363,
2023A03J0571, 2024A03J0672, 2024A03J0598 and 2024A03J0603), the
Guangzhou Traditional Chinese Medicine and Integrated Traditional
Chinese and Western Medicine Project (grant. no. 20242A011014) and
the Technology Project of Guangzhou Municipal Health Commission
(grant. no. 20261A010032).
Availability of data and materials
Not applicable.
Authors' contributions
YL, XL and XX prepared the original draft. XL, BH
and XL drew figures and tables, and participated in the concept and
design of the manuscript. BY and XX reviewed and revised the
manuscript. YL, XX, LZ and HM were responsible for conceptual
design. HM and LZ carried out supervision and management. All
authors read and approved the final manuscript. Data authentication
is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
β2-AR
|
β2-adrenergic receptor
|
|
AMPK
|
AMP- activated protein kinase
|
|
AUC
|
area under the curve
|
|
BA
|
bile acid
|
|
BBB
|
blood brain barrier
|
|
COX-2
|
cyclooxygenase-2
|
|
EGCG
|
epigallocatechin-3-gallate
|
|
EMT
|
epithelial-to-mesenchymal
transition
|
|
ER−
|
estrogen receptor-negative
|
|
H2O2
|
hydrogen peroxide;
|
|
HBXIP
|
hepatitis B virus X-interacting
protein
|
|
HCC
|
hepatocellular carcinoma
|
|
HFD
|
high-fat diet
|
|
MCAO
|
middle cerebral artery occlusion
|
|
NAFLD
|
non-alcoholic fatty liver disease
|
|
NFEG-microgel
|
NF-EGCG double-encapsulated
microgel
|
|
NF
|
nuciferine
|
|
NO
|
nitric oxide
|
|
Nrf2
|
nuclear factor erythroid 2-related
factor 2
|
|
OA
|
osteoarthritis
|
|
OSCC
|
oral squamous cell carcinoma
|
|
PC
|
pancreatic cancer
|
|
PCP
|
phencyclidine
|
|
PLA
|
polylactic acid
|
|
PPAR
|
peroxisome proliferator-activated
receptor
|
|
RA
|
rheumatoid arthritis
|
|
PTGS2
|
prostaglandin-endoperoxide synthase
2
|
|
ROS
|
reactive oxygen species
|
|
SPR
|
surface plasmon resonance
|
|
STZ
|
streptozocin
|
|
TAS2R46
|
taste 2 receptor member 46
|
|
TFEB
|
transcription factor EB
|
|
TLR4
|
Toll-like receptor 4
|
|
VEGF
|
vascular endothelial growth
factor
|
|
VSMCs
|
vascular smooth muscle cells
|
References
|
1
|
Newman DJ and Cragg GM: Natural Products
as Sources of New Drugs over the Nearly Four Decades from 01/1981
to 09/2019. J Nat Prod. 83:770–803. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen G, Zhu M and Guo M: Research advances
in traditional and modern use of Nelumbo nucifera: Phytochemicals,
health promoting activities and beyond. Crit Rev Food Sci Nutr. 59
(sup1):S189–S209. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhao T, Zhu Y, Zhao R, Xiong S, Sun J,
Zhang J, Fan D, Deng J and Yang H: Structure-activity relationship,
bioactivities, molecular mechanisms, and clinical application of
nuciferine on inflammation-related diseases. Pharmacol Res.
193:1068202023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Justino AB, Florentino RM, Franca A, Filho
ACML, Franco RR, Saraiva AL, Fonseca MC, Leite MF and Salmen
Espindola F: Alkaloid and acetogenin-rich fraction from Annona
crassiflora fruit peel inhibits proliferation and migration of
human liver cancer HepG2 cells. PLoS One. 16:e02503942021.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yeh YT, Huang JC, Kuo PL and Chen CY:
Bioactive constituents from Michelia champaca. Nat Prod Commun.
6:1251–1252. 2011.PubMed/NCBI
|
|
6
|
de Lima BR, da Silva FMA, Soares ER, de
Almeida RA, da Silva-Filho FA, Barison A, Costa EV, Koolen HHF, de
Souza ADL and Pinheiro MLB: Integrative approach based on leaf
spray mass spectrometry, HPLC-DAD-MS/MS, and NMR for comprehensive
characterization of isoquinoline-derived alkaloids in leaves of
onychopetalum amazonicum. E. Fr. J Braz Chem Soc. 31:79–89.
2020.
|
|
7
|
Tungmunnithum D, Pinthong D and Hano C:
Flavonoids from nelumbo nucifera gaertn., a medicinal plant: Uses
in traditional medicine, phytochemistry and pharmacological
activities. Medicines (Basel). 5:1272018.PubMed/NCBI
|
|
8
|
Ren X, Chen H, Wang H, Wang Y, Huang C and
Pan H: Advances in the pharmacological effects and mechanisms of
Nelumbo nucifera gaertn. J Ethnopharmacol. 331:1182622024.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhu Y, Zhao Q, Huang Q, Li Y, Yu J, Zhang
R, Liu J, Yan P, Xia J, Guo L, et al: Nuciferine regulates immune
function and gut microbiota in DSS-induced ulcerative colitis.
Front Vet Sci. 9:9393772022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wu H, Yang Y, Guo S, Yang J, Jiang K, Zhao
G, Qiu C and Deng G: Nuciferine ameliorates inflammatory responses
by inhibiting the TLR4-Mediated pathway in
lipopolysaccharide-induced acute lung injury. Front Pharmacol.
8:9392017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen C, Duan F, Xie Y, Wan Q, Liu H, Gong
J, Huang L and Song Z: Nuciferine attenuates acute ischemic stroke
in a rat model: a metabolomic approach for the mechanistic study.
Mol Omics. 18:765–778. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Guo WH, Zhen GQ, Wu F, Lv YP, Yan JL, Zu
JN and Song CC: Nuciferine inhibits osteoclast formation through
suppressing glycolysis metabolic programming and ROS production.
Kaohsiung J Med Sci. 40:1057–1067. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Xie W, Chen S, Wang W, Qin X, Kong C and
Wang D: Nuciferine reduces vascular leakage and improves cardiac
function in acute myocardial infarction by regulating the PI3K/AKT
pathway. Sci Rep. 14:70862024. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wan Y, Xia J, Xu JF, Chen L, Yang Y, Wu
JJ, Tang F, Ao H and Peng C: Nuciferine, an active ingredient
derived from lotus leaf, lights up the way for the potential
treatment of obesity and obesity-related diseases. Pharmacol Res.
175:1060022022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhu X, Si F, Hao R, Zheng J and Zhang C:
Nuciferine protects against obesity-induced nephrotoxicity through
its hypolipidemic, anti-inflammatory, and antioxidant effects. J
Agric Food Chem. 71:18769–18779. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xu J, Ying A and Shi T: Nuciferine
inhibits skin cutaneous melanoma cell growth by suppressing
TLR4/NF-κB signaling. Anticancer Agents Med Chem. 20:2099–2105.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xie JR, Chen XJ and Zhou G: Nuciferine
inhibits oral squamous cell carcinoma partially through suppressing
the STAT3 signaling pathway. Int J Mol Sci. 24:145322023.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu W, Yi DD, Guo JL, Xiang ZX, Deng LF
and He L: Nuciferine, extracted from Nelumbo nucifera Gaertn,
inhibits tumor-promoting effect of nicotine involving Wnt/β-catenin
signaling in non-small cell lung cancer. J Ethnopharmacol.
165:83–93. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhou Y, Chen Z, Lin Q, Yang Y, Hang Y,
Zhou X, Wu C and Xie Z: Nuciferine reduced fat deposition by
controlling triglyceride and cholesterol concentration in broiler
chickens. Poult Sci. 99:7101–7108. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ma C, Li G, He Y, Xu B, Mi X, Wang H and
Wang Z: Pronuciferine and nuciferine inhibit lipogenesis in 3T3-L1
adipocytes by activating the AMPK signaling pathway. Life Sci.
136:120–125. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yu Y, Lu J, Sun L, Lyu X, Chang XY, Mi X,
Hu MG, Wu C and Chen X: Akkermansia muciniphila: A potential novel
mechanism of nuciferine to improve hyperlipidemia. Biomed
Pharmacother. 133:1110142021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Farrell MS, McCorvy JD, Huang XP, Urban
DJ, White KL, Giguere PM, Doak AK, Bernstein AI, Stout KA, Park SM,
et al: In vitro and in vivo characterization of the alkaloid
nuciferine. PLoS One. 11:e01506022016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ye LH, He XX, You C, Tao X, Wang LS, Zhang
MD, Zhou YF and Chang Q: Pharmacokinetics of nuciferine and
n-nornuciferine, two major alkaloids from nelumbo nucifera leaves,
in rat plasma and the brain. Front Pharmacol. 9:9022018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu Y, Wu X, Mi Y, Zhang B, Gu S, Liu G
and Li X: PLGA nanoparticles for the oral delivery of nuciferine:
preparation, physicochemical characterization and in vitro/in vivo
studies. Drug Deliv. 24:443–451. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bharathi R, Harini G, Sankaranarayanan A,
Shanmugavadivu A, Vairamani M and Selvamurugan N: Nuciferine-loaded
chitosan hydrogel-integrated 3D-printed polylactic acid scaffolds
for bone tissue engineering: A combinatorial approach. Int J Biol
Macromol. 253((Pt 7)): 1274922023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhu S, Xu W, Liu J, Guan F, Xu A, Zhao J
and Ge J: Preparation of microgel co-loaded with nuciferine and
epigallocatechin-3-gallate for the regulation of lipid metabolism.
Front Nutr. 9:10697972022. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ye LH, He XX, Kong LT, Liao YH, Pan RL,
Xiao BX, Liu XM and Chang Q: Identification and characterization of
potent CYP2D6 inhibitors in lotus leaves. J Ethnopharmacol.
153:190–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hu L, Xu W, Zhang X, Su J, Liu X, Li H and
Zhang W: In-vitro and in-vivo evaluations of cytochrome P450 1A2
interactions with nuciferine. J Pharm Pharmacol. 62:658–662. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu XT, Liu TT, Xu HL, Chen QX and Wang Q:
Inhibitory kinetics and bioactivities of Nuciferine and Methyl
Ganoderate on Mucor miehei lipase and 3T3-L1 preadipocytes. Int J
Biol Macromol. 163:1719–1728. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li T, Chen H, Guo Y, Huang M, Liu P,
Aikemu A, Mohammadtursun N, Pan X and Yang X: Nuciferine restores
autophagy via the PI3K-AKT-mTOR pathway to alleviate renal fibrosis
in diabetic kidney disease. J Agric Food Chem. 73:5223–5235. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rapuru R, Begum RF, Singh SA, Vellapandian
C, Ali N, AlAsmari AF and Prajapati BG: Exploring the therapeutic
potential of leriodenine and nuciferine from Nelumbo nucifera for
renal fibrosis: An In-silico analysis. Z Naturforsch C J Biosci.
80:673–689. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xiong X, Tang N, Lai X, Zhang J, Wen W, Li
X, Li A, Wu Y and Liu Z: Insights into amentoflavone: A natural
multifunctional biflavonoid. Front Pharmacol. 12:7687082021.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang MX, Zhao XJ, Chen TY, Liu YL, Jiao
RQ, Zhang JH, Ma CH, Liu JH, Pan Y and Kong LD: Nuciferine
alleviates renal injury by inhibiting inflammatory responses in
fructose-fed rats. J Agric Food Chem. 64:7899–7910. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li D, Liu B, Fan Y, Liu M, Han B, Meng Y,
Xu X, Song Z, Liu X, Hao Q, et al: Nuciferine protects against
folic acid-induced acute kidney injury by inhibiting ferroptosis.
Br J Pharmacol. 178:1182–1199. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen X, Zheng X, Zhang M, Yin H, Jiang K,
Wu H, Dai A and Yang S: Nuciferine alleviates LPS-induced mastitis
in mice via suppressing the TLR4-NF-kappaB signaling pathway.
Inflamm Res. 67:903–911. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kulhari U, Kundu S, Mugale MN and Sahu BD:
Nuciferine alleviates intestinal inflammation by inhibiting
MAPK/NF-ĸB and NLRP3/Caspase 1 pathways in vivo and in vitro. Int
Immunopharmacol. 115:1096132023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li J, Dong S, Quan S, Ding S, Zhou X, Yu
Y, Wu Y, Huang W, Shi Q and Li Q: Nuciferine reduces inflammation
induced by cerebral ischemia-reperfusion injury through the
PI3K/Akt/NF-kappaB pathway. Phytomedicine. 125:1553122024.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang L, Gao J, Tang P, Chong L, Liu Y,
Liu P, Zhang X, Chen L and Hou C: Nuciferine inhibits LPS-induced
inflammatory response in BV2 cells by activating PPAR-ү. Int
Immunopharmacol. 63:9–13. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kim SM, Park EJ and Lee HJ: Nuciferine
attenuates lipopolysaccharide-stimulated inflammatory responses by
inhibiting p38 MAPK/ATF2 signaling pathways. Inflammopharmacology.
30:2373–2383. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Kashiwada Y, Aoshima A, Ikeshiro Y, Chen
YP, Furukawa H, Itoigawa M, Fujioka T, Mihashi K, Cosentino LM,
Morris-Natschke SL and Lee KH: Anti-HIV benzylisoquinoline
alkaloids and flavonoids from the leaves of Nelumbo nucifera, and
structure-activity correlations with related alkaloids. Bioorg Med
Chem. 13:443–448. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Yang DD, Chutiwitoonchai N, Wang F, Tian
P, Sureram S, Lei X, Mahidol C, Ruchirawat S and Kittakoop P:
Effects of organic salts of virucidal and antiviral compounds from
Nelumbo nucifera and Kaempferia parviflora against SARS-CoV-2. Sci
Rep. 15:63802025. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sies H: Oxidative stress: A concept in
redox biology and medicine. Redox Biol. 4:180–183. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Chen X, Zhang A, Zhao K, Gao H, Shi P,
Chen Y, Cheng Z, Zhou W and Zhang Y: The role of oxidative stress
in intervertebral disc degeneration: Mechanisms and therapeutic
implications. Ageing Res Rev. 98:1023232024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jelic MD, Mandic AD, Maricic SM and
Srdjenovic BU: Oxidative stress and its role in cancer. J Cancer
Res Ther. 17:22–28. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Harishkumar R, Manjari MS, Rose C and
Selvaraj CI: Protective effect of Nelumbo nucifera (Gaertn.)
against H(2)O(2)-induced oxidative stress on H9c2 cardiomyocytes.
Mol Biol Rep. 47:1117–1128. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Charoensin S and Weera W: Preventive
effect of nuciferine on H(2)O(2)-Induced fibroblast senescence and
pro-inflammatory cytokine gene expression. Molecules. 27:81482022.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Xue K, Zhang G, Zhou Y, Wang K, Yao Z,
Chen J, Zhang Y, Li Z, Li Z, Zheng Z, et al: Nuciferine improves
random skin flap survival via TFEB-mediated activation of
autophagy-lysosomal pathway. Int Immunopharmacol. 119:1102042023.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Veerichetty V, Saravanabavan I, Pradeep A
and Abiraamasundari R: Development of gummy bear supplements and in
vitro exploration of antioxidant and antiproliferative potential of
Nuciferine. J Ayurveda Integr Med. 15:1008682024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Qiu J, Le Y, Liu N, Chen L, Jiang Y, Wang
Y, Fan X, Rong X, Yu Z, Li S and Dou X: Nuciferine Alleviates
High-Fat Diet- and ApoE(−/-)-induced hepatic steatosis and
ferroptosis in NAFLD Mice via the PPARα signaling pathway. J Agric
Food Chem. 72:24417–24431. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Fang Z, Jiang X, Wang S, Tai W, Jiang Q,
Loor JJ, Yu H, Hao X, Chen M, Shao Q, et al: Nuciferine protects
bovine hepatocytes against free fatty acid-induced oxidative damage
by activating the transcription factor EB/peroxisome
proliferator-activated receptor gamma coactivator 1 alpha pathway.
J Dairy Sci. 107:625–640. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Nguyen KH, Ta TN, Pham TH, Nguyen QT, Pham
HD, Mishra S and Nyomba BL: Nuciferine stimulates insulin secretion
from beta cells-an in vitro comparison with glibenclamide. J
Ethnopharmacol. 142:488–495. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Wang MX, Liu YL, Yang Y, Zhang DM and Kong
LD: Nuciferine restores potassium oxonate-induced hyperuricemia and
kidney inflammation in mice. Eur J Pharmacol. 747:59–70. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ye LH, Xiao BX, Liao YH, Liu XM, Pan RL
and Chang Q: Metabolism profiles of nuciferine in rats using
ultrafast liquid chromatography with tandem mass spectrometry.
Biomed Chromatogr. 30:1216–1222. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wu XL, Wu MJ, Chen XZ, Ma HL, Ding LQ, Qiu
F, Pan Q and Zhang DQ: Metabolic profiling of nuciferine in rat
urine, plasma, bile and feces after oral administration using
ultra-high performance liquid chromatography-diode array
detection-quadrupole time-of-flight mass spectrometry. J Pharm
Biomed Anal. 140:71–80. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gao H, Zhang L, Zhu A, Liu X, Wang T, Wan
M, Yang X and Zhang Y and Zhang Y: Metabolic profiling of
nuciferine in vivo and in vitro. J Agric Food Chem. Novemebr
18–2020.(Epub ahead of print). View Article : Google Scholar
|
|
56
|
Li L, Lei H, Wang W, Du W, Yuan J, Tu M,
Zhou H, Zeng S and Jiang H: Co-administration of nuciferine reduces
the concentration of metformin in liver via differential inhibition
of hepatic drug transporter OCT1 and MATE1. Biopharm Drug Dispos.
39:411–419. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wang LM, Wang P, Teka T, Zhang YC, Yang
WZ, Zhang Y, Wang T, Liu LX, Han LF and Liu CX: 1H NMR
and UHPLC/Q-Orbitrap-MS-based metabolomics combined with 16S rRNA
gut microbiota analysis revealed the potential regulation mechanism
of nuciferine in hyperuricemia rats. J Agric Food Chem.
68:14059–14070. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Xu H, Lyu X, Guo X, Yang H, Duan L, Zhu H,
Pan H, Gong F and Wang L: Distinct AMPK-Mediated FAS/HSL pathway is
implicated in the alleviating effect of nuciferine on obesity and
hepatic steatosis in HFD-Fed mice. Nutrients. 14:18982022.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Tang Z, Luo T, Huang P, Luo M, Zhu J, Wang
X, Lin Q, He Z, Gao P and Liu S: Nuciferine administration in
C57BL/6J mice with gestational diabetes mellitus induced by a
high-fat diet: The improvement of glycolipid disorders and
intestinal dysbacteriosis. Food Funct. 12:11174–11189. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Du X, Di Malta C, Fang Z, Fang Z, Shen T,
Niu X, Chen M, Jin B, Yu H, Lei L, et al: Nuciferine protects
against high-fat diet-induced hepatic steatosis and insulin
resistance via activating TFEB-mediated autophagy-lysosomal
pathway. Acta Pharm Sin B. 12:2869–2886. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Sun J, Fan J, Li T, Yan X and Jiang Y:
Nuciferine protects against high-fat diet-induced hepatic steatosis
via modulation of gut microbiota and bile acid metabolism in rats.
J Agric Food Chem. 70:12014–12028. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Li J, Zhao C, Liu M, Chen L, Zhu Y, Gao W,
Du X, Song Y, Li X, Liu G, et al: Nuciferine ameliorates
nonesterified fatty acid-induced bovine mammary epithelial cell
lipid accumulation, apoptosis, and impaired migration via
activating LKB1/AMPK signaling pathway. J Agric Food Chem.
71:443–456. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Li F, Zhao X, Xie F, Wang Z, Ding H, Wang
W, Jiao R, Pan Y and Kong L: Nuciferine blocks MIB2-mediated CARD6
polyubiquitination and degradation in the amelioration of high
fructose-induced liver lipid accumulation. Food Funct.
14:4706–4721. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Yan C, Zhan Y, Yuan S, Cao Y, Chen Y, Dong
M, Zhang H, Chen L, Jiang R, Liu W, et al: Nuciferine prevents
obesity by activating brown adipose tissue. Food Funct. 15:967–976.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Guo F, Yang X, Li X, Feng R, Guan C, Wang
Y, Li Y and Sun C: Nuciferine prevents hepatic steatosis and injury
induced by a high-fat diet in hamsters. PLoS One. 8:e637702013.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Yin J, Luo Y, Deng H, Qin S, Tang W, Zeng
L and Zhou B: Hugan Qingzhi medication ameliorates hepatic
steatosis by activating AMPK and PPARalpha pathways in L02 cells
and HepG2 cells. J Ethnopharmacol. 154:229–239. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Zhang DD, Zhang JG, Wu X, Liu Y, Gu SY,
Zhu GH, Wang YZ, Liu GL and Li XY: Nuciferine downregulates
Per-Arnt-Sim kinase expression during its alleviation of
lipogenesis and inflammation on oleic acid-induced hepatic
steatosis in HepG2 cells. Front Pharmacol. 6:2382015. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Zhang C, Deng J, Liu D, Tuo X, Xiao L, Lai
B, Yao Q, Liu J, Yang H and Wang N: Nuciferine ameliorates hepatic
steatosis in high-fat diet/streptozocin-induced diabetic mice
through a PPARα/PPARү coactivator-1α pathway. Br J Pharmacol.
175:4218–4228. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Ning Q, Wang Y, Zhang Y, Shen G, Xie Z and
Pang J: Nuciferine prevents hepatic steatosis by regulating lipid
metabolismin diabetic rat model. Open Life Sci. 14:699–706. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Xu H, Wang L, Yan K, Zhu H, Pan H, Yang H,
Liu M and Gong F: Nuciferine inhibited the differentiation and
lipid accumulation of 3T3-L1 preadipocytes by regulating the
expression of lipogenic genes and adipokines. Front Pharmacol.
12:6322362021. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Li J, Liu M, Fan M, Tian Q, Wang J, Du Y,
Yu J, Li X, Yang L, Zhao M, et al: Nuciferine ameliorates
blood-brain barrier disruption post-ischemic stroke via inhibiting
the JAK2/STAT3 pathway. Phytomedicine. 143:1568292025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Wu L, Chen C, Li Y, Guo C, Fan Y, Yu D,
Zhang T, Wen B, Yan Z and Liu A: UPLC-Q-TOF/MS-based serum
metabolomics reveals the anti-ischemic stroke mechanism of
nuciferine in MCAO rats. ACS Omega. 5:33433–33444. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Khan S, Khan HU, Khan FA, Shah A, Wadood
A, Ahmad S, Almehmadi M, Alsaiari AA, Shah FU and Kamran N:
Anti-Alzheimer and antioxidant effects of nelumbo nucifera L.
Alkaloids, nuciferine and norcoclaurine in alloxan-induced diabetic
albino rats. Pharmaceuticals (Basel). 15:12052022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Chen C, Ma Q, Jiang J, Wang T, Qiu L and
Liu A: Protective effects of nuciferine in middle cerebral artery
occlusion rats based on transcriptomics. Brain Sci. 12:5722022.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Nguyen A, Lee P, Rodriguez EK, Chahal K,
Freedman BR and Nazarian A: Addressing the growing burden of
musculoskeletal diseases in the ageing US population: Challenges
and innovations. Lancet Healthy Longev. 6:1007072025. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Song C, Cao J, Lei Y, Chi H, Kong P, Chen
G, Yu T, Li J, Kumar Prajapati R, Xia J and Yan J: Nuciferine
prevents bone loss by disrupting multinucleated osteoclast
formation and promoting type H vessel formation. FASEB J.
34:4798–4811. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Wen H, Zhou S, Li J, Peng F, Han X and Li
H: Nuciferine attenuates the progression of osteoarthritis by
targeting PI3K/Akt/NF-κB signaling pathway. J Funct Food.
86:1046822021. View Article : Google Scholar
|
|
78
|
Peng M, Shen G, Tu Q, Zhang W and Wang J:
Nuciferine ameliorates osteoarthritis: An in vitro and in vivo
study. Int Immunopharmacol. 142((Pt B)): 1130982024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wang H, Geng X, Ai F, Yu Z, Zhang Y, Zhang
B, Lv C, Gao R, Yue B and Dou W: Nuciferine alleviates
collagen-induced arthritic in rats by inhibiting the proliferation
and invasion of human arthritis-derived fibroblast-like
synoviocytes and rectifying Th17/Treg imbalance. Chin J Nat Med.
22:341–355. 2024.PubMed/NCBI
|
|
80
|
Kulhari U, Ambujakshan A, Ahmed M,
Washimkar K, Kachari J, Mugale MN and Sahu BD: Nuciferine inhibits
TLR4/NF-ĸB/MAPK signaling axis and alleviates adjuvant-induced
arthritis in rats. Eur J Pharmacol. 982:1769402024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wang X, Cheang WS, Yang H, Xiao L, Lai B,
Zhang M, Ni J, Luo Z, Zhang Z, Huang Y and Wang N: Nuciferine
relaxes rat mesenteric arteries through endothelium-dependent and
-independent mechanisms. Br J Pharmacol. 172:5609–5618. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Deng H, Xu Q, Sang XT, Huang X, Jin LL,
Chen FE, Shen QK, Quan ZS and Cao LH: Study on the vasodilatory
activity of lotus leaf extract and its representative substance
nuciferine on thoracic aorta in rats. Front Pharmacol.
13:9464452022. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Cho S, Cho HW, Woo KW, Jeong J, Lim J,
Park S, Seo M and Lim S: Nelumbo nucifera Receptaculum extract
suppresses angiotensin II-induced cardiomyocyte hypertrophy.
Molecules. 24:16472019. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Harishkumar R, Christopher JG, Ravindran R
and Selvaraj CI: Nuciferine attenuates doxorubicin-induced
cardiotoxicity: An in vitro and in vivo study. Cardiovasc Toxicol.
21:947–963. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Harishkumar R and Selvaraj CI: Nuciferine
from Nelumbo nucifera Gaertn. attenuates isoproterenol-induced
myocardial infarction in Wistar rats. Biotechnol Appl Biochem.
69:1176–1189. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Li R, Qin X, Yue L, Liu W, Gao Y, Zhu F,
Wang D and Zhou Q: Nuciferine improves cardiac function in mice
subjected to myocardial ischemia/reperfusion injury by upregulating
PPAR-ү. Heliyon. 9:e136302023. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Xiao M, Xian C, Wang Y, Qi X, Zhang R, Liu
Z and Cheng Y: Nuciferine attenuates atherosclerosis by regulating
the proliferation and migration of VSMCs through the
Calm4/MMP12/AKT pathway in ApoE((−/-)) mice fed with High-Fat-Diet.
Phytomedicine. 108:1545362023. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Wei H, Yin Y, Yang W, Zhu J, Chen L, Guo
R, Yang Z and Li S: Nuciferine induces autophagy to relieve
vascular cell adhesion molecule 1 activation via repressing the
Akt/mTOR/AP1 signal pathway in the vascular endothelium. Front
Pharmacol. 14:12643242023. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Zhou YX, Wang WP, Ke J, Ou HP, Chen LY,
Hou AG, Li P, Ma YS and Bin Jin W: Nuciferine analogs block
voltage-gated sodium, calcium and potassium channels to regulate
the action potential and treat arrhythmia. Biomed Pharmacother.
179:1174222024. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Mukherjee PK, Mukherjee D, Maji AK, Rai S
and Heinrich M: The sacred lotus (Nelumbo nucifera) - phytochemical
and therapeutic profile. J Pharm Pharmacol. 61:407–422. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Nakamura S, Nakashima S, Tanabe G, Oda Y,
Yokota N, Fujimoto K, Matsumoto T, Sakuma R, Ohta T, Ogawa K, et
al: Alkaloid constituents from flower buds and leaves of sacred
lotus (Nelumbo nucifera, Nymphaeaceae) with melanogenesis
inhibitory activity in B16 melanoma cells. Bioorg Med Chem.
21:779–787. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Wu PF, Chiu CC, Chen CY and Wang HM:
7-Hydroxydehydronuciferine induces human melanoma death via
triggering autophagy and apoptosis. Exp Dermatol. 24:930–935. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Bai X, Liu X, Li S, An H, Kang X and Guo
S: Nuciferine inhibits TMEM16A in dietary adjuvant therapy for lung
cancer. J Agric Food Chem. 70:3687–3696. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Gautam RK, Bhardwaj S and Kushwaha S:
Nuciferine inhibits epithelial-mesenchymal transition (EMT) in A549
cells and ameliorates bleomycin-induced pulmonary fibrosis in mice
via the TGF-β1/Smad2/3 signaling pathway. Mol Nutr Food Res.
69:e701312025. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Qi Q, Li R, Li HY, Cao YB, Bai M, Fan XJ,
Wang SY, Zhang B and Li S: Identification of the anti-tumor
activity and mechanisms of nuciferine through a network
pharmacology approach. Acta Pharmacol Sin. 37:963–972. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Okayama M, Matsumoto T, Kitagawa T,
Nakamura S, Ohta T, Yoshida T and Watanabe T: Cytotoxic activities
of alkaloid constituents from the climbing stems and rhizomes of
Sinomenium acutum against cancer stem cells. J Nat Med. 78:226–235.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Liu RM, Xu P, Chen Q, Feng SL and Xie Y: A
multiple-targets alkaloid nuciferine overcomes paclitaxel-induced
drug resistance in vitro and in vivo. Phytomedicine. 79:1533422020.
View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Kang EJ, Lee SK, Park KK, Son SH, Kim KR
and Chung WY: Liensinine and nuciferine, bioactive components of
nelumbo nucifera, inhibit the growth of breast cancer cells and
breast cancer-associated bone loss. Evid Based Complement Alternat
Med. 2017:15831852017. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Tong Y, Li Z, Wu Y, Zhu S, Lu K and He Z:
Lotus leaf extract inhibits ER(−) breast cancer cell migration and
metastasis. Nutr Metab (Lond). 18:202021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Zheng L, Jiang H, Li R, Song L, Chen R and
Dong H: The pharmacological mechanisms of xiaochaihutang in
treating breast cancer based on network pharmacology. Contrast
Media Mol Imaging. 2022:39006362022. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Zhou L, Wang Q, Zhang H, Li Y, Xie S and
Xu M: YAP inhibition by nuciferine via AMPK-Mediated Downregulation
of HMGCR sensitizes pancreatic cancer cells to gemcitabine.
Biomolecules. 9:6202019. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Li QH, Sui LP, Zhao YH, Chen BG, Li J, Ma
ZH, Hu ZH, Tang YL and Guo YX: Tripartite motif-containing 44 is
involved in the tumorigenesis of laryngeal squamous cell carcinoma,
and its expression is downregulated by nuciferine. Tohoku J Exp
Med. 254:17–23. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Li T, Wang W, Zhu M, Dong T, Jiang J, Li
Z, An T, Chen X, Mao Y, Chen Y and Yang X: The antineoplastic
effects of nuciferine on hepatocellular carcinoma are associated
with suppression of the HER2-AKT/ERK1/2 signaling pathway. Discov
Oncol. 16:8222025. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Glaviano A, Foo ASC, Lam HY, Yap KCH,
Jacot W, Jones RH, Eng H, Nair MG, Makvandi P, Geoerger B, et al:
PI3K/AKT/mTOR signaling transduction pathway and targeted therapies
in cancer. Mol Cancer. 22:1382023. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
An X, Ma L, Bai Y, Chen C, Liu J, Dawuti
A, Zeng K, Yang B, Han B and Abulizi A: Nuciferine attenuates
cancer cachexia-induced muscle wasting in mice via HSP90AA1. J
Cachexia Sarcopenia Muscle. 16:e137772025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Oeckinghaus A and Ghosh S: The NF-kappaB
family of transcription factors and its regulation. Cold Spring
Harb Perspect Biol. 1:a0000342009. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Mao H, Zhao X and Sun SC: NF-ĸB in
inflammation and cancer. Cell Mol Immunol. 22:811–839. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Yu H, Lin L, Zhang Z, Zhang H and Hu H:
Targeting NF-ĸB pathway for the therapy of diseases: Mechanism and
clinical study. Signal Transduct Target Ther. 5:2092020. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Rawla P, Sunkara T and Gaduputi V:
Epidemiology of pancreatic cancer: Global trends, etiology and risk
factors. World J Oncol. 10:10–27. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Wang J, Xiang H, Lu Y and Wu T: Role and
clinical significance of TGF-β1 and TGF-βR1 in malignant tumors
(Review). Int J Mol Med. 47:552021. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Su E, Han X and Jiang G: The transforming
growth factor beta 1/SMAD signaling pathway involved in human
chronic myeloid leukemia. Tumori. 96:659–666. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Song P, Gao Z, Bao Y, Chen L, Huang Y, Liu
Y, Dong Q and Wei X: Wnt/β-catenin signaling pathway in
carcinogenesis and cancer therapy. J Hematol Oncol. 17:462024.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Zou S, Tong Q, Liu B, Huang W, Tian Y and
Fu X: Targeting STAT3 in cancer immunotherapy. Mol Cancer.
19:1452020. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Zhang P, Wang J, Zhao Y, Zhang X, Qu L,
Wang C, Feng J, Wang A, Zhou W, Liu Y, et al: Discovery of novel
antagonists on β(2)-adrenoceptor from natural products using a
label-free cell phenotypic assay. Naunyn Schmiedebergs Arch
Pharmacol. 391:1411–1420. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Heng HL, Chee CF, Chin SP, Ouyang Y, Wang
H, Buckle MJC, Herr DR, Paterson IC, Doughty SW, Abd Rahman N and
Chung LY: Synthesis and evaluation of nuciferine and roemerine
enantiomers as 5-HT(2) and alpha(1) receptor antagonists.
Medchemcomm. 9:576–582. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Guan JW, Xu Y, Yu W, Wei LH, Xue R, Yu H,
Fan JM, Wang HZ, Abulizi A, Chen W and Han B: Biosensor-based
active ingredient recognition system for screening TNF-α inhibitors
from lotus leaves. Anal Bioanal Chem. 415:1641–1655. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Ding C, Ruan J, Huang J, Liu L, Li Y, Du Y
and Zhao Y: Nuciferine activates intestinal TAS2R46 to attenuate
metabolic disorders and hyperlipidemia via hepatic VLDL regulation.
Phytomedicine. 142:1568002025. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Zhang H, Chen G, Zhang Y, Yang M, Chen J
and Guo M: Potential hypoglycemic, hypolipidemic, and
anti-inflammatory bioactive components in Nelumbo nucifera leaves
explored by bioaffinity ultrafiltration with multiple targets. Food
Chem. 375:1318562022. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Zhang YK, Chen JS, Wang MM, Wang CZ, Wang
MX, Wang Z, Yang QL, Sun B, Sun JY, Liu YF and Liu C: Synthesis and
bioactivity evaluation of novel nuciferine derivatives with
antihyperuricemia and nephroprotective effects. Bioorg Chem.
126:1059162022. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Ishrat N, Gupta A, Khan MF, Shahab U, Khan
MS, Ahmad N, Kaur K, Ahmad S and Mahdi AA: Phytoconstituents of
Nymphaea rubra flowers and their anti-diabetic metabolic targets.
Fitoterapia. 176:1060142024. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Veerichetty V and Saravanabavan I:
Molecular docking study of nuciferine as a tyrosinase inhibitor and
its therapeutic potential for hyperpigmentation. Genomics Inform.
21:e432023. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Zhao X, Hou T, Zhou H, Liu Z, Liu Y, Wang
C, Guo Z, Yu D, Xu Q, Wang J and Liang X: Multi-effective
components and their target mechanism of Ziziphi Spinosae Semen in
the treatment of insomnia. Fitoterapia. 171:1057122023. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Zhou Z, Qi J, Wu Y, Li C, Bao W, Lin X and
Zhu A: Nuciferine effectively protects mice against
acetaminophen-induced liver injury. Antioxidants. 12:9492023.
View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Li H, Li W, Wu Y, Wu H and Cai X:
Integrating network pharmacology and animal experimental validation
to investigate the mechanism of lotus leaf in obesity. Int
Immunopharmacol. 145:1137192025. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Lu J, Xu YT, Qian XL, Zhu DX, Lu JY, Ma H
and Liu J: Preparation, pharmacokinetics and anti-obesity effects
on dogs of nuciferine liposomes. Vet Med Sci. 10:e700172024.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Huang X, Hao N, Chen G, Liu S and Che Z:
Chemistry and biology of nuciferine. Industrial Crops and Products.
179:1146942022. View Article : Google Scholar
|
|
127
|
Zhao D, Ma L, Brownlie J, Tonissen K, Pan
Y and Feng Y: Neuroprotective potential of major alkaloids from
nelumbo nucifera (Lotus): Mechanisms and therapeutic implications.
Int J Mol Sci. 26:82802025. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Yang X, Yu MF, Lei J, Peng YB, Zhao P, Xue
L, Chen W, Ma LQ, Liu QH and Shen J: Nuciferine relaxes tracheal
rings via the blockade of VDLCC and NSCC channels. Planta Med.
84:83–90. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Xiong WT, Liao JB, Yang ZX, Cui HT, Zhang
ZY, Wen WB and Wang HW: Effect of nuciferine on gut microbiota and
inflammatory response in obese model mice. Zhongguo Zhong Yao Za
Zhi. 46:2104–2111. 2021.(In Chinese). PubMed/NCBI
|
|
130
|
Cui H, Li Y, Cao M, Liao J, Liu X, Miao J,
Fu H, Song R, Wen W, Zhang Z and Wang H: Untargeted metabolomic
analysis of the effects and mechanism of nuciferine treatment on
rats with nonalcoholic fatty liver disease. Front Pharmacol.
11:8582020. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Liu CM, Kao CL, Wu HM, Li WJ, Huang CT, Li
HT and Chen CY: Antioxidant and anticancer aporphine alkaloids from
the leaves of Nelumbo nucifera Gaertn. cv. Rosa-plena. Molecules.
19:17829–17838. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Liu W, Chen X, Ge Y, Wang H, Phosat C, Li
J, Mao HP, Gao XM and Chang YX: Network pharmacology strategy for
revealing the pharmacological mechanism of pharmacokinetic target
components of San-Ye-Tang-Zhi-Qing formula for the treatment of
type 2 diabetes mellitus. J Ethnopharmacol. 260:1130442020.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Wang Y, Yao W, Li B, Qian S, Wei B, Gong
S, Wang J, Liu M and Wei M: Nuciferine modulates the gut microbiota
and prevents obesity in high-fat diet-fed rats. Exp Mol Med.
52:1959–1975. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Shi Z, Fang ZY, Gao XX, Yu H, Zhu YW,
Ouyang HL, Song YX, Du XL, Wang Z, Li XW and Liu GW: Nuciferine
improves high-fat diet-induced obesity via reducing intestinal
permeability by increasing autophagy and remodeling the gut
microbiota. Food Funct. 12:5850–5861. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Chan JD, Acharya S, Day TA and Marchant
JS: Pharmacological profiling an abundantly expressed schistosome
serotonergic GPCR identifies nuciferine as a potent antagonist. Int
J Parasitol Drugs Drug Resist. 6:364–370. 2016. View Article : Google Scholar : PubMed/NCBI
|