Introduction
Modalities such as endocrine therapy, chemotherapy
and therapy using antibodies are widely used for early operable
breast cancer to prevent relapse after surgery. The guidelines
established at the St. Gallen Consensus Conference (1) propose therapeutic approaches based on
treatment thresholds that are stratified by hormone receptor
expression [including estrogen receptors (ER) and progesterone
receptors (PgR)] and HER-2 overexpression or amplification
(1). Moreover, it is crucial to use
chemotherapy for any patient with breast cancer without these
targets, but with a high risk of recurrence, even if patient
results are positive for hormone receptors. Chemotherapy is a
therapeutic option for patients with recurrence risks, notably
those with early-stage disease. However, no specific markers exist
to forecast the effectiveness of anti-cancer drugs.
In recent studies on prognostic factors and factors
predicting the effects of chemotherapy, enzymes such as thymidylate
synthase (TS), dihydropyrimidine dehydrogenase (DPD), thymidine
phosphorylase (TP) and orotate phosphoribosyl transferase (OPRT)
have shown notable effects on 5-FU as a chemotherapeutic agent.
These studies have mainly focused on cancers of the digestive
system (2–9).
TS is a target enzyme for chemotherapy with
5-fluorouracil (5-FU). Numerous studies have found that 5-FU
therapy for metastatic and advanced colon cancer is more effective
in patients with low levels of intratumoral TS mRNA or TS protein
expression compared to patients with high levels of expression
(2–6). However, certain studies report that
5-FU post-operative adjuvant chemotherapy is effective in patients
with colon cancers expressing high TS levels (7–9).
Attention has been focused on 5-FU therapy and TS expression levels
in breast cancer. Patients with breast cancers that express low TS
levels and who receive 5-FU-based chemotherapy have an ideal
prognosis (10,11). However, the addition of
tegafur-uracil (UFT), an oral 5-FU agent, to hormone monotherapy
given as post-operative adjuvant therapy for breast cancer is
likely to offer longer relapse-free survival (RFS) time in patients
with high levels of TS expression (12).
DPD is a 5-FU degrading enzyme. It has been reported
that 5-FU therapy for advanced head and neck cancers is more
effective in patients with lower ratios of tumor/non-tumor DPD
enzymatic activities (13). Another
study on adjuvant chemotherapy following surgery for breast cancer
also showed 5-FU to be associated with more a favorable prognosis
in patients with lower DPD expression (14).
TP, also known as platelet-derived endothelial cell
growth factor, appears to behave in a manner similar to TS. In
metastatic colon cancer, 5-FU therapy is less effective for
patients with high levels of intratumoral TP mRNA expression
(15). Alternatively, TP expression
was mainly observed in the tumor stroma of colorectal carcinoma,
where patients with a high TP expression showed a more favorable
prognosis and a lower rate of metastasis than patients with a low
TP expression (16).
OPRT is a phosphorylating enzyme of 5-FU. OPRT
activity in tumor tissue is reportedly related to prognosis and
5-FU sensitivity in colon cancer (17–19).
Additionally, whether these molecular markers
predict 5-FU efficacy in breast cancer is controversial, since the
histological variety of breast cancer specimens contains large
amounts of cancerous stroma and non-tumorous tissues. Gene
expression levels of TS, DPD and TP in carcinoma cells and stroma
were shown to vary in breast cancer (20). Laser capture microdissection (LCM)
and real-time polymerase chain reaction (RT-PCR) are effective
techniques for measuring gene expression in carcinoma cells.
However, few reports exist regarding the expression of these
enzymes in breast carcinoma cells analyzed by LCM plus RT-PCR in
association with the prognosis and effect of 5-FU.
In this study, a retrospective approach was adopted
to investigate whether markers such as intratumoral TS, DPD, TP and
OPRT analyzed by LCM plus RT-PCR in formalin-fixed
paraffin-embedded specimens may be used as prognostic or
sensitivity-defining factors (5-FU efficacy-determining
factors).
Materials and methods
Case series
A total of 217 patients who had undergone surgery
between 1988 and 2006 at Ito Surgical Mammary Gland Clinic and
Tokushima Breast Care Clinic, Japan, for primary breast cancer were
recruited. The median duration of observation was 7.6 years. This
study was approved by the Ethics Committee of the Tokushima
University Hospital and the patients gave written consent. Patient
characteristics and stage grouping corresponding to pTNM categories
by UICC are shown in Table I. Oral
5-FU agents were administered to 147 patients, of whom 127 were
able to continue the medication for ≥6 months. UFT, doxifluridine
(5′-DFUR) and 5-FU tablets were used as oral 5-FU agents. A total
of 20 patients stayed on the medication for <6 months. A total
of 70 patients did not receive any oral 5-FU agents. Postoperative
endocrine therapy was carried out in 112 cases.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| No. of patients | 217 |
| Age |
| Median | 53 |
| Range | 24–83 |
| Stage |
| I | 109 |
| II | 88 |
| III | 20 |
| Menopausal
status |
| Premenopausal | 85 |
| Postmenopausal | 132 |
| Histological
classification |
| Papillotubular
carcinoma | 64 |
| Solid-tubular
carcinoma | 24 |
| Scirrhous
carcinoma | 96 |
| Mucinous
carcinoma | 6 |
| Medullary
carcinoma | 10 |
| Others | 17 |
| ER |
| − | 61 |
| + | 147 |
| PgR |
| − | 83 |
| + | 125 |
| HER-2 |
| 0,1 | 136 |
| 2 | 23 |
| 3 | 24 |
| Axillary lymph node
metastasis |
| − | 141 |
| + | 73 |
| Nuclear grade |
| 1 | 109 |
| 2 | 18 |
| 3 | 35 |
| Hormonal therapy |
| Not received | 105 |
| Received | 112 |
| Chemotherapy of oral
5-FU |
| Not received | 70 |
| Received (<6
months) | 20 |
| Received (≥6
months) | 127 |
Determination of gene expression levels
related to 5-FU- metabolizing enzymes using RT-PCR
Following the evaluation of
hematoxylin-eosin-stained slides by pathologists, 5 slides of 5-μm
sections were prepared from each formalin-fixed paraffin-embedded
specimen. The Danenberg Tumor Profile (DTP) method (Response
Genetics, Inc., Los Angeles, CA, USA, United States Patent Number
6,248,535), a system combining LCM technique and quantitative
RT-PCR through Taq Man (Applied Biosystems) chemistry, was employed
to measure quantities of mRNA for nucleic acid-metabolizing enzymes
in tumor tissues on the slides. Following RNA extraction, the cDNA
was prepared as previously described (21). The sequence of the target cDNA was
amplified using RT-PCR by the Taq Man method [ABI PRISM 7900
Sequence Detection System (Taq Man); Applied Biosystems, Foster
City, CA, USA]. The PCR reaction mixture contained the primers:
dATP, dCTP, dGTP, dUTP, MgCl2 and Taq Man buffer
(reagents from Applied Biosystems). PCR conditions were 10 sec at
50°C; 10 min at 95°C, followed by 42 reaction cycles (each cycle
lasting 15 sec at 95°C and 1 min at 60°C). Each mRNA expression
level was computed from its ratio to the expression of β-actin.
Immunohistochemical analysis
Immunohistochemical analysis for ER, PgR and HER-2
was performed on the patients. Positive responses to ER or PgR were
determined when ≥10% of the tumor tissues were stained. HER-2
responses were classified into four levels (0–3) based on the
HercepTest: 0, no staining; 1+, weak partial membranous staining in
≥10% of cells; 2+, weak to moderate complete membranous staining in
≥10% of cells and 3+, strong complete membranous staining in ≥10%
of cells.
Statistical analysis
mRNA expression levels of each enzyme were converted
into the value for log10 (mRNA) and the values were
assessed using the Student's t-test (two-sided) or ANOVA.
Univariate and multivariate survival analyses based on Cox's
proportional hazard regression model were used to evaluate
independent contributions of each variable to RFS for 70 patients
not treated with 5-FU. The appropriate cut-off value for each mRNA
was calculated using receiver operating characteristic (ROC)
analysis. Sensitivity and specificity were computed from
relationships with each mRNA, designating relapse as positive and
relapse-free states as negative. Factors were investigated in the
127 patients who were on 5-FU agents for ≥6 months to predict the
efficacy, based on the cut-off value obtained through ROC analysis.
The patients were divided into a low or high group, using the
cut-off values of each mRNA. An estimate of RFS was calculated
using the Kaplan-Meier method, and differences were assessed using
the log-rank test. RFS was analyzed between patients who received
5-FU for ≥6 months and those who received it for <6 months or
not at all. 5-FU may not have exhibited an adequate effect in the
20 patients who had discontinued it after less than 6 months due to
factors including adverse events (10 patients) and patient refusal
(5 patients). Significance was set at P<0.05. JMP ver7.0.1,
provided by SAS Institute, Inc., was used for the statistical
analysis. As the event number was small, overall survival (OS) was
not calculated.
Results
Evaluation of mRNA expression levels and
correlation to clinicopathological factors
The expression of mRNA in breast cancer was
successfully determined in 166, 172, 174 and 167 patients, and the
median mRNA expression levels were 3.05, 1.35, 11.9 and 1.39 for
TS, DPD, TP and OPRT, respectively. The results failed to indicate
any correlation between TS and OPRT expression and various
clinicopathological factors (Table
II). DPD expression was reduced, and inversely related to the
nuclear grade. A low DPD expression was detected in postmenopausal
patients and in hormone receptor-negative tumors. A low TP
expression was detected in patients with lymph node metastasis and
with hormone receptor-negative tumors.
 | Table IIAssociation of TS, DPD, TP and OPRT
mRNA expression with various clinicopathological factors. |
Table II
Association of TS, DPD, TP and OPRT
mRNA expression with various clinicopathological factors.
| mRNA (mean) | TS | P-value | DPD | P-value | TP | P-value | OPRT | P-value |
|---|
| Stage | | NS | | <0.05 | | NS | | NS |
| I | 3.72 | | 1.79 | | 14.3 | | 1.57 | |
| II | 3.24 | | 1.39 | | 14.5 | | 1.54 | |
| III | 4.07 | | 1.74 | | 12.8 | | 1.84 | |
| Menopausal
status | | NS | | <0.05 | | NS | | NS |
| Premenopausal | 3.88 | | 1.82 | | 12.2 | | 1.63 | |
| Postmenopausal | 3.33 | | 1.48 | | 15.5 | | 1.55 | |
| Axillary lymph node
metastasis | | NS | | NS | | <0.05 | | NS |
| − | 3.58 | | 1.69 | | 15.5 | | 1.58 | |
| + | 3.45 | | 1.44 | | 11.7 | | 1.55 | |
| Nuclear grade | | NS | | <0.01 | | NS | | NS |
| 1 | 3.34 | | 1.73 | | 13.9 | | 1.61 | |
| 2 | 3.93 | | 1.63 | | 14.6 | | 1.47 | |
| 3 | 4.10 | | 1.25 | | 15.4 | | 1.56 | |
| Histological
classification | | NS | | <0.05 | | NS | | NS |
| Papillotubular
carcinoma | 3.26 | | 1.51 | | 12.7 | | 1.65 | |
| Scirrhous
carcinoma | 3.39 | | 1.71 | | 13.7 | | 1.36 | |
| ER | | NS | | <0.01 | | <0.01 | | NS |
| − | 3.62 | | 1.28 | | 12.7 | | 1.47 | |
| + | 3.55 | | 1.76 | | 15.0 | | 1.63 | |
| PgR | | NS | | <0.01 | | <0.05 | | NS |
| − | 3.55 | | 1.37 | | 13.4 | | 1.48 | |
| + | 3.58 | | 1.78 | | 15.0 | | 1.66 | |
| HER-2 | | NS | | NS | | NS | | NS |
| 0, 1 | 3.66 | | 1.63 | | 13.7 | | 1.61 | |
| 2 | 3.76 | | 1.70 | | 17.2 | | 1.84 | |
| 3 | 2.93 | | 1.51 | | 15.2 | | 1.23 | |
Correlation between mRNA expression
levels and RFS in patients without 5-FU agents
Univariate analysis focusing on RFS showed the
nuclear grade and TS expression level to be significant prognostic
factors. Multivariate analysis identified the presence (or absence)
of axillary lymph node metastases, nuclear grade and the TS
expression level as independent prognostic factors for RFS
(Table III).
 | Table IIIUnivariate and multivariate analysis
of clinicopathological factors and mRNA expression. |
Table III
Univariate and multivariate analysis
of clinicopathological factors and mRNA expression.
| Prognostic
factor | Univariate
analysis | Multivariate
analysis |
|---|
| Hazard ratio | P-value | Hazard ratio | P-value |
|---|
| Stage | | NS | | |
| I | 1 | | | |
| II, III | 0.91 | | | |
| Menopausal
status | | NS | | |
| Premenopausal | 1 | | | |
|
Postmenopausal | 1.30 | | | |
| Axillary lymph node
metastasis | | NS | | <0.05 |
| − | 1 | | 1 | |
| + | 3.18 | | 11.6 | |
| Nuclear grade | | <0.05 | | <0.05 |
| 1, 2 | 1 | | 1 | |
| 3 | 5.38 | | 6.12 | |
| Histological
classification | | NS | | |
| Papillotubular
carcinoma | 1 | | | |
| Scirrhous
carcinoma | 0.76 | | | |
| ER | | NS | | |
| − | 1 | | | |
| + | 0.81 | | | |
| Pgr | | NS | | |
| − | 1 | | | |
| + | 0.91 | | | |
| HER-2 | | NS | | |
| 0, 1 | 1 | | | |
| 2, 3 | 1.02 | | | |
| TS | | <0.01 | | <0.01 |
| Low
(<4.57) | 1 | | 1 | |
| High (≥4.57) | 6.67 | | 10.9 | |
| DPD | | NS | | |
| Low (<1.8) | 1 | | | |
| High (≥1.8) | 0.66 | | | |
| TP | | NS | | |
| Low
(<11.8) | 1 | | | |
| High (≥11.8) | 1.78 | | | |
| OPRT | | NS | | |
| Low
(<1.66) | 1 | | | |
| High (≥1.66) | 3.56 | | | |
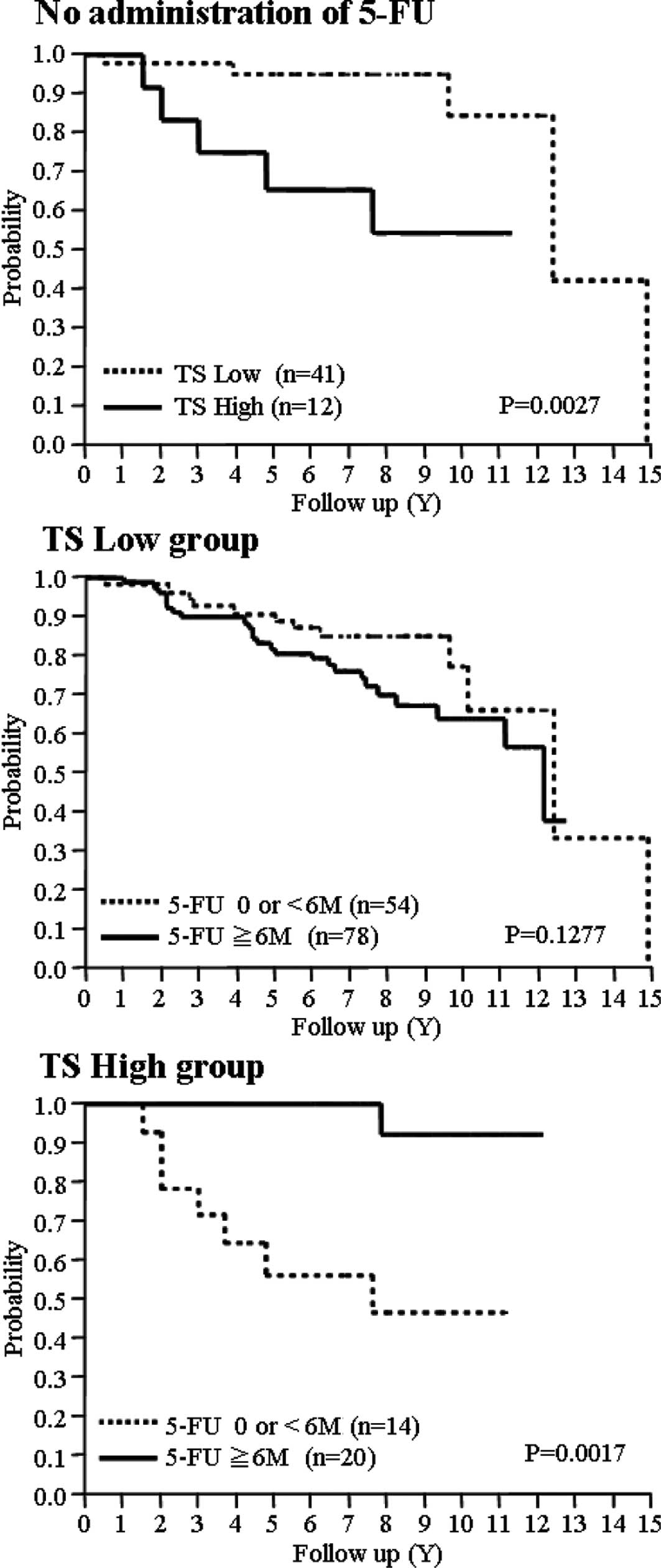
TS expression level as prognostic factor,
predicting the effect of 5-FU agents
The prognosis was significantly better in the low TS
group where no 5-FU was administered (Fig. 1A). In addition, prognosis of the low
TP group was significantly better, but no correlation was found
between DPD or OPRT expression levels and RFS time (data not
shown). No difference was noted between patients who received 5-FU
administration for ≥6 months and those who had <6 months or none
in the low TS group, while the high TS group showed a significantly
more favorable prognosis when 5-FU was administered for 6 months or
more (Fig. 1B and C). No
differences were found on completion of similar analyses for DPD,
TP and OPRT to that of TS (data not shown).
Discussion
This study presented novel information regarding
correlations between TS mRNA expression and prognosis in 217 breast
cancer patients. Part of our data was included in the results of a
recent report on a large number of test subjects with regard to TS,
DPD and OPRT mRNA and protein expression levels in certain tumors
(22).
Positive TS immunoreactivity in breast cancer tissue
is associated with aggressive phenotypes, including large tumor
size, high histological grade and nodal metastasis (10). Failure to correlate TS mRNA
expression with clinicopathological factors in the present study
may be due to variations in the methods regarding detection and
evaluations. Results of the multivariate analysis suggest that the
TS mRNA expression level was an independent factor.
Observation of the RFS time stratified by the level
of TS mRNA suggest that TS is an effective prognostic factor for
patients that did not receive 5-FU. These results suggest a
favorable prognosis in patients whose level of TS mRNA expression
is less than 4.57, while the effect of oral 5-FU is believed to be
minimal. Among patients with TS mRNA levels exceeding 4.57, on the
other hand, the prognosis was relatively poor. Such patients may
require an alternative form of treatment.
Factors that predict the efficacy of oral 5-FU
agents showed their recurrence-preventing effects, particularly in
patients expressing TS mRNA levels of 4.57 or more. The findings of
this study coincide with other findings, including the effect of an
oral 5-FU agent (UFT) on hormone monotherapy applied as
postoperative adjuvant therapy for breast cancer (12), and longer RFS time in the group with
a high TS expression, as examined by immunohistochemistry.
Therapies using infusional 5-FU, such as CMF
(cyclophosphamide, methotrexate and fluorouracil) and CEF
(cyclophosphamide, epirubicin and fluorouracil) regimens, have
resulted in a favorable prognosis for patients with low TS cancers
(10,11). Our data, however, were not in
agreement with these results. Oral administration of 5-FU showed a
higher peak level in blood than did continuous intravenous
injection of 5-FU at an equal molar ratio (23). The reliability of TS as a predictor
for 5-FU efficacy may thus differ between oral and infusional
5-FU.
The estimation of TS mRNA expression by
microdissection and RT-PCR from formalin-fixed paraffin-embedded
specimens may be more useful than immunohistochemistry, as cut-off
values are clearly calculated. We calculated a useful cut-off value
and patient ratio for prognostic predictions and 5-FU effect
forecast using ROC analysis. This study showed that the proportion
of cases expressing high TS levels was as much as 20% of all
patients. Such patients are expected to achieve a high relapse
prevention effect following oral 5-FU administration for 6 months
or more as postoperative adjuvant chemotherapy. Pestalozzi et
al analyzed TS expression in early breast cancer by
immunohistochemistry and reported that node-negative patients with
high TS levels showed a significant improvement in disease-free
survival and OS when treated with six cycles of adjuvant CMF
therapy, compared with those receiving only one cycle (24). Our result coincided with this
report, suggesting that completion of chemotherapy leads to a
favorable prognosis.
In this study, high DPD or TP levels in breast
cancer tissue correlated with a positive expression of hormone
receptors. These results imply that UFT including the DPD inhibitor
is efficacious as an oral 5-FU derivative for the hormone
receptor-positive patient.
The present study found prognostic value only in the
expression of TS and TP, and an efficacy-predicting value in TS
expression. However, other investigators showed the expression of
DPD, TP or OPRT to be correlated with various clinicopathological
factors, prognosis and efficacy of 5-FU agents (14,25–28).
Although the reason for these discrepancies was unclear, trials to
verify prognosis and efficacy of the 5-FU agents while taking
clinicopathological factors into account are a crucial strategic
objective to further validate useful biomarkers.
In conclusion, TS mRNA is effective for breast
cancer prognosis and as a predictor of efficacy for oral 5-FU
anticancer agents among the mRNA expression of 5-FU-metabolizing
enzymes. Thresholds for systemic treatment modality indications
were discussed in St. Gallen (1),
and those for cytotoxic chemotherapy use have yet to be clearly
defined. Increasingly, greater expectation is to be placed on the
use of individualized medicine, using prognostic and
efficacy-predicting markers. If TS mRNA expression levels can be
utilized in this manner, more selective and efficacious treatments
may become possible. Our results were obtained with short follow-up
times in a small-scale population analysis. Larger-scale studies
are required in prospective clinical trials for patients with
breast cancer before using TS expression as a predictive
factor.
Acknowledgements
This study was partially supported by a Grant-in Aid
from Taiho Pharmaceutical Co., Cancer Research, Japan. We also
thank Ms. Kume, Ms. Umemoto and Ms. Takai for their technical
assistance.
References
|
1
|
Goldhirsch A, Ingle JN, Gelber RD, Coates
AS, Thürlimann B and Senn HJ: (Panel members). Thresholds for
therapies: highlights of the St Gallen International Expert
Consensus on the primary therapy of early breast cancer 2009. Ann
Oncol. 20:1319–1329. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bathe OF, Franceschi D, Livingstone AS,
Moffat FL, Tian E and Ardalan B: Increased thymidylate synthase
gene expression in liver metastases from colorectal carcinoma:
implications for chemotherapeutic options and survival. Cancer J
Sci Am. 5:34–40. 1999.PubMed/NCBI
|
|
3
|
Cascinu S, Aschele C, Barni S, Debernardis
D, Baldo C, Tunesi G, Catalano V, Staccioli MP, Brenna A, Muretto P
and Catalano G: Thymidylate synthase protein expression in advanced
colon cancer: correlation with the site of metastasis and the
clinical response to leucovorin-modulated bolus 5-fluorouracil.
Clin Cancer Res. 5:1996–1999. 1999.
|
|
4
|
Leichman CG, Lenz HJ, Leichman L,
Danenberg K, Baranda J, Groshen S, Boswell W, Metzger R, Tan M and
Danenberg PV: Quantitation of intratumoral thymidylate synthase
expression predicts for disseminated colorectal cancer response and
resistance to protracted-infusion fluorouracil and weekly
leucovorin. J Clin Oncol. 15:3223–3229. 1997.
|
|
5
|
Lenz HJ, Hayashi K, Salonga D, Danenberg
KD, Danenberg PV, Metzger R, Banerjee D, Bertino JR, Groshen S,
Leichman LP and Leichman CG: p53 point mutations and thymidylate
synthase messenger RNA levels in disseminated colorectal cancer: an
analysis of response and survival. Clin Cancer Res. 4:1243–1250.
1998.PubMed/NCBI
|
|
6
|
Paradiso A, Simone G, Petroni S, Leone B,
Vallejo C, Lacava J, Romero A, Machiavelli M, De Lena M, Allegra CJ
and Johnston PG: Thymidilate synthase and p53 primary tumour
expression as predictive factors for advanced colorectal cancer
patients. Br J Cancer. 82:560–567. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Edler D, Glimelius B, Hallström M,
Jakobsen A, Johnston PG, Magnusson I, Ragnhammar P and Blomgren H:
Thymidylate synthase expression in colorectal cancer: a prognostic
and predictive marker of benefit from adjuvant fluorouracil-based
chemotherapy. J Clin Oncol. 20:1721–1228. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Johnston PG, Fisher ER, Rockette HE,
Fisher B, Wolmark N, Drake JC, Chabner BA and Allegra CJ: The role
of thymidylate synthase expression in prognosis and outcome of
adjuvant chemotherapy in patients with rectal cancer. J Clin Oncol.
12:2640–2647. 1994.PubMed/NCBI
|
|
9
|
Takenoue T, Nagawa H, Matsuda K, Fujii S,
Nita ME, Hatano K, Kitayama J, Tsuruo T and Muto T: Relation
between thymidylate synthase expression and survival in colon
carcinoma, and determination of appropriate application of
5-fluorouracil by immunohistochemical method. Ann Surg Oncol.
7:193–198. 2000. View Article : Google Scholar
|
|
10
|
Yu Z, Sun J, Zhen J, Zhang Q and Yang Q:
Thymidylate synthase predicts for clinical outcome in invasive
breast cancer. Histol Histopathol. 20:871–878. 2005.PubMed/NCBI
|
|
11
|
Hakamada Y, Tsuchida A, Arima M, Kubouchi
T, Tokita H, Ota D, Kaise H and Aoki T, Kusama M and Aoki T:
Prognostic predictors in breast cancer patients with postoperative
5-fluorouracil-based chemotherapy. Int J Mol Med. 16:309–314.
2005.PubMed/NCBI
|
|
12
|
Toi M, Ikeda T, Akiyama F, Kurosumi M,
Tsuda H, Sakamoto G and Abe O: Predictive implications of
nucleoside metabolizing enzymes in premenopausal women with
node-positive primary breast cancer who were randomly assigned to
receive tamoxifen alone or tamoxifen plus tegafur-uracil as
adjuvant therapy. Int J Oncol. 31:899–906. 2007.
|
|
13
|
Etienne MC, Chéradame S, Fischel JL,
Formento P, Dassonville O, Renée N, Schneider M, Thyss A, Demard F
and Milano G: Response to fluorouracil therapy in cancer patients:
the role of tumoral dihydropyrimidine dehydrogenase activity. J
Clin Oncol. 13:1663–1670. 1995.PubMed/NCBI
|
|
14
|
Horiguchi J, Yoshida T, Koibuchi Y, Iijima
K, Ninomiya J, Takei H, Yokoe T, Iino Y and Morishita Y: DPD
activity and immunohistochemical DPD expression in human breast
cancer. Oncol Rep. 11:65–72. 2004.PubMed/NCBI
|
|
15
|
Metzger R, Danenberg K, Leichman CG,
Salonga D, Schwartz EL, Wadler S, Lenz HJ, Groshen S, Leichman L
and Danenberg PV: High basal level gene expression of thymidine
phosphorylase (platelet-derived endothelial cell growth factor) in
colorectal tumors is associated with nonresponse to 5-fluorouracil.
Clin Cancer Res. 4:2371–2376. 1998.
|
|
16
|
Saito S, Tsuno N, Nagawa H, Sunami E,
Zhengxi J, Osada T, Kitayama J, Shibata Y, Tsuruo T and Muto T:
Expression of platelet-derived endothelial cell growth factor
correlates with good prognosis in patients with colorectal
carcinoma. Cancer. 88:42–49. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ochiai T, Sugitani M, Nishimura K, Noguchi
H, Watanabe T, Sengoku H, Komiyama H and Okano M: Correlation
between clinical pathophysiologic factors and expression of orotate
phosphoribosyl transferase (OPRT), thymidylate synthase (TS), and
dihydropyrimidine dehydrogenase (DPD) in colorectal cancer (in
Japanese). Gan To Kagaku Ryoho. 29:413–420. 2002.
|
|
18
|
Ochiai T, Nishimura K, Noguchi H, Kitajima
M, Tsukada A, Watanabe E, Nagaoka I and Futagawa S: Prognostic
impact of orotate phosphoribosyl transferase among 5-fluorouracil
metabolic enzymes in resectable colorectal cancers treated by oral
5-fluorouracil-based adjuvant chemotherapy. Int J Cancer.
118:3084–3088. 2006. View Article : Google Scholar
|
|
19
|
Ichikawa W, Uetake H, Shirota Y, Yamada H,
Takahashi T, Nihei Z, Sugihara K, Sasaki Y and Hirayama RK: Both
gene expression for orotate phosphoribosyltransferase and its ratio
to dihydropyrimidine dehydrogenase influence outcome following
fluoropyrimidine-based chemotherapy for metastatic colorectal
cancer. Br J Cancer. 89:1486–1492. 2003. View Article : Google Scholar
|
|
20
|
Kakimoto M, Uetake H, Osanai T, Shirota Y,
Takagi Y, Takeshita E, Toriya Y, Danenberg K, Danenberg PV and
Sugihara K: Thymidylate synthase and dihydropyrimidine
dehydrogenase gene expression in breast cancer predicts 5-FU
sensitivity by a histocultural drug sensitivity test. Cancer Lett.
223:103–111. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Makino H, Uetake H, Danenberg KD,
Danenberg PV and Sugihara K: Efficacy of laser capture
microdissection plus RT-PCR technique in analyzing gene expression
levels in human gastric cancer and colon cancer. BMC Cancer.
8:2102008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fukui Y, Oka T, Nagayama S, Danenberg PV,
Danenberg KD and Fukushima M: Thymidylate synthase,
dihydropyrimidine dehydrogenase, orotate phosphoribosyltransferase
mRNA and protein expression levels in solid tumors in large scale
population analysis. Int J Mol Med. 22:709–716. 2008.
|
|
23
|
Ho DH, Pazdur R, Covington W, Brown N, Huo
YY, Lassere Y and Kuritani J: Comparison of 5-fluorouracil
pharmacokinetics in patients receiving continuous 5-fluorouracil
infusion and oral uracil plus
N1-(2′-tetrahydrofuryl)-5-fluorouracil. Clin Cancer Res.
4:2085–2088. 1998.PubMed/NCBI
|
|
24
|
Pestalozzi BC, Peterson HF, Gelber RD, et
al: Prognostic importance of thymidylate synthase expression in
early breast cancer. J Clin Oncol. 15:1923–1931. 1997.PubMed/NCBI
|
|
25
|
Kurebayashi J, Yamamoto Y, Udagawa K,
Okubo S, Fukushima M and Sonoo H: Establishment of enzyme-linked
immunosorbent assays for thymidylate synthase and dihydropyriminide
dehydrogenase in cancer tissues. Oncol Rep. 11:973–979. 2004.
|
|
26
|
Ioachim E: Thymidine phoshorylase
expression in breast cancer: the prognostic significance and its
association with other angiogenesis related proteins and
extracellular matrix components. Histol Histopathol. 23:187–196.
2008.
|
|
27
|
Bronckaers A, Gago F, Balzarini J and
Liekens S: The dual role of thymidine phosphorylase in cancer
development and chemotherapy. Med Res Rev. 29:903–953. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tsunoda Y, Suzuki K, Sakamoto MA, Date Y,
Sawada T, Tsunoda A and Kusano M: Evaluation of
5-fluorouracil-related genes in breast cancer to predict the effect
of adjuvant therapy with CMF (in Japanese). Gan To Kagaku Ryoho.
36:51–55. 2009.PubMed/NCBI
|