Introduction
Breast cancer is the most commonly occurring cancer
among women. A family history of breast cancer is one of the main
risk factors for developing the disease. It is currently estimated
that approximately 5–10% of breast cancers are due to an inherited
predisposition, and approximately 20–25% of this risk is explained
by two high-penetrance susceptibility genes: BRCA1 (17q21)
(MIM no. 113705) (1) and
BRCA2 (13q12–13) (MIM no. 600185) (2). Both are tumor suppressor genes
involved mainly in DNA repair. Mutations in BRCA1 or
BRCA2 account for a lifetime risk of approximately 40–65%
for breast cancer and 11–40% for ovarian cancer (3). Extensive analyses of the BRCA1
and BRCA2 genes have resulted in the identification of a
large number of different disease-causing germline mutations
(4).
However, limited information exists regarding
BRCA1 and BRCA2 genetic testing in the majority of
non-Caucasian ethnicities and there is a paucity of data regarding
the genetic diversity in BRCA1 and BRCA2 in
populations of African descent. We report a novel mutation that
consists of a deletion of 2 bp (c.1949_1950delTA) in the exon 11 of
the BRCA1 gene. This is a frameshift mutation that causes
the disruption of the translational reading frame, resulting in a
stop codon downstream in the 671 position of the BRCA1 protein. The
mutation was present in a Senegalese woman with triple-negative
breast cancer and a family history of the disease.
Patient and methods
Patient
As part of genetic studies carried out on
breast/ovarian families in the University Hospital Vall d’Hebron
(Barcelona, Spain), germ-line BRCA1 and BRCA2
mutations were screened in the proband of the family reported in
this study.
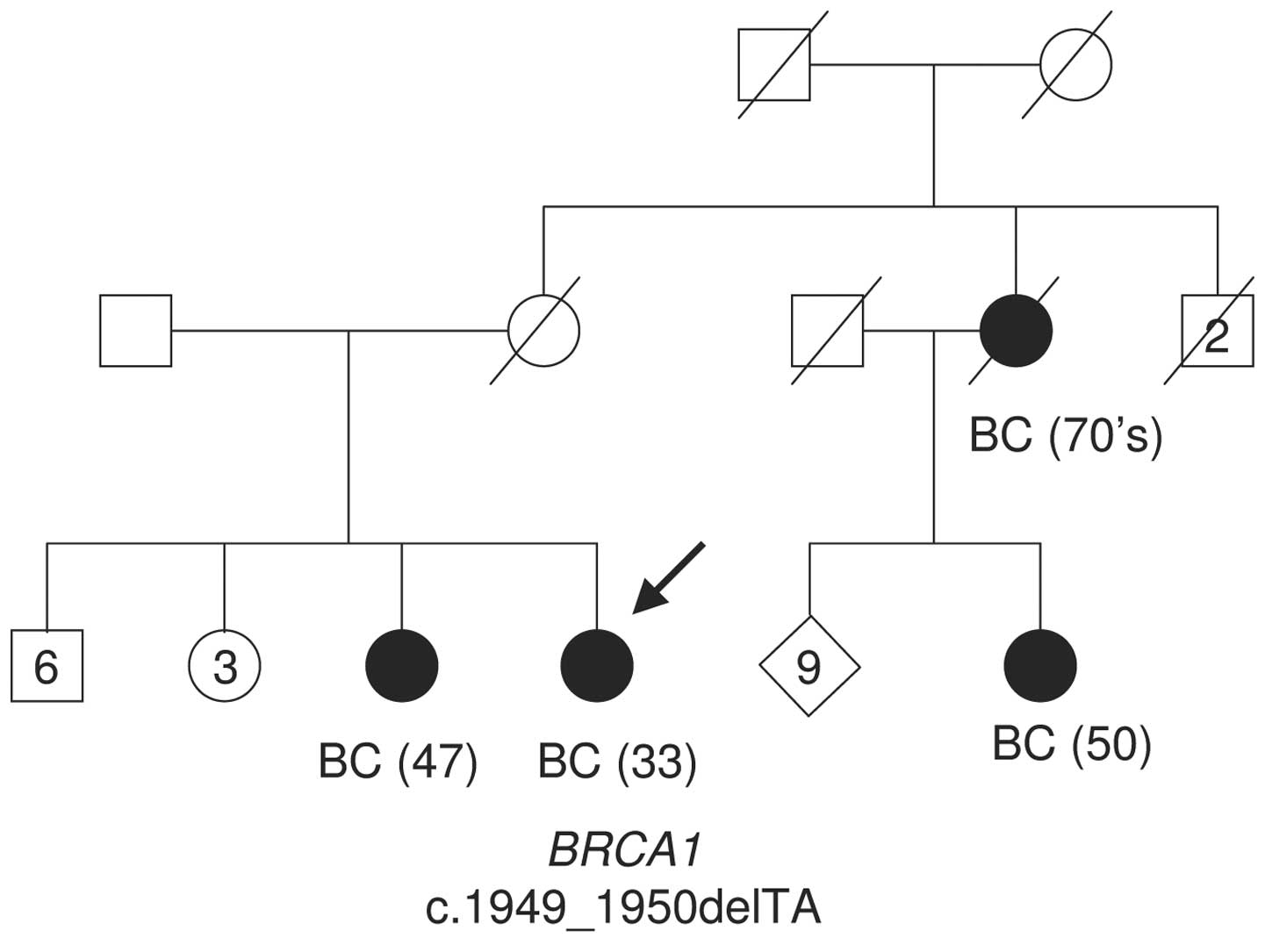
The proband was a 33 year-old premenopausal
Senegalese female of Wolof ethnicity, diagnosed with a
triple-negative (ER-, PgR-, HER2-) stage IV breast ductal
carcinoma. She had six male and four female siblings. One sister, a
maternal cousin and a maternal aunt succumbed to breast cancer at
the ages of 47, 50, and approximately 70, respectively (Fig. 1 shows the pedigree of the affected
family). The proband underwent genetic counseling and signed
informed consent for gene testing of germ-line BRCA1 and
BRCA2 mutations. The study was approved by the ethics
committee of the University Hospital Vall d’Hebron.
Methods
Genomic DNA was extracted from whole blood using a
Puregene Genome DNA purification kit (Gentra System, Minneapolis,
MN, USA). DNA was amplified by PCR using primers specific for the
coding sequence and intron/exon boundaries of the two genes.
Analysis of the entire coding and flanking sequences was carried
out by sequencing using a BigDye terminator V3.1 cycle sequencing
kit on a 3130xl Genetic Analyzer (Applied Biosystems, Foster City,
CA, USA). Both forward and reverse strands were sequenced. The
variant sequences described in our study were named according to
GenBank (BRCA1 NM_007294; BRCA2 NM_000059).
Results
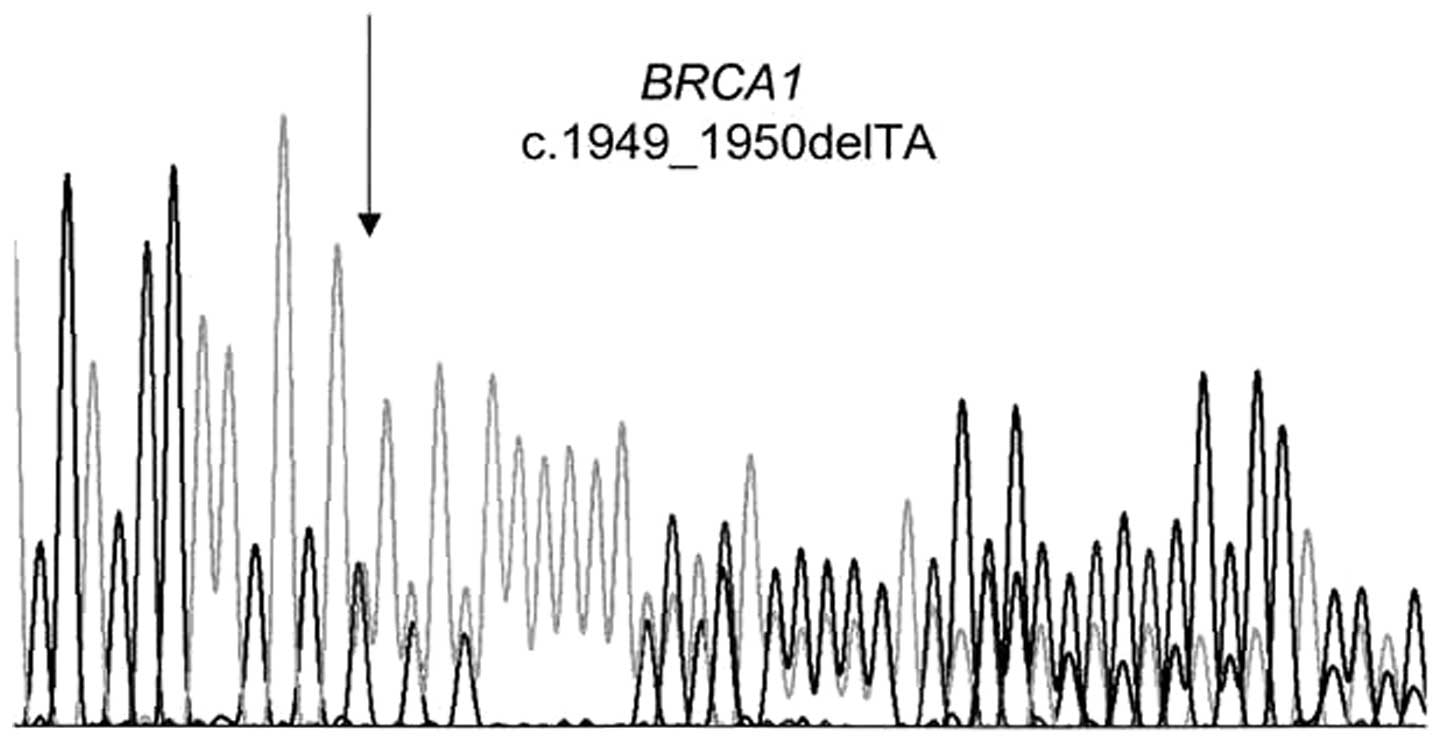
We identified in the exon 11 of the BRCA1
gene the novel mutation c.1949_1950delTA (p.Ile650LysfsX22) (HGVS
nomenclature) or 2068delTA (BIC database nomenclature) (Fig. 2) in the index case of a family with
hereditary breast cancer from Senegal.
To the best of our knowledge, this two-base pair
deletion is not present in the BIC database and has not previously
been published. Furthermore, the index case presented two
unclassified variants in BRCA2. The first is the novel
variant c.296-7dupT in the intron 2 of the gene, which is localized
in the poly T(10) of the splicing
acceptor site. This variant may cause the same effect as the
IVS2-7T>A known variant, which generates an alternative
transcript with the in-frame skipping of the exon 3, and is not
considered to be completely pathogenic due to its presence in
healthy populations (5). An RNA
sample from the proband and DNA from other family members to study
this variant were not available. The second variant, c.5710C>G
in exon 11 (5938C>G, BIC nomenclature), causes the p.L1904V
change in the BRCA2 protein. This variant has been reported
eight times in the BIC database. Although it has a weak probability
of altering splicing or protein functionality (Alamut Software
1.54, Interactive Biosoftware), this variant remains
unclassified.
Discussion
The majority of studies on BRCA1 and
BRCA2 genes have been performed in Caucasian women of
European descent, while little information exists regarding other
ethnicities. Nevertheless, several reports have described
BRCA1 and BRCA2 sequence variants in women of African
or African-American descent and have characterized a different
spectrum of BRCA1 and BRCA2 mutations from that of
the North American or European Caucasian population (1,6–18).
Such studies have revealed ancient mutations
originating in the African continent, such as the BRCA1
recurrent 943ins10 mutation. Haplotype analyses of 943ins10
(c.815_824dup10) carriers indicate that this mutation, which has
been identified in African-American and Latin-American/Caribbean
families, has an ancient African origin (8,13) in
the Ivory Coast in West Africa (7).
However, it is unclear whether some of the mutations reported are
unique to the African-American population. Of note, the 3036del4
mutation (c.2808_2811delACAA) in BRCA2, which was
recurrently present in different European populations (19) has also been identified in young
African women with breast cancer from Ibadan, Nigeria (14).
As of the end of 2010, the BIC database contains 38
BRCA1 and 31 BRCA2 different deleterious mutations
and a high number of sequence variants of unknown significance
identified in families of African descent. Moreover, other
mutations and variants identified in African/African-American
families have also been published. These data suggest a significant
level of genetic variation in BRCA1 and BRCA2 in
populations of African descent. The novel c.1949_1950delTA mutation
reported in the present study extends the knowledge of genetic
susceptibility to breast cancer in sub-Saharan African populations.
Furthermore, the finding of this mutation in a Senegalese family
can be of interest for other populations originating from western
Africa, as is the case for the vast majority of
African-Americans.
The incidence and the age of onset of breast cancer
vary across geographic regions and ethnic groups. Incidence and
average age of onset are lower in African-American women than among
Caucasian women (20,21). These differences may be attributed
to multiple environmental factors. Breast cancer incidence has
increased in the last decades in African populations (22), most likely due to the better
reporting and the adoption of a Western lifestyle in urban cities.
However, biological and genetic factors may also explain
variability between ethnicities. Studies in the United States have
documented that breast cancers in young pre-menopausal
African-American women were more aggressive, leading to a decrease
in the overall survival rates compared to Caucasian women (23–25).
However, little data is currently available
concerning the prevalence of BRCA1/2 mutations among
breast cancer patients indigenous to sub-Saharan countries. With
the exception of the Ashkenazi population, African-American women
diagnosed with breast cancer under the age of 35 years have been
reported to have a higher prevalence of BRCA1 mutations than
any age-matched racial/ethnic group (26). The higher BRCA1 mutation
prevalence correlates with their elevated rates of triple-negative
or basal-like breast cancer profile (27). These phenotypic traits are present
in the female Senegalese patient carrying the BRCA1
c.1949_1950delTA mutation described in our report, who was
diagnosed at 33 years of age with an ER-, PgR-, Her2-breast
tumor.
Bearing in mind that there is a significant
racial/ethnic variation in the spectrum of BRCA1/2
mutations, new data regarding BRCA1 and BRCA2 genetic
mutation prevalence and penetrance in African women is likely to
improve the performance of risk-assessment tools and
mutation-prediction models, and affect the clinical genetic testing
process and follow-up for individuals of African ancestry (28).
Acknowledgements
This article is dedicated to Dr A. Pelegrí, who
passed away during the preparation of the manuscript.
References
|
1
|
Miki Y, Swensen J, Shattuck-Eidens D, et
al: A strong candidate for the breast and ovarian cancer
susceptibility gene BRCA1. Science. 266:66–71. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wooster R, Bignell G, Lancaster J, et al:
Identification of the breast cancer susceptibility gene BRCA2.
Nature. 378:789–792. 1995. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Antoniou A, Pharoah PDP, Narod S, et al:
Average risks of breast and ovarian cancer associated with BRCA1 or
BRCA2 mutations detected in case series unselected for family
history: a combined analysis of 22 studies. Am J Hum Genet.
72:1117–1130. 2003. View
Article : Google Scholar
|
|
4
|
BIC. http://research.nhgri.nih.gov/bic.
|
|
5
|
Santarosa M, Viel A and Boiocchi M: Splice
variant lacking the transactivation domain of the BRCA2 gene and
mutations in the splice acceptor site of intron. Genes Chromosomes
Cancer. 26:381–382. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Futreal PA, Liu Q, Shattuck-Eidens D, et
al: BRCA1 mutations in primary breast and ovarían
carcinomas. Science. 266:120–122. 1994. View Article : Google Scholar
|
|
7
|
Arena JF, Smith S, Plewinska M, et al:
BRCA1 mutations in African American women (abstract). Am J
Hum Genet. (Suppl)59:A341996.
|
|
8
|
Stoppa-Lyonnet D, Laurent-Puig P, Essioux
L, et al: BRCA1 sequence variations in 160 individuals referred to
a breast/ovarian family cancer clinic. Institut Curie Breast Cancer
Group. Am J Hum Genet. 60:1021–1030. 1997.
|
|
9
|
Gao Q, Neuhausen S, Cummings S, et al:
Recurrent germ-line BRCA1 mutations in extended African American
families with early-onset breast cancer. Am J Hum Genet.
60:1233–1236. 1997.PubMed/NCBI
|
|
10
|
Newman B, Mu H, Butler LM, et al:
Frequency of breast cancer attributable to BRCA1 in a
population-based series of American women. JAMA. 279:915–921. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Panguluri RCK, Brody LC, Modali R, et al:
BRCA1 mutations in African Americans. Hum Genet. 105:28–31.
1999. View Article : Google Scholar
|
|
12
|
Dangel J, Wagner-Costalas J, Bove B, et
al: Novel germline BRCA1 mutation (155del4) in an African American
with early-onset breast cancer. Hum Mutat. 14:5451999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mefford HC, Baumbach L, Panguluri RC, et
al: Evidence for a BRCA1 founder mutation in families of
West African ancestry. Am J Hum Genet. 65:575–578. 1999.
|
|
14
|
Gao Q, Adebamowo CA, Fackenthal J, et al:
Protein truncating BRCA1 and BRCA2 mutations in African women with
pre-menopausal breast cancer. Hum Genet. 107:192–194. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gao Q, Tomlinson G, Das S, et al:
Prevalence of BRCA1 and BRCA2 mutations among clinic-based African
American families with breast cancer. Hum Genet. 107:186–191. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shen D, Wu Y, Subbarao M, Bhat H, Chillar
R and Vadgama JV: Mutation analysis of BRCA1 gene in
African-American patients with breast cancer. J Natl Med Assoc.
92:29–35. 2000.PubMed/NCBI
|
|
17
|
Nanda R, Schumm LP, Cummings S, et al:
Genetic testing in an ethnically diverse cohort of high-risk women:
a comparative analysis of BRCA1 and BRCA2 mutations in American
families of European and African ancestry. JAMA. 294:1925–1933.
2005. View Article : Google Scholar
|
|
18
|
Haffty DH, Choi S, Goyal A, et al: Breast
cancer in young women (YBC): prevalence of BRCA1/2 mutations and
risk of secondary malignancies across diverse racial groups. Ann
Oncol. 20:1653–1659. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Neuhausen SL, Godwin AK, Gershoni-Baruch
R, et al: Haplotype and phenotype analysis of nine recurrent BRCA2
mutations in 111 families: results of an international study. Am J
Hum Genet. 62:1381–1388. 1998. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
El-Tamer MB and Wait RB: Age at
presentation of African-American and Caucasian breast cancer
patients. J Am Coll Surg. 188:237–240. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jemal A, Siegel R, Ward E, et al: Cancer
statistics. CA Cancer J Clin. 56:106–130. 2006.
|
|
22
|
International Agency for Research on
Cancer. GLOBOCAN. 2002, http://www-dep.iarc.fr/.
|
|
23
|
MacMahon B: Epidemiology and the causes of
breast cancer. Int J Cancer. 118:2373–2378. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Adebamowo CA and Adekunle OO:
Case-controlled study of the epidemiological risk factors for
breast cancer in Nigeria. Br J Surg. 86:665–668. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Eley JW, Hill HA, Chen VW, et al: Racial
differences in survival from breast cancer: results of the National
Cancer Institute Black/White Cancer Survival Study. JAMA.
272:947–954. 1994. View Article : Google Scholar
|
|
26
|
John EM, Miron A, Gong G, et al:
Prevalence of pathogenic BRCA1 mutation carriers in 5 US
racial/ethnic groups. JAMA. 298:2869–2876. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Carey LA, Perou CM, Livasy CA, et al:
Race, breast cancer subtypes, and survival in the Carolina Breast
Cancer Study. JAMA. 295:2492–2502. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kurian AW, Gong GD, John EM, et al:
Performance of prediction models for BRCA mutation carriage in
three racial/ethnic groups: findings from the Northern California
Breast Cancer Family Registry. Cancer Epidemiol Biomarkers Prev.
18:1084–1091. 2009. View Article : Google Scholar
|