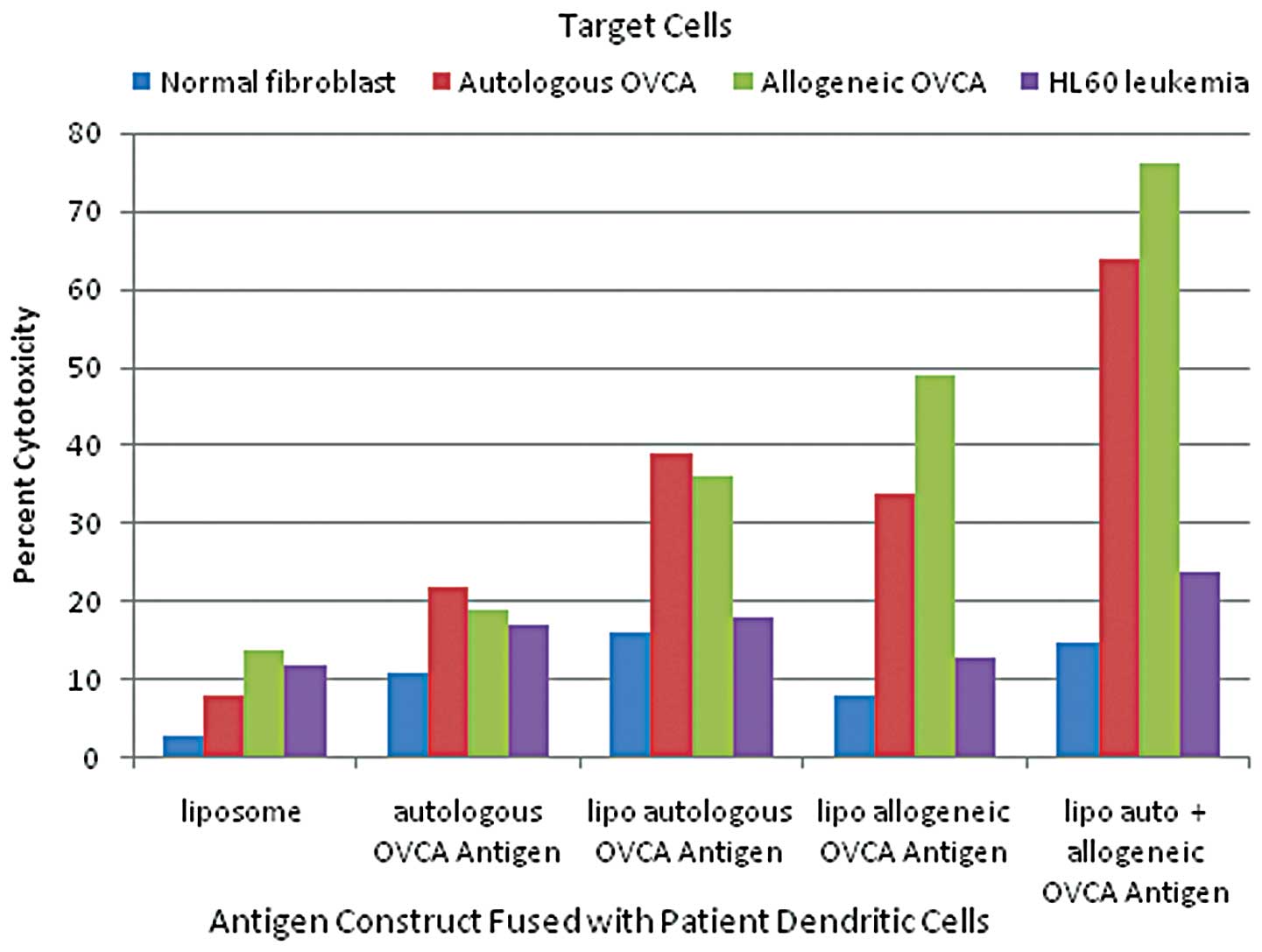
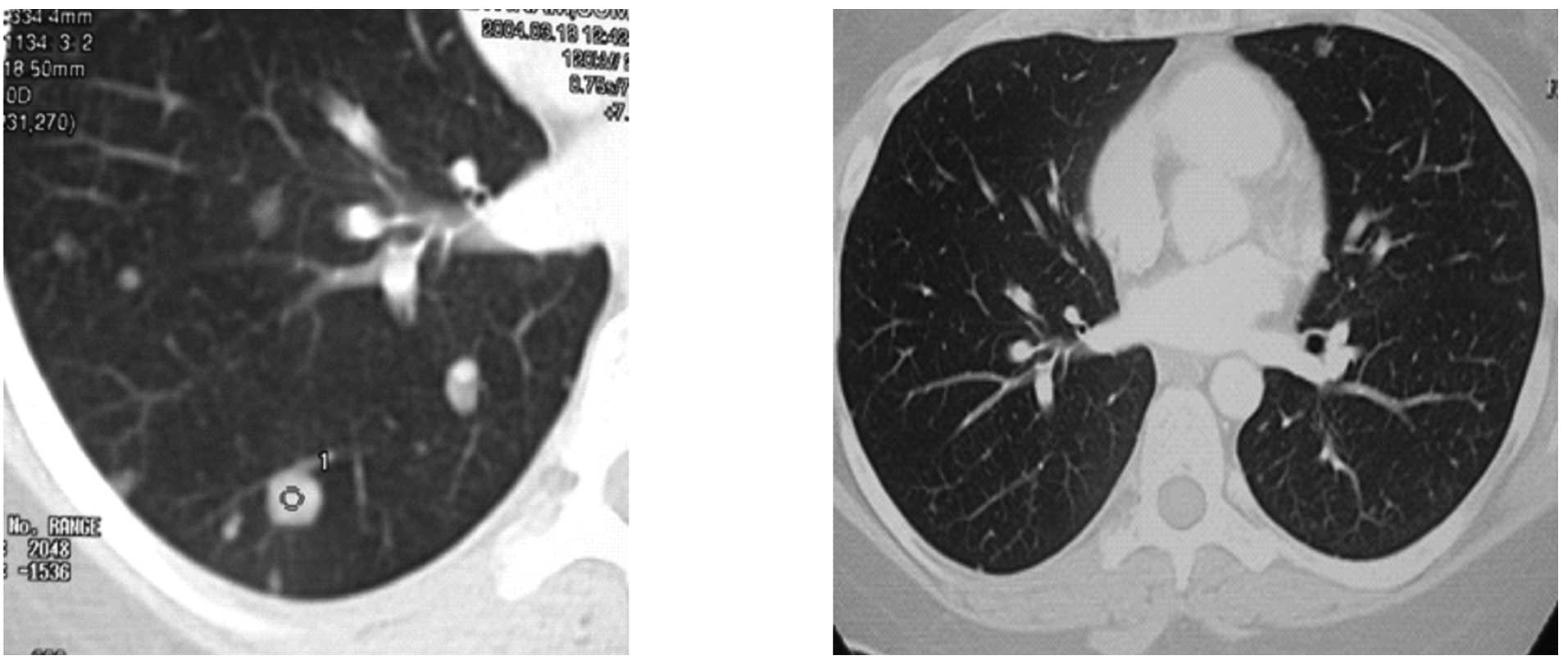
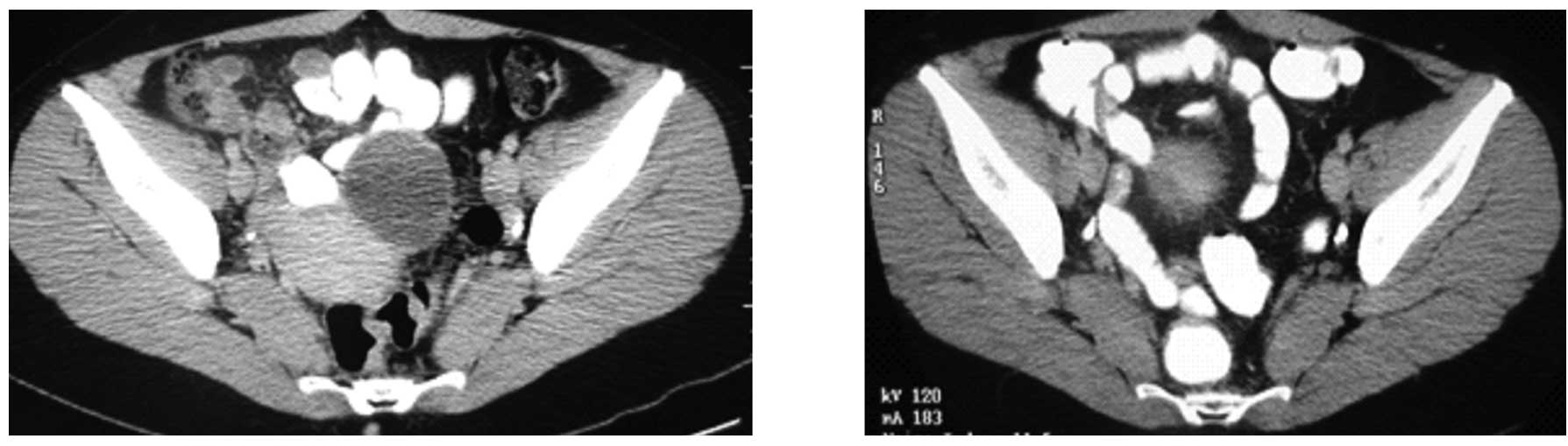
|
1
|
Jemal A, Murray T, Samuels A, Ghafoor A,
Ward E and Thun MJ: Cancer statistics. CA Cancer J Clin. 53:5–26.
2003.
|
|
2
|
McGuire WP, Hoskins WJ, Brady MF, Kucera
PR, Partridge EE, Look KY, Clarke-Pearson DL and Davidson M:
Cyclophosphamide and cisplatin compared with paclitaxel and
cisplatin in patients with stage III and stage IV ovarian cancer. N
Engl J Med. 334:1–6. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kirby TO, Huh W and Alvarez R:
Immunotherapy of ovarian cancer. Exp Opin Biol Ther. 2:409–417.
2002. View Article : Google Scholar
|
|
4
|
Ichige K, Perey L, Vogel CA, Buchegger F
and Kufe D: Expression of the DF3-P epitope in human ovarian
carcinomas. Clin Cancer Res. 1:565–571. 1995.PubMed/NCBI
|
|
5
|
Lu KH, Patterson AP, Wang L, et al:
Selection of potential markers for epithelial ovarian cancer with
gene expression arrays and recursive descent partition analysis.
Clin Cancer Res. 10:3291–3300. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
McKenzie SJ, DeSombre KA, Bast BS, et al:
Serum levels of HER-2 neu (C-erbB-2) correlate with overexpression
of p185neu in human ovarian cancer. Cancer. 71:3942–3946. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Peoples GE, Goedegebuure PS, Smith R,
Linehan DC, Yoshino I and Eberlein TJ: Breast and ovarian
cancer-specific cytotoxic T lymphocytes recognize the same
HER2/neu-derived peptide. Proc Nat Acad Sci USA. 92:432–436. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wagner U, Schlebusch H, Kohler S,
Schmolling J, Grunn U and Krebs D: Immunological responses to the
tumor-associated antigen CA125 in patients with advanced ovarian
cancer induced by the murine monoclonal anti-idiotype vaccine
ACA125. Hybridoma. 16:33–40. 1997. View Article : Google Scholar
|
|
9
|
Jacobs I and Bast RC Jr: The CA 125
tumour-associated antigen: a review of the literature. Hum Reprod.
4:1–12. 1989.PubMed/NCBI
|
|
10
|
Gusdon JP Jr, Homesley HD, Jobson VW and
Muss HB: Treatment of advanced ovarian malignancy with
chemo-immunotherapy using autologous tumor and Corynebacterium
parvum. Obstet Gynecol. 62:728–735. 1983.PubMed/NCBI
|
|
11
|
Dillman RO, Nayak SK, Brown JV, Mahdavi K
and Beutel LD: The feasibility of using short-term cultures of
ovarian cancer cells for use as autologous tumor cell vaccines as
adjuvant treatment of advanced ovarian cancer. Cancer Biother
Radiopharm. 14:443–449. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bachmann MF, Speiser DE, Mak TW and Ohashi
PS: Absence of costimulation and not the intensity of TCR signaling
is critical for the induction of T cell unresponsiveness in vivo.
Eur J Immunol. 29:2156–2166. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen L, Ashe S, Brady WA, et al:
Costimulation of antitumor immunity by the B7 counterreceptor for
the T lymphocyte molecules CD28 and CTLA-4. Cell. 71:1093–1102.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zheng P, Sarma S, Guo Y and Liu Y: Two
mechanisms for tumor evasion of preexisting cytotoxic T-cell
responses: lessons from recurrent tumors. Cancer Res. 59:3461–3467.
1999.PubMed/NCBI
|
|
15
|
Steinman RM: The dendritic cell system and
its role in immunogenicity. Annu Rev Immunol. 9:271–296. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Steinman RM: Dendritic cells and the
control of immunity: enhancing the efficiency of antigen
presentation. Mt Sinai J Med. 68:106–166. 2001.PubMed/NCBI
|
|
17
|
Young JW, Koulova L, Soergel SA, Clark EA,
Steinman RM and Dupont B: The B7/BB1 antigen provides one of
several costimulatory signals for the activation of CD4+ T
lymphocytes by human blood dendritic cells in vitro. J Clin Invest.
90:229–237. 1992.PubMed/NCBI
|
|
18
|
Inaba K, Witmer-Pack M, Inaba M, et al:
The tissue distribution of the B7-2 costimulator in mice: abundant
expression on dendritic cells in situ and during maturation in
vitro. J Exp Med. 180:1849–1860. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gong J, Nikrui N, Chen D, et al: Fusions
of human ovarian carcinoma cells with autologous or allogeneic
dendritic cells induce antitumor immunity. J Immunol.
165:1705–1711. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Koido S, Ohana M, Liu C, et al: Dendritic
cells fused with human cancer cells: morphology, antigen expression
and T cell stimulation. Clin Immunol. 113:261–269. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kaye S: Chemotherapy for ovarian cancer:
yesterday, today and tomorrow. Br J Cancer. 89:S1–S2. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dadmarz RD, Ordoubadi A, Mixon A, et al:
Tumor-infiltrating lymphocytes from human ovarian cancer patients
recognize autologous tumor in an MHC Class II-restricted fashion.
Cancer J Sci Am. 2:2631996.
|
|
23
|
Hayashi K, Yonamine K, Masuko-Hongo K, et
al: Clonal expansion of T cells that are specific for autologous
ovarian tumor among tumorinfiltrating T cells in humans. Gynecol
Oncol. 74:86–92. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang L, Conejo-Garcia JR, Katsaros D, et
al: Intratumoral T cells, recurrence, and survival in epithelial
ovarian cancer. N Engl J Med. 348:203–213. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Fujita K, Ikarashi H, Takakuwa K, et al:
Prolonged disease-free period in patients with advanced epithelial
ovarian cancer after adoptive transfer of tumor-infiltrating
lymphocytes. Clin Cancer Res. 1:501–507. 1995.
|
|
26
|
Nijman H, van Diest P, Poort-Keesom R, et
al: T cell infiltration and MHC I and II expression in the presence
of tumor antigens: an immunohistochemical study in patients with
serous epithelial ovarian cancer. Eur J Obstet Gynecol Reprod Biol.
94:114–120. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Freedman R and Platsoucas C: Immunotherapy
for peritoneal ovarian carcinoma metastasis using ex vivo expanded
tumor infiltrating lymphocytes. Cancer Treat Res. 82:115–146. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Schlienger K, Chu C, Woo E, et al: TRANCE-
and CD40 ligand matured dendritic cells reveal MHC class
I-restricted T cells specific for autologous tumor in late-stage
ovarian cancer patients. Clin Cancer Res. 9:1517–1527. 2003.
|
|
29
|
Pardoll D: Spinning molecular immunology
into successful immunotherapy. Nat Rev Immunol. 2:227–238. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Koidoa S, Nikruib N, Ohanac M, Xiac J,
Tanaka Y, Liuc C, Durfeec J, Lernerc A and Gong J: Assessment of
fusion cells from patient-derived ovarian carcinoma cells and
dendritic cells as a vaccine for clinical use. Gynecologic
Oncology. 99:462–471. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Adams M, Navabi H, Croston D, Coleman S,
Tabi Z, Clayton A, Jasani B and Mason MD: The rationale for
combined chemo/immunotherapy using a Toll-like receptor 3 (TLR3)
agonist and tumour-derived exosomes in advanced ovarian cancer.
Vaccine. 23:2374–2378. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Rosenberg S, Yang J and Restifo N: Cancer
immunotherapy: moving beyond current vaccines. Nat Med. 10:909–915.
2004. View
Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nowak A, Robinson B and Lake R: Synergy
between chemotherapy and immunotherapy in the treatment of
established murine solid tumors. Cancer Res. 63:4490–4496.
2003.PubMed/NCBI
|
|
34
|
Fattorossi A, Battaglia A, Ferrandina G,
et al: Neoadjuvant therapy changes the lymphocyte composition of
tumor-draining lymph nodes in cervical carcinoma. Cancer.
100:1418–1428. 2004. View Article : Google Scholar : PubMed/NCBI
|