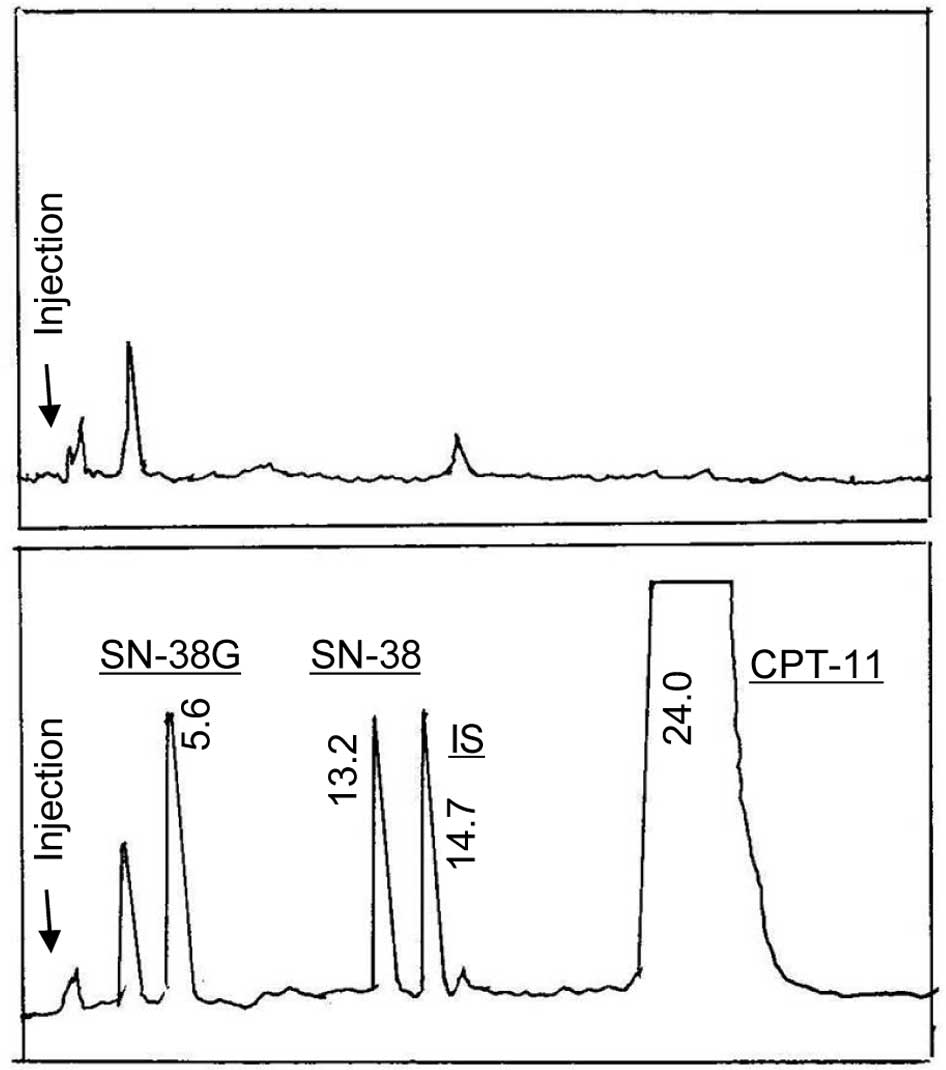
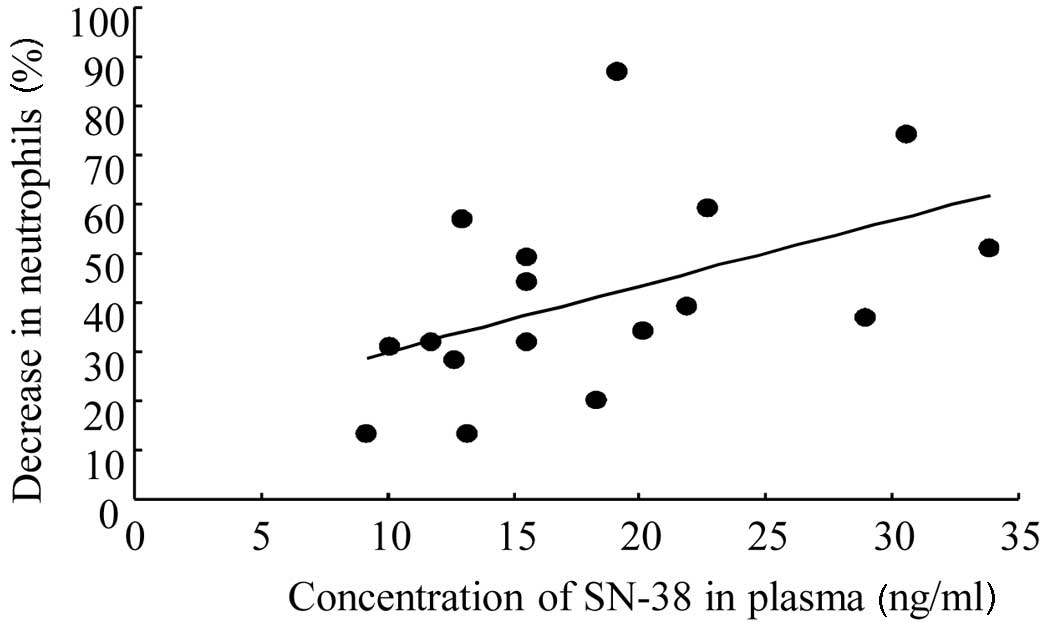
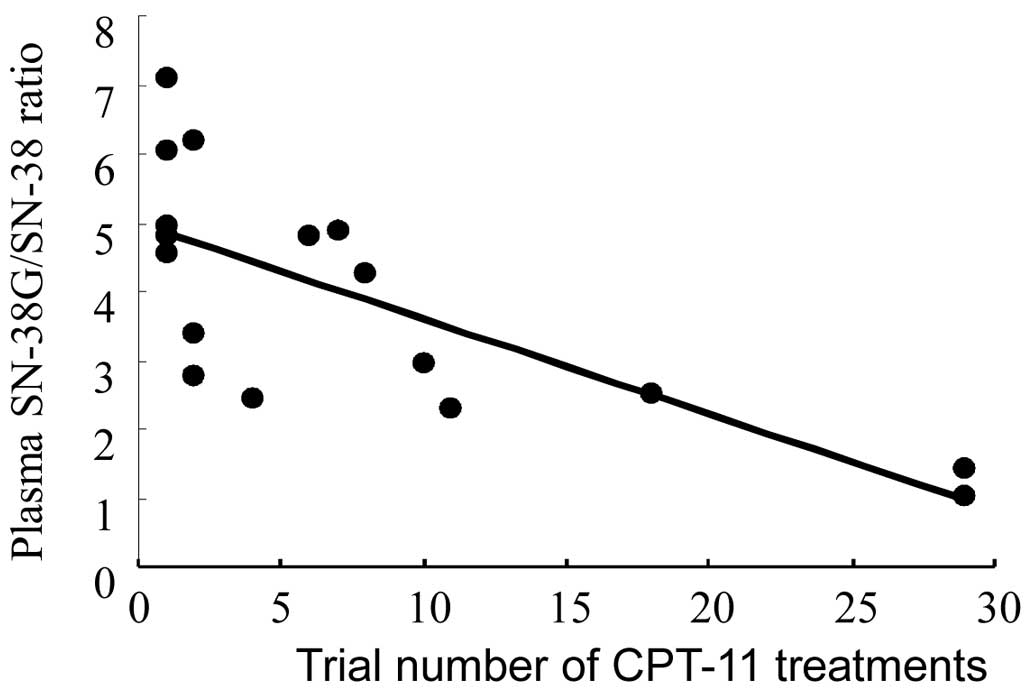
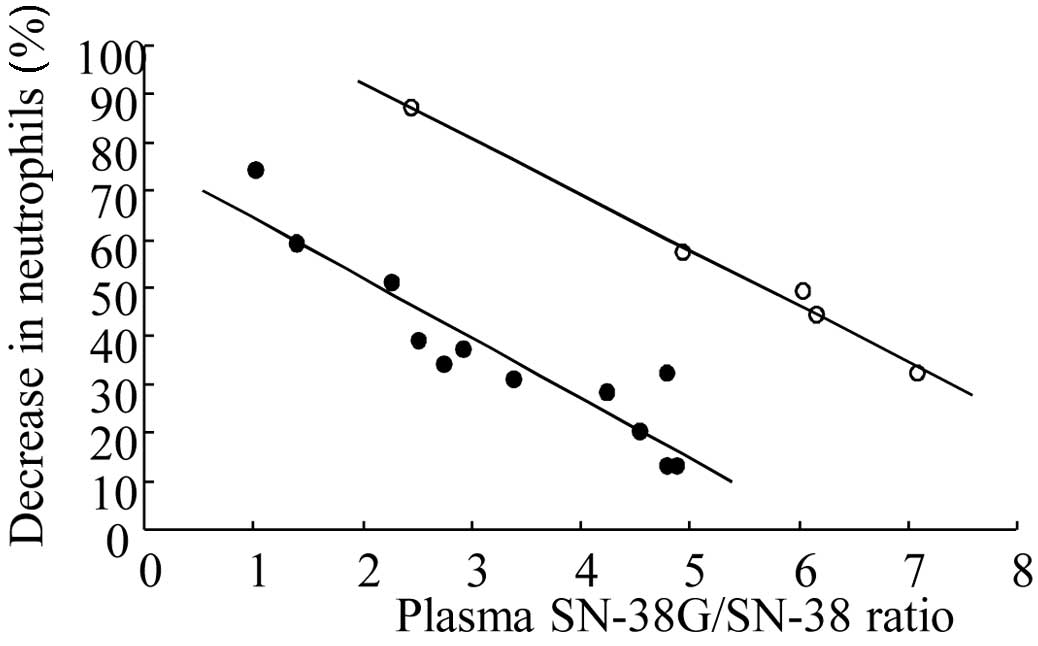
Spandidos Publications style
Hirose K, Kozu C, Yamashita K, Maruo E, Kitamura M, Hasegawa J, Omoda K, Murakami T and Maeda Y: Correlation between plasma concentration ratios of SN-38 glucuronide and SN-38 and neutropenia induction in patients with colorectal cancer and wild-type UGT1A1 gene. Oncol Lett 3: 694-698, 2012.
APA
Hirose, K., Kozu, C., Yamashita, K., Maruo, E., Kitamura, M., Hasegawa, J. ... Maeda, Y. (2012). Correlation between plasma concentration ratios of SN-38 glucuronide and SN-38 and neutropenia induction in patients with colorectal cancer and wild-type UGT1A1 gene. Oncology Letters, 3, 694-698. https://doi.org/10.3892/ol.2011.533
MLA
Hirose, K., Kozu, C., Yamashita, K., Maruo, E., Kitamura, M., Hasegawa, J., Omoda, K., Murakami, T., Maeda, Y."Correlation between plasma concentration ratios of SN-38 glucuronide and SN-38 and neutropenia induction in patients with colorectal cancer and wild-type UGT1A1 gene". Oncology Letters 3.3 (2012): 694-698.
Chicago
Hirose, K., Kozu, C., Yamashita, K., Maruo, E., Kitamura, M., Hasegawa, J., Omoda, K., Murakami, T., Maeda, Y."Correlation between plasma concentration ratios of SN-38 glucuronide and SN-38 and neutropenia induction in patients with colorectal cancer and wild-type UGT1A1 gene". Oncology Letters 3, no. 3 (2012): 694-698. https://doi.org/10.3892/ol.2011.533