Introduction
Concurrent chemoradiotherapy (CCRT) for oral
squamous cell carcinoma (OSCC) was recently established as an
effective treatment (1–5). In particular, therapy employing
super-selective intra-arterial infusion for head and neck cancers
has been shown to contribute to organ preservation and an increase
in the survival rate (6–12). However, a number of problems remain,
for example, the technical difficulty of intra-arterial infusion,
limitations due to the anatomical vascular distribution, local
tissue necrosis, edema, swelling and pain (6–8). S-1
is an oral anticancer drug in which the prodrug of 5-fluorouracil
(5-FU), tegafur, is combined with gimeracil, a potent antagonist of
the 5-FU-degrading enzyme dihydropyrimidine dehydrogenase, to
increase the antitumor effect of 5-FU, and oteracil potassium to
reduce digestive organ toxicity (1). Since the drug was approved for the
treatment of malignant tumors in the head and neck region in 2001,
the efficacy of the drug alone and in concurrent combination with
radiotherapy has been reported (13–18).
Previously, we performed chemoradiotherapy employing
super-selective intra-arterial infusion for patients with primary
OSCC as a preoperative treatment. However, we experienced the
complications of super-selective intra-arterial infusion, including
pharyngeal edema and necrosis, therefore we introduced CCRT with
S-1 from October 2005. In this study, to evaluate the significance
of CCRT with S-1 as a preoperative treatment, we compared the
efficacy and safety of the CCRTs employing S-1 and super-selective
intra-arterial infusion.
Patients and methods
Patients
Of the patients with primary OSCC who underwent
radical therapy at our department, 21 patients who underwent
neoadjuvant chemoradiotherapy employing super-selective
intra-arterial infusion of cisplatin (CDDP) or carboplatin (CBDCA)
between July 1997 and April 2006 [intra-arterial infusion (AI)
group] and 19 patients who underwent concurrent neoadjuvant
chemoradiotherapy with S-1 between October 2005 and December 2009
(S-1 group) participated in this study. There were slightly more
males in the AI group, but no significant difference was noted in
the mean age. The most frequent location of the OSCC in the S-1
group was the tongue, observed in 8 patients (42%), followed by the
gingiva in the lower jaw in 5 patients (26%). The gingiva in the
lower jaw was the most frequent OSCC location in the AI group,
observed in 9 patients (43%), followed by the tongue in 6 patients
(28%). No patient in the AI group had an upper gingival lesion.
Regarding the T and stage classifications, our department applies
preoperative CCRT in late T2 or worse cases. Accordingly, all cases
were T2 or worse in the S-1 group and only one case was T1 in the
AI group, showing no significant difference in the distribution of
T or stage classification between the two groups. The clinical
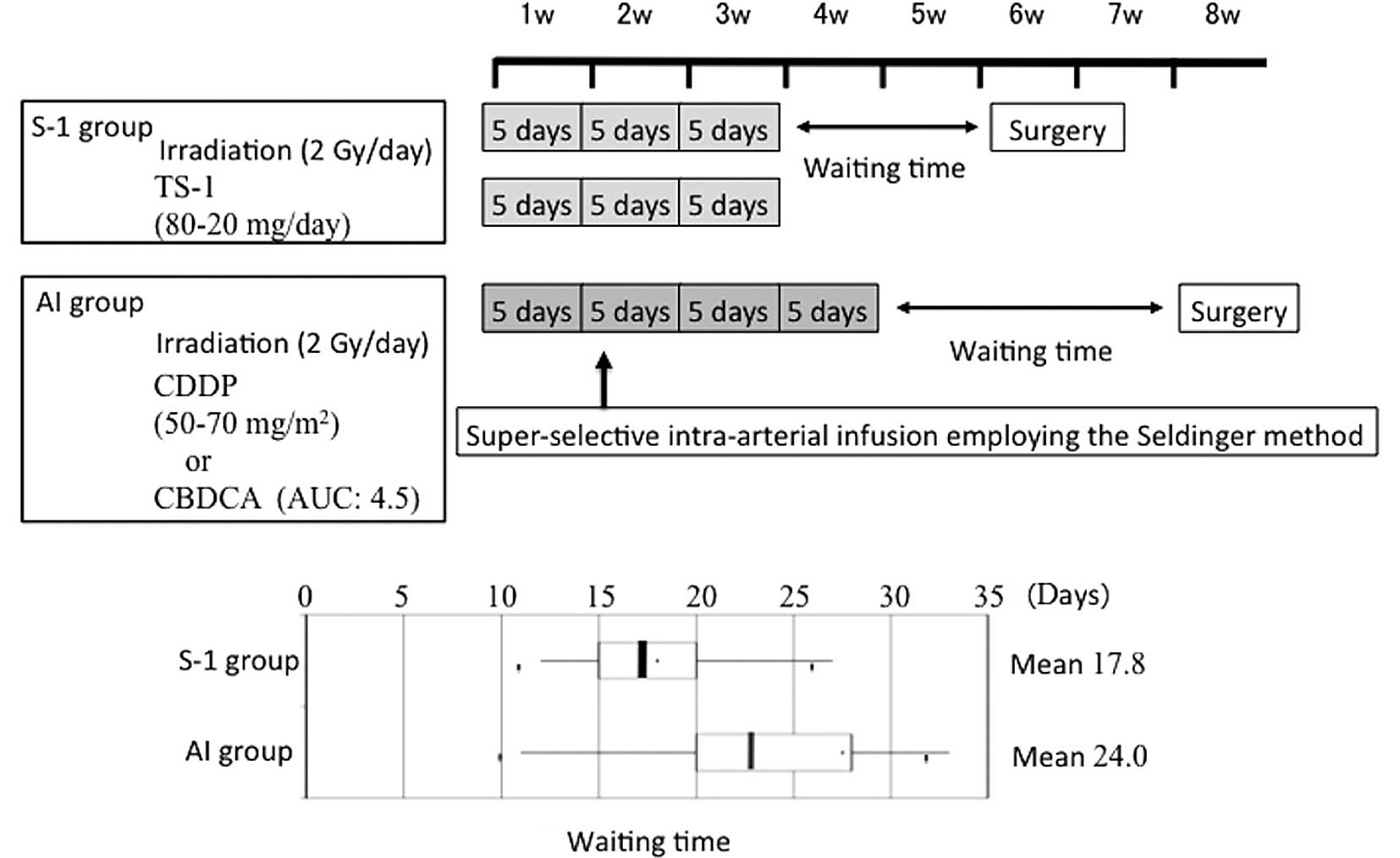
characteristics of the study population are shown in Table I. Fig.
1 shows the preoperative treatment schedules at the Department
of Oral and Maxillofacial Surgery, Kagoshima University. In CCRT
with S-1, external X-ray irradiation employing linac at 2 Gy was
performed 5 times a week for a total of 15 times, S-1 was orally
administered with irradiation and radical surgery was performed ~2
weeks after the preoperative treatment. In the AI group, external
X-ray irradiation employing linac at 2 Gy was performed 5 times a
week for a total of 20 times and super-selective intra-arterial
infusion was performed in the second week of radiotherapy, using
the Seldinger method. CDDP was typically used as the
chemotherapeutic drug, but CBDCA was administered to patients who
were suspected of having renal dysfunction. Radical surgery was
performed ~3 weeks after the completion of the preoperative
treatment. The actual waiting time required for surgery is shown in
the box plot of Fig. 1.
 | Table IClinical characteristics of
patients. |
Table I
Clinical characteristics of
patients.
| Characteristics | S-1 group (%) | AI group (%) |
|---|
| No. of patients | 19 | 21 |
| Gender
(male/female) | 10/9 | 13/8 |
| Median age in years
(range) | 68.5 (37–83) | 66.9 (39–84) |
| Primary tumor
site |
| Tongue | 8 (42) | 6 (28) |
| Lower gingiva | 5 (26) | 9 (43) |
| Upper gingiva | 2 (11) | 0 (0) |
| Buccal mucosa | 1 (5) | 2 (10) |
| Soft palate | 1 (5) | 1 (5) |
| Oral floor | 2 (11) | 3 (14) |
| Tumor
classification |
| T1 | 0 (0) | 1 (5) |
| T2 | 14 (73) | 11 (52) |
| T3 | 2 (11) | 3 (14) |
| T4 | 3 (16) | 6 (29) |
| Stage |
| I | 0 (0) | 1 (5) |
| II | 3 (16) | 7 (33) |
| III | 9 (47) | 6 (29) |
| IV | 7 (37) | 7 (33) |
The study was approved by the ethics committee of
Kagoshima University. Written informed consent was obtained from
all patients.
Areas of investigation
Histological effect following
neoadjuvant chemoradiotherapy
By employing the classification established by
Shimosato et al (Table II)
(19), the histological effect
following preoperative treatment was evaluated in each group.
Grades of IIb or higher were judged to be histologically
effective.
 | Table IIHistological gradings of the effects
of chemoradiotherapy. |
Table II
Histological gradings of the effects
of chemoradiotherapy.
| Grade | Histological
findings |
|---|
| I | Characteristic
changes are noted in the tumor cells, but the tumor structures have
not been destroyed. There is no detection of tumor nests due to the
lysis of individual tumor cells. |
| II | In addition to the
characteristic cell changes, the tumor structures have been
destroyed as a result of the disappearance of tumor cells. However,
a variable number of viable cells remain. |
| IIa | The destruction of
the tumor structures is mild; viable tumor cells are frequently
observed. |
| IIb | The destruction of
the tumor structures is severe; viable tumor cells are few in
number. |
| III | Markedly altered and
presumably non-viable tumor cells are present singly or in small
clusters and viable tumor cells are rarely observed. |
| IV | No tumor cells remain
in any sections (local cure). |
Residual tumor pattern
The residual tumor pattern in cases showing a GrII
histological effect was investigated following the classification
reported by Böheim et al (20). Malignant alveolar lesions
distributed directly under the mucoepithelium with a decreasing
size towards the superficial mucosal layer are designated as
superficial-type and conditions with cancer cells distributed in
the deep mucosa and mixed with degenerative necrotic lesions are
known as deep-type.
Disease-specific survival rate
The disease-specific survival rate, as determined
using the Kaplan-Meier method, was compared between the groups.
Adverse events
Adverse events of Gr2 or worse were compared between
the groups following the National Cancer Institute’s Common
Toxicity Criteria (NCI-CTC) v.3.0.
Statistical analysis
The Student’s t-test was used to compare the
response rates following neoadjuvant chemoradiotherapy of the S-1
and AI groups. A generalized Wilcoxon test and the log-rank test
were used to compare the disease-specific survival rate of the
groups. Statistical evaluations were performed with JMP®
statistical discovery software. P<0.05 was considered to
indicate a statistically significant result.
Results
Histological effect following neoadjuvant
chemoradiotherapy
The effects of the preoperative treatments in the
two groups, graded using the classification established by
Shimosato et al (19) were
determined (Fig. 2). The effect was
GrIIa or higher in all patients. In the S-1 group, GrIIb was most
frequently observed (10 cases, 52.6%) followed by GrIIa in 5 cases,
GrIII in 3 and GrIV in 1 case. In the AI group, the effect was
rated GrIII or GrIV in 11 cases, accounting for 52.4% of the AI
group patients and indicating a high histological therapeutic
effect, but this effect was rated GrIIa in 6 cases (25.6%),
exhibiting a bimodal histological effect. Regarding grades of IIb
or higher as effective, the response rates were 73.7 and 71.4% in
the S-1 and AI groups, respectively, revealing no significant
difference between the two groups.
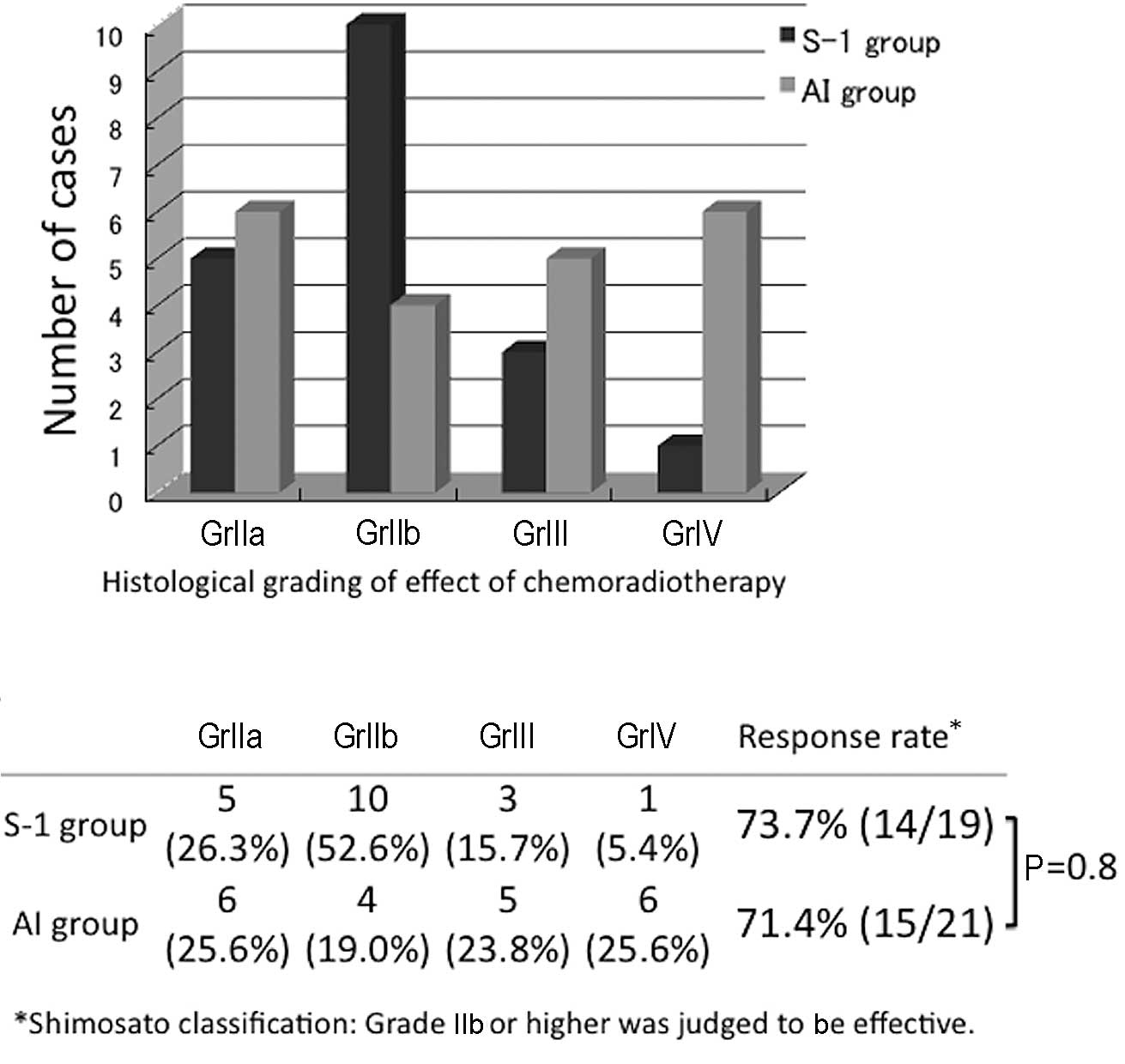 | Figure 2Comparison of the histological
gradings of the effects of chemoradiotherapy of the S-1 and AI
groups. In the S-1 group, GrIIb was most frequently observed (10
cases, 52.6%). In the AI group, the effect was rated GrIII or GrIV
in 11 cases, accounting for 52.4%, showing a high histological
therapeutic effect, but a bimodal histological effect. Regarding
grades of IIb or above as effective, the response rates were 73.7
and 71.4% in the S-1 and AI groups, respectively, showing no
significant difference between the two groups. S-1, concurrent
neoadjuvant chemoradiotherapy with S-1 and a total external
irradiation dose of 30 Gy; AI, concurrent neoadjuvant
chemoradiotherapy using super-selective intra-arterial infusion
with cisplatin or carboplatin and a total external irradiation dose
of 40 Gy; Shimosato classification carried out according to the
criteria of Shimosato et al (19). |
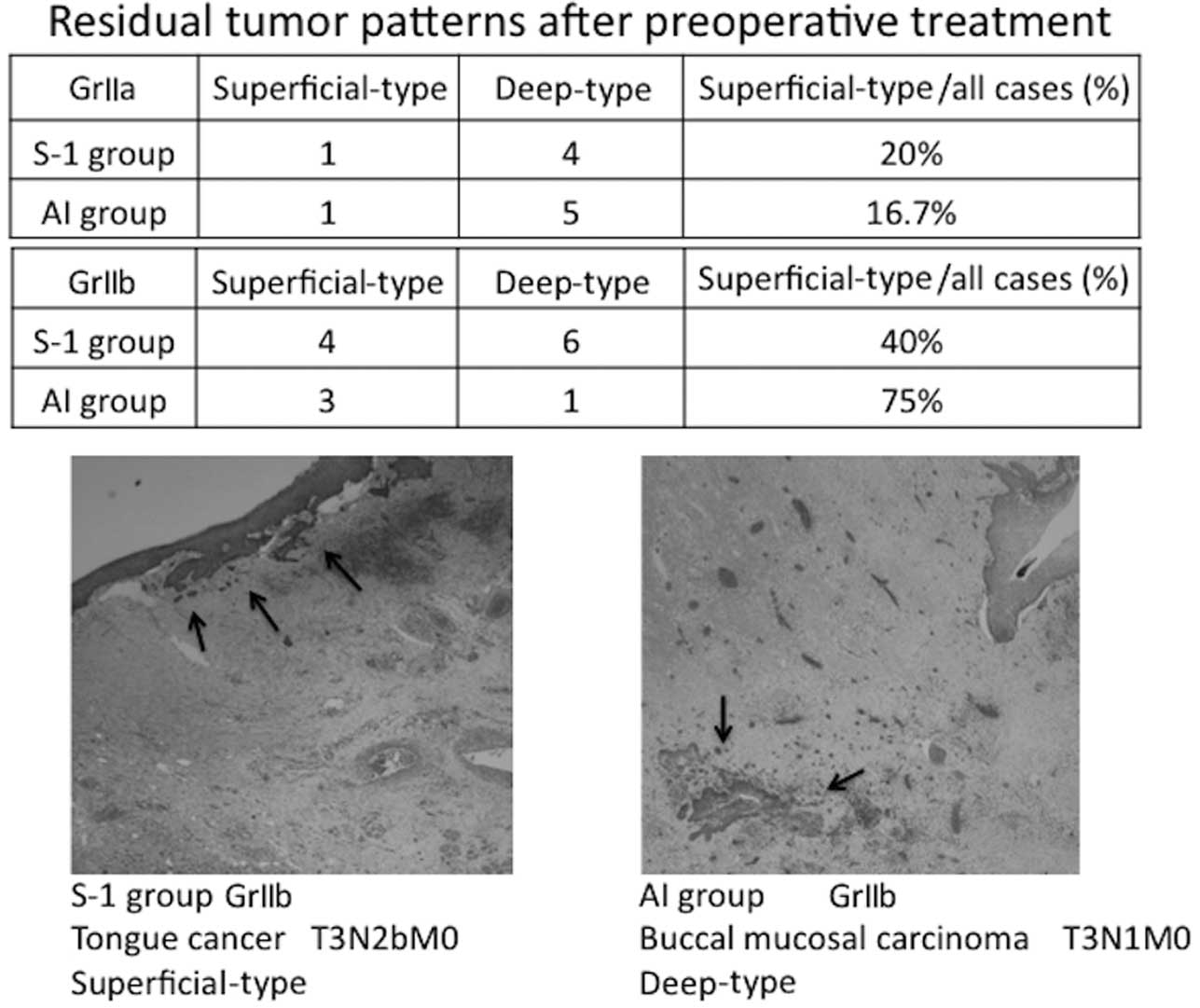
Residual tumor pattern following
preoperative treatment
The residual tumor patterns following the
preoperative treatment are shown in Fig. 3.
The residual tumor patterns of the GrII cases, in
which viable cancer cells remained, were classified as superficial-
or deep-type based on the classification reported by Böheim et
al (20). The GrIIa cases were
mostly deep-type in the two groups. The evaluation of the GrIIb
cases was limited due to the small number of cases, but 4 and 6
cases were superficial- and deep-type in the S-1 group,
respectively, showing that the superficial type accounted for 40%
of the GrIIb cases in the S-1 group. In the AI group, 3 cases were
superficial-type, accounting for 75% of the GrIIb cases. The lower
panel of Fig. 3 shows the
pathological findings in a superficial-type GrIIb case in the S-1
group and a deep-type GrIIb case in the AI group. The
superficial-type tumor was reduced towards the mucoepithelium,
whereas the deep-type tumor cells remained in the degenerative
tissue.
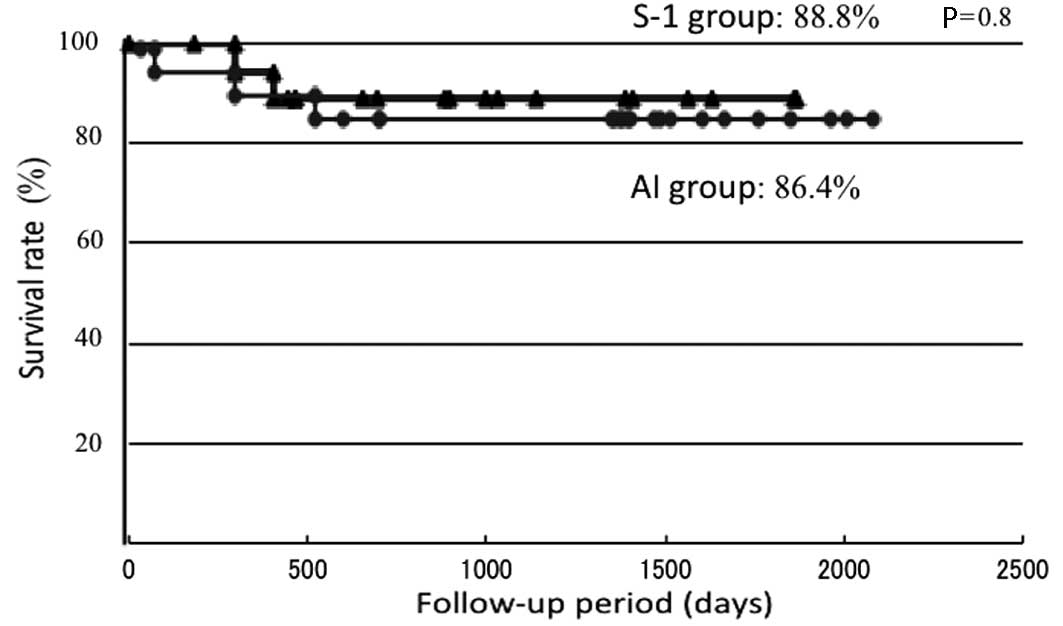
Disease-specific survival rate
The disease-specific survival rates were determined
using the Kaplan-Meier method (Fig.
4). The disease-specific survival rates were 88.8 and 86.4% in
the S-1 and AI groups, respectively, showing a similar therapeutic
outcome. Regarding the causes of mortality, 1 patient succumbed to
the primary lesion and another to distant metastasis in the S-1
group and 3 patients succumbed to the primary lesion in the AI
group.
Adverse events
S-1 was withdrawn due to neutropenia (1 case) and
diarrhea (1 case) in the S-1 group. All patients recovered
following the discontinuation of administration and no patients had
their surgery postponed or canceled.
Table III shows
NCI-CTC v.3.0 Gr2 or above adverse events. The most frequent
adverse event in the two groups was stomatitis, with incidences of
73.7 and 85.7% in the S-1 and AI groups, respectively. Gr3
stomatitis was noted in 1 patient in the S-1 group. Regarding
hematological toxicity, leukopenia occurred in 2 patients (10.5%)
and neutropenia in 1 patient (5.2%) in the S-1 group, whereas
hypochromia occurred in 2 patients (14.2%), leukopenia in 2 (14.2%)
and neutropenia in 2 (14.2%) in the AI group, and Gr3 neutropenia
was noted in 1 patient. In addition, complications of pharyngeal
edema and necrosis were noted in the AI group.
 | Table IIINCI-CTC v.3.0 grade 2 or above adverse
events. |
Table III
NCI-CTC v.3.0 grade 2 or above adverse
events.
| Adverse events | Gr2 | Gr3 | Gr4 | % |
|---|
| S-1 group |
| Leukopenia | 2 | 0 | 0 | 10.5 |
| Neutropenia | 1a | 0 | 0 | 5.2 |
| Fatigue | 3 | 0 | 0 | 15.7 |
| Vomiting | 2 | 0 | 0 | 10.5 |
| Diarrhea | 0 | 1a | 0 | 5.2 |
| Stomatitis | 13 | 1 | 0 | 73.7 |
| Elevation of
bilirubin level | 1 | 0 | 0 | 5.2 |
| AI group |
| Hypochromia | 2 | 0 | 0 | 14.2 |
| Leukopenia | 2 | 0 | 0 | 14.2 |
| Neutropenia | 1 | 1 | 0 | 14.2 |
|
Thrombocytopenia | 1 | 0 | 0 | 7.1 |
| Fatigue | 3 | 0 | 0 | 21.4 |
| Nausea | 2 | 0 | 0 | 14.2 |
| Vomiting | 0 | 1 | 0 | 7.1 |
| Stomatitis | 12 | 0 | 0 | 57.1 |
| Soft tissue
impairment | 1b | 1c | 0 | 9.5 |
Discussion
Successful treatment for oral cancer should retain
esthetic appearance and conserve function, unlike the treatment of
malignant tumors in other regions, in addition to facilitating a
high survival rate. The response rate of primary lesions and the
organ conservation rate have increased with the development of
potent chemotherapeutic drugs and regimens for concomitant drug
administration and advancements in administration methods, for
example chemotherapy by super-selective intra-arterial infusion
(8–12). According to the oral cancer
treatment guidelines in Japan, platinum-based drugs, including
CDDP, are widely used in chemotherapy as the standard, but S-1 is
also used. In a meta-analysis of 32 clinical studies on CCRT for
advanced head and neck cancers reported in Germany in 2006
(18), 5-FU alone and in
combination with other drugs, including CDDP, exhibited a marked
effect. However, the incidence of oral cancer in the elderly has
increased in Japan. Selecting the effective dose and administration
method of chemotherapeutic drugs for elderly patients who may have
a number of underlying problems is difficult due to the severe
adverse events and complications caused by super-selective
intra-arterial infusion, including hemorrhage and edema (21). S-1 is an oral anticancer drug in
which the antitumor effect of 5-FU is strengthened and digestive
organ toxicity is reduced. Since the administration of S-1 is
relatively simple, the drug has been administered for advanced and
non-advanced oral cancer in elderly patients, and cases which
responded to S-1 alone and CCRT (13) and its efficacy as a preoperative
treatment (16) have been reported.
However, there have been few studies that have performed a
comparison with other chemoradiotherapies, for instance
chemoradiotherapy using super-selective intra-arterial infusion. To
evaluate CCRT with S-1 as preoperative treatment for OSCC, we
compared the efficacy and safety of CCRT employing super-selective
intra-arterial infusion previously administered at our department
and CCRT with S-1.
Several conclusions were derived from our clinical
analyses: i) no significant difference was found in the
histological response rate of the two groups; ii) the residual
tumor pattern was slightly superior in the AI group, although we
should be cautious due to the limited number of surgeries; iii) no
significant difference in the disease-specific survival rates of
the groups was found; and iv) no serious adverse event
occurred.
In the S-1 group, the histological effect was rated
GrIII or GrIV in only 4 patients (21.4%), but GrIIb effects were
observed in 10 cases, accounting for the greatest proportion
(52.6%). Regarding grades of IIb or higher as effective, the
histological response rate was 73.7%, which is similar to the
findings reported by Matsui et al (16). Enhancement of the effect of
radiotherapy by S-1 in clinical head and neck cancer cases has been
reported (13,16) and Zeng et al (22) investigated its mechanism in a basic
experiment in which S-1 inhibited the activation of
irradiation-induced HIF-1. This inhibition resulted in a reduced
microvascular density and increased apoptosis, which significantly
increased the irradiation sensitivity in the presence of S-1
compared with that in radiotherapy alone. GrIII and IV curative
effects were observed in more patients in the AI compared to the
S-1 group, but GrIIa with a residual tumor was also noted in a
number of cases. This finding is not contradictory to improvements
in the response rate of primary lesions and the organ conservation
rate, but the effect of the chemotherapeutic drug may have been
insufficient in certain regions due to the technical difficulty of
intra-arterial infusion depending on the location of the primary
lesion and limitations caused by the anatomical vascular
distribution and the number of feeder blood vessels. Therefore, the
histological response rate to CCRT with S-1 is high, although fewer
cases showed a marked improvement compared with the AI group. The
potentiation mechanism of concomitant treatment with irradiation
has been elucidated and suggests that the therapy may be used to
enhance the histological therapeutic effect.
Regarding the residual tumor pattern following CDDP-
based preoperative CCRT, Kirita et al (23) reported that the residual tumor cells
were mostly localized in the central superficial layer of the
primary lesion in complete response cases of tongue carcinoma,
suggesting the possibility of limited surgery. By contrast, Böheim
et al (20) analyzed the
residual pattern of viable cancer cells and found that it could be
classified into 2 types: superficial, in which the tumor cell
distribution narrows towards the mucosal superficial layer, and
deep, in which the tumor cells are present in the deep mucosa. In
our GrII cases, the deep type was most frequently observed in cases
that achieved a GrIIa histological effect in the AI and S-1 groups,
showing no significant difference between the groups. However, in
GrIIb cases, 4 (40%) were superficial-type in the S-1 group,
whereas the superficial type accounted for 75% in the AI group,
although the number of cases was small. The therapeutic effect on
the primary lesion was suggested to be higher in the AI group when
an effect was obtained, retaining the possibility of limited
surgery. However, caution should be exercised with regard to its
application in the two groups, as indicated by the findings.
The disease-specific survival rates during the
follow-up period were 88.8 and 86.4% in the S-1 and AI groups,
respectively, showing a similar survival curve with no significant
difference in the survival rate (Fig.
4). Few studies have compared two regimens of chemoradiotherapy
performed at the same facility during the same period in Japan. In
their study, Kuratomi et al (24) compared chemoradiotherapies with S-1
and low-dose CDDP venous injection for resectable pharyngeal and
laryngeal cancers and the disease-specific survival rates were
found to be 77 and 76%, respectively. Although a simple comparison
with the results of this study is impossible as the location of the
primary lesions was different, the effect of chemoradiotherapy with
S-1 for head and neck cancer may be comparable to therapy employing
super- selective intra-arterial infusion.
Regarding adverse events, stomatitis had the highest
incidence in the S-1 group, as previously reported (16,17),
with Gr2 or worse stomatitis noted in 73.7% of the patients
(Table III). Gr3 stomatitis was
noted in 1 patient, but it was reversible and treatment was not
discontinued due to stomatitis in any patient during the
observation period. In the AI group, the frequency of stomatitis
was 57.1%, similar to that in the S-1 group, but no Gr3 case was
present. Regarding hematological toxicity in the S-1 group, Ohnishi
et al (17) reported that
the incidences of Gr3 or worse leukopenia and anemia were 5.2 and
2.6%, respectively, and Shirasaki et al (15) reported that Gr2 adverse events were
hypochromia (35%), leukopenia (35%) and thrombocytopenia (5%), but
no Gr3 or worse event occurred. In our patients, no Gr3 or worse
hematological toxicity was observed and the frequency of adverse
events was slightly lower than those reported. Kishimoto et
al (21) reported that
treatment of dermatitis and stomatitis was necessary in CCRT with
S-1 for elderly patients, but the treatment method was suitable for
elderly patients as the drug could be administered while observing
adverse events. However, since renal function is generally reduced
in the elderly, the lower level of renal excretion of gimeracil
results in an increased 5-FU level. This may then influence the
therapeutic effect and lead to severe bone marrow inhibition and
lung disorders, including interstitial pneumonia (25), demonstrating that S-1 administration
should be carefully managed. In the AI group, the effects of bone
marrow inhibition, including anemia, leukopenia, neutropenia and
thrombocytopenia, were observed. Gr3 neutropenia was noted in 1
patient, but the condition was improved with the administration of
a G-CSF preparation. Bone marrow inhibition was also widely
observed in a study reported by Furutani et al (26), in which numerous Gr3 and 4 adverse
events occurred, showing the necessity of investigating the optimum
dose of chemotherapeutic drugs. Moreover, severe complications
induced by super-selective intra-arterial infusion chemotherapy,
including laryngeal edema and mucosal necrosis, have been reported
(6,7). As pharyngeal edema and necrosis were
also evident in our previous study (?), sufficient attention should
be paid to the application of super-selective intra-arterial
infusion chemotherapy. Regarding the S-1 administration method,
Harada et al (13) altered
the regimen from a 4-week administration followed by a 2-week
withdrawal to a 2-week administration followed by a 1-week
withdrawal, thereby improving the adverse events and avoiding the
suspension or dose reduction of S-1 and the withdrawal of
irradiation. In our S-1 group, the drug was administered for 15
days with irradiation and the mean waiting time for surgery was
17.8 days. A wait of longer than 3 weeks was necessary for certain
patients, but the treatment was mostly completed as scheduled,
suggesting that 3-week administration does not cause marked changes
in adverse events.
The comparison between the S-1 and AI groups was
limited as the number of cases was small and the location and stage
of the primary lesion varied. An analysis involving an increased
number of cases is necessary. However, the response and survival
rates were not significantly different between the S-1 and AI
groups, although the proportion of markedly improved cases was
slightly different. The frequency of hematological toxicity and the
incidence of complications were low in CCRT with S-1, suggesting
that this therapy was effective and sufficiently beneficial for
oral cancer as a preoperative treatment.
References
|
1
|
Shirasaka T: Development history and
concept of an oral anticancer agent S-1(TS-1®): its
clinical usefulness and future vistas. Jpn J Clin Oncol. 39:2–15.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fuwa N: Current and future state of
chemoradiotherapy for head and neck cancer. Nihon Igaku Hoshasen
Gakkai Zasshi. 62:65–72. 2002.(In Japanese).
|
|
3
|
Choong N and Vokes E: Expanding role of
the medical oncologist in the management of head and neck cancer.
CA Cancer J Clin. 58:32–58. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bernier J, Domenge C, Ozsahlin M, et al:
Postoperative irradiation with or without concomitant chemotherapy
for locally advanced head and neck cancer. N Engl J Med.
350:1945–1952. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Klug C, Berzaczy D, Voracek M, et al:
Preoperative chemoradiotherapy in the management of oral cancer: A
review. J Craniomaxillofac Surg. 36:75–88. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yoshizaki T, Tanaka F, Shiga H, et al:
Superselective intra-arterial chemotherapy for head and neck
squamous cell carcinoma. Head and Neck Cancer. 29:445–449. 2003.
View Article : Google Scholar
|
|
7
|
Shiga K, Yokoyama J, Tateda M, et al:
Superselective intraarterial chemotherapy for patients with head
and neck tumor. Head and Neck Cancer. 29:457–462. 2003. View Article : Google Scholar
|
|
8
|
Imai S, Gyoten M, Kajihara Y, et al:
Superselective intraarterial chemotherapy using
cisplatin(CDDP)-carboplatin(CBDCA) combined with radiotherapy for
head and neck cancer. Head and Neck Cancer. 29:463–467. 2003.
View Article : Google Scholar
|
|
9
|
Endo S, Suzuki S, Tsuji K, et al:
Intraarterial concomitant chemoradiation for tongue cancer:
analysis of 20 patients. Nippon Jibiinkoka Gakkai Kaiho.
108:689–693. 2005.(In Japanese).
|
|
10
|
Tohnai I: Intra-arterial chemotherapy for
head and neck cancer. Gan To Kagaku Ryoho. 32:2024–2029. 2005.(In
Japanese).
|
|
11
|
Tonai I, Mitsudo K, Nishiguchi H, et al:
Daily concurrent chemoradiotherapy using superselective
intra-arterial infusion via superficial temporal
artery-preoperative therapy for stage III, IV oral cancer. Head and
Neck Cancer. 31:413–418. 2005.
|
|
12
|
Mitsudo K, Shigetomi T, Fukui T, et al:
Daily concurrent chemoradiotherapy with docetaxel (DOC) and
cisplatin (CDDP) using superselective intra-arterial infusion via
superficial temporal artery for advanced oral cancer. J Clin Oncol.
abse170202009.
|
|
13
|
Harada K, Kawashima Y, Uchida D, et al: A
case of advanced oral squamous cell carcinoma responding to
concurrent radiotherapy. Gan To Kagaku Ryoho. 34:745–747. 2007.(In
Japanese).
|
|
14
|
Tsukuda M, Kida A, Fujii M, et al:
Long-term results of S-1 administration as adjuvant chemotherapy
for advanced head and neck cancer. Gan To Kagaku Ryoho.
34:1215–1225. 2007.(In Japanese).
|
|
15
|
Shirasaki T, Maruya S, Namba A, et al:
Treatment results of chemotherapy with S-1 for head and neck
cancer. Gan To Kagaku Ryoho. 36:237–240. 2009.(In Japanese).
|
|
16
|
Matsui R, Mukai H, Imamura H, et al:
Clinical evaluation of anti-tumor effect in concurrent radiotherapy
with TS-1 for oral cancer (abstract). Jpn J Oral Maxillofac Surg.
55:572009.
|
|
17
|
Ohnishi K, Shioyama Y, Nakamura K, et al:
Concurrent chemoradiotherapy with S-1 as first-line treatment for
patients with oropharyngeal cancer. J Radiat Res. 52:47–53. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Budach W, Hehr T, Budach V, et al: A
meta-analysis of hyperfractionated and accelerated radiotherapy and
combined chemotherapy and radiotherapy regimens in unresectable
locally advanced squamous cell head and neck cancer. BMC Cancer.
6:282006. View Article : Google Scholar
|
|
19
|
Shimosato Y, Oboshi S and Baba K:
Histological evaluation of effects of radiotherapy and chemotherapy
for carcinoma. Jpn J Clin Oncol. 1:19–35. 1971.
|
|
20
|
Böheim K and Spoendkin H: The effect of
chemotherapy in relation to pathohistological tumor grading in head
and neck cancer. Acta Otorhinolaryngol. 238:197–204.
1983.PubMed/NCBI
|
|
21
|
Kishimoto K, Yoshida S, Domaem S, et al:
Concurrent chemoradiotherapy with TS-1 for advanced oral
cancer-Feasibility, problems, and countermeasures for elderly
patients with complication. Jpn J Oral Maxillofac Surg. 55:177–183.
2009.(In Japanese).
|
|
22
|
Zeng L, Ou G, Itasaka S, et al: TS-1
enhances the effect of radiotherapy by suppressing
radiation-induced hypoxia-inducible factor-1 activation and
inducing endothelial cell apoptosis. Cancer Sci. 99:2327–2335.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kirita T, Ohgi K, Kawakami M, et al:
Primary tumor resection of tongue carcinoma based on response to
preoperative therapy. Int J Oral Maxillofac Surg. 31:267–272. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kuratomi Y, Satoh S, Monji M, et al: A
comparative study of concurrent chemoradiotherapy with S-1 or CDDP
for pharyngeal or laryngeal cancer. Gan To Kagaku Ryoho.
37:1471–1476. 2010.(In Japanese).
|
|
25
|
Yamamo N, Ohshima T, Sato T, et al: A case
of interstitial pneumonia after S-1 administration for gastric
cancer. Gan To Kagaku Ryoho. 35:1935–1937. 2008.(In Japanese).
|
|
26
|
Furutani K, Fuwa N, Kodaira T, et al:
Combination therapy of continuous intra-arterial chemotherapy and
radiotherapy for stage III, IV tongue cancer. Head and Neck Cancer.
37:419–423. 2005.
|