Introduction
Angiogenesis is the process of new blood vessel
formation from pre-existing vasculature, and is essential for new
organ development, differentiation and growth (1,2).
Angiogenesis plays an important role in the growth and metastasis
of solid tumors (1,3). It is a complex process which must be
accurately regulated by pro- and antiangiogenic factors (4). Among the large number of proangiogenic
factors, vascular endothelial growth factor (VEGF) has been
confirmed to be key in angiogenesis in a number of preclinical and
clinical studies (5,6). VEGF is often overexpressed in most
tumors (7) and its expression is
linked to tumor growth, metastasis and angiogenesis. The
overexpression of VEGF has been associated with a poor prognosis in
non-small cell lung cancer (NSCLC) (8). Lung cancer has become one of the most
common malignancies and is the leading cause of cancer-related
mortality in males and females throughout the world (9). NSCLC accounts for 85% of all cases of
lung cancer. However, chemotherapy combinations appear to have
reached a therapeutic plateau of activity in the treatment of
advanced NSCLC (10).
In recent years, a number of antiangiogenic agents
have been developed, including monoclonal antibodies (mAbs),
targeting specifically the proangiogenic factors (11,12),
and synthetic tyrosine kinase inhibitors, targeting multiple
proangiogenic factors (13,14). A number of clinical studies have now
verified the antiangiogenic agents used in various types of cancer
and many patients treated with angiogenesis inhibitors survive for
a longer period (15). Therefore,
it is important to study antitumor activity which occurs via the
inhibition of tumor angiogenesis (16).
Taspine, isolated from Radix et Rhizoma
Leonticsi, has been shown to have antitumor angiogenesis
activity (17,18). HMQ1611, a taspine derivative, was
prepared in our laboratory to obtain a better water-solubility and
a weaker toxicity. In this study, we aimed to investigate the in
vivo effect of HMQ1611 on the growth of human lung cancer by
establishing an A549 animal-transplanted lung cancer tumor model.
We also evaluated the inhibition of microvessel formation in the
lung tissue and used ECV304 cells to study the in vitro
antiangiogenic effect of HMQ1611 on tube formation. We also
analyzed the antiangiogenic effect in vitro using a tissue
model. To further understand the molecular mechanism, we
investigated the HMQ1611 inhibitory effect on VEGF secretion and
KDR, which play key roles in tumor angiogenesis.
Materials and methods
Reagents
HMQ1611 was provided by the Natural Drug Research
and Engineering Center of Xi’an Jiaotong University (Shaanxi,
China). Stock concentration of HMQ1611 (20 mM) was prepared with
dimethyl sulfoxide (DMSO) and stored at 4°C. The stock solution was
further diluted with the serum-free RPMI-1640 medium immediately
before use. WST-1 reagent was purchased from Roche Diagnostics
(Indianapolis, IN, USA). DMSO, RPMI-1640, DMEM and trypsin were
purchased from Sigma-Aldrich (St. Louis, MO, USA). Matrigel was
obtained from BD Biosciences (Franklin Lakes, NJ, USA). Thrombin
was obtained from Guoao Pharmaceutical (Changchun, China).
Enzyme-linked immunosorbent assay (ELISA) kits were purchased from
R&D Systems (Minneapolis, MN, USA). KDR kinase was obtained
from Carna Biosciences (Kobe, Japan). HTRF® package
insert was purchased from Cisbio Bioassays (Bedford, MA, USA).
Cells
The A549 human NSCLC cell line and ECV304 cells were
purchased from the Shanghai Institute of Cell Biology in the
Chinese Academy of Sciences (Shanghai, China). A549 and ECV304
cells were cultured in RPMI-1640 supplemented with 10% FBS and
incubated at 37°C in a 5% CO2 atmosphere.
Mice
BALB/c mice (4–6 weeks old) were purchased from the
Shanghai Institute of Experimental Animals in the Chinese Academy
of Sciences. Kunming mice (15–18 g) were purchased from Animal
Research center of Xi’an Jiaotong University. The mice were
maintained under laminar air flow conditions with a 12-h light
(6:00–18:00)/12-h dark (18:00-6:00) cycle. Laboratory food and
water were freely available. Animal care was in accordance with the
National Institute of Health guidelines and the Animal Research
Committee of Xi’an Jiaotong University.
Antitumor effect of HMQ1611 on A549 cell
line xenografted in athymic mice
BALB/c immunodeficient mice were purchased from the
Shanghai Institute of Experimental Animals in the Chinese Academy
of Sciences. A549 cells (2×107 cells/ml) were implanted
into the right axilla of athymic mice (0.2 ml/mouse) to form solid
tumors. Athymic mice with solid tumors were randomly divided into
groups which received HMQ1611 (100 mg/kg and 200 mg/kg in 0.5%
CMC-Na; n=8) or vehicle alone (normal saline; n=8). Drugs were
administered once a day for two weeks beginning when the tumor
volumes became noticeable. The tumors were measured with calipers
three times and tumor volume was calculated as: tumor volume =
(length x width2)/2. The weight of the mice and the
tumor volumes were recorded when the mice were sacrificed.
Cell proliferation assay
A549 (1×105 cells/well) cells were seeded
in a 96-well plate (in RPMI-1640 medium with 10% FBS) and
cultivated for 24 h. Then a series of different concentrations of
HMQ1611 in serum-free RPMI-1640 medium was added to the 96-well
plate for 48 h. After 48 h, 10 μl/well WST-1 was added to the
96-well plate, which was then incubated for variable time periods
(0.5 to 4 h) in a humidified atmosphere (at 37°C under 5%
CO2). The plate was agitated thoroughly for 1 min on a
shaker prior to every measurement. The absorbance was measured at
450 nm in a microplate reader (Bio-Rad, Hercules, CA, USA). The
results are expressed as a percentage of the proliferation ratio:
percentage of proliferation ratio = [(ODtreatment group
- ODblank group) / (ODcontrol group -
ODblank group)] × 100%.
Tube formation assay
The tube formation assay was performed to determine
the effect of HMQ1611 on angiogenesis in vitro. Briefly,
matrigel was diluted in serum-free DMEM to 3 mg/ml of the final
concentration. A 48-well plate was coated with 200 μl/well matrigel
and 5 μl/well thrombin (50 U/ml) and incubated at 37°C for 30 min
to form a gel layer. Following gel formation, 1×105
ECV304 cells were added to each well in 500 μl 10% FBS-containing
medium and various concentrations of HMQ1611 were applied to each
well. The plates were incubated at 37°C for 18 h. Images of the
formation of capillary tubes were then captured randomly under a
microscope. The area of the tubes was measured with using Graphpad
Prism 5 software, with three images from separate experiments for
each data point. The inhibition rate of tube formation was
calculated as: [1 - (tube areatreated / tube
areacontrol)] × 100%.
Inhibition of angiogenesis of lung
tissue
An assay of mouse lung tissue was used as the in
vitro angiogenesis model. For preparation of fibrin matrices, 3
mg/ml solution of fibrinogen containing 300 μg/ml ε-aminocaproic
acid (in DMEM) was incubated on ice for 10 min. An aliquot of this
solution (250 μl) was mixed with 1 unit of thrombin and quickly
pipetted into the wells of a 24-well plate (Costar, Corning, NY,
USA). The mixture was incubated at 37°C for 30 min, during which
time a male mouse was anesthetized with diethyl ether and
sacrificed. The lung tissues were cut into 0.5–1.0 mm3
sections and placed on top of the previously prepared fibrin
matrix. Lung tissues were then covered with a second layer of
fibrin matrix that was covered with DMEM containing 20% fetal
bovine serum. The lung tissues were incubated with the growth
media, which consisted of DMEM with or without HMQ1611 (0, 5, 10
and 45 μM). The plates were stored in an incubator at 37°C and 5%
CO2. The number of sprouting vessels was analyzed with
the image analysis program Image Pro Plus (IPP, version 5.1, Media
Cybernetics, Bethesda, MD, USA).
ELISA
A549 cells were seeded at a density of
1×105 cells/well in a 6-well plate and grown to
confluence. The various concentrations of HMQ1611 were added to the
well for 48 h and then the supernatant was removed and immediately
assayed. The quantitation of VEGF was determined by ELISA, which
was performed according to the manufacturer’s instructions.
Kinase assay
The ability of HMQ1611 to inhibit the
phosphorylation of a peptide substrate by KDR kinase was evaluated
in a microtiter plate format using homogeneous time-resolved
fluorescence (HTRF). Initially, 2 μl kinase (Km=0.003767 ng/μl) and
2 μl substrate (Km=121.4 nM) were separately added to a 384-well
plate, and variable concentrations of HMQ1611 (diluted in kinase
buffer) were then added to the assay plate. ATP (2 μl, Km=1.332 μM)
was added and the reaction was allowed to proceed at 37°C for 30
min. The TK-Antibody labeled with Eu3+-cryptate and
streptavidin-XL665 were then added with EDTA to detect the
phosphorylated product at room temperature for 1 h. Then the
fluorescence was measured at 615 nm (cryptate) and 665 nm (XL665)
using the Perkin-Elmer victor 2030 multilabel plate reader.
Finally, the results were calculated as follows: ratio =
(OD665 nm / OD615 nm) × 104.
Statistical analysis
All data are expressed as mean ± SEM (standard error
of the mean). The statistical software SPSS 18.0 was used to
analyze statistical data and ANOVA was used to analyze differences
between groups of data. P<0.05 was considered to indicate a
statistically significant result.
Results
Effect of HMQ1611 on the growth of A549
lung cancer cells in athymic mice
The antitumor properties of HMQ1611 were evaluated
using human tumor models xenografted in athymic mice. HMQ1611
significantly inhibited tumor growth in A549-xenografted athymic
mice. Compared with the control group, the group treated with
HMQ1611 significantly inhibited the tumor growth at a rate of
28.28% and 54.76%, respectively. Furthermore, there was no
substantial change in the body weight of the athymic mice during
the experiment (the mean body weight at the start was 20.79±2.15,
20.21±1.56 and 20.43±1.89 g in the control, 100 mg/kg and 200 mg/kg
groups; at the end the mean body weight was 23.89±3.49, 24.48±4.02
and 23.93±4.29 g, respectively), which could be considered as the
antitumor activity of HMQ1611 overcoming the toxicity in athymic
mice.
Effect of HMQ1611 on proliferation of
A549 cells
The WST-1 assay demonstrated that the HMQ1611
significantly inhibited the growth of A549 cells in a
dose-dependent manner. HMQ1611 had hardly any effect on A549
proliferation at 2 μM. The inhibition increased with increasing
concentrations, with an inhibitory percentage of 98.79% at the 50
μM dose. The IC50 was 26.70 μM.
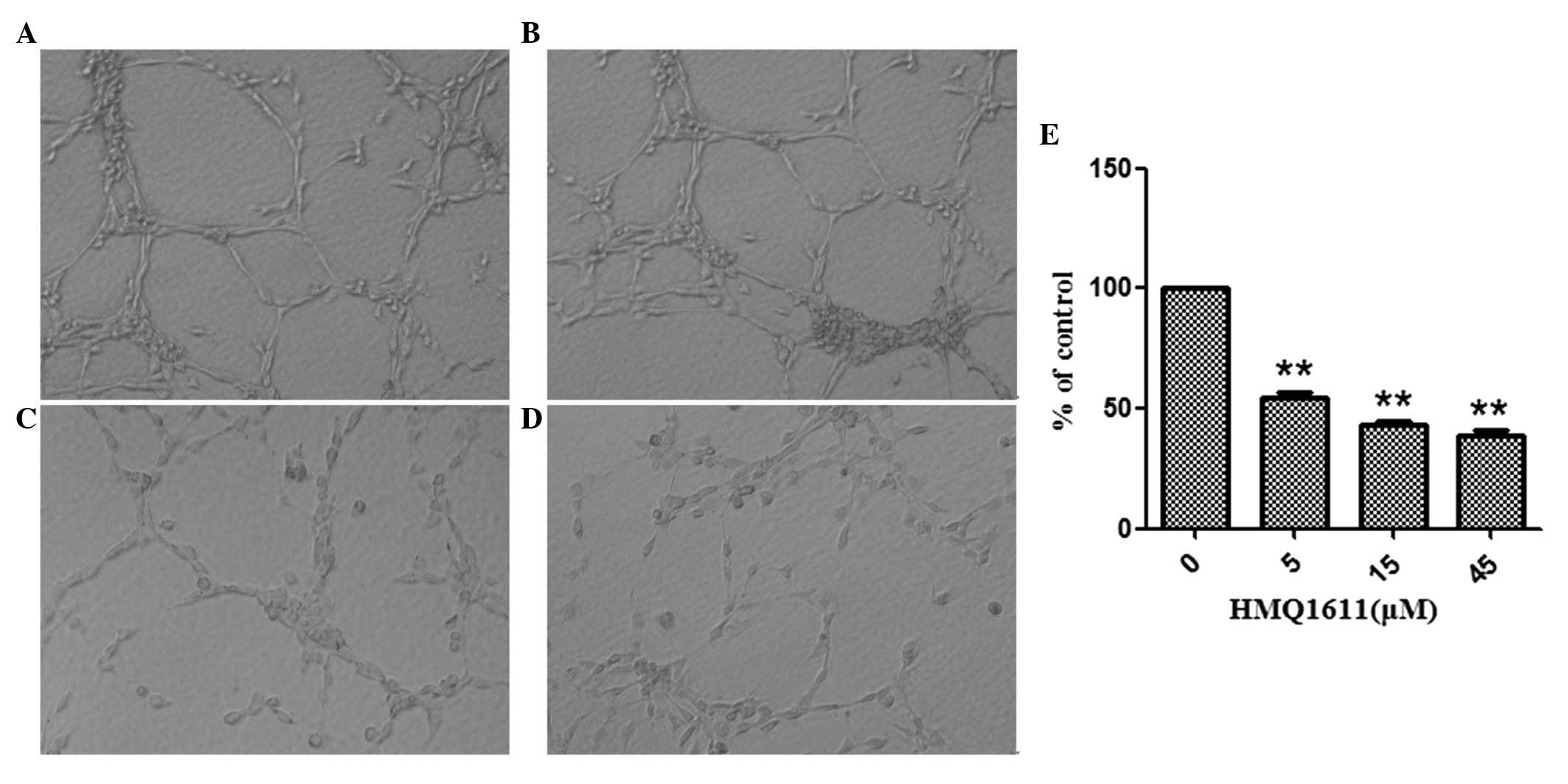
Effect of HMQ1611 on tube formation of
ECV304 cells
Tube formation is an important process in the
differentiation of resting endothelial cells into new vessels.
Fig. 1B, C and D showed that
treatment with HMQ1611, ranging from 5 to 45 μM, markedly inhibited
the tube formation in a dose-dependent manner. The inhibitory
percentages were 45.55, 56.96 and 61.39% at 5, 15 and 45 μM,
respectively.
Effect of HMQ1611 on angiogenesis in the
lung tissue model
The lung tissue model was established to imitate
angiogenesis in vivo. As shown in Fig. 2, HMQ1611 markedly inhibited the
formation of new blood vessels compared with the control group. The
quantitative data of the number of blood vessel indicated that
HMQ1611 significantly reduced vascularization of the lung tissue at
the concentrations of 5, 15 and 45 μM in a dose-dependent
manner.
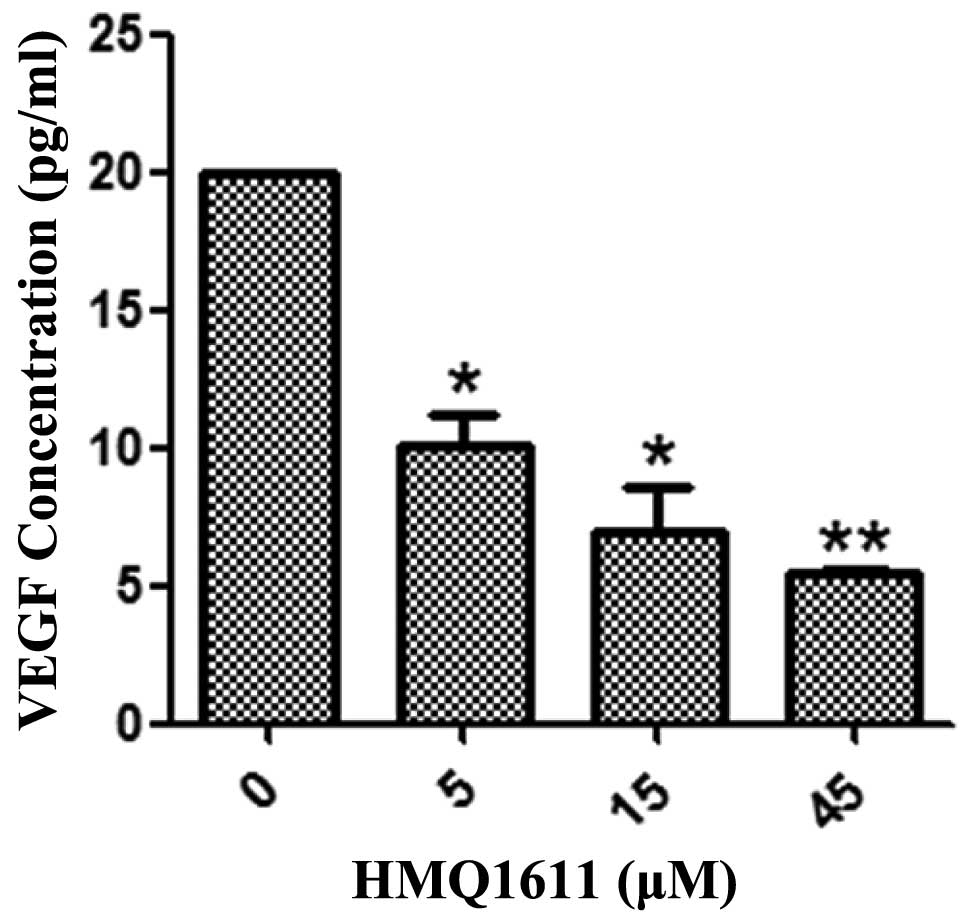
Effect of HMQ1611 on VEGF secretion
A sandwich ELISA was used to determine the level of
VEGF released from A549 cells. In A549 cells that were cultured
with or without HMQ1611, Fig. 3
showed that HMQ1611 inhibited VEGF production in a dose-dependent
manner from 5 to 45 μM compared with the negative control. The VEGF
amount was significantly decreased at all used concentrations.
Kinase assay
The LANCE™ assay was used to assess the
effects of HMQ1611 on KDR kinase activity. The IC50 of
HMQ1611 on KDR kinase activity was >5,000 nM, suggesting that
HMQ1611 did not markedly alter KDR kinase activity.
Discussion
Previous studies in our laboratory have revealed
that taspine had antitumor activity and was able to markedly
inhibit the proliferation of ECV304 cells and angiogenesis
(17,19). In the current study, we investigated
HMQ1611, a taspine derivative, its action on NSCLC and its
potential antiangiogenic mechanisms.
We initially evaluated the effect of HMQ1611 on
tumor growth in vivo by xenografts of A549 cells in athymic
mice. It was shown that the tumor development of BALB/c mice with
the implantation of A549 cells was inhibited by the administration
of HMQ1611. The tumor formation of BALB/c mice was significantly
suspended in the HMQ1611 group. The average tumor volume and weight
were decreased in the administration group compared with the
control group; however, the final body weight of the mice in the
two groups was essentially identical. Meanwhile, xenografts in
athymic mice treated with HMQ1611 at different concentrations
exhibited different inhibition rates. These results demonstrated
the antitumor effect of HMQ1611 and that it had no marked
toxicity.
We then used the WST-1 assay to observe the effect
of HMQ1611 on A549 cell proliferation in vitro. The results
showed that HMQ1611 exhibited significant inhibition of A549 cell
proliferation in a dose-dependent manner. This assay also provided
an effective non-toxic concentration for use in further study.
Angiogenesis has been recognized as an important driver in the
initiation and progression of several types of human cancer
(20), while endothelial cell tube
formation is the final stage of angiogenesis (21,22).
Therefore, we used the tube formation of endothelial cell on
matrigel to validate the in vitro antiangiogenic properties
of HMQ1611. The results showed that the tubes exhibited different
numbers at different concentrations of HMQ1611. Compared with the
control group, the formation of tubes in animals treated with
HMQ1611 was postponed, indicating that the compound inhibited the
tube formation. The tissue vessel model established in our
laboratory was used to verify the role of HMQ1611 in angiogenesis
at the tissue level. Following treatment with HMQ1611, new vessels
on the periphery of the lung tissues grew slower than in the
control group; moreover, the number of vessels reduced markedly.
These facts indicate that HMQ1611 effectively disturbed the vessel
formation that is critical for the supply of nutrients and oxygen
to the tumor. Therefore, we hypothesized that the antitumor
activity of HMQ1611 most likely occurs via the inhibition of tumor
angiogenesis. VEGF, recognized as one of the key proangiogenic
factors in angiogenesis, is an endothelial cell-specific mitogenic
and chemotactic agent (23) and the
inhibition of VEGF in endothelial cells should block the process of
angiogenesis. Subsequently, we explored the potential mechanism of
HMQ1611 inhibition of angiogenesis by detecting VEGF secretion and
KDR kinase activity in vitro. The data showed that HMQ1611
inhibited VEGF secretion and did not markedly inhibit KDR kinase.
These results indicate that HMQ1611 most likely only reduced VEGF
secretion and did not block the receptor of KDR.
In conclusion, the present study demonstrated that
HMQ1611 inhibits tumor growth in xenografted A549 cells in nude
mice by inhibiting the growth of neovessels. In other words, the
results show that HMQ1611 is an inhibitor of angiogenesis which
functions by downregulating VEGF. Our study suggests that HMQ1611
is a promising candidate for a treatment strategy in
angiogenesis-related disease.
Acknowledgements
This study was supported by National
Natural Science Foundation of China (Grant No. 81001447) and
Shaanxi young star of science and technology Program (Grant No.
2012KJXX-06).
References
|
1.
|
J FolkmanAnti-angiogenesis: a new concept
for therapy of solid tumorsAnn
Surg175409416197210.1097/00000658-197203000-000145077799
|
|
2.
|
N FerraraVascular endothelial growth
factor as a target for anticancer
therapyOncologist9210200410.1634/theoncologist.9-suppl_1-2
|
|
3.
|
J FolkmanWhat is the evidence that tumors
are angiogenesis dependent?J Natl Cancer
Inst8246199010.1093/jnci/82.1.41688381
|
|
4.
|
H HirteNovel developments in angiogenesis
cancer therapyCurr Oncol165054200910.3747/co.v16i3.44419526086
|
|
5.
|
N FerraraHP GerberJ LeCouterThe biology of
VEGF and its receptorsNat
Med9669676200310.1038/nm0603-66912778165
|
|
6.
|
KJ KimB LiJ WinerM ArmaniniN GillettHS
PhillipsN FerraraInhibition of vascular endothelial growth
factor-induced angiogenesis suppresses tumor growth in
vivoNature362841844199310.1038/362841a07683111
|
|
7.
|
N FerraraVascular endothelial growth
factor: basic science and clinical progressEndocr
Rev25581611200410.1210/er.2003-002715294883
|
|
8.
|
H ImotoT OsakiS TagaA OhgamiY IchiyoshiK
YasumotoVascular endothelial growth factor expression in
non-small-cell lung cancer: prognostic significance in squamous
cell carcinomaJ Thorac Cardiovasc
Surg11510071014199810.1016/S0022-5223(98)70398-89605068
|
|
9.
|
DM ParkinGlobal cancer statistics in the
year 2000Lancet Oncol2533543200111905707
|
|
10.
|
DN CarneyLung cancer - time to move on
from chemotherapyN Engl J
Med346126128200210.1056/NEJM20020110346021111784881
|
|
11.
|
ME HalatschU SchmidtJ Behnke-MurschA
UnterbergCR WirtzEpidermal growth factor receptor inhibition for
the treatment of glioblastoma multiforme and other malignant brain
tumorsCancer Treat
Rev327489200610.1016/j.ctrv.2006.01.00316488082
|
|
12.
|
J HardingB BurtnessCetuximab: an epidermal
growth factor receptor chimeric human-murine monoclonal
antibodyDrugs Today
(Barc)41107127200510.1358/dot.2005.41.2.88266215821783
|
|
13.
|
J Glade BenderEM CooneyJJ KandelDJ
YamashiroVascular remodeling and clinical resistance to
antiangiogenic cancer therapyDrug Resist
Updat7289300200415533766
|
|
14.
|
P CarmelietAngiogenesis in life, disease
and medicineNature438932936200510.1038/nature0447816355210
|
|
15.
|
D RibattiNovel angiogenesis inhibitors:
addressing the issue of redundancy in the angiogenic signaling
pathwayCancer Treatment
Rev37344352201110.1016/j.ctrv.2011.02.00221435792
|
|
16.
|
AP JekunenKJA KairemoInhibition of
malignant angiogenesisCancer Treat
Rev23263266199710.1016/S0305-7372(97)90014-19377596
|
|
17.
|
Y ZhangL HeL MengW LuoTaspine isolated
from Radix et Rhizoma Leonticis inhibits proliferation and
migration of endothelial cells as well as chicken chorioallantoic
membrane neovascularisationVascul Pharmacol1481291372008
|
|
18.
|
YM ZhangLC HeHY WangInhibitory effect of
taspine on mouse S180 sarcoma and its mechanismZhongguo Zhong Yao
Za Zhi329539562007(In Chinese)
|
|
19.
|
Y ZhangL HeY ZhouTaspine isolated from
Radix et Rhizoma Leonticis inhibits growth of human
umbilical vein endothelial cell (HUVEC) by inducing its
apoptosisPhytomedicine151121192008
|
|
20.
|
R GrepinG PagesMolecular mechanisms of
resistance to tumor antiangiogenic strategiesJ
Oncol2010835680201010.1155/2010/83568020224655
|
|
21.
|
LM EllisEpidermal growth factor receptor
in tumor angiogenesisHematol Oncol Clin North
Am1810071021200410.1016/j.hoc.2004.06.00215474332
|
|
22.
|
RK JainMolecular regulation of vessel
maturationNat Med9685693200310.1038/nm0603-68512778167
|
|
23.
|
S RousseauF HouleJ LandryJ Huotp38 MAP
kinase activation by vascular endothelial growth factor mediates
actin reorganization and cell migration in human endothelial
cellsOncogene1521692177199710.1038/sj.onc.12013809393975
|