Introduction
An accurate assessment of renal function is
essential for cancer patients receiving cytotoxic agents, which are
primarily eliminated through the kidney in an unchanged form. Among
these drugs is carboplatin, for which dosing is based on renal
function rather than on body surface area (BSA) and is calculated
using the Calvert formula: dose (mg) = AUC (GFR + 25) (1). Most protocols use an estimated
glomerular filtration rate (eGFR), which is calculated using the
Cockroft-Gault equation (CG), based on the serum creatinine (Scr)
level, age and weight (2).
Targeting the carboplatin dose based on the AUC leads to more
predictable toxicity compared with dose determination based on BSA.
However, CG may be unreliable in cancer patients due to confounding
factors such as muscle mass, rate of metabolism of the muscle
protein creatine to creatinine, absorption of dietary creatine,
filtration of creatinine by the renal glomeruls and its secretion
by the proximal renal tubules (3).
In particular, the blood creatinine level may decrease in patients
with advanced cancer due to a reduction of muscle mass, leading to
false increases in the CG eGFR and, therefore, to unjustified
increases in carboplatin dosing calculated according to the Calvert
formula.
We hypothesized that the CG eGFR may be higher than
the actual GFR in a considerable number of patients with advanced
non-small cell lung carcinoma (NSCLC) treated with carboplatin,
potentially leading to carboplatin overdosing. To test this
hypothesis, fluctuations in Scr level, CG eGFR and the theoretical
carboplatin dose derived from the Calvert formula, and CG eGFR
based on actual Scr were retrospectively analyzed in repeated
cycles of carboplatin-based chemotherapy in patients with advanced
NSCLC. In addition, the dose of carboplatin resulting from the
Calvert formula and the recent Scr was compared with the actual
carboplatin dose prescribed by the physicians.
Materials and methods
Patients
Following approval of the study protocol by the
institutional ethics committee, a retrospective analysis of all
medical records of adult patients treated for advanced NSCLC in the
Division of Oncology at Rambam Health Care Campus (RHCC), Haifa,
Israel, between January 2007 and June 2010 was undertaken. Patients
who had been treated with a carboplatin-based combination as
first-line chemotherapy treatment were eligible for the study if
they had received at least two cycles of carboplatin-containing
combinations. Patients with elevated pretreatment Scr (i.e.,
>1.3 mg/dl) were excluded.
Patient data recorded included gender, age, weight,
height, NSCLC histological subtype, ECOG performance status (PS),
disease stage, type of chemotherapy combination and Scr and blood
urea nitrogen, which were routinely recorded prior to each
chemotherapy cycle. Since malnutrition could be associated with
decreased creatinine production, the body mass index (BMI) was
calculated using the body weight measured at the beginning of the
first cycle of carboplatin. Patients were classified as
underweight, healthy weight, overweight or obese, according to the
criteria of the Centers for Disease Control and Prevention
(4).
Treatment
Three different carboplatin-based regimens were
used: i) carboplatin (AUC5) and pemetrexed 500 mg/m2,
administered intravenously on day 1 every 21 days; ii) carboplatin
(AUC6) and paclitaxel 200 mg/m2, administered
intravenously on day 1 every 21 days; and iii) carboplatin (AUC5)
administered intravenously on day 1 and gemcitabine 1,000
mg/m2 administered intravenously on days 1 and 8, with
cycles repeated every 21 days.
The eGFR was calculated at the beginning of each
cycle according to the CG formula: 140 - age (yrs) x body weight
(kg) / 72 x Scr (mg/dl) (for females: x 0.85) (2). Scr is routinely determined in our
center before each dose of carboplatin, and the relevant Scr was
used for GFR calculation.
Renal function was graded in accordance with
clinical practice guidelines published by the working group of the
National Kidney Foundation (5):
Stage 1, GFR ≥90 ml/min; Stage 2, GFR 60–89 ml/min; Stage 3, GFR
30–59 ml/min; Stage 4, GFR 15–29 ml/min; and Stage 5, GFR <15
ml/min.
Patient data, including body weight at the beginning
of each chemotherapy line and Scr level at the beginning of each
carboplatin-containing cycle, are routinely introduced into a
computerized system used at our center (Prometheus), and the
carboplatin dose, based on the Calvert formula and the CG equation,
is routinely computed by that system. The maximal doses of
carboplatin prescribed by the computerized system were those
defined by the treatment protocol. The dose could be reduced at the
discretion of the physician, who could determine that less than
100% of the dose would be prescribed. Moreover, the physician could
further reduce the dose by inputting an Scr level higher than that
measured prior to the relevant cycle. Modifications of the Scr were
made at the physician's discretion and were not based on defined
rules.
The theoretical dose of carboplatin was defined as
the dose that would have been derived from the Calvert formula, in
which eGFR is calculated by CG using actual Scr.
Statistics
The difference in eGFR based on demographics (gender
and age) was analyzed using the Mann-Whitney non-parametric test.
Changes in Scr and GFR at different time points based on BMI, PS,
stage, histology and chemotherapy medication were analyzed using
one-way ANOVA (Bonferroni) post-hoc multiple comparisons. Repeated
measures analysis was used to compare the test results of eGFR in
the first three chemotherapy cycles for all patients and for
different groups segmented by the various parameters (BMI, PS,
stage, histology and chemotherapy medication). The data were
processed using SPSS for Windows (version 18), and a P-value
<0.05 was considered to indicate a statistically significant
result.
Results
Patient characteristics and renal
function
The files of 126 consecutive patients with advanced
NSCLC who had been treated with carboplatin-based combinations
between January 2007 and June 2010 were reviewed. Nine patients
were excluded from the study due to baseline creatinine levels
>1.3 mg/dl. The main characteristics of the remaining 117
patients are presented in Table I.
Most (85%) of the patients were male, and most (76%) of these had
good PS; i.e., grade 0–1. Only four patients (3.5%) were
underweight.
 | Table ICharacteristics of 117 NSCLC patients
treated with carboplatin-based chemotherapy. |
Table I
Characteristics of 117 NSCLC patients
treated with carboplatin-based chemotherapy.
| Characteristic | Value |
|---|
| Patients (M/F) | 99/18 |
| Median age
(range) | 62 (41–86) years |
| Median baseline
creatinine | |
| (M/F) | 0.85 (0.88/0.73)
mg/dl |
| Range | 0.40–1.3 mg/dl |
| Median GFR (Cockroft
and Gault) | |
| (M/F) | 81.3 (81.3/82.5)
ml/min/1.73m2 |
| Range | 31–168
ml/min/1.73m2 |
| GFR, n (%) | |
| 30–59
ml/min/1.73m2 | 19 (16) |
| 60–89
ml/min/1.73m2 | 55 (47) |
| 90–119
ml/min/1.73m2 | 26 (22) |
| >120
ml/min/1.73m2 | 17 (15) |
| Performance status
(ECOG), n (%) | |
| 0 | 15 (13) |
| 1 | 74 (63) |
| 2 | 27 (23) |
| Unknown | 1 (0.9) |
| Body mass index
(BMI), n (%) | |
| Underweight (BMI
<18.5) | 4 (3.5) |
| Normal range
(18.5≥BMI≤24.9) | 50 (42.5) |
| Overweight
(25≥BMI≤29.9) | 49 (42) |
| Obese (BMI
≥30) | 14 (12) |
| Histology, n (%) | |
| Squamous cell
carcinoma | 31 (26.5) |
| Adenocarcinoma | 51 (43.5) |
| NSCLC
non-specified | 35 (30) |
| Drug used in
combination with carboplatin, n (%) | |
| Gemcitabine | 41 (35) |
| Paclitaxel | 60 (51) |
| Pemetrexed | 16 (14) |
| Stage (AJCC), n
(%) | |
| IIIA/IIIB | 66 (56.5) |
| IV | 51 (43.5) |
The median baseline creatinine level was 0.8 mg/dl
for 45 males aged <60 years and 0.92 mg/dl for 61 males aged ≥60
years. The median level was 0.85 mg/dl for six females aged <60
years and 0.73 mg/dl for 14 females aged >60 years.
The median baseline GFR calculated according to
Cockcroft and Gault was 82 ml/min, with no significant gender
difference (P=0.9). The GFR was significantly higher in patients
<60 years than in older patients (97 versus 72 ml/min; P=0.001).
There was no significant gender difference in the average eGFR
values. Since poor PS and weight loss may be associated with muscle
waste, the correlation between those parameters and GFR (adjusted
to ml/min/1.73 m2) was tested. No significant difference
was noted between PS 0–1 and PS 2. The number of patients with BMI
<18.5 was too small to allow for statistical analysis.
Fluctuations in the full theoretical
carboplatin doses
The number of chemotherapy cycles ranged between two
and seven (median, 4). The main reason for treatment
discontinuation was tumor progression. Changes in the full
theoretical doses of carboplatin that would have resulted from
prescribing 100% of the dose and using recent Scr levels for dose
calculation are shown in Table II.
Since a change of >10% was considered clinically significant,
the results are expressed as changes of 10–20% and >20%. A total
of 320 chemotherapy cycles were administered between cycles 2–5 for
the entire study population. If treatment had been administered
according to the CG formula only, 79/320 of the cycles (24.7%)
would have been administered in doses >10% higher, and 53/320
(16.6%) in doses >10% lower than the theoretical baseline dose
of carboplatin. This difference between the rate of dose increase
of >10% and dose decrease of >10% was statistically
significant (P=0.015) and correlated with changes in creatinine
levels during the course of treatment. Compared with the baseline
level, Scr increased by >10% in 69/320 (21.5%) cycles and
decreased by >10% in 69/320 (28.7%) cycles.
 | Table IIFluctuations in theoretical
carboplatin doses derived from Calvert formulaa. |
Table II
Fluctuations in theoretical
carboplatin doses derived from Calvert formulaa.
| Number of cycles | Number of
patients | Increase of
20–50% | Increase 10–20% | No change ±10% | Reduction 10–20% | Reduction 20–40% |
|---|
| 2 | 113 | 14 (12.4%) | 10 (8.8%) | 69 (61.1%) | 14 (12.4%) | 6 (5.3%) |
| 3 | 94 | 12 (12.8%) | 9 (9.6%) | 62 (66%) | 8 (8.5%) | 3 (3.2%) |
| 4 | 73 | 9 (12.3%) | 15 (20.5%) | 36 (49.3%) | 8 (11%) | 5 (6.8%) |
| 5 | 40 | 0 | 10 (25%) | 21 (52.5%) | 4 (10%) | 5 (12.5%) |
Effect of various pretreatment parameters
on fluctuations in eGFR
The trend for change in CG eGFR over the course of
chemotherapy was assessed in 94 patients who completed at least
three cycles of treatment. Fluctuations in eGFR during the
treatment period were evaluated in correlation with the following
parameters: BMI, PS, chemotherapy regimen, stage and histology.
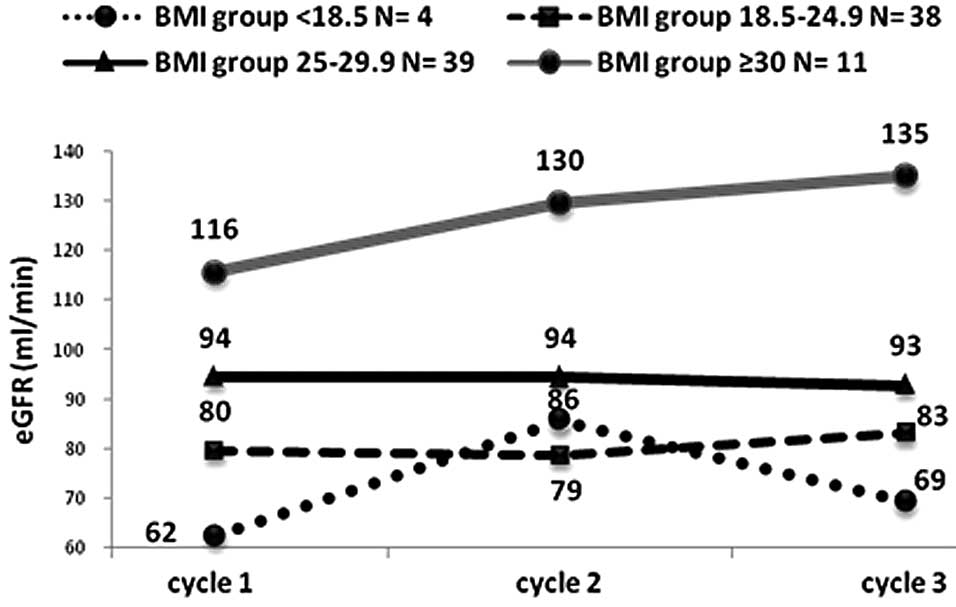
BMI is the only parameter to predict changes in eGFR
at a significant level of confidence. Patients with BMI ≥30 had a
significant tendency for increased eGFR during subsequent
chemotherapy treatments, with P=0.02, P=0.001 and P=0.01 for BMI
<18.5, 18.5≥BMI≤24.9 and 25≥BMI≤29.9, respectively, and P=0.009
for all groups combined (Fig. 1).
The trend for fluctuations in GFR did not differ significantly
between patients with PS grade 2 and PS grade 0–1.
Physicians' decisions on carboplatin
doses
The impact of using a modified Scr according to the
physicians' discretion was evaluated by comparing actual prescribed
doses to theoretical doses derived from the Calvert formula, which
would have resulted had the recent Scr levels been used in the CG
eGFR calculation. Differences of ≥10% were considered clinically
significant. As shown in Table III,
the prescribed dose was changed by >10% in 43.1% of the doses
due to Scr modification. Clinicians tended to decrease the given
dose rather than increase it. In approximately one-third of the
chemotherapy cycles, the oncologist preferred to artificially input
a higher Scr value into the computerized system than that actually
measured, resulting in a dose decrease of >10%.
 | Table IIIDifference between actual prescribed
carboplatin doses and theoretical carboplatin dosesa. |
Table III
Difference between actual prescribed
carboplatin doses and theoretical carboplatin dosesa.
| Number of
cycles | Number of given
cycles | Prescribed dose
higher than theoretical by ≥10% | Difference of
<10% between prescribed dose and dose derived from actual
Scr | Prescribed dose
lower than dose derived from actual Scr by ≥10% |
|---|
| 1 | 117 | 12 (9%) | 73 (63%) | 32 (28%) |
| 2–5 | 320 | 35 (11%) | 176 (55%) | 109 (34%) |
| Total | 437 | 47 (10.8%) | 249 (56.9%) | 141 (32.3%) |
Discussion
Carboplatin dosing according to the Calvert formula
is based on the assumption that the main mechanism of clearance of
this drug is glomerular filtration. Although creatinine clearance
consists of both glomerular filtration and tubular secretion, the
use of creatinine clearance as a surrogate for GFR for drug dose
calculation is generally accepted. GFR measurement based on
radiolabeled isotopes is accurate but expensive, and a 24-h urine
collection is inaccurate and cumbersome. Therefore, several
mathematical equations have been developed to estimate GFR based on
the assumption that serum creatinine is a marker for estimating
GFR. Of these equations, CG is widely used in the clinical setting,
including for the estimation of GFR in the Calvert formula.
Although widely used in daily practice, CG, as well as other
formulas for GFR estimation, has significant limitations in cancer
patients (6). Recently, the new
Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula
was found to have failed as an estimate of GFR in cancer patients
and as a clinical alternative to isotope tests (7). The current study was specifically
aimed at evaluating the reliability of CG for carboplatin dose
calculation according to the Calvert formula in patients with
advanced NSCLC.
CG assumes that muscle mass is proportional to body
weight. However, since creatinine production is influenced by
several factors, including muscle mass, physical condition and
nutrition, this equation may not accurately reflect the actual
cortical collecting tubule. Muscle waste in these patients could
result in an overestimation of GFR and, therefore, in an overdose
of carboplatin when relying on the Calvert formula using CG and
actual Scr for GFR calculation. To test this hypothesis we
retrospectively analyzed the Calvert formula-calculated carboplatin
doses, based on actual Scr and CG eGFR in patients with advanced
NSCLC.
Due to the current study design it is impossible to
determine whether the baseline CG eGFR was indeed overestimated.
However, the median GFR of 81.3 ml/min/1.73 m2 prior to
the onset of chemotherapy was not obviously lower than that
expected for a similar non-cancer population, as was found in the
JUPITER study (Justification for the Use of Statins in Prevention:
An Intervention Trial Evaluating Rosuvastatin). This trial included
16,279 participants, with a median GFR of 73.6 ml/min/1.73
m2 calculated using the MDRD equation (8). The apparent lack of underestimation of
baseline eGFR could be explained by the fact that only 3.5% of the
patients were underweight and the majority (77%) had a good PS.
The range of eGFR among cancer patients at the
beginning of chemotherapy treatment was evaluated in the
retrospective analysis of the renal insufficiency and anticancer
medications (IRMA) study (9). In a
subgroup analysis of 445 patients with lung carcinoma from the IRMA
study, the rate of GFR <60 ml/min was 23.5% and the rate of GFR
>90 ml/min was 31.5% (10). A
similar distribution of baseline GFR was found in the current
study, with 16% with eGFR <60 ml/min and 37% with eGFR >90
ml/min.
In the current study, CG eGFR varied widely during
the course of chemotherapy, resulting in clinically significant
changes (i.e., >10%) in the theoretical Calvert formula-derived
carboplatin dose in 41% of subsequent cycles. Furthermore, the dose
of carboplatin derived from the Calvert formula and CG eGFR was
increased by >10% in approximately a quarter of subsequent
cycles, while it was decreased by >10% in only 17% of cycles
(P=0.015).
No apparent association was found between the
baseline CG eGFR and the tendency for fluctuations in eGFR in
subsequent cycles. Notably, the tendency for increases in CG eGFR
was most pronounced in obese patients (BMI >30) due to more
significant decreases in Scr levels during the course of
chemotherapy in these patients.
Carboplatin is an important drug in the treatment of
ovarian carcinoma patients. The correlation between BMI and
carboplatin dosing and treatment outcome was tested in several
studies. Recently, in the Scottish Randomized Trial in Ovarian
Cancer (SCOTROC) study, more than 1,000 patients were recruited and
no association was found between BMI and survival or BMI and
carboplatin dose intensity (11).
In the SCOTROC study, one of the inclusion criteria was GFR
measurement by plasma clearance of EDTA or 24-h urine collection
for creatinine clearance. This demand was based on the conclusion
of certain studies that eGFR is not reliable in obese patients
(12). In our study, 12% of
patients had severe obesity and high eGFR prior to and during
chemotherapy treatment. A call for greater caution in estimating
GFR in these patients, and possibly a recommendation for urine
collection for creatinine clearance, is reasonable.
During the study period there were no firm
guidelines at our institution on how to compensate for fluctuations
in Scr levels in the actual computerized prescription of
carboplatin. In approximately one-third of chemotherapy cycles, we
found that there was a reduction of more than 10% in the actual
carboplatin doses prescribed by the physician compared with the
doses calculated according to the Calvert formula. Whether or not
the physician's preference was to artificially lower the prescribed
carboplatin dose is unknown.
In conclusion, Calvert formula-derived carboplatin
doses fluctuate widely during repeated cycles in patients with
advanced NSCLC where actual Scr is used for CG GFR estimation.
Discrepancies of >10% compared with the initial dose were found
in more than 40% of subsequent doses. Approximately a quarter of
theoretical subsequent doses were more than 10% higher than the
initial dose due to decreases in Scr.
Physicians tend to prescribe a lower dose than that
derived from the Calvert formula by using a higher Scr than that
actually measured. Thus, reconsidering the measurement of 24-h
creatinine clearance as a feasible alternative in selected patients
with observed Scr reduction during therapy is recommended.
References
|
1.
|
AH CalvertSJ HarlandDR NewellEarly
clinical studies with cis-diammine-1,1-cyclobutane dicarboxylate
platinum IICancer Chemother
Pharmacol9140147198210.1007/BF002577426761010
|
|
2.
|
DW CockcroftHM GaultPrediction of
creatinine clearance from serum
creatinineNephron163141197610.1159/0001805801244564
|
|
3.
|
RE BleilerHP SchedlCreatinine excretion:
Variability and relationships to diet and body sizeJ Lab Clin
Med59945955196213869979
|
|
4.
|
Clinical Guidelines on the Identification,
Evaluation, and Treatment of Overweight and Obesity in Adults:
National Heart, Lung, and Blood InstituteNIH PublicationBethesda,
MD, USA2007
|
|
5.
|
National Kidney FoundationK/DOQI clinical
practice guidelines for chronic kidney disease: evaluation,
classification, and stratificationAm J Kidney Dis39Suppl
1S1S266200211904577
|
|
6.
|
SG PooleMJ DooleyD RischinA comparison of
bedside renal function estimates and measured glomerular filtration
rate (Tc99mDTPA clearance) in cancer patientsAnn
Oncol13949955200210.1093/annonc/mdf236
|
|
7.
|
B Redal-BaigorriKH StokholmK RasmussenN
JeppesenEstimation of kidney function in cancer patientsDan Med
Bull58A4236201121299923
|
|
8.
|
DG VidtPM RidkerJT MonyakMJ SchreiberMD
Cressmanf Clin Ther33717725201110.1016/j.clinthera.2011.05.004
|
|
9.
|
V Launay-VacherS OudardN JanusRenal
Insufficiency and Cancer Medications (IRMA) Study Group: Prevalence
of renal insufficiency in cancer patients and implications for
anti-cancer drug management: the renal insufficiency and anticancer
medications (IRMA) studyCancer11013761384200710.1002/cncr.22904
|
|
10.
|
V Launay-VacherR EtessamiN JanusRenal
Insufficiency Anticancer Medications (IRMA) Study Group: Lung
cancer and renal insufficiency: prevalence and anticancer drug
issuesLung1876974200910.1007/s00408-008-9123-518941834
|
|
11.
|
SV BarrettJ PaulA HayPA VaseySB KayeRM
GlasspoolScottish Gynaecological Cancer Trials GroupDoes body mass
index affect progression-free or overall survival in patients with
ovarian cancer? Results from SCOTROC I trialAnn
Oncol19898902200810.1093/annonc/mdm60618272913
|
|
12.
|
JC VerhaveP FeslerJ RibsteinG du CailarA
MimranEstimation of renal function in subjects with normal serum
creatinine levels: influence of age and body mass indexAm J Kidney
Dis46233241200510.1053/j.ajkd.2005.05.01116112041
|