Introduction
While there have been many criteria for determining
the prognosis of patients with malignant tumors, such as the TNM
staging criteria, these serve only to provide physicians with
information regarding the outcome of patients during daily clinical
analysis. In a previous study, we demonstrated that the
Pathological Prognostic Score (PPS), determined based on
histopatological data including depth of tumor, lymph node
metastasis, lymphatic invasion and venous invasion, clearly
classified the prognosis of patients with colorectal carcinoma
(1).
Although undifferentiated carcinoma of the colon and
rectum have been reputed to possess a more aggressive potential,
which leads to a worse prognosis of patients (2), the criteria for determining the
prognosis of patients with colorectal carcinoma reflected by this
pathological type of tumor has not yet been presented. In this
study, we investigated the significance of a novel staging
criterion (a five-point scoring system; FPSS) for determining the
prognosis of patients with colorectal carcinoma. The FPSS comprised
five histopathological categories of patients with colorectal
carcinoma: Depth of tumor, lymph node metastasis, lymphatic
invasion, venous invasion and histopathological tumor type.
Patients and methods
Patients
In total, 357 patients with colorectal carcinoma,
who had been treated by surgical resection at the Fukuoka Higashi
Medical Center from January 1997 to January 2011, were evaluated.
Forty-two patients had been treated with palliative resection due
to the presence of distant metastasis and/or peritoneal
dissemination. No patients had been treated with neoadjuvant
therapy. Patients were aged between 24 and 91 years (mean, 69) and
the group comprised 214 males and 143 females. The study was
approved by the Ethics Committee of Fukuoka Higashi Medical Center,
Koga, Japan. Written infomed consent was obtained from the
patient.
Pathological research
The clinicopathological factors were determined
according to the general rules for clinical and pathological
studies on cancer of the colon, rectum and anus, outlined by the
Japanese Research Society for Cancer of the Colon and Rectum
(3). Additionally, TNM tumor stages
were determined by the TNM classification of malignant tumors
prescribed by the International Union Against Cancer (4).
FPSS
FPSS scores were determined by assigning one point
to a more advanced result in each of the following categories:
Tumor depth (T1 and 2 vs. T3 and 4); lymph node metastasis
(positive vs. negative); lymphatic invasion (positive vs.
negative); venous invasion (positive vs. negative) and
histopathological tumor type (differentiated vs. undifferentiated
tumor). Subsequently, FPSS scores were determined by an aggregate
of points for each category and ranged from 0–5.
Patient follow-up
Follow-up of patients was continued until mortality
and only patients whose cause of death was colorectal carcinoma
were included in the tumor-related deaths. The time period between
surgery and death was termed the survival time.
Statistical analysis
All statistical analyses were conducted using
StatView version 5.0 (SAS Institute Inc, Cary, NC, USA). Then, a
χ2 test was used to compare the difference in proportion
values between FPSS scores. A Mann-Whitney U test was used to
compare the mean ages of patients. Survival curves were conducted
using the Kaplan-Meier method and a Mantel-Cox test was used to
analyze their equality. P<0.05 was considered to indicate a
statistically significant difference.
Results
Each factor included in the FPSS (tumor depth, nodal
metastasis, lymphatic invasion, venous invasion and
histopathological tumor type) was found to be an indicator of worse
prognosis in patients with colorectal carcinoma (Table I).
 | Table IAnalysis of survival rates based on
five pathological factors. |
Table I
Analysis of survival rates based on
five pathological factors.
| Characteristic | No. of patients | 5-year SR (%) | P-value |
|---|
| Depth of tumor | | | |
| T1 and T2 | 106 | 98.7 | <0.0001 |
| T3 and T4 | 251 | 74.5 | |
| Nodal metastasis | | | |
| No | 205 | 91.4 | <0.0001 |
| Yes | 152 | 68.3 | |
| Lymphatic
invasion | | | |
| No | 214 | 91.4 | <0.0001 |
| Yes | 143 | 67.1 | |
| Venous invasion | | | |
| No | 259 | 88.5 | <0.0001 |
| Yes | 98 | 63.2 | |
| Histologic type | | | |
| Differentiated | 325 | 86.3 | <0.0001 |
|
Undifferentiated | 32 | 37.5 | |
The study population was divided into three groups
according to the FPSS score: 0 and 1 (153 patients, 42.9%); 2 and 3
(150 patients, 42.0%); and 4 and 5 (54 patients, 15.1%). A
significant correlation was observed between FPSS score and the
following tumor-related factors: Tumor depth, lymph node (nodal)
metastasis, lymphatic invasion, venous invasion and proportion of
curative resection (P<0.0001 for each factor; Table II).
 | Table IICorrelation between FPSS score and
clinicopathological characteristics of patients. |
Table II
Correlation between FPSS score and
clinicopathological characteristics of patients.
| No. of patients (%)
| |
|---|
| Characteristic | FPSS 0 and 1
(n=153) | FPSS 2 and 3
(n=150) | FPSS 4 and 5
(n=54) | P-value |
|---|
| Gender | | | | |
| Male | 95 (62.1) | 92 (61.3) | 27 (50.0) | 0.267 |
| Female | 58 (37.9) | 58 (38.7) | 27 (50.0) | |
| Age (years ± SD) | 69.6±10.7 | 69.8±10.9 | 68.8±12.1 | 0.906 |
| Location of
tumor | | | | |
| Colon | 108 (70.6) | 106 (70.7) | 38 (70.4) | 0.999 |
| Rectum | 45 (29.4) | 44 (29.3) | 16 (29.6) | |
| Depth of tumor | | | | |
| T1 | 46 (30.1) | 0 | 0 | <0.0001 |
| T2 | 52 (34.0) | 8 (5.3) | 0 | |
| T3 | 55 (35.9) | 134 (89.4) | 52 (96.3) | |
| T4 | 0 | 8 (5.3) | 2 (3.7) | |
| Nodal metastasis | | | | |
| No | 147 (96.1) | 55 (36.7) | 3 (5.6) | <0.0001 |
| Yes | 6 (3.9) | 95 (63.3) | 51 (94.4) | |
| Lymphatic
invasion | | | | |
| No | 146 (95.4) | 67 (44.7) | 1 (1.9) | <0.0001 |
| Yes | 7 (4.6) | 83 (55.3) | 53 (98.1) | |
| Venous invasion | | | | |
| No | 144 (94.1) | 107 (71.3) | 8 (14.8) | <0.0001 |
| Yes | 9 (5.9) | 43 (28.7) | 46 (85.2) | |
| Resection | | | | |
| Curative | 152 (99.3) | 126 (84.0) | 37 (68.5) | <0.0001 |
| Non-curative | 1 (0.7) | 24 (16.0) | 17 (31.5) | |
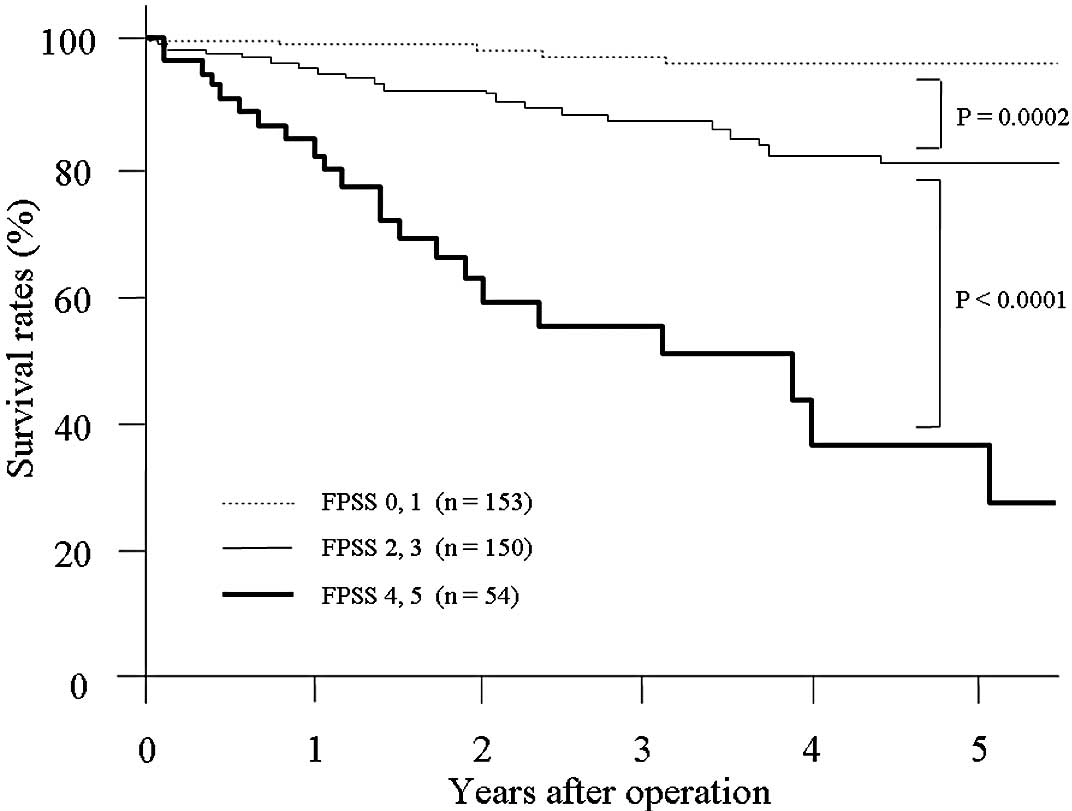
The 1-, 3- and 5-year survival rates of patients
with FPSS scores of 0 and 1 were 99.3, 97.4 and 96.2%,
respectively. The rates were 95.5, 87.1 and 80.5%, respectively, in
patients with FPSS scores of 2 and 3; and 83.8, 54.7 and 35.8%,
respectively, in patients with FPSS scores of 4 and 5. A
statistically significant difference was observed between the
survival of patients with FPSS scores of 0 and 1, and 2 and 3 (P=
0.0002); as well as FPSS scores of 2 and 3, and 4 and 5
(P<0.0001; Fig. 1).
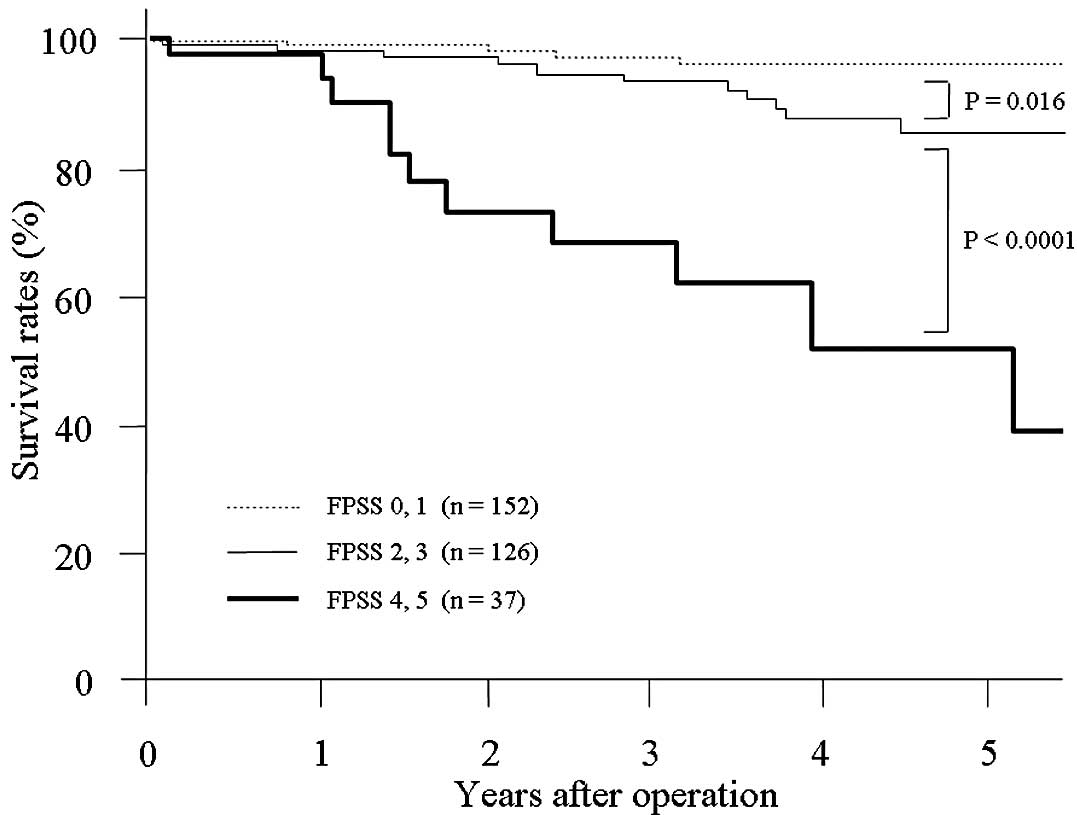
Subsequently, an investigation restricted to 315
patients who had been treated with curative resection was
performed. Similarly, a significant correlation was observed
between FPSS scores and certain investigated tumor-related factors
(Table III). The 1-, 3- and 5-year
survival rates of patients with FPSS scores of 0 and 1 were 99.3,
97.4 and 96.2%, respectively. Such rates were 98.2, 93.6 and 85.8%,
respectively, in patients with FPSS scores of 2 and 3; and 97.2,
67.7 and 51.3%, respectively, in patients with FPSS scores of 4 and
5. Additionally, a significant difference was observed between
survival of patients with FPSS scores of 0 and 1, and 2 and 3 (P=
0.016); and FPSS scores of 2 and 3, and 4 and 5 (P<0.0001;
Fig. 2).
 | Table IIICorrelation between FPSS score and
clinicopathological characteristics of patients treated with
curative resection. |
Table III
Correlation between FPSS score and
clinicopathological characteristics of patients treated with
curative resection.
| No. of patients (%)
| |
|---|
| Characteristic | FPSS 0 and 1
(n=152) | FPSS 2 and 3
(n=126) | FPSS 4 and
5(n=37) | P-value |
|---|
| Gender | | | | |
| Male | 95 (62.5) | 79 (62.7) | 19 (51.4) | 0.419 |
| Female | 57 (37.5) | 47 (37.3) | 18 (48.6) | |
| Age (years ± SD) | 69.6±10.8 | 70.2±11.0 | 71.8±10.9 | 0.268 |
| Location of
tumor | | | | |
| Colon | 108 (71.1) | 92 (73.0) | 24 (64.9) | 0.630 |
| Rectum | 44 (28.9) | 34 (27.0) | 13 (35.1) | |
| Depth of tumor | | | | |
| T1 | 46 (30.3) | 0 | 0 | <0.0001 |
| T2 | 52 (34.2) | 6 (4.8) | 0 | |
| T3 | 54 (35.5) | 114 (90.4) | 35 (94.6) | |
| T4 | 0 | 6 (4.8) | 2 (5.4) | |
| Nodal metastasis | | | | |
| No | 146 (96.1) | 48 (38.1) | 3 (8.1) | <0.0001 |
| Yes | 6 (3.9) | 78 (61.9) | 34 (91.9) | |
| Lymphatic
invasion | | | | |
| No | 145 (95.4) | 56 (44.4) | 1 (2.7) | <0.0001 |
| Yes | 7 (4.6) | 70 (55.6) | 36 (97.3) | |
| Venous
invasion | | | | |
| No | 143 (94.1) | 93 (73.8) | 4 (10.8) | <0.0001 |
| Yes | 9 (5.9) | 33 (26.2) | 33 (89.2) | |
Discussion
We have previously demonstrated the prognostic
significance of the Pathologic Prognostic Score (PPS), determined
by pathological tumor-related factors including depth of the tumor,
lymph node metastasis, lymphatic invasion and venous invasion,
which has provided a useful prognostic stratification for patients
with gastric carcinoma (5) and
colorectal carcinoma (1).
Poorly differentiated or undifferentiated carcinoma,
including poorly differentiated adenocarcinoma and mucinous
carcinoma of the colon and rectum, has been reported to possess a
more aggressive biological potential compared with differentiated
carcinomas (2,6–9).
Certain clinical and experimental studies have been conducted to
identify the subtype among colorectal poorly differentiated
carcinoma that has a more progressive potential or causes a more
unfavorable prognosis of patients (10–13).
However, to the best of our knowledge, there have been no studies
regarding a criterion for determining the tumor stage of poorly
differentiated or undifferentiated colon and rectal carcinoma.
Therefore, we set out to create an evolved
criterion, FPSS, based on data regarding the histopathological
tumor type, separating poorly differentiated and differentiated
carcinoma, in addition to PPS, to potentially determine the
prognosis of patients with colorectal carcinoma. A significant
difference in prognosis was found between patients who had FPSS
scores of 0 and 1, and 2 and 3; and FPSS scores of 2 and 3, and 4
and 5. Therefore, there is evidence to suggest that the quality of
stratification observed in the classification system was useful.
Moreover, an analysis of patients who had been treated with
curative resection demonstrated similar results.
As emphasized in previous studies, a novel criterion
for determining the prognosis of cancer patients has the potential
for simple and useful application (1,5,14).
While the five histopathological tumor-related factors comprising
the FPSS are relatively common and the majority of medical
institutes are capable of examining them, surgeons would benefit
from the convenience of the clinical application of the FPSS in
treating patients with colorectal carcinoma. This is due to the
fact that the FPSS provides useful information regarding the
clinical outcomes of patients. In conclusion, the FPSS may be a
useful criteria for predicting the clinical outcome of patients
with colorectal carcinoma.
References
|
1
|
Nozoe T, Mori E, Iguchi T, Kohno M, Maeda
T, Ezaki T and Sueishi K: Pathological prognostic score; A rational
criteria to stratify survival in colorectal carcinoma. J Surg
Oncol. 106:243–247. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nozoe T, Mori E, Takahashi I and Ezaki T:
Preoperative elevation of serum C-reactive protein as an
independent prognostic indicator of colorectal carcinoma. Surg
Today. 38:597–602. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Japanese Society for Cancer of the Colon
and Rectum: Japanese classification of colorectal carcinoma. 7th
English edition. Kanehara Company; Tokyo: pp. 6–33. 2009
|
|
4
|
Sobin L, Gospodarowicz M and Wittekind C:
International Union Against Cancer TNM classification of malignant
tumours. 7th edition. Wiley-Blackwell; New York: pp. 100–105.
2009
|
|
5
|
Nozoe T, Iguchi T, Egashira A, Adachi E,
Matsukuma A and Ezaki T: Pathological prognostic score as a simple
criterion to predict outcome in gastric carcinoma. J Surg Oncol.
102:73–76. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chung CK, Zaino RJ and Stryker JA:
Colorectal carcinoma: evaluation of histologic grade and factors
influencing prognosis. J Surg Oncol. 21:143–148. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nozoe T, Anai H, Nasu S and Sugimachi K:
Clinicopathological characteristics of mucinous carcinoma of the
colon and rectum. J Surg Oncol. 75:103–107. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nozoe T, Inutsuka S, Honda M, Ezaki T and
Korenaga D: Clinicopathologic significance of cyclin A expression
in colorectal carcinoma. J Exp Clin Cancer Res. 23:127–133.
2004.PubMed/NCBI
|
|
9
|
Takeuchi K, Kuwano H, Tsuzuki Y, Ando T,
Sekihara M, Hara T and Asao T: Clinicopathological characteristics
of poorly differentiated adenocarcinoma of the colon and rectum.
Hepatogastroenterology. 51:1698–1702. 2004.PubMed/NCBI
|
|
10
|
Kazama Y, Watanabe T, Kanazawa T, Tanaka
J, Tanaka T and Nagawa H: Microsatellite instability in poorly
differentiated adenocarcinomas of the colon and rectum:
relationship to clinicopathological features. J Clin Pathol.
60:701–704. 2007. View Article : Google Scholar
|
|
11
|
Ogawa M, Watanabe M, Eto K, et al: Poorly
differentiated adenocarcinoma of the colon and rectum: clinical
characteristics. Hepatogastroenterology. 55:907–911.
2008.PubMed/NCBI
|
|
12
|
Ueno H, Mochizuki H, Hashiguchi Y, et al:
Histological grading of colorectal cancer: a simple and objective
method. Ann Surg. 247:811–818. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ishihara S, Watanabe T, Akahane T, et al:
Tumor location is a prognostic factor in poorly differentiated
adenocarcinoma, mucinous adenocarcinoma, and signet-ring cell
carcinoma of the colon. Int J Colorectal Dis. 27:371–379. 2012.
View Article : Google Scholar
|
|
14
|
Nozoe T and Sugimachi K: Simple staging
criteria for esophageal carcinoma: classification with a strict
prognostic stratification. Am J Clin Oncol. 26:307–311. 2003.
View Article : Google Scholar : PubMed/NCBI
|