Introduction
Approximately 34–86% of neoplasms of the salivary
glands are located in the parotid gland (1,2).
Parotidectomy is the first-line treatment for parotid gland tumors
(3,4). However, complications experienced
following surgery significantly reduce patients’ quality of life
(5). The most common complications
are cosmetic deformity and Frey syndrome.
Owing to all or part of the parotid gland being
removed, facial depression is observed in almost 100% of patients
who have undergone parotidectomy. Frey syndrome was first described
by Łucja Frey in 1923 (6), with an
incidence of between 11 and 95% (7,8). It is
characterized by flushing or sweating on one side of the forehead,
face, scalp and neck occurring soon following the ingestion of
food, in response to salivatory stimulation (9).
The use of autogenous tissue interposition for the
prevention of Frey syndrome and cosmetic deformity during
parotidectomy are considered as simple, safe and effective
approaches by clinicians. The sternocleidomastoid myocutaneous flap
(SCMF) is one of the most commonly used autogenous tissues
(10). Numerous studies have been
performed to examine the effectiveness of the SCMF for the
prevention of cosmetic deformity and Frey syndrome, however, they
provide contradictory results and have small samples sizes with
consequently low statistical power.
In 2010, Sanabria et al (10) conducted a meta-analysis
investigating the effectiveness of the SCMF for the prevention of
cosmetic deformity and Frey syndrome. The study included two
randomized controlled trials (RCTs) (11,12).
At the time of writing this manuscript, eleven RCTs have been
published. A comprehensive systematic review and meta-analysis is
therefore required to provide an updated review of the
effectiveness of the SCMF for the prevention of cosmetic deformity
and Frey syndrome.
Materials and methods
Report terms
We attempted to follow the proposed PRISMA
(Preferred Reporting Items for Systematic Reviews and
Meta-Analyses) guidelines (13) to
report the present systematic review and meta-analysis.
Literature search
We conducted a PubMed and CNKI database search in
August 2012 for relevant studies that examined the effectiveness of
the SCMF for the prevention of cosmetic deformity and/or Frey
syndrome. The following search terms were used: i) Frey’s syndrome,
Frey syndrome, gustatory sweating, auriculotemporal syndrome,
cosmetic deformity, facial depression and cosmetic disfigurement;
ii) sternocleidomastoid. These two search terms were combined using
the Boolean operator ‘and’. No restrictions were imposed. In
addition, we examined the reference lists of the retrieved RCTs and
recently published reviews.
Study selection
We conducted an initial screening of titles or
abstracts. Following this, we performed a second screening based on
full-text review. Studies were considered eligible if they met the
following criteria: i) the study design was an RCT; ii) the study
included patients with benign or malignant parotid tumors who
underwent partial or total parotidectomy with facial nerve
preservation, and without a history of previous surgical procedures
in the parotid area or previous radiotherapy; iii) interventions
included the SCMF, and the control group constituted no SCMF or
other prophylactic measures; iv) the main outcome was incidence of
cosmetic deformity and/or Frey syndrome, determined with subjective
or objective measures; v) data of each outcome were reported or
obtained by contacting the corresponding author of the study.
Data extraction
Two authors (XJ Tian and YH Xiong) independently
extracted the following data for each eligible study: first
author’s last name, year of publication, site of origin, sample
size, characteristics of the SCMF and control groups, outcome
evaluation methods, length of follow-up, incidence of cosmetic
deformity and Frey syndrome. Any disagreements were resolved by
consulting a third author (XT Zeng).
Assessment of methodological quality
The methodological quality of each study was
assessed using the Cochrane collaboration’s tool for assessing risk
of bias (14), which contains the
following seven criteria: i) details of the randomization method;
ii) allocation concealment; iii) blinding of participants and
personnel; iv) blinding of outcome assessment; v) incomplete
outcome data; vi) selective outcome reporting and vii) other
sources of bias. Each study was assessed by two authors (DY Liu and
XJ Tian) independently and any disagreements were resolved by
consulting a third author (XT Zeng).
Data synthesis and analysis
We calculated risk ratios (RRs) and 95% confidence
intervals (CIs) for all studies with sufficient data. Heterogeneity
was examined using the Cochrane Q test and quantified with the
I2 statistic (15). The
value of the I2 statistic was used to select the
appropriate pooling method: if the I2 value was <50%,
the fixed-effects meta-analysis was applied; if the I2
value was ≥50%, the random-effects meta-analysis was used.
In the presence of heterogeneity, we performed
sensitivity analyses by removal of each study in turn in order to
examine the robustness of the main results. Potential publication
bias was investigated by visual assessment using a funnel plot and
further examined using a combination of the Egger regression test
(16) and the ‘trim and fill’
method (17).
Statistical analyses were conducted with
Comprehensive Meta-Analysis software, version 2.2 (Biostat,
Englewood, New Jersey, USA) (18).
For all comparisons, except those for heterogeneity, P<0.05 was
considered to indicate a statistically significant result. All
tests were two-sided.
Results
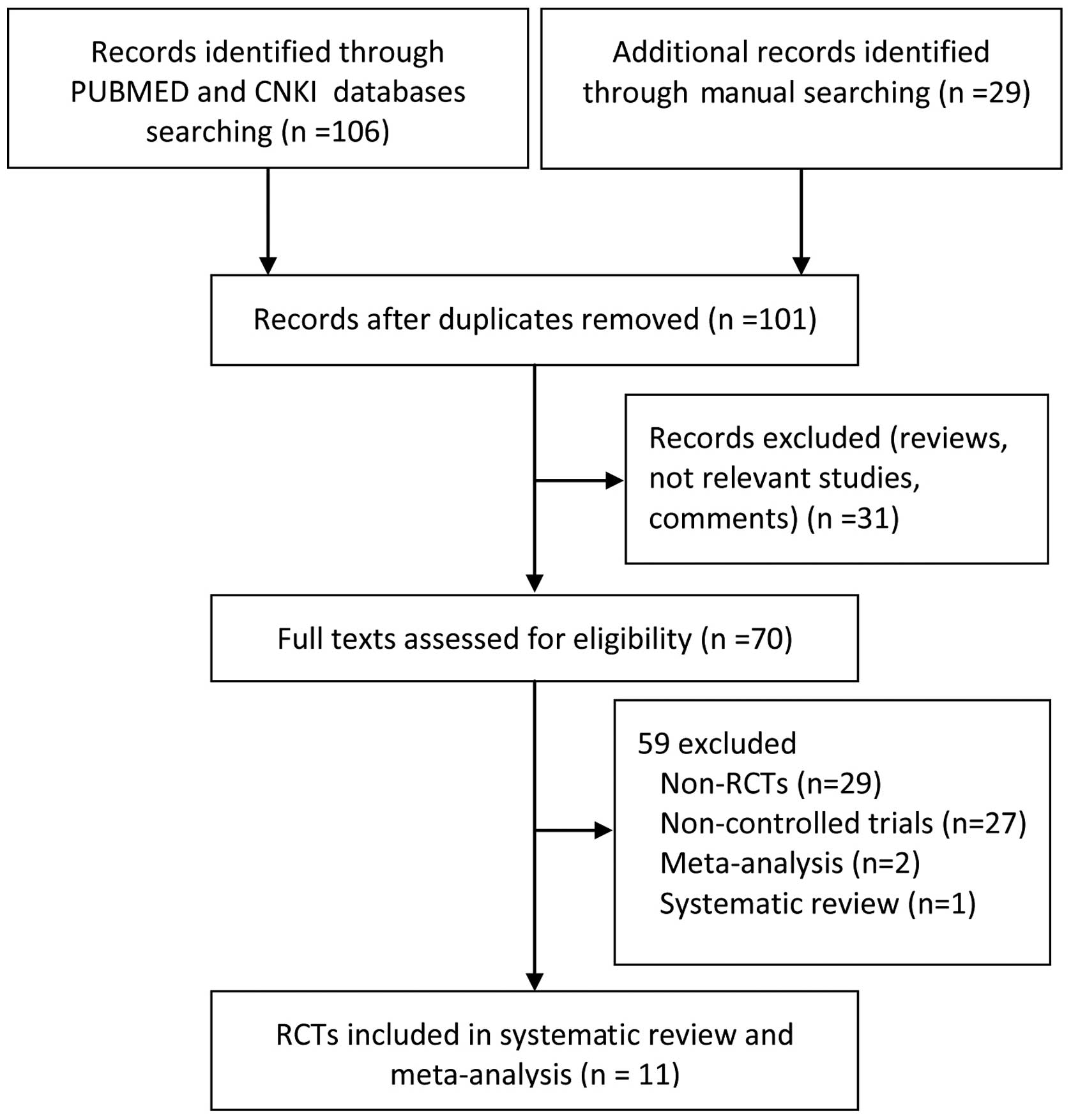
Search results
An initial search yielded 135 potentially relevant
studies and 11 RCTs (11,12,19–27)
were selected for the purpose of our analysis. Fig. 1 depicts a flowchart showing the
study selection process and their characteristics are listed in
Table I.
 | Table ICharacteristics of included RCTs. |
Table I
Characteristics of included RCTs.
| Author (ref.) | Year | Country | Sample size
(T/C) | Age (years) | Gender
(male:female) | Intervention
| Follow-up | Outcome |
|---|
| T | C |
|---|
| Kerawala et al
(11) | 2002 | UK | 21/15 | range, 26–81 | 23:13 | SCMF | Blank | 1–6 years | Cosmetic result; Frey
syndrome (objective and subjective) |
| Chen and Yang
(20) | 2004 | China | 108/64 | range,
14–78
mean, 43.2 | 96:76 | SCMF | Blank | 6 months–5 years | Cosmetic result; Frey
syndrome (objective) |
| Asal et al
(12) | 2005 | Turkey | 12/12 | mean, 49
(T)
mean, 50 (C) | 7:5/5:7 | SCMF | Blank | 9–48 months | Cosmetic result; Frey
syndrome (objective and subjective) |
| Zhao et al
(19) | 2005 | China | 57/33 | range,
12–79
mean, 33.92 | 32:58 | SCMF | Blank | 2 years | Cosmetic result; Frey
syndrome (objective and subjective) |
| Zhi et al
(21) | 2007 | China | 38/47 | range,
14–68
mean, 35.2 | 32:53 | SCMF | Blank | 1 year | Cosmetic result;
Frey syndrome (objective and subjective) |
| Ding et al
(22) | 2010 | China | 60/60 | range,
20–83
mean, 53.6 | 69:51 | SCMF | Blank | 18 months–5
years | Cosmetic result;
Frey syndrome (objective) |
| Liao et al
(23) | 2010 | China | 48/20 | mean, 45
(T)
mean, 48 (C) | 26:22/11:9 | SCMF | Blank | 6 months–3
years | Cosmetic result;
Frey syndrome (subjective) |
| Deng et al
(24) | 2011 | China | 52/95 | range,
21–72
mean, 52.5 | NA | SCMF | Blank | 6 months–5
years | Cosmetic result;
Frey syndrome (objective) |
| Peng and Chen
(25) | 2011 | China | 22/23 | range, 19–72 | 26:19 | SCMF | Blank | 3 months–2
years | Frey syndrome
(subjective) |
| Zhao et al
(26) | 2011 | China | 23/15 | range, 18–65 | NA | SCMF | Blank | 1 year–25
months | Frey syndrome
(objective and subjective) |
| Li and Xu (27) | 2012 | China | 20/20 | mean ± SD,
35.3±4.6 | 22:18 | SCMF | Blank | 10 months–3
years | Cosmetic result;
Frey syndrome (objective) |
Methodological quality
Table II shows the
quality of RCTs according to the Cochrane collaboration’s tool. It
refers to randomization only, lacking information with regard to
allocation concealment and blinding; however, no incomplete outcome
data, no selective outcome reporting and other sources of bias were
observed. Therefore, there was a moderate risk of bias.
 | Table IIQuality of included RCTs according to
the Cochrane collaboration’s tool. |
Table II
Quality of included RCTs according to
the Cochrane collaboration’s tool.
| Author (ref.) | Year | Randomization
method | Allocation
concealment | Incomplete outcome
data | Blinding of
participants and personnel | Blinding of outcome
assessment | Selective outcome
reporting | Other sources of
bias |
|---|
| Kerawala et
al (11) | 2002 | Low | Unclear | Low | Low | Low | Low | Low |
| Chen and Yang
(20) | 2004 | Unclear | Unclear | Unclear | Low | Low | Low | Low |
| Asal et al
(12) | 2005 | Unclear | Unclear | Low | Low | Low | Low | Low |
| Zhao et al
(19) | 2005 | Unclear | Unclear | Unclear | Unclear | Low | Low | Low |
| Zhi et al
(21) | 2007 | Unclear | Unclear | Unclear | Low | Low | Low | Low |
| Ding et al
(22) | 2010 | Unclear | Unclear | Unclear | Low | Low | Low | Low |
| Liao et al
(23) | 2010 | High | Unclear | Unclear | Unclear | Low | Low | Low |
| Deng et al
(24) | 2011 | Unclear | Unclear | Unclear | Low | Low | Low | Low |
| Peng and Chen
(25) | 2011 | Unclear | Unclear | Unclear | Low | Low | Low | Low |
| Zhao et al
(26) | 2011 | Unclear | Unclear | Unclear | Low | Low | Low | Low |
| Li and Xu (27) | 2012 | Unclear | Unclear | Unclear | Unclear | Low | Low | Low |
Frey syndrome
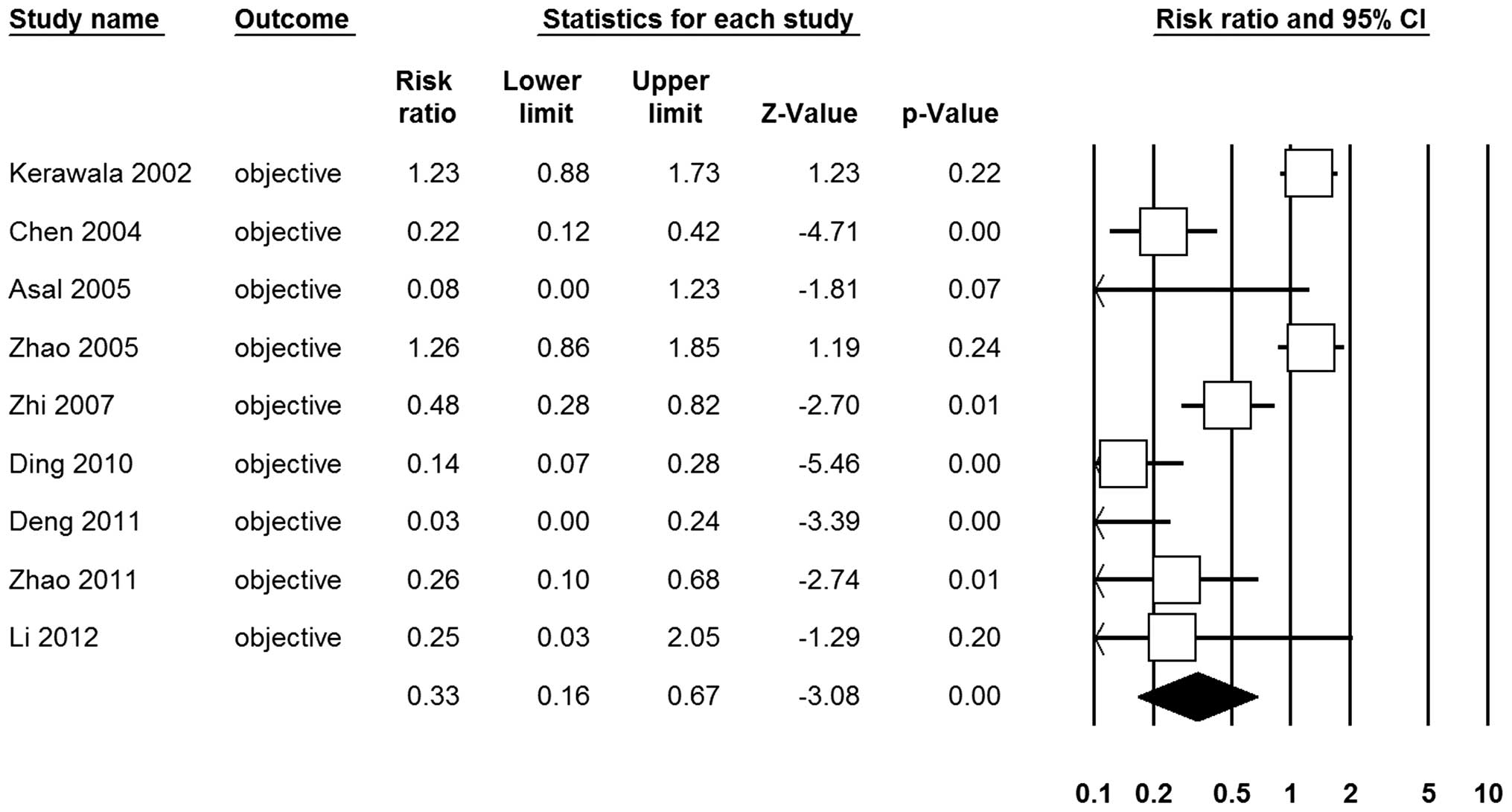
Nine RCTs (11,12,19–22,24,26,27)
reported the incidence of objective Frey syndrome by performing the
starch-iodine test. A significant heterogeneity was observed
(I2=88.79%, P<0.10), therefore we used a
random-effects model. The meta-analysis demonstrated that the SCMF
markedly decreased the risk of incidence of Frey syndrome (67%; RR,
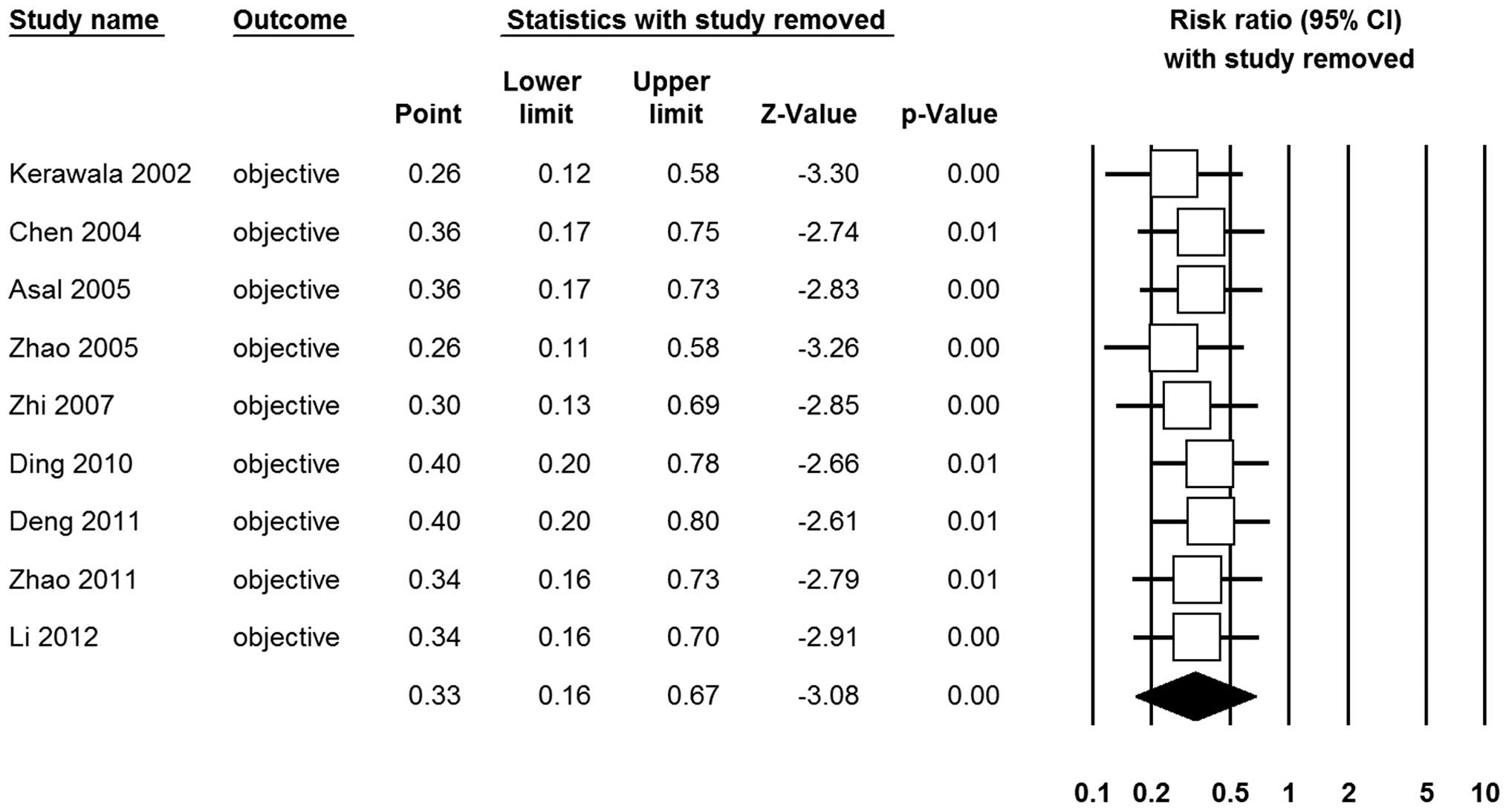
0.33; 95% CI, 0.16–0.67; P<0.01; Fig. 2). Sensitivity analysis was performed
by sequential removal of each study and the significance of pooled
RR was not influenced by omitting any single study, suggesting that
the result of this meta-analysis was stable (Fig. 3).
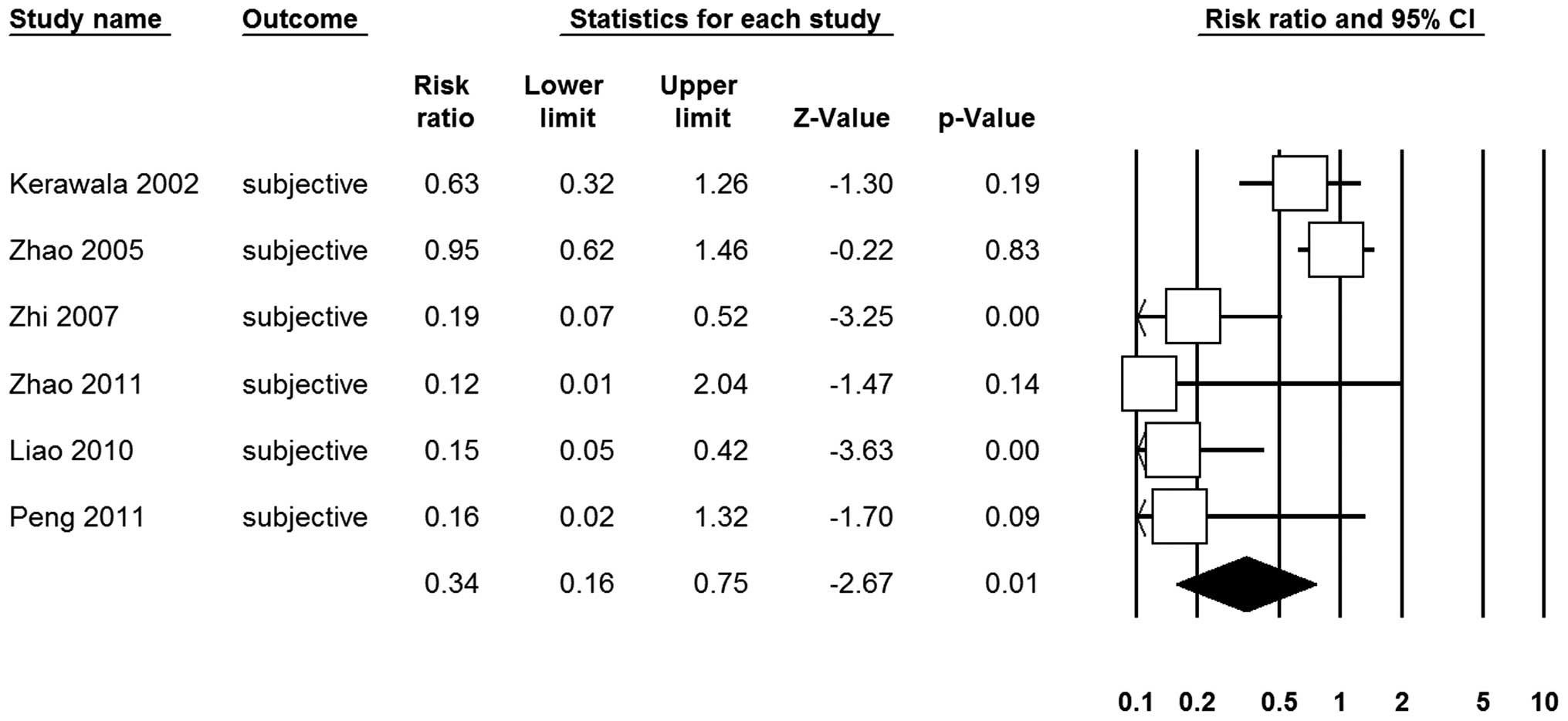
Seven RCTs (11,12,19,21,23,26,28)
reported the subjective incidence of Frey syndrome. A significant
heterogeneity (I2=74.24%, P<0.10) was observed,
therefore, the random-effects model was used. The result also
demonstrated that there was a significant correlation towards a
lower risk of incidence in the SCMF group (66%; RR, 0.34; 95% CI,
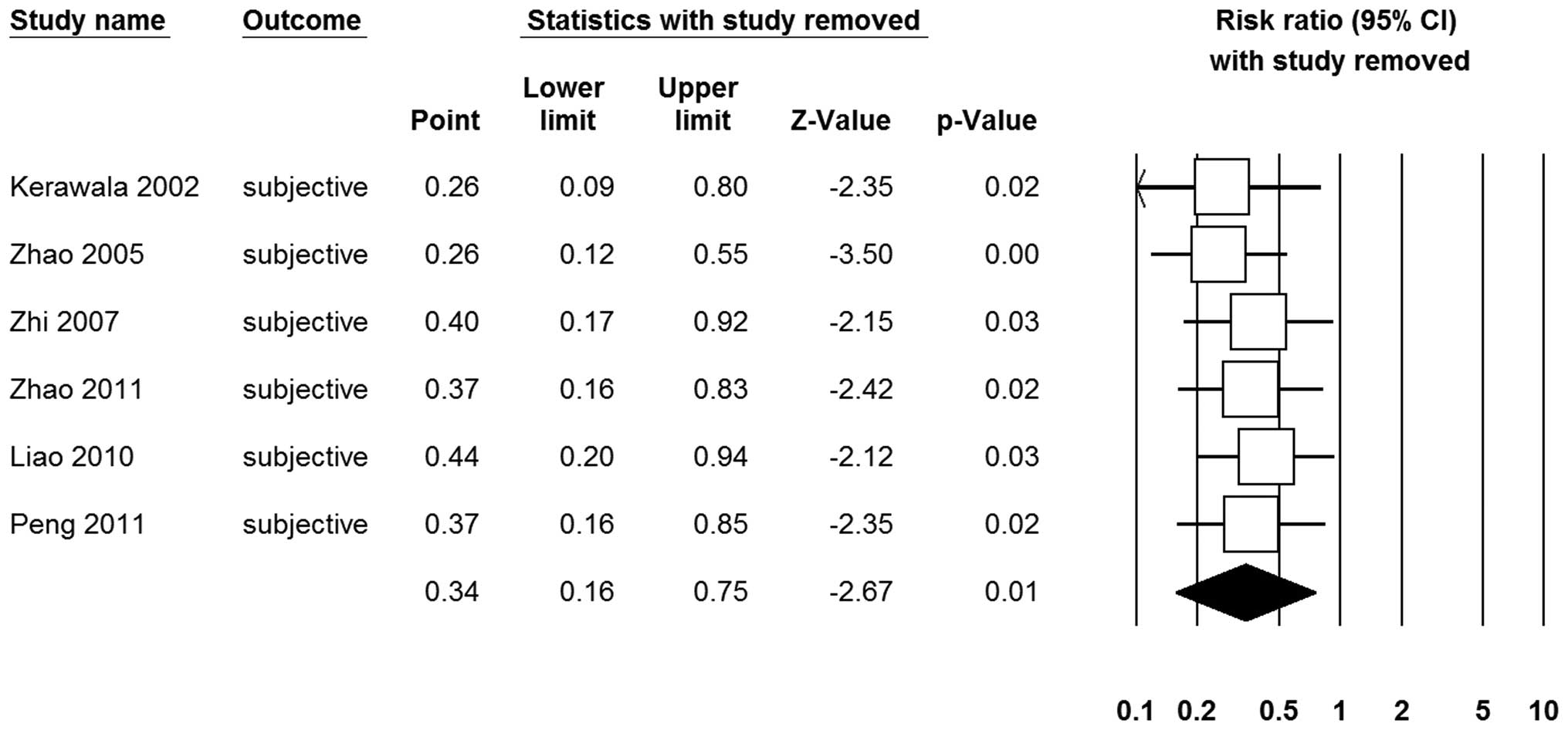
0.16–0.75; P= 0.01; Fig. 4). The
sensitivity analysis also indicated this result was significant
(Fig. 5).
Cosmetic results
Nine RCTs (11,12,19–24,27)
observed cosmetic deformity. Due to the considerable variation
among included studies, meta-analysis was not applicable. Two RCTs
(11,12) demonstrated that the differences
between the SCMF and no SCMF group were not statistically
significant, while the other seven RCTs demonstrated that the
cosmetic appearance was statistically significant for the SCMF
group compared with the no SCMF group. Table III indicates a qualitative analysis
of the evidence.
 | Table IIICosmetic result of included RCTs. |
Table III
Cosmetic result of included RCTs.
| Author (ref.) | Year | Assessment
method | Result (C/T) | Conclusion |
|---|
| Kerawala et
al (11) | 2002 | VAS | Subjective:
1.5±1.6/2.6±2.1, P=0.13; objective: 2.8±1.3/3.5±1.3, P=0.12 | Insignificant
difference |
| Chen and Yang
(20) | 2004 | Doctor
observed |
Marked/inconspicuous, P<0.01 | Significant
difference |
| Asal et al
(12) | 2005 | Questionnaire and
doctor observed | Subjective: all
patients were pleased with the cosmetic result; objective: the
facial contours of 7/6 patients were not unsatisfied to the
otolaryngologist | Insignificant
difference |
| Zhao et al
(19) | 2005 | Questionnaire | 22/4 patients felt
unsatisfied, P<0.05 | Significant
difference |
| Zhi et al
(21) | 2007 | Questionnaire | 26/4 patients
experienced earlobe depression, P<0.05 | Significant
difference |
| Ding et al
(22) | 2010 | Doctor and patient
observed | 60/2 patients
experienced facial depression, P=0.0014 | Significant
difference |
| Liao et al
(23) | 2010 | Doctor
observed | 18/8 patients
experienced facial depression, P<0.01 | Significant
difference |
| Deng et al
(24) | 2011 | Doctor observed and
examined | 79/6 patients
experienced facial depression, P<0.01 | Significant
difference |
| Li and Xu (27) | 2012 | Reported by
patient | 6/0 patients felt
unsatisfied, P<0.05 | Significant
difference |
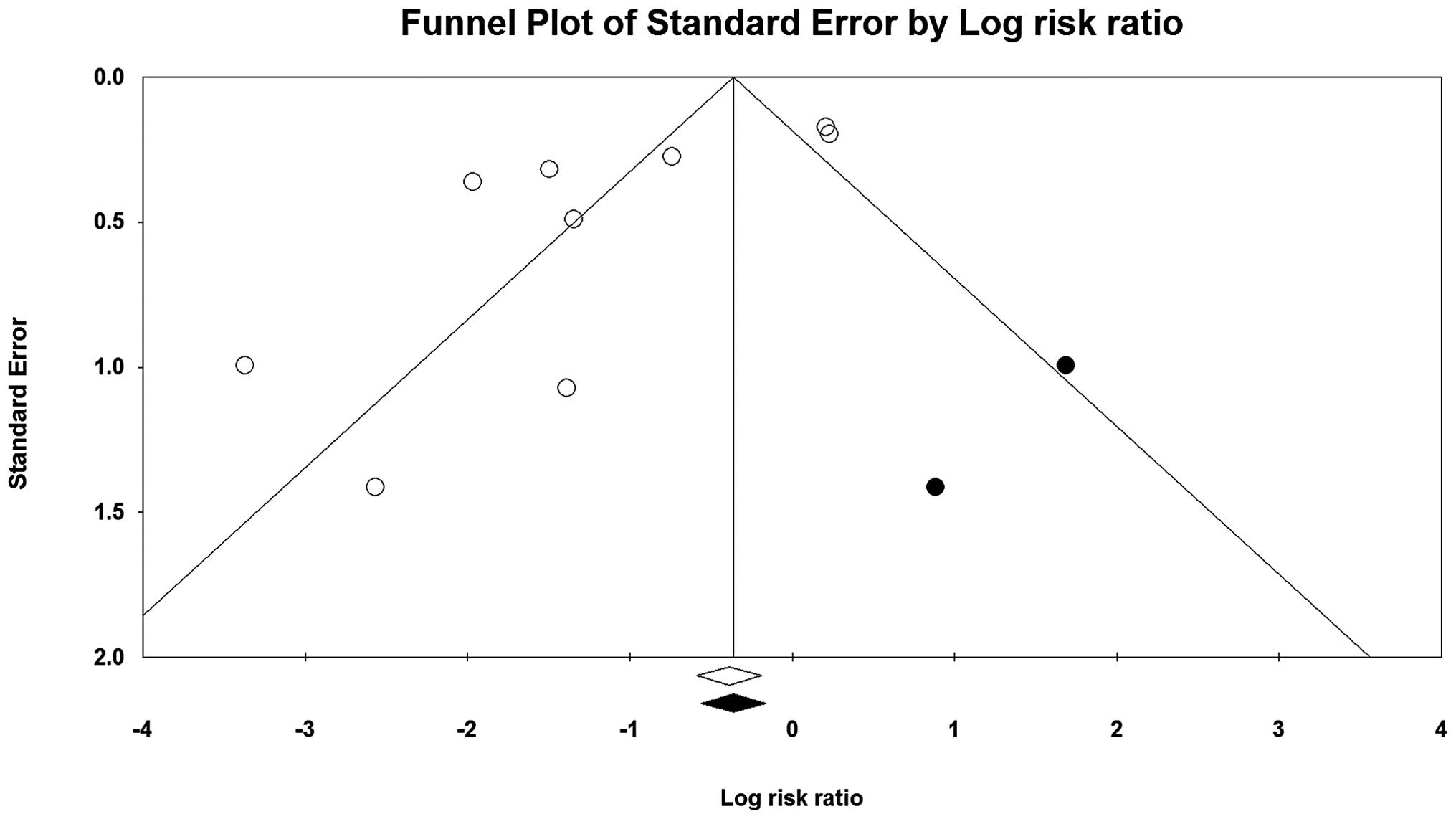
Publication bias
Fig. 6 demonstrates
that the funnel plot was asymmetrical (based on the evaluation of
objective Frey syndrome), which indicated that publication bias
existed (white circles). The Egger linear regression also detected
moderate publication bias among studies (Egger, P=0.023). As
evidence of bias may be due to inadequate statistical power, we
used a non-parametric method of ‘trim and fill’ to estimate two
possible missing studies (black spots in Fig. 6), the estimated RR including the
‘missing’ studies was not substantially different from our estimate
with an adjustment for the missing studies (RR, 0.43; 95% CI,
0.22–0.88).
Discussion
In 1927, Andre Thomas described the pathophysiology
of Frey syndrome as the aberrant regeneration of sectioned
para-sympathetic fibers, which regrow to innervate the vessels and
sweat glands of the skin overlying the parotid (29). Accepting this pathophysiology and
mechanism, oral and maxillofacial surgeons proposed to interpose
any tissue between the parotid bed and the skin, including the
temporoparietal fascia rotational flap (30), the superficial muscular aponeurotic
system (SMAS) (31), the SCMF and
AlloDerm (32), with the aim of
inhibiting aberrant innervation in order to prevent Frey
syndrome.
Compared with other autogenous tissue flaps, the
SCMF has several advantages: i) it is easy to rotate into the
parotid region without another incision; ii) it is long enough to
cover all the branches of the facial nerve; iii) it decreases the
depression of the surgical area following parotid gland resection;
iv) there is a low risk of necrosis of the flap due to its
vascularization and v) there is a low risk of complications,
primarily, spinal accessory nerve injury (10). Compared with AlloDerm, the major
advantage of the SCMF is that it is more cost-effective.
The first RCT of the SCMF was published by Kerawala
et al (11) in 2002.
Following that, a number of additional RCTs have been published.
However, the results are inconsistent. In 2010, Sanabria et
al (10) performed a
meta-analysis of the SCMF for the prevention of Frey syndrome,
including two RCTs and nine non-RCTs. The authors concluded that
the result of their meta-analysis was inconclusive with regard to
the use of the SCMF as an intervention for the prevention of Frey
syndrome following parotid surgery. It is widely considered that a
non-RCT design introduces a higher degree of bias compared with a
RCT design. Therefore, we conducted a meta-analysis which only
included RCTs in order to obtain a more accurate result. The
meta-analysis demonstrated that the SCMF is capable of clearly
decreasing the incidence of objective and subjective Frey syndrome.
The sensitivity analysis indicated that the result was robust.
Some studies have indicated that the SCMF evidently
improves cosmetic appearance compared with no flap and some studies
have indicated that this function was limited. Due to the
considerable variation of the assessment methods used among the
included studies, a meta-analysis was not performed. Our systematic
review did not obtain a conclusive result.
However, there are some limitations with regard to
our systematic review and meta-analysis that should be stated.
Firstly, the sample size of the studies contributing a significant
amount of data to this meta-analysis was small, therefore, we were
not able to adequately assess the effects of prevention. Secondly,
the methodological quality of the included RCTs demonstrated a
moderate risk of bias, and a lack of information with regard to
randomization, allocation concealment and blinding (some RCTs) may
have introduced bias. Thirdly, a significant heterogeneity between
RCTs existed, although sensitivity analysis revealed that it did
not exert a significant influence. However, its potential influence
should be considered. Fourthly, publication bias was detected, the
‘file-drawer’ effect may exist and our ‘trim and fill’ analysis
also estimated two possible missing studies, which indicated that
studies that reported negative results may be more likely to remain
unpublished. Lastly, a meta-analysis of the cosmetic result was not
conducted, so we are not able to obtain a quantitative result for
this outcome.
For future studies, we suggest that the same
measurment tools are applied, including the 10-cm visual analog
scale (11) and blinding to the
evalution of the cosmetic result. We also suggest to perform RCTs
to compare the effectiveness of the SCMF and AlloDerm, as AlloDerm
is presently widely used for the prevention of Frey syndrome
(32). We recommend the use of the
starch-iodine test and blinding in studies with regard to Frey
syndrome in future studies. As the method of parotidectomy
(4) and prognosis (33) are influenced by benign and malignant
tumors, diagnosis of parotid gland lesions prior to parotidectomy
is important. A well accepted, safe, reliable, minimally invasive
and cost-effective method, including fine needle aspiration
cytology (34) is recommended.
In conclusion, based on currently available
evidence, the use of the SCMF is beneficial for the prevention of
Frey syndrome, however, whether it is additionally benefical for
cosmetic deformity remains inconclusive.
Acknowledgements
This study was supported by the
Foundation of Education and Science Planning Project of Hubei
Province (No. 2012A050), the Nature Science Foundation of Hubei
Province (2012FFB03902) and the Intramural Research Program of the
Hubei University of Medicine (No. 2011CZX01), without commercial or
not-for-profit sectors.
References
|
1
|
Vuhahula EA: Salivary gland tumors in
Uganda: clinical pathological study. Afr Health Sci. 4:15–23.
2004.PubMed/NCBI
|
|
2
|
Pinkston JA and Cole P: Incidence rates of
salivary gland tumors: results from a population-based study.
Otolaryngol Head Neck Surg. 120:834–840. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Clayman MA, Clayman SM and Seagle MB: A
review of the surgical and medical treatment of Frey syndrome. Ann
Plast Surg. 57:581–584. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ali NS, Nawaz A, Rajput S and Ikram M:
Parotidectomy: a review of 112 patients treated at a teaching
hospital in Pakistan. Asian Pac J Cancer Prev. 11:1111–1113.
2010.PubMed/NCBI
|
|
5
|
Nitzan D, Kronenberg J, Horowitz Z, et al:
Quality of life following parotidectomy for malignant and benign
disease. Plast Reconstr Surg. 114:1060–1067. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dulguerov P, Marchal F and Gysin C: Frey
syndrome before Frey: the correct history. Laryngoscope.
109:1471–1473. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Koch M, Zenk J and Iro H: Long-term
results of morbidity after parotid gland surgery in benign disease.
Laryngoscope. 120:724–730. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
de Bree R, van der Waal I and Leemans CR:
Management of Frey syndrome. Head Neck. 29:773–778. 2007.
|
|
9
|
Dulguerov P, Quinodoz D, Vaezi A, Cosendai
G, Piletta P and Lehmann W: New objective and quantitative tests
for gustatory sweating. Acta Otolaryngol. 119:599–603. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sanabria A, Kowalski LP, Bradley PJ, et
al: Sternocleidomastoid muscle flap in preventing Frey’s syndrome
after parotidectomy: a systematic review. Head Neck. 34:589–598.
2012.
|
|
11
|
Kerawala CJ, McAloney N and Stassen LF:
Prospective randomised trial of the benefits of a
sternocleidomastoid flap after superficial parotidectomy. Br J Oral
Maxillofac Surg. 40:468–472. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Asal K, Köybaşioğlu A, Inal E, et al:
Sternocleidomastoid muscle flap reconstruction during parotidectomy
to prevent Frey’s syndrome and facial contour deformity. Ear Nose
Throat J. 84:173–176. 2005.
|
|
13
|
Liberati A, Altman DG, Tetzlaff J, et al:
The PRISMA statement for reporting systematic reviews and
meta-analyses of studies that evaluate health care interventions:
explanation and elaboration. J Clin Epidemiol. 62:e1–e34. 2009.
View Article : Google Scholar
|
|
14
|
Zeng XT, Bao CP, Cao SY and Liu JY: The
third part in meta-analysis series: Tools for assessing the quality
of randomized controlled trials. Chin J Evid Based Cardiovasc Med.
4:183–185. 2012.(In Chinese).
|
|
15
|
Higgins JP and Thompson SG: Quantifying
heterogeneity in a meta-analysis. Stat Med. 21:1539–1558. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Egger M, Davey Smith G, Schneider M and
Minder C: Bias in meta-analysis detected by a simple, graphical
test. BMJ. 315:629–634. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Duval S and Tweedie R: Trim and fill: A
simple funnel-plot-based method of testing and adjusting for
publication bias in meta-analysis. Biometrics. 56:455–463. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Borenstein M, Hedges L and Rothstein H:
Comprehensive Meta-analysis. Biostat; Englewood, New Jersey: 2005,
(computer software).
|
|
19
|
Zhao HW, Li LJ, Han B, Liu H and Pan J: A
retrospective study on the complications after modified
parotidectomy in benign tumors of parotid gland. Hua Xi Kou Qiang
Yi Xue Za Zhi. 23:53–56. 2005.(In Chinese).
|
|
20
|
Chen R and Yang K: Clinical observation of
sternocleidomastoid muscle flap to repair tissue defect and in
preventing Frey syndrome after parotidectomy. Chongqing Yi Yao.
33:94–95. 2004.(In Chinese).
|
|
21
|
Zhi KQ, Wen YM and Li LJ: Prevention and
treatment of complications following benign parotid neoplasms
dissection. Xi’an Jiao Tong Da Xue Xue Bao Yi Xue Ban. 28:116–117.
2007.(In Chinese).
|
|
22
|
Ding YF, Zhou PG, Gu YC and Chen Y:
Applicatian of sterno-cleidomastoid muscle flap in benign
parotidectomies. J Dental Preven Treat. 18:88–90. 2010.(In
Chinese).
|
|
23
|
Liao SK, Li JC, Zhang K, Xu JC, Xu T and
Chen YF: Clinical study of 48 cases of sternocleidomastoid muscle
flap in parotidectomy. Shi Yong Kou Qiang Yi Xue Za Zhi. 3:106–108.
2010.(In Chinese).
|
|
24
|
Deng YQ, Zheng CS and Zhu YM: Application
of sternocleidomastoid muscle flap and parotid fascia flap in
operation on parotid benign tumor. J Oral Sci Res. 27:893–895.
2011.
|
|
25
|
Peng YF and Chen YD: Clinical analysis of
45 cases of parotid gland tumor. Zhong Wai Jian Kang Wen Zhai.
8:2412011.(In Chinese).
|
|
26
|
Zhao J, Yu HQ and Liu AG: Comparative
study of using heterogeneous acellular dermal matrix and
reconstruction of parotid masseter faseial in prevention of Frey’s
syndrome after parotidectomy. Stomatology. 31:356–359. 2011.(In
Chinese).
|
|
27
|
Li MD and Xu JM: Clinical study of veiled
incision and sternocleidomastoid flap in the operation of parotid
gland tumor. Zhong Wai Yi Liao. 31:44–46. 2012.(In Chinese).
|
|
28
|
Li C, Fan JC, Li B, et al: Meta-analysis
of surgical techniques for preventing Frey syndrome and a concave
facial deformity after parotidectomy. Zhonghua Er Bi Yan Hou Tou
Jing Wai Ke Za Zhi. 46:580–585. 2011.(In Chinese).
|
|
29
|
Glaister DH, Hearnshaw JR, Heffron PF,
Peck AW and Patey DH: The mechanism of post-parotidectomy gustatory
sweating (the auriculo-temporal syndrome). Br Med J. 2:942–946.
1958. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rubinstein RY, Rosen A and Leeman D: Frey
syndrome: treatment with temporoparietal fascia flap interposition.
Arch Otolaryngol Head Neck Surg. 125:808–811. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Moulton-Barrett R, Allison G and Rappaport
I: Variation’s in the use of SMAS (superficial musculoaponeurotic
system) to prevent Frey’s syndrome after parotidectomy. Int Surg.
81:174–176. 1996.
|
|
32
|
Zeng XT, Tang XJ, Wang XJ, et al: AlloDerm
implants for prevention of Frey syndrome after parotidectomy: a
systematic review and meta-analysis. Mol Med Rep. 5:974–980.
2012.PubMed/NCBI
|
|
33
|
Oliveira LR, Soave DF, Oliveira-Costa JP,
Zorgetto VA and Ribeiro-Silva A: Prognostic factors in patients
with malignant salivary gland neoplasms in a Brazilian population.
Asian Pac J Cancer Prev. 12:363–368. 2011.PubMed/NCBI
|
|
34
|
Nguansangiam S, Jesdapatarakul S, Dhanarak
N and Sosrisakorn K: Accuracy of fine needle aspiration cytology of
salivary gland lesions: routine diagnostic experience in Bangkok,
Thailand. Asian Pac J Cancer Prev. 13:1583–1588. 2012. View Article : Google Scholar
|