Introduction
A solid pseudopapillary tumor of the pancreas (SPTP)
is an uncommon pancreatic neoplasm with low malignancy that
accounts for 0.1–3% of all exocrine pancreatic tumors, with marked
adolescent female predominance. Since first described by Frantz in
1959 (1), there have been >700
well-documented cases of SPTP reported in the published English
literature (2). However, SPTs that
occur as primary tumors outside the pancreas are exceedingly rare.
The most common extrapancreatic site is the mesocolon or omentum,
where the tumor arises in ectopic pancreatic tissue (3–6).
Recently, Miyazaki et al reported one case of SPT without an
ectopic pancreas in the retroperitoneum, a site not previously
recognized to harbor this type of tumor (7). The present study reports an additional
case of this rare tumor located in the retroperitoneum and the
histological observations of a pancreatic rest. A detailed review
of the literature concerning extrapancreatic STPs is also
discussed. Written informed consent was obtained from the
patient.
Case report
A 22-year-old female was referred to the First
Affiliated Hospital of Zhejiang University School of Medicine,
Zheijang, China, due to the presence of a retroperitoneal
5.9×5.3-cm mass in the left adrenal region that was detected by B
ultrasound (BUS) imaging during a routine annual examination
(Fig. 1). The patient did not
complain of fatigue, fever or pain, and the patient’s body weight
and blood pressure were within the corresponding normal ranges. The
complete blood count, chemistry panel, urine analysis and liver
function test results were also normal. An endocrinological
evaluation of the adrenal gland, including an analysis of the
plasma rennin activity and aldosterone, steroid and catecholamine
levels, excluded a functional adenoma. Abdominal computed
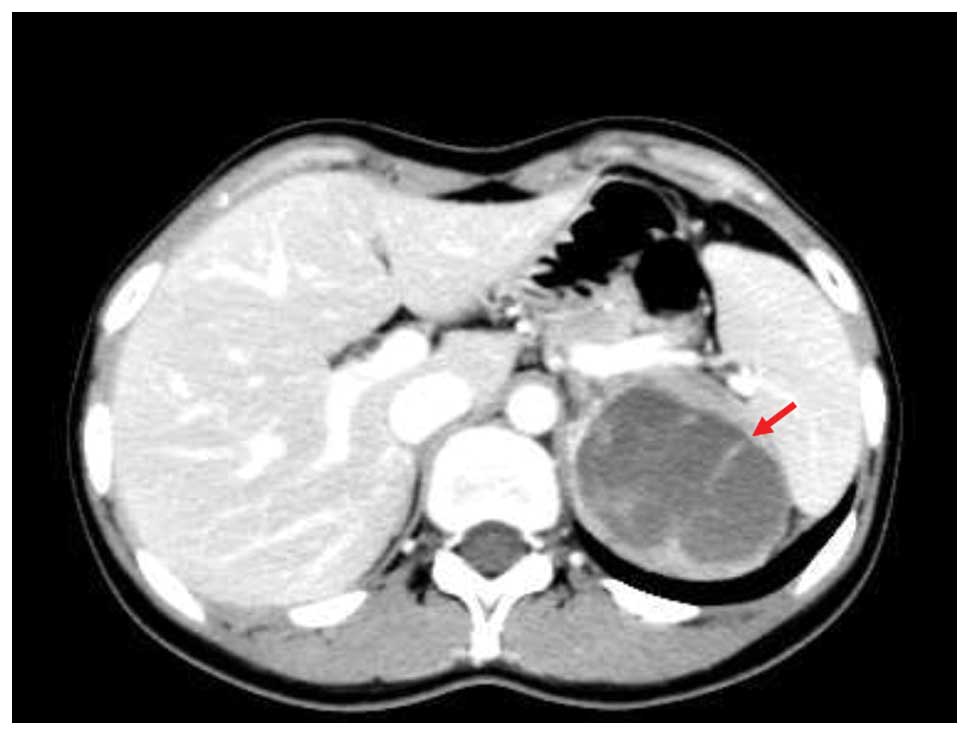
tomography (CT) revealed that the retroperitoneal mass was solid,
encapsulated and ∼6 cm in diameter, with low attenuation and
containing solid and cystic components. The mass was located in the
adrenal region; positioned close to the pancreatic tail, behind the
stomach and between the spleen and the aorta (Fig. 2). Since the diagnosis of an adrenal
tumor was not excludable based on the results of the BUS and CT
examinations, the patient was scheduled for laparoscopic
adrenalectomy. During the surgery, the pancreas and left adrenal
gland were in contact and clearly separated from the tumor. The
main blood supplies originated from branches of the splenic artery.
The tumor was completely removed from the retroperitoneum during
this laparoscopy.
Upon gross examination, the tumor was measured as
6×6×5 cm in size, with a wight of 80 g. The external surface was
smooth, and the cut surfaces showed that the solid tumor was
interspersed with cystic and necrotic spaces. Microscopically, the
tumor was traversed by fibrovascular septa, forming
broad-to-delicate pseudopapillae and hemorrhagic foci. The cores of
certain pseudopapillae had a myxohyaline appearance. The tumor
cells were cuboidal with round nuclei, finely granular chromatin
and an eosinophilic cytoplasm. Mitotic figures were not observed.
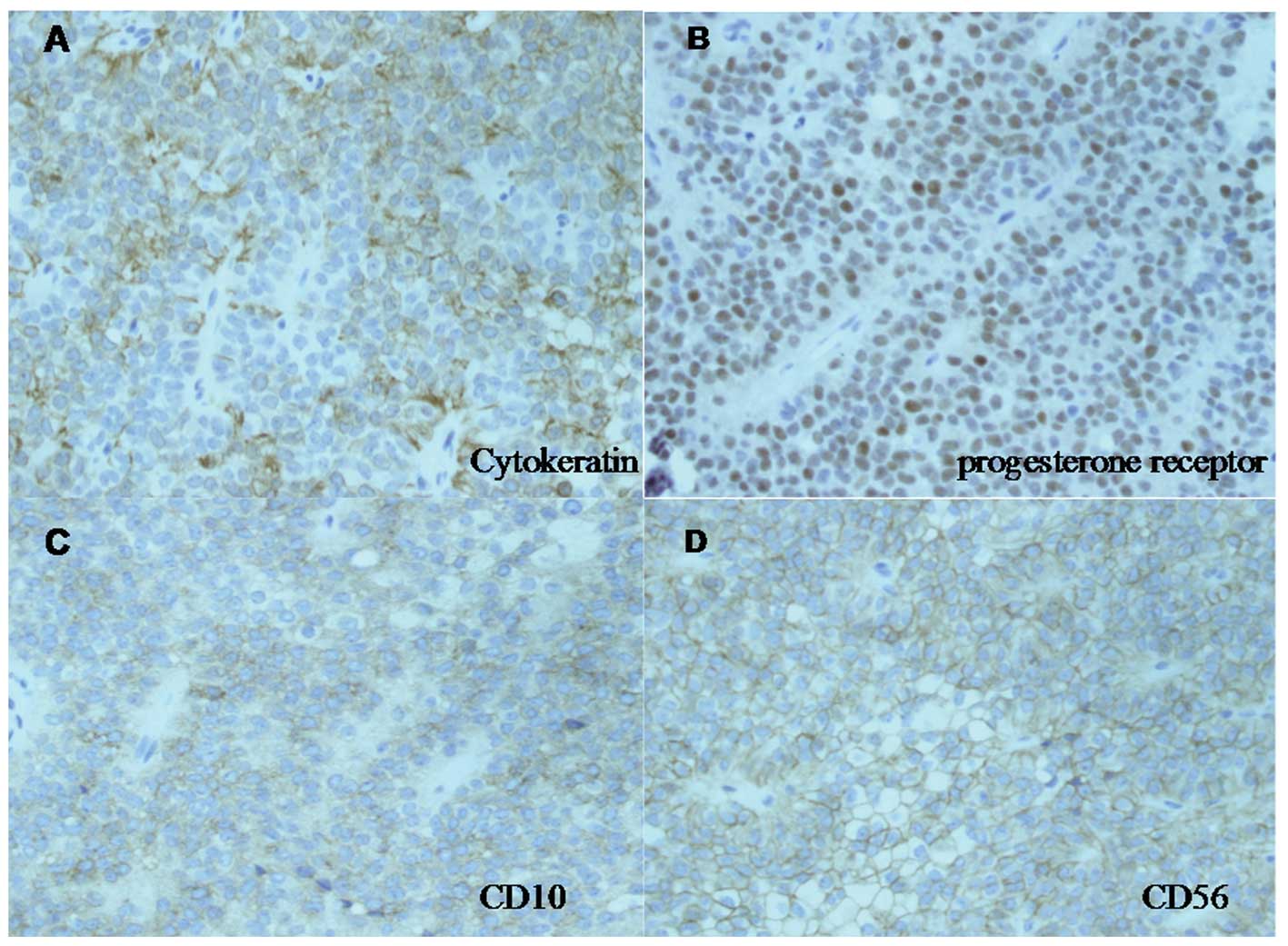
Immunostaining for the progesterone receptor and CD56 was positive.
Cytokeratin and CD10 were focally positive. Staining for CD34,
synaptophysin, α-1-antitrypsin and vimentin was negative (Fig.3). Ectopic pancreatic tissue was also
observed histologically within the resected tumor. Based on these
observations, SPT arising in a pancreatic rest of the
retroperitoneum was diagnosed. At present, 14 months subsequent to
the surgery, the patient is healthy without evidence of recurrence
of SPT.
Discussion
SPT is a relatively rare neoplasm, which is usually
found in the body or tail of the pancreas. It has been described
using various terms, including solid and cystic tumor, papillary
epithelial neoplasm, papillary cystic neoplasm and Frantz’s tumor.
In 2000, the World Health Organization classified this clinical
entity as a ‘solid pseudopapillary tumor’ of the pancreas and
further defined malignant SPT as present when tumor cells show
angio-invasion, perineural invasion or deep invasion into the
surrounding pancreatic parenchyma (8). The tumor may present at all ages and
is observed to occur more often in Asian and African-American
adolescent females (9). The mean
age at diagnosis of the >700 cases reported to date is 28 years
(range, 7–79 years), and 89% of the patients are female (10).
Tumors presenting as SPT but arising from
extrapancreatic sites are extremely rare in the literature. A
review of the English literature published since 1990 (using
PUBMED) revealed only 12 cases of extrapancreatic SPT. These cases,
along with that of the present study, are summarized in Table I. Among these 13 cases, there are 11
female patients and 2 male patients, with a mean age of 32 years
(range, 13–73 years), who presented with large tumors (mean size, 8
cm) similar to the clinical profile of the pancreatic counterpart.
The clinical presentation of extrapancreatic SPT is varied.
Abdominal pain and a palpable mass were the most common symptoms;
however, certain patients were asymptomatic and the tumor was
incidentally detected. Notably, 3 out of the 13 cases involved
tumors that arose from the ectopic pancreas, a well-developed and
normally organised pancreatic tissue lying outside its normal
location with no anatomical or vascular connections to the proper
pancreas. Ectopic pancreases are reported to be detected at a
frequency of 1–2% by autopsy. The most common sites for an ectopic
pancreas are the stomach, 25–38%, the duodenum, 17–36%, and the
jejunum, 15–22% (11). An ectopic
pancreas may also be found in Meckel’s diverticulum, the biliary
tract, the gallbladder, the liver and the spleen. To the best of
our knowledge, the retroperitoneum is an extremely rare location
for an ectopic pancreas and our case was the first report of
extrapancreatic SPT with evidence of a pre-existing ectopic
pancreas in the retroperitoneum.
 | Table IClinical features of reported cases of
extrapancreatic solid pseudopapillary neoplasms. |
Table I
Clinical features of reported cases of
extrapancreatic solid pseudopapillary neoplasms.
| Age (years) | Gender | Location | Size (cm) | Presentation | Ectopic pancreas | Therapy | Recurrence | Follow-up
(months) | Outcome | Reference |
|---|
| 13 | F | Mesocolon | 8 | Abdominal pain | + | Open surgery | - | 36+ | Living/NED | 3 |
| 15 | F | Mesocolon | 21 | Abdominal pain | + | Open surgery | NA | NA | NA | 5 |
| 46 | F | Omentum | 5 | Asymptomatic | - | Laparotomy | - | 3+ | Living/NED | 12 |
| 45 | M | Omentum | 18 | Abdominal mass | - | Laparotomy | Liver and
peritoneum | 98 | Succumbed to primary
disease | 13 |
| 31 | F | Liver | 30 | Abdominal
distention | - | Open surgery | - | 13+ | Living/NED | 14 |
| 17 | F | Left ovary | 25.5 | Abdominal mass | - | Open surgery | - | 72+ | Living/NED | 15 |
| 57 | F | Right ovary | 3 | Asymptomatic | - | Open surgery | NA | NA | NA | 15 |
| 21 | F | Left ovary | 14 | Abdominal
swelling | - | Open surgery | NA | NA | NA | 15 |
| 25 | F | Right ovary | 16.5 | Abdominal
fullness | - | Open surgery | - | 144+ | Living/NED | 16 |
| 73 | M | Duodenum | 14 | Vomiting, fatigue and
weight loss | - | Open surgery | Liver | 3 | Succumbed | 17 |
| 32 | F | Stomach | 10 | Asymptomatic | - | Open surgery and
chemotherapy | Liver, ovary and
peritoneum | 120+ | Living | 17 |
| 22 | F | Retroperitoneum | 8 | Abdominal
discomfort | - | Laparotomy | - | 6+ | Living/NED | 7 |
| 22 | F | Retroperitoneum | 6 | Asymptomatic | + | Laparotomy | - | 14+ | Living/NED | Present case |
The etiology of SPT is not well established. The
immunohistological features of these tumors favor an epithelial
origin, but certain cases of cytokeratin and vimentin coexpression
raise the question of whether these tumors are of a mesenchymal
nature (18). Increasing evidence
supports the hypothesis that SPT may be associated with
hormonally-sensitive tissues from the gonads. The exclusive
occurrence of these tumors in young females, along with the
positive presence of progesterone receptor markers, supports the
theory that hormones have affects on the tumor development
(19). Furthermore, the
identification of extrapancreatic SPT in the ovaries also indicates
a possible genital link, substantiated by the closeness of the
genital ridges to the pancreatic anlage during embryogenesis
(18). Thus, it is currently
proposed that SPT originates from omnipotent cells associated with
the genital ridges; a few of these cells may be entrapped in the
pancreatic anlage during organogenesis, while others follow the
normal migration pathway to the definitive location of the ovaries
(15). According to this theory,
extrapancreatic SPT may occur at any point along the route(s)
followed by the migrating cells, including in the retroperitoneal
space.
The clinical course of SPT is usually favorable as
the tumors are indolent with <5% demonstrating aggressive
behavior (20). Aggressiveness is
generally correlated with cellular atypia, mitotic activity,
prominent necrobiotic nests and invasion of the vascular spaces,
perineural interstitium or neighbouring organs (21,22).
Metastases occur with a low incidence of 14.7% and the majority are
metastases of the liver (10). The
optimal treatment for SPT remains as radical surgical resection,
even in cases of metastatic disease, as the clinical progression
following metastasis is slow and most lesions can be treated by
complete surgical excision of the metastatic tumors (23). In patients with unresectable forms
of the disease, there is limited evidence supporting the use of
chemotherapy (24,25). Additionally, it is difficult to
assess whether the long-term survival occasionally observed is
attributable to the clinical benefits associated with chemotherapy,
rather than the natural course of a poorly aggressive tumor. In the
literature reviewed previously, metastasis was reported in 3 out of
the 13 cases with extrapancreatic tumors; however, the majority of
patients demonstrated long-term survival with no evidence of
disease. The benign outcome is reassuring in that extrapancreatic
STPs are likely to have a favorable clinical course similar to
their pancreatic counterparts.
In conclusion, tumors presenting as SPT but arising
from extrapancreatic sites are extremely rare. Certain SPTs may
arise from the ectopic pancreas. The present study reported a case
of this tumor type with a pancreatic rest that was located in the
retroperitoneum. The etiology was also discussed. STPs are
presently considered to originate from omnipotent cells associated
with the genital ridges. A certain number of these cells may be
entrapped in the pancreatic anlage to form SPTPs, while
extrapancreatic SPTs may occur at any point along the route
followed by the migrating cells during organogenesis, including the
retroperitoneal space. The clinical course of SPT is usually
favorable and extrapancreatic SPTs are likely to have a favorable
clinical course that is similar to their pancreatic
counterparts.
Acknowledgements
The authors would like to thank Dr
Yanmin Zhao for editing the original manuscript.
References
|
1
|
Frantz VK: Tumors of the pancreas. Atlas
of Tumor Pathology. 1st edition. US Armed Forces Institute of
Pathology; Washington DC, USA: pp. 32–33. 1959
|
|
2
|
Papavramidis T and Papavramidis S: Solid
pseudopapillary tumors of the pancreas: review of 718 patients
reported in English literature. J Am Coll Surg. 200:965–972. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ishikawa O, Ishiguro S, Ohhigashi H, et
al: Solid and papillary neoplasm arising from an ectopic pancreas
in the mesocolon. Am J Gastroenterol. 85:597–601. 1990.PubMed/NCBI
|
|
4
|
Elorza Orúe JL, Ruiz Díaz I, Tubia
Landaberea J and San Vicente Leza M: Solid and papillary tumor on
ectopic pancreas in transversal mesocolon. Rev Esp Enferm Dig.
79:429–431. 1991.(In Spanish).
|
|
5
|
Tornóczky T, Kálmán E, Jáksó P, et al:
Solid and papillary epithelial neoplasm arising in heterotopic
pancreatic tissue of the mesocolon. J Clin Pathol. 54:241–245.
2001.PubMed/NCBI
|
|
6
|
Kövári E, Járay B and Pulay I: Papillary
cystic neoplasms in the ectopic pancreas. Orv Hetil. 137:923–925.
1996.(In Hungarian).
|
|
7
|
Miyazaki Y, Miyajima A, Maeda T, et al:
Extrapancreatic solid pseudopapillary tumor: case report and review
of the literature. Int J Clin Oncol. 17:165–168. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Coleman KM, Doherty MC and Bigler SA:
Solid-pseudopapillary tumor of the pancreas. Radiographics.
23:1644–1648. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mortenson MM, Katz MH, Tamm EP, et al:
Current diagnosis and management of unusual pancreatic tumors. Am J
Surg. 196:100–113. 2008. View Article : Google Scholar
|
|
10
|
Mao C, Guvendi M, Domenico DR, Kim K,
Thomford NR and Howard JM: Papillary cystic and solid tumors of the
pancreas: a pancreatic embryonic tumor? Studies of three cases and
cumulative review of the world’s literature. Surgery. 118:821–828.
1995.PubMed/NCBI
|
|
11
|
Christodoulidis G, Zacharoulis D, Barbanis
S, Katsogridakis E and Hatzitheofilou K: Heterotopic pancreas in
the stomach: a case report and literature review. World J
Gastroenterol. 13:6098–6100. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fukunaga M: Pseudopapillary solid cystic
tumor arising from an extrapancreatic site. Arch Pathol Lab Med.
125:1368–1371. 2001.PubMed/NCBI
|
|
13
|
Hibi T, Ojima H, Sakamoto Y, et al: A
solid pseudopapillary tumor arising from the greater omentum
followed by multiple metastases with increasing malignant
potential. J Gastroenterol. 41:276–281. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim YI, Kim ST, Lee GK and Choi BI:
Papillary cystic tumor of the liver. A case report with
ultrastructural observation. Cancer. 65:2740–2746. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Deshpande V, Oliva E and Young RH: Solid
pseudopapillary neoplasm of the ovary: a report of 3 primary
ovarian tumors resembling those of the pancreas. Am J Surg Pathol.
34:1514–1520. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cheuk W, Beavon I, Chui DT and Chan JK:
Extrapancreatic solid pseudopapillary neoplasm: report of a case of
primary ovarian origin and review of the literature. Int J Gynecol
Pathol. 30:539–543. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Walter T, Hommell-Fontaine J, Hervieu V,
et al: Primary malignant solid pseudopapillary tumors of the
gastroduodenal area. Clin Res Hepatol Gastroenterol. 35:227–233.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kosmahl M, Seada LS, Jänig U, Harms D and
Klöppel G: Solid-pseudopapillary tumor of the pancreas: its origin
revisited. Virchows Arch. 436:473–480. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Santini D, Poli F and Lega S:
Solid-papillary tumors of the pancreas: histopathology. JOP.
7:131–136. 2006.PubMed/NCBI
|
|
20
|
Tang LH, Aydin H, Brennan MF and Klimstra
DS: Clinically aggressive solid pseudopapillary tumors of the
pancreas: a report of two cases with components of undifferentiated
carcinoma and a comparative clinicopathologic analysis of 34
conventional cases. Am J Surg Pathol. 29:512–519. 2005. View Article : Google Scholar
|
|
21
|
Canzonieri V, Berretta M, Buonadonna A, et
al: Solid pseudo-papillary tumour of the pancreas. Lancet Oncol.
4:255–256. 2003. View Article : Google Scholar
|
|
22
|
Nishihara K, Nagoshi M, Tsuneyoshi M,
Yamaguchi K and Hayashi I: Papillary cystic tumors of the pancreas.
Assessment of their malignant potential. Cancer. 71:82–92. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Reddy S, Cameron JL, Scudiere J, et al:
Surgical management of solid-pseudopapillary neoplasms of the
pancreas (Franz or Hamoudi tumors): a large single-institutional
series. J Am Coll Surg. 208:950–959. 2009. View Article : Google Scholar
|
|
24
|
Maffuz A, Bustamante Fde T, Silva JA and
Torres-Vargas S: Preoperative gemcitabine for unresectable, solid
pseudopapillary tumour of the pancreas. Lancet Oncol. 6:185–186.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hah JO, Park WK, Lee NH and Choi JH:
Preoperative chemotherapy and intraoperative radiofrequency
ablation for unresectable solid pseudopapillary tumor of the
pancreas. J Pediatr Hematol Oncol. 29:851–853. 2007. View Article : Google Scholar : PubMed/NCBI
|