Introduction
Cervical carcinoma is the second most frequently
diagnosed cancer among females and is also one of the leading
causes of cancer-associated mortality in females in developing
countries (1). The mechanisms of
cervical carcinoma formation remain largely unknown (2). At present, the first-line therapy is
radical surgery with adjuvant chemotherapy. In recent years, along
with increases in rate of diagnosis, improvements to surgical
methods and the application of comprehensive therapy have caused
improvements in the cure rates of cervical carcinoma. However,
cervical carcinoma remains a significant health problem in both
developed and developing countries. Therefore, it is important to
identify a novel non-surgical targeting intervention or drug target
for treating cervical carcinoma (3,4).
MicroRNAs (miRNAs) are a class of non-coding,
single-stranded RNA molecules that are ~19–25 nucleotides in length
and have key roles in the regulation of gene expression in plants
and animals. They function as post-transcriptional gene regulators
by pairing with the 3′-untranslated regions (3′-UTRs) of specific
target messenger RNAs (mRNAs), resulting in their degradation or
the repression of translation (5–8).
miRNAs have been shown to be involved in cell growth,
proliferation, apoptosis and stress responses (9,10). In
addition, previous studies have indicated that numerous miRNAs are
vital in cancer initiation and progression, through the regulation
of their target gene pathways involved in cancer pathogenesis, as
oncogenes or tumor suppressors (11–14).
However, the role of miRNAs in regulating tumor metastasis has only
been recently studied and there remains much to be investigated. It
is important to study the association between target genes and
miRNAs to understand the regulatory mechanism of miRNAs in animal
development, tumor invasion and cell proliferation (6).
Tribbles (TRIBs) are a gene family which control the
specificity of the activation of mitogen-activated protein kinases.
A study demonstrated that rats treated with the TRIB2 gene
contracted acute myeloid leukemia (AML) and the TRIB2 gene appears
to be underexpressed when the growth of leukemia cells is inhibited
(15). However, increases in the
expression of TRIB2 were observed in AML patients (16,17).
It has been demonstrated that TRIB2 induces AML through a series of
mechanisms, including inhibiting C/EBPα (18). However, the biological role of TRIB2
in cervical carcinoma remains unclear.
The phenomenon of downregulation, as well as
upregulation, of miRNAs is most frequently observed in cancer,
suggesting that miRNAs function as tumor suppressor genes or
oncogenes (19). It has been
demonstrated that the miR-99 family regulates stress responses,
apoptosis, proliferation and angiogenesis (20). Generally, miR-99 functions as a
tumor suppressor gene. In the present study, we hypothesized that
miR-99 binds to the 3′-UTR of TRIB2 to negatively regulate TRIB2
expression, and we constructed an miR-99 gene expression vector
(pU6.1/miR-99) to investigate the effects of miR-99 on HeLa cell
proliferation and apoptosis. The results demonstrated that miR-99,
as a tumor suppressor gene, was able to induce HeLa cell
apoptosis.
Materials and methods
Construction of miRNA expression vector
(pU6.1/miR-99)
The miR-99 gene (NW_001794337) was amplified by PCR
from human genomic DNA of human whole blood. Written informed
consent was obtained from the patients. The forward primer was
5′-CATC GGATCCTACTATTGA AACAAAAGCAG-3′, while the reverse primer
was 5′-ATCGAAGCTTCTATTGT TGAACGGCACT-3′. The amplification
conditions were as follows: 5 min initial denaturation at 95°C
followed by 28 cycles of 45-sec denaturation at 95°C, 30-sec
annealing at 56°C and 45-sec elongation at 72°C. The obtained
363-bp fragment of miR-99 was cloned into the T-vector (Takara Bio,
Inc., Otsu, Japan) to construct the T-miR-99 vector. Subsequently,
the fragments were subcloned into pRNAi-U6.1/Neo (Biomics
Biotechnologies, Nantong, China) using BamHI and
HindIII (Takara Bio, Inc.) restriction sites. The identity
of the human miR-99 sequence in the plasmid was confirmed by using
the constructed plasmid as a template for the generation of PCR and
via automated DNA sequencing (Biosune, Shanghai, China; data not
shown).
Cell culture and miRNA-expressing plasmid
transfection
HeLa cells (Shanghai Institute of Cell Biology,
Shanghai, China) were maintained in RPMI 1640 medium (Gibco,
Carlsbad, CA, USA) supplemented with 10% fetal calf serum (Hyclone,
Logan, UT, USA) and 10 U/ml penicillin-streptomycin (Sigma, St.
Louis, MO, USA). pRNAi-U6.1/Neo (Biomics Biotechnologies), using U6
promoter-driven green fluorescent protein (GFP) expression, was
used as an miRNA expression vector. For cotransfection, cells were
treated with 0.5 μg miRNA and 0.5 μg pcDNA-GFP-UTR using
Lipofectamine 2000 (Invitrogen Life Technologies, Carlsbad, CA,
USA) according to the manufacturer’s instructions. The GFP
expression in cells was observed under a fluorescent microscope
(BX43, Olympus, Inc., Japan) at 48 h after transfection. The
percentage of positive cells was detected by flow cytometry (FACS,
Beckman Coulter, Inc., Miami, FL, USA).
Western blotting
The cells were collected at 72 h after transfection.
Cell lysates were prepared in RIPA buffer (Beyotime Institute of
Biotechnology, Haimen, China). The concentrations of the extracted
protein were determined by BCA protein assay (Beyotime Institute of
Biotechnology). Following SDS-PAGE, the samples were transferred
onto nitrocellulose membranes (Millipore, Bedford, MA, USA).
Following incubation with TBS containing 5% non-fat dry milk and
0.1% Tween-20 (TBST) with agitation at room temperature for 2 h,
the membranes were incubated with the primary antibody (anti-TRIB2;
Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) at 4°C
overnight, then washed three times with TBST. The membranes were
incubated with the corresponding secondary antibody (1:3000;
Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd., Beijing,
China) for 1 h at room temperature. Bands were visualized using an
ECL kit (Millipore).
RNA isolation and qPCR
Total RNA was isolated with TRIzol reagent
(Invitrogen Life Technologies), then cDNA was synthesized and
detected via reverse transcription-PCR (RT-PCR) and qPCR. The PCR
products were analyzed by electrophoresis on a 1% agarose gel
containing ethidium bromide. miRNA was isolated with the mirVana
miRNA kit (Ambion, Austin, TX, USA). Following isolation, miRNAs
were polyadenylated using poly(A) polymerase (Ambion). The cDNA was
synthesized with an RT primer, 5′-AACATGTACAGTCCATGGATGd(T)30N(A,
G, C or T)-3′. The forward primer of miR-99 used to amplify the
miRNA was 5′-CCCGTAGATCCGATCTTGTG-3′, while the reverse primer was
5′-AACATGTACAGTCCATGGATG-3′. qPCR was then performed using SuperTaq
Polymerase (Takara Bio, Inc.) according to the manufacturer’s
instructions. The expression of miRNAs was detected with an RG3000
system (Corbett Research, Cambridge, UK) using the Quantitect
SYBR-Green Kit (Qiagen, Hilden, Germany), as follows: initial
denaturation at 95°C for 2 min, followed by 40 cycles of 95°C for
20 sec, 55°C annealing for 15 sec and extension at 72°C for 30 sec.
Fluorescence was observed at 585 nm at each extension step at 72°C.
Human 5S ribosomal RNA (rRNA) served as a control. The forward
primer of 5S rRNA was 5′-GCCATACCACCCTGAACG-3′ and the reverse
primer was 5′-AACATGTACAGTCCATGGATG-3′.
Trypan blue exclusion test of cell
viability
Tryplan blue is a dye that binds to DNA when the
cell membrane is disrupted (dead cells), and it cannot enter into
living cells with intact membranes (21). Briefly, HeLa cells
(4×104/well) were seeded in 24-well tissue culture
plates (Corning Inc., Corning, NY, USA). Following transfection and
stimulation, the cells were trypsinized and the cell pellets were
collected by centrifugation at 1,806 × g for 2 min. Finally, the
cells were resuspended in 50 μl medium per well and stained with 10
μl 0.4% Trypan blue solution for 4 min. Blue cells were counted as
dead cells and the relative death rate was calculated as the number
of dead cells divided by the total number of cells.
Cell proliferation assay with 3-(4,
5-dimethylthiazol-2-yl)-2,5diphenyltetrazolium bromide
Cell proliferation was assayed with colorimetric
MTT. Briefly, HeLa cells were seeded in 96-well plates. Cells were
treated as indicated for 24 h, then 10 μl MTT was added to 100 μl
culture media and cultured for 4 h at 37°C. Subsequently, 100 μl
dimethyl sulfoxide was added to each well to dissolve the formazan
completely. The optical density was detected at 570 nm on a
microplate reader (Bio-Rad, Hercules, CA, USA).
Flow cytometry analysis for cell
apoptosis
At 48 h after transfection, the HeLa cells were
harvested from the plates by trypsinization and collected by
centrifugation at 1,806 × g for 2 min, then washed twice with PBS.
The cells were then resuspended in 1 ml PBS and incubated with 1 ml
propidium iodide at 4°C for 30 min, at a cell density of
1×106. The cells were then analyzed using a flow
cytometer (FACS, Beckman Coulter, Inc.).
Electron microscopy analysis of HeLa
cells
Following transfection for 48 h, the HeLa cells were
trypsinized and centrifuged at 4515 × g for 2 min. The cell pellets
were harvested and fixed with a 37°C solution of 2%
paraformaldehyde, 2.5% glutaraldehyde (Ted Pella, Inc., Redding,
CA, USA) in 0.1 M sodium cacodylate (pH 7.4) and incubated for an
additional 30 min on ice. The cells were then rinsed three times
for 3 min each with 0.1 M sodium cacodylate containing 3 mM calcium
chloride (pH 7.4) on ice, and post-fixed with 1% osmium tetroxide,
0.8% potassium ferrocyanide and 3 mM calcium chloride (pH 7.4) for
1 h. The cultures were stained overnight with 2% uranyl acetate at
4°C and embedded in Durcupan resin (Fluka, St. Louis, MO, USA).
Ultrathin (70-nm) sections were evaluated by transmission electron
microscopy (EM; JEM-100cx; JEOL Ltd, Tokyo, Japan) operated at 80
kV. Images were recorded at a magnification of ×8,000.
Statistics
SAS software (Chicago, IL, USA) was used to analyze
the significance of all results and Student’s t-test was used for
inter-group comparisons. P<0.05 was considered to indicate a
statistically significant difference.
Results
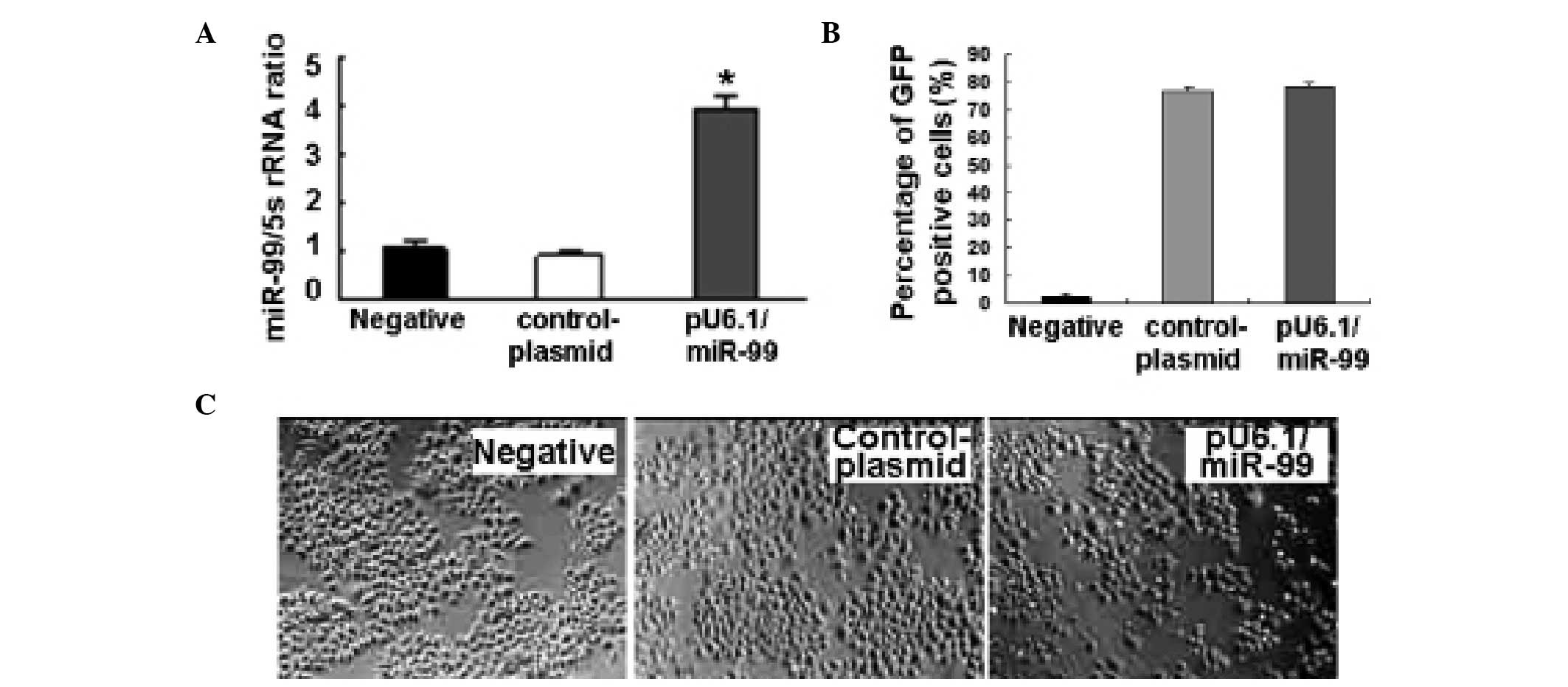
Overexpression of miR-99 after treating
HeLa cells with pU6.1/miR-99
Initially, the quality of the isolated total RNA was
tested by electrophoresis. The bands of 28S, 18S and 5S were
clearly shown and not degraded. miR-99 was observed to be
upregulated after HeLa cells were treated with the pU6.1/miR-99
vector, according to qPCR (Fig.
1A).
Morphological changes following
pU6.1/miR-99 treatment
After HeLa cells were transfected with the
pU6.1/miR-99 vector that expresses GFP to reflect the transfection
rate, the results showed that the transfection rate was >70%
(Fig. 1B) and miR-99 was
overexpressed in pU6.1/miR-99 cultures compared with the controls
(Fig. 1A). The
pU6.1/miR-99-transfected HeLa cells exhibited clear cell volume
reduction, shrinkage and increasing numbers of floating cells,
while the control cultures (pRNAi-U6.1/Neo-NC-treated cells) were
polygonal or spindle shaped and adhered tightly to the well
(Fig. 1C).
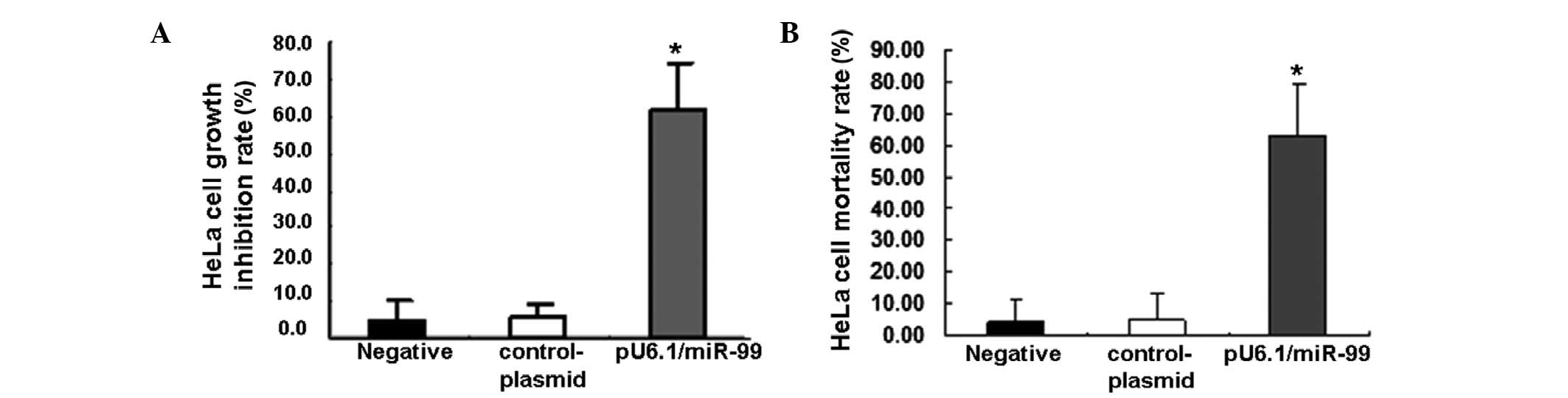
Inhibiting HeLa cell proliferation by
miR-99
The previously mentioned results demonstrated that
the overexpression of miR-99 may suppress HeLa cell growth. To
further investigate, HeLa cell proliferation was detected following
treatment with the pU6.1/miR-99 plasmid using MTT. The results
showed that the overexpression of miR-99 markedly inhibited cell
proliferation in pU6.1/miR-99-treated cells compared with that in
the control cells (Fig. 2A,
P<0.01). A greater number of dead cells were also observed in
pU6.1/miR-99-treated cultures when Trypan blue was used to reflect
the cell mortality rate (Fig. 2B,
P<0.01).
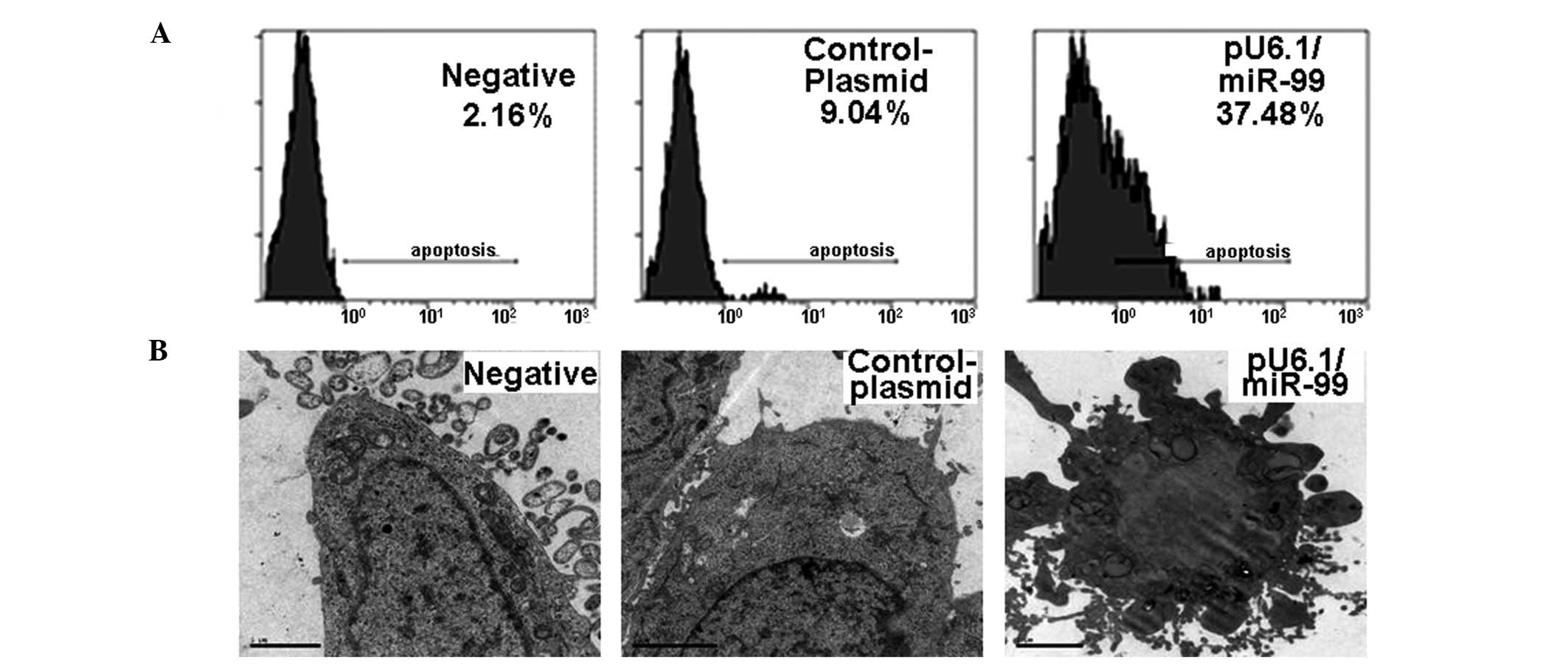
Induction of HeLa cell apoptosis with
miR-99
To further study the effect of miR-99 on HeLa cell
growth, cell apoptosis was analyzed by flow cytometry and electron
microscopy following pU6.1/miR-99 treatment. The HeLa cell
apoptotic rate was 37.48% in pU6.1/miR-99-treated cultures, which
was significantly higher than that in pRNAi-U6.1/Neo-NC control
(9.04%) or negative control cultures (2.16%) (Fig. 3A). Under electron microscopy, the
negative control and pRNAi-U6.1/Neo-NC-treated cells showed rich
microvilli on the cell surface, intact cells and nuclear membranes,
visible bilayers, rich organelles in the cells and visible large
numbers of rough endoplasmic reticulum and ribosomes in the
cytosol. However, the pU6.1/miR-99-transfected cells showed
increases in intracellular electron density and the proportion of
nuclear plasma, as well as patchy nuclear material densification or
plaques, while the nucleoli had almost disappeared (Fig. 3B). Blebbing phenomena and apoptotic
bodies were also observed in pU6.1/miR-99-transfected cells
(Fig. 3B).
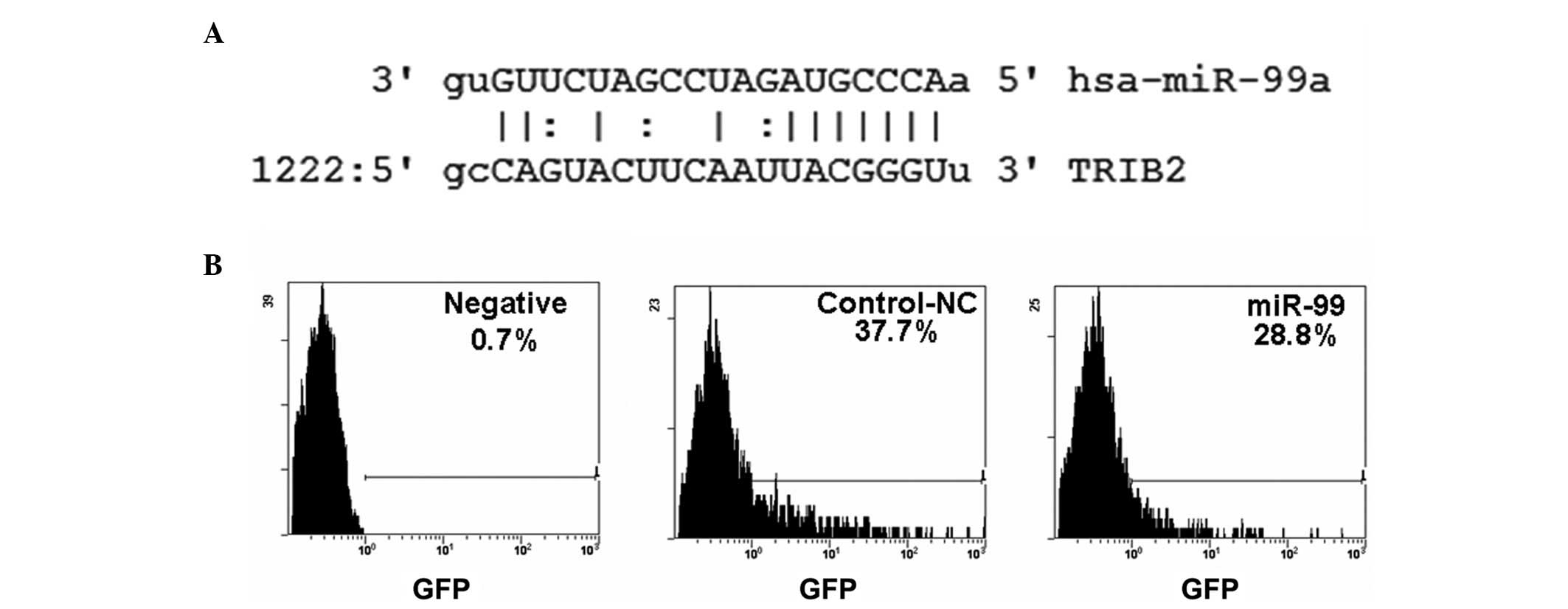
miR-99 suppresses TRIB2 expression in
apoptotic HeLa cells
To investigate the mechanism by which apoptotic HeLa
cells are induced by miR-99, the miR-99 targeting gene was
predicted using microRNA Targetscan software (http://www.targetscan.org/index.html)
and microRNA.org (http://www.microrna.org/microrna/getGeneForm.do)
online. It was observed that TRIB2 was one of the genes targeted by
miR-99 (Fig. 4A). Subsequently, the
pcDNA-GFP-TRIB2-3′-UTR vector [which includes the TRIB2-3′-UTR and
is described in our previous study (22)] was cotransfected with miR-99 into
HeLa cells. The results showed that the number of GFP-positive
cells was noticeably decreased in miR-99-treated cells compared
with control plasmid-treated cultures (Fig. 4B), indicating that the TRIB2-3′-UTR
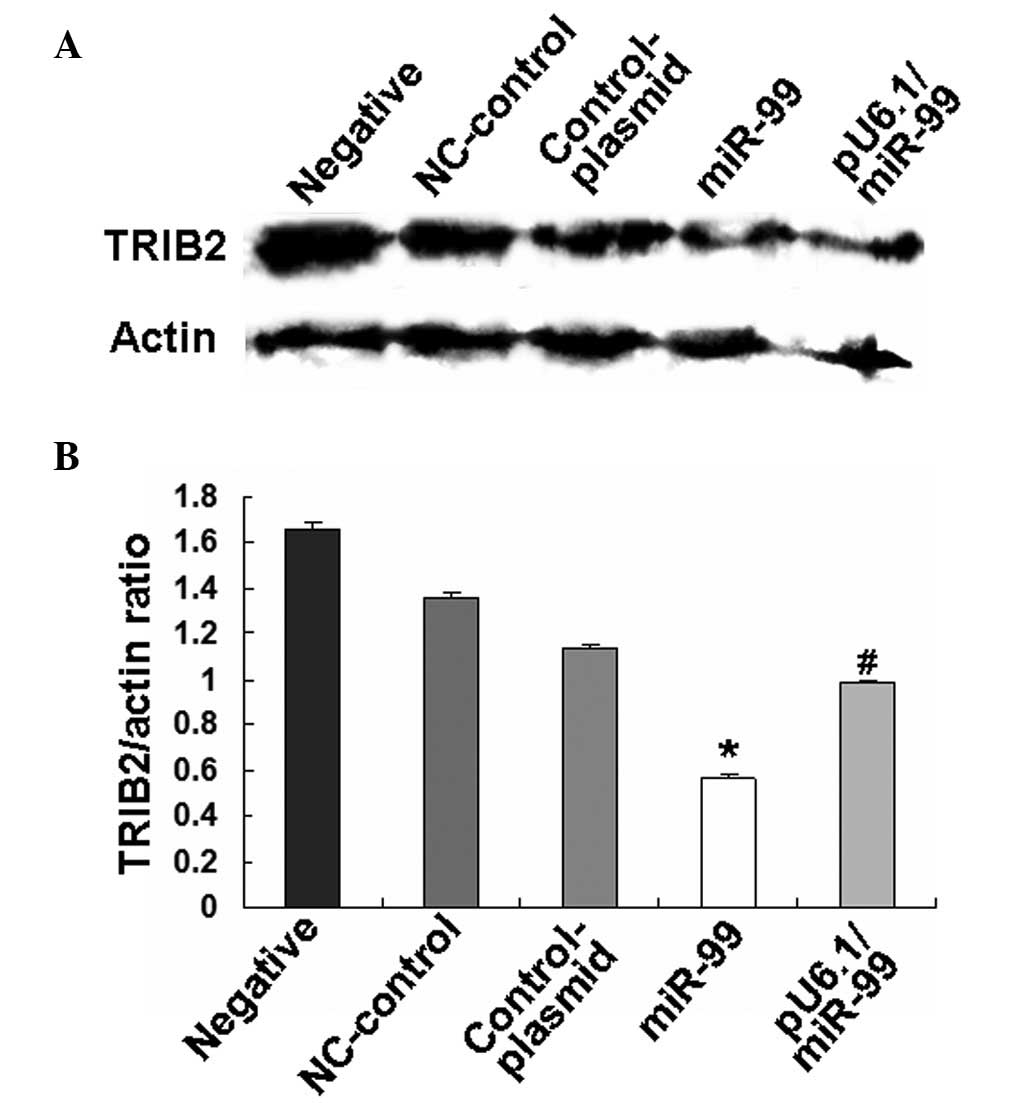
was regulated by miR-99. Western blotting further demonstrated that
the overexpression of miR-99 was able to down-regulate TRIB2
expression in HeLa cells following miR-99 or pU6.1/miR-99 vector
treatment (Fig. 5).
Discussion
miRNAs regulate signaling molecules through
translational repression or gene splicing and modulate at least
one-third of all human gene expression (23). miRNAs have important roles in cell
proliferation and differentiation, and altered miRNA expression may
lead to cancer. The majority of miRNAs are highly conserved and
timing-, tissue- and cell-specific, as well as being subject to
developmental, spatial and temporal regulation. miRNAs are also
involved in cell differentiation, proliferation and apoptosis.
Numerous studies have demonstrated that miRNAs are closely
associated with the presence of a variety of tumors. Their targets
may be tumor suppressor genes or oncogenes involved in
tumorigenesis. In our previous study, >200 abnormal miRNA
expression patterns were identified in DMC-induced apoptotic A549
cells. Further cluster analysis (by Cluster 3.0, CapitalBio
Corporation, Beijing, China) and qPCR showed increases in the
expression of miR-16, miR-34a, miR-34b, miR-34c, miR-17-5p and
miR-125, whereas the expression levels of miR-106, miR-150, let-7c
and miR-99 were decreased.
In the present study, the pU6.1/miR-99 expression
vector, which could express miR-99 effectively in HeLa cells, was
constructed first. Moreover, the overexpression of miR-99 induced
HeLa cell apoptosis and inhibited HeLa cell proliferation following
transfection with the pU6.1/miR-99 expression vector. The
pRNAi-U6.1/Neo plasmid was selected to construct the pU6.1/miR-99
expression vector, due to two main advantages: i) it contains the
neomycin gene, which make it is easy to establish stable cell lines
by G418; and ii) it expresses GFP, which makes it is easy to
observe its transfection rate. Additionally, these proteins do not
affect cell growth and function.
The miR-99 family contains three members, miR-99a,
miR-99b and miR-100 (24), which
regulate cell stress responses, apoptosis, proliferation and
angiogenesis. The expression of the miR-99 family has been observed
to be increased following exposure to a single dose of DNA damage
to induce the DNA repair pathways (25). It has been shown that miR-99a is
able to change the efficiency of DNA repair by regulating SNF2H
(26). Porkka et al reported
that the expression of the miR-99 family was reduced in the
majority of advanced prostate cancer compared with normal prostate
epithelium (27). In the present
study, it was demonstrated that miR-99 acted as a tumor suppressor
gene and was able to induce HeLa cell apoptosis by regulating TRIB2
expression.
The TRIB genes were first identified in Drosophila
(28) and comprise a family of
kinase-like proteins containing a single kinase-like domain.
Mammals have three homologs of TRIB, TRIB1, TRIB2 and TRIB3
(29,30). It has been observed that TRIB2 is
highly expressed in several acute myeloid leukemias lacking C/EBP-α
mutations (16). TRIB2 causes fatal
transplantable AML when introduced in murine hematopoietic stem
cells in vivo(31). In a
previous study, we showed that TRIB2 was an oncogene, which was
more highly expressed in lung adenocarcinoma compared with
paracancerous tissue controls (22). In the present study, using microRNA
Targetscan software, the TRIB2 3′-UTR was observed to be targeted
by miR-99 and the results further demonstrated that miR-99 was able
to negatively regulate TRIB2 expression in HeLa cells.
In conclusion, the present study studied miR-99
expression and its roles in regulating HeLa cell proliferation at
the cellular level, and demonstrated that the overexpression of
miR-99 induced HeLa cell apoptosis. The study identified that
miR-99 has a tumor suppressor function, suggesting a theoretical
basis for its application in cancer therapeutics.
Acknowledgements
This study was supported by the NCET-10-0919 and
National Natural Science Foundation (nos. 30801324, 81141114 and
81200601), Shandong Science and Technology Committee (nos.
ZR2009CQ033 and ZR2012HQ035) and the Foundation of Shandong
Educational Committee of China (nos. J10LC60 and J11LC01).
Abbreviations:
|
miRNAs
|
microRNAs
|
|
mRNA
|
messenger RNAs
|
|
AML
|
acute myeloid leukemia
|
|
RT-PCR
|
reverse transcription-PCR
|
|
MTT
|
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
|
|
DMSO
|
dimethyl sulfoxide
|
|
TRIB
|
Tribbles
|
References
|
1
|
De Bacco F, Luraghi P, Medico E, et al:
Induction of MET by ionizing radiation and its role in
radioresistance and invasive growth of cancer. J Natl Cancer Inst.
103:645–661. 2011.PubMed/NCBI
|
|
2
|
Dursun P, Ayhan A and Kuscu E: New
surgical approaches for the management of cervical carcinoma. Eur J
Surg Oncol. 34:487–496. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar
|
|
4
|
Morice P and Castaigne D: Advances in the
surgical management of invasive cervical cancer. Curr Opin Obstet
Gynecol. 17:5–12. 2005. View Article : Google Scholar
|
|
5
|
Leite KR, Sousa-Canavez JM, Reis ST, et
al: Change in expression of miR-let7c, miR-100, and miR-218 from
high grade localized prostate cancer to metastasis. Urol Oncol.
29:265–269. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Long MJ, Wu FX, Li P, Liu M, Li X and Tang
H: MicroRNA-10a targets CHL1 and promotes cell growth, migration
and invasion in human cervical cancer cells. Cancer Lett.
324:186–196. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xu XM, Wang XB, Chen MM, et al:
MicroRNA-19a and -19b regulate cervical carcinoma cell
proliferation and invasion by targeting CUL5. Cancer Lett.
322:148–158. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Winter J, Jung S, Keller S, Gregory RI and
Diederichs S: Many roads to maturity: microRNA biogenesis pathways
and their regulation. Nat Cell Biol. 11:228–234. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Aigner A: MicroRNAs (miRNAs) in cancer
invasion and metastasis: therapeutic approaches based on
metastasis-related miRNAs. J Mol Med (Berl). 89:445–457. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cho WC: MicroRNAs: potential biomarkers
for cancer diagnosis, prognosis and targets for therapy. Int J
Biochem Cell Biol. 42:1273–1281. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Imam JS, Buddavarapu K, Lee-Chang JS,
Ganapathy S, Camosy C, Chen Y and Rao MK: MicroRNA-185 suppresses
tumor growth and progression by targeting the Six1 oncogene in
human cancers. Oncogene. 29:4971–4979. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Greenberg E, Hershkovitz L, Itzhaki O, et
al: Regulation of cancer aggressive features in melanoma cells by
microRNAs. PLoS One. 6:e189362011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Suzuki HI, Yamagata K, Sugimoto K, Iwamoto
T, Kato S and Miyazono K: Modulation of microRNA processing by p53.
Nature. 460:529–533. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Majid S, Dar AA, Saini S, et al:
MicroRNA-1280 inhibits invasion and metastasis by targeting ROCK1
in bladder cancer. PLoS One. 7:e467432012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Naiki T, Saijou E, Miyaoka Y, et al: TRB2,
a mouse Tribbles ortholog, suppresses adipocyte differentiation by
inhibiting AKT and C/EBPbeta. J Biol Chem. 282:24075–24082. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Keeshan K, Shestova O, Ussin L and Pear
WS: Tribbles homolog 2 (Trib2) and HoxA9 cooperate to accelerate
acute myelogenous leukemia. Blood Cells Mol Dis. 40:119–121. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gilby DC, Sung HY, Winship PR, Goodeve AC,
Reilly JT and Kiss-Toth E: Tribbles-1 and -2 are tumour
suppressors, down-regulated in human acute myeloid leukaemia.
Immunol Lett. 130:115–124. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Grandinetti KB, Stevens TA, Ha S, et al:
Overexpression of TRIB2 in human lung cancers contributes to
tumorigenesis through downregulation of C/EBPα. Oncogene.
30:3328–3335. 2011.PubMed/NCBI
|
|
19
|
Michael MZ, O’ Connor SM, van Holst
Pellekaan NG, Young GP and James RJ: Reduced accumulation of
specific microRNAs in colorectal neoplasia. Mol Cancer Res.
1:882–891. 2003.PubMed/NCBI
|
|
20
|
Chen Z, Jin Y, Yu D, et al:
Down-regulation of the microRNA-99 family members in head and neck
squamous cell carcinoma. Oral Oncol. 48:686–691. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Arunkumar R, Nair SA and Subramoniam A:
Induction of cell-specific apoptosis and protection of mice from
cancer challenge by a steroid positive compound from Zornia
diphylla (L.) Pers. J Pharmacol Pharmacother. 3:233–241. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang C, Chi YL, Wang PY, et al: miR-511
and miR-1297 inhibit human lung adenocarcinoma cell proliferation
by targeting oncogene TRIB2. PLoS One. 7:e460902012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang B, Pan X, Cobb GP and Anderson TA:
microRNAs as oncogenes and tumor suppressors. Dev Biol. 302:1–12.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Houbaviy HB, Murray MF and Sharp PA:
Embryonic stem cell-specific MicroRNAs. Dev Cell. 5:351–358. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sun D, Lee YS, Malhotra A, et al: miR-99
family of MicroRNAs suppresses the expression of prostate-specific
antigen and prostate cancer cell proliferation. Cancer Res.
71:1313–1324. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mueller AC, Sun D and Dutta A: The miR-99
family regulates the DNA damage response through its target SNF2H.
Oncogene. 32:1164–1172. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Porkka KP, Pfeiffer MJ, Waltering KK,
Vessella RL, Tammela TL and Visakorpi T: MicroRNA expression
profiling in prostate cancer. Cancer Res. 67:6130–6135. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Grosshans J and Wieschaus E: A genetic
link between morphogenesis and cell division during formation of
the ventral furrow in Drosophila. Cell. 101:523–531. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hegedus Z, Czibula A and Kiss-Toth E:
Tribbles: a family of kinase-like proteins with potent signalling
regulatory function. Cell Signal. 19:238–250. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hegedus Z, Czibula A and Kiss-Toth E:
Tribbles: novel regulators of cell function; evolutionary aspects.
Cell Mol Life Sci. 63:1632–1641. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Keeshan K, He Y, Wouters BJ, et al:
Tribbles homolog 2 inactivates C/EBPa and causes acute myelogenous
leukemia. Cancer Cell. 10:401–411. 2006. View Article : Google Scholar : PubMed/NCBI
|