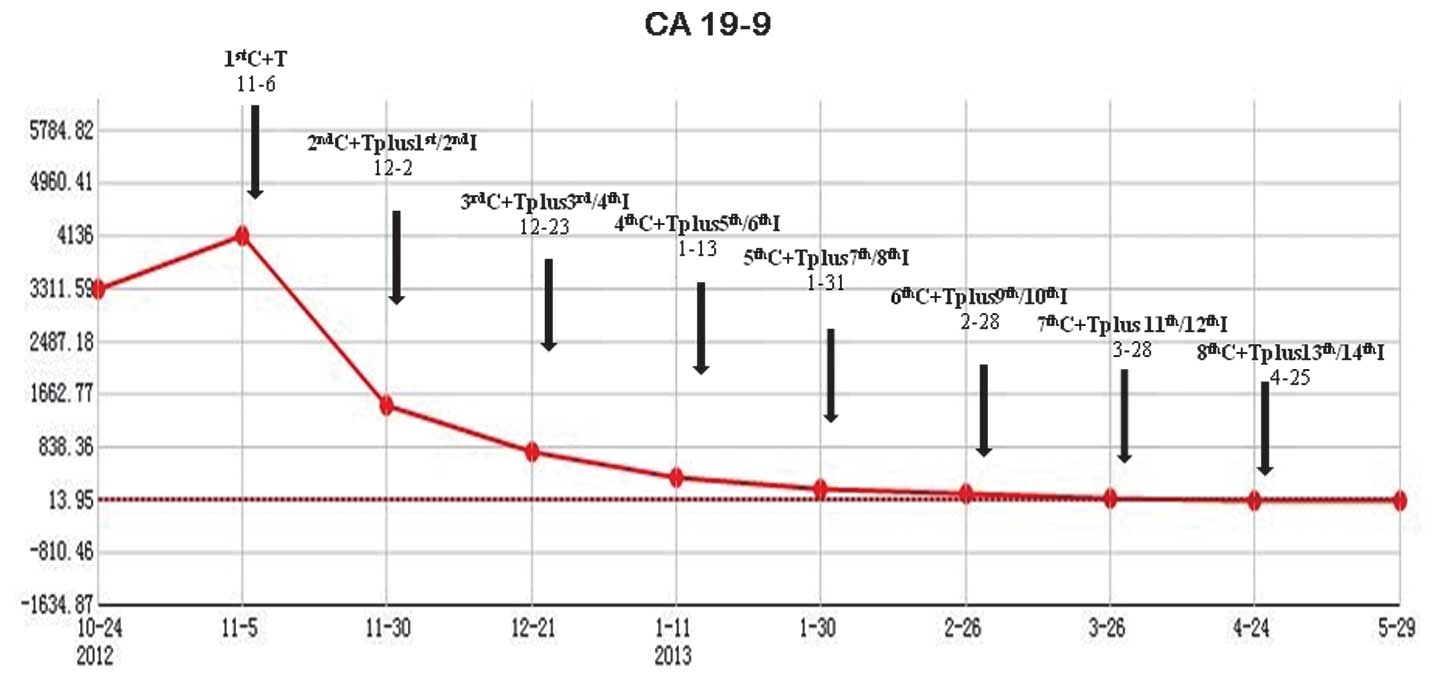
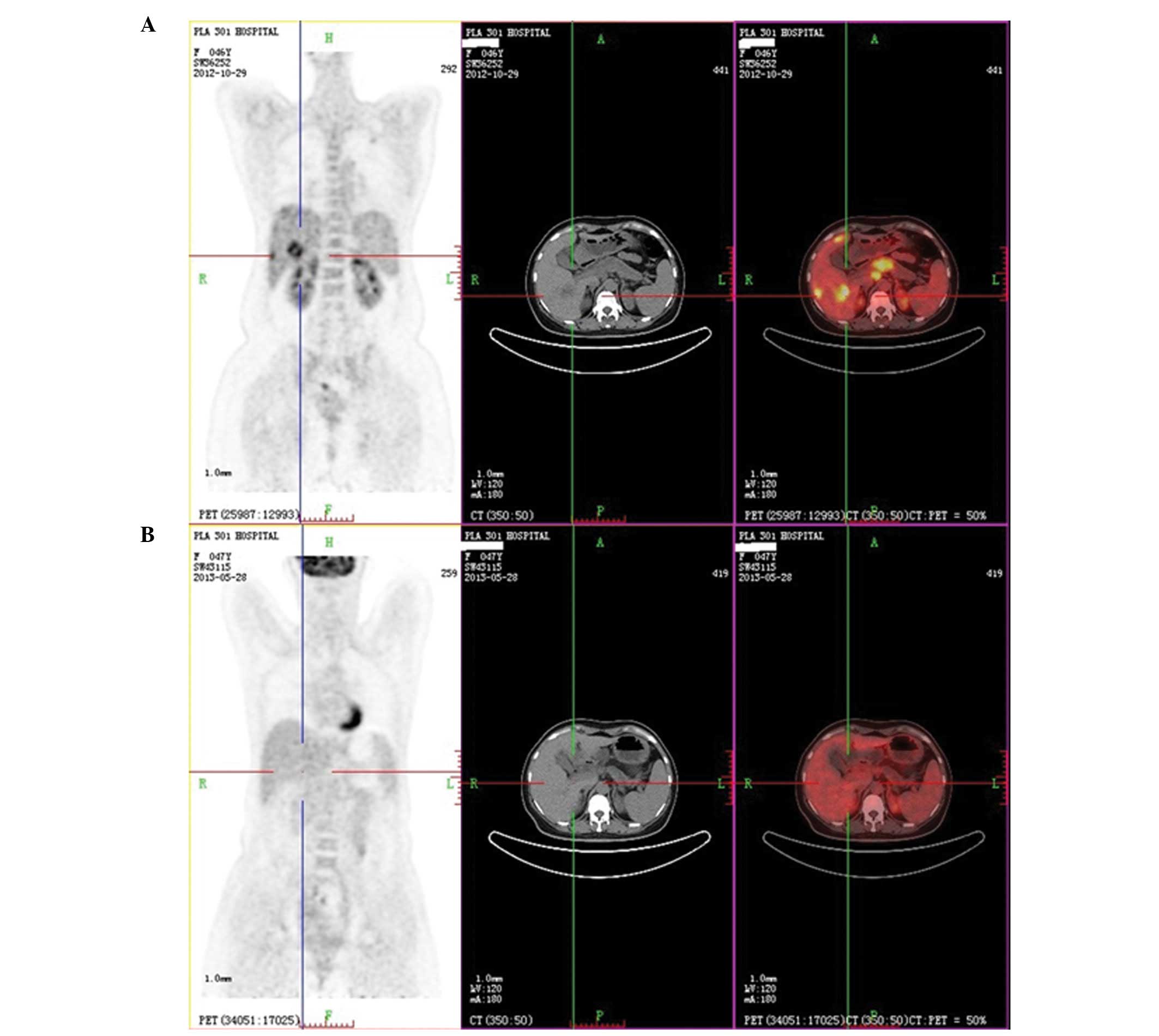
|
1
|
Jemal A, Siegel R, Ward E, et al: Cancer
statistics, 2008. CA Cancer J Clin. 58:71–96. 2008. View Article : Google Scholar
|
|
2
|
Li D, Xie K, Wolff R and Abbruzzese JL:
Pancreatic cancer. Lancet. 363:1049–1057. 2004. View Article : Google Scholar
|
|
3
|
Wong HH and Lemoine NR: Pancreatic cancer:
molecular pathogenesis and new therapeutic targets. Nat Rev
Gastroenterol Hepatol. 6:412–422. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pancreatric Section, British Society of
Gastroenterology; Pancreatic Society of Great Britain and Ireland;
Association of Upper Gastrointestinal Surgeons of Great Britain and
Ireland; Royal College of Pathologists; Special Interest Group for
Gastro-Intestinal Radiology. Guidelines for the management of
patients with pancreatic cancer periampullary and ampullary
carcinomas. Gut. 54(Suppl 5): v1–v16. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Burris HA III, Moore MJ, Andersen J, et
al: Improvements in survival and clinical benefit with gemcitabine
as first-line therapy for patients with advanced pancreas cancer: a
randomized trial. J Clin Oncol. 15:2403–2413. 1997.PubMed/NCBI
|
|
6
|
Hidalgo M: Pancreatic cancer. N Engl J
Med. 362:1605–1617. 2010. View Article : Google Scholar
|
|
7
|
Sultana A, Smith CT, Cunningham D,
Starling N, Neoptolemos JP and Ghaneh P: Meta-analyses of
chemotherapy for locally advanced and metastatic pancreatic cancer.
J Clin Oncol. 25:2607–2615. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Heinemann V, Boeck S, Hinke A, Labianca R
and Louvet C: Meta-analysis of randomized trials: evaluation of
benefit from gemcitabine-based combination chemotherapy applied in
advanced pancreatic cancer. BMC Cancer. 8:822008. View Article : Google Scholar
|
|
9
|
Moore MJ, Goldstein D, Hamm J, et al:
Erlotinib plus gemcitabine compared with gemcitabine alone in
patients with advanced pancreatic cancer: a phase III trial of the
National Cancer Institute of Canada Clinical Trials Group. J Clin
Oncol. 25:1960–1966. 2007. View Article : Google Scholar
|
|
10
|
Strumberg D, Schultheis B, Scheulen ME, et
al: Phase II study of nimotuzumab, a humanized monoclonal
anti-epidermal growth factor receptor (EGFR) antibody, in patients
with locally advanced or metastatic pancreatic cancer. Invest New
Drugs. 30:1138–1143. 2012. View Article : Google Scholar
|
|
11
|
Vonderheide RH, Bajor DL, Winograd R,
Evans RA, Bayne LJ and Beatty GL: CD40 immunotherapy for pancreatic
cancer. Cancer Immunol Immunother. 62:949–954. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sekine T, Takayama T, Konomi Y and Kakizoe
T: Prevention of cancer recurrence by infusion of activated
autologous lymphocytes. Hum Cell. 7:121–124. 1994.(In
Japanese).
|
|
13
|
Dudley ME and Rosenberg SA:
Adoptive-cell-transfer therapy for the treatment of patients with
cancer. Nat Rev Cancer. 3:666–675. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ostrand-Rosenberg S: Looking to the future
of cancer immunotherapy: many questions to answer and many
therapeutic opportunities. Cancer Immunol Immunother. 62:1–2. 2013.
View Article : Google Scholar
|
|
15
|
Baxevanis CN, Perez SA and Papamichail M:
Combinatorial treatments including vaccines, chemotherapy and
monoclonal antibodies for cancer therapy. Cancer Immunol
Immunother. 58:317–324. 2009. View Article : Google Scholar
|
|
16
|
Zhang L, Feng D, Yu LX, Tsung K and Norton
JA: Preexisting antitumor immunity augments the antitumor effects
of chemotherapy. Cancer Immunol Immunother. 62:1061–1071. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ramakrishnan R and Gabrilovich DI: Novel
mechanism of synergistic effects of conventional chemotherapy and
immune therapy of cancer. Cancer Immunol Immunother. 62:405–410.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mitchell MS: Combining chemotherapy with
biological response modifiers in treatment of cancer. J Natl Cancer
Inst. 80:1445–1450. 1988. View Article : Google Scholar
|
|
19
|
Sekine T, Shiraiwa H, Yamazaki T, Tobisu K
and Kakizoe T: A feasible method for expansion of peripheral blood
lymphocytes by culture with immobilized anti-CD3 monoclonal
antibody and interleukin-2 for use in adoptive immunotherapy of
cancer patients. Biomed Pharmacother. 47:73–78. 1993. View Article : Google Scholar
|
|
20
|
Sun Z, Shi L, Zhang H, et al: Immune
modulation and safety profile of adoptive immunotherapy using
expanded autologous activated lymphocytes against advanced cancer.
Clin Immunol. 138:23–32. 2011. View Article : Google Scholar
|
|
21
|
Takayama T, Sekine T, Makuuchi M, et al:
Adoptive immunotherapy to lower postsurgical recurrence rates of
hepatocellular carcinoma: a randomised trial. Lancet. 356:802–807.
2000. View Article : Google Scholar
|
|
22
|
Schmidt-Wolf IG, Negrin RS, Kiem HP, Blume
KG and Weissman IL: Use of a SCID mouse/human lymphoma model to
evaluate cytokine-induced killer cells with potent antitumor cell
activity. J Exp Med. 174:139–149. 1991. View Article : Google Scholar
|
|
23
|
Therasse P, Arbuck SG, Eisenhauer EA, et
al: New guidelines to evaluate the response to treatment in solid
tumors. European Organization for Research and Treatment of Cancer,
National Cancer Institute of the United States, National Cancer
Institute of Canada. J Natl Cancer Inst. 92:205–216. 2000.
View Article : Google Scholar
|
|
24
|
Holmboe L, Andersen AM, Mørkrid L, Slørdal
L and Hall KS: High dose methotrexate chemotherapy:
pharmacokinetics, folate and toxicity in osteosarcoma patients. Br
J Clin Pharmacol. 73:106–114. 2012. View Article : Google Scholar
|