Introduction
Cholangiocarcinoma is a malignancy that arises from
the neoplastic transformation of cholangiocytes, and currently,
this disease accounts for between 10 and 15% of all hepatobiliary
tumors and the incidence is rising globally (1,2). Despite
previous advances in illustrating the molecular characteristics of
cholangiocarcinoma, the treatment remains challenging, and there
has been no significant change in the overall five-year survival
rate of patients over previous decades (3,4).
Therefore, exploration of the molecular events underlying the
initiation and progression of cholangiocarcinoma is urgently
required.
Apoptosis, also termed programmed cell death, plays
an important role in controlling cellular proliferation as part of
normal development (5). Numerous
previous studies have revealed that downregulated apoptotic
pathways contribute to chemoresistance in cancer cells, and
enhanced apoptosis is a key mechanism by which chemotherapeutic
agents exert their antitumor effects (6,7). Thus, the
induction of apoptosis is currently considered to be an index for
selecting novel anticancer drugs (7,8).
Guggulsterone is a polyphenol that is derived from
Commiphora mukul, a plant native to India. For thousands of
years, guggulsterone has been used to treat various human diseases,
including inflammation, arthritis and cardiovascular disease
(9–11). The active components of this herbal
extract are the trans Z- and cis E-isomers of
guggulsterone (12).
Previous studies have reported that guggulsterone
possesses anticancer potential due to the anti-proliferative and
apoptosis-inducing effects exerted by this agent on a panel of
human cancers, including leukemia, breast carcinoma, prostate
cancer, colorectal cancer, pancreatic carcinoma and esophageal
cancer (13–18).
However, to the best of our knowledge, the effect of
guggulsterone on human cholangiocarcinoma remains to be elucidated.
In the present study, the effects of guggulsterone on cellular
proliferation and apoptosis in cholangiocarcinoma Sk-ChA-1 and
Mz-ChA-1 cells were explored, and the underlying mechanism was
investigated.
Materials and methods
Reagents
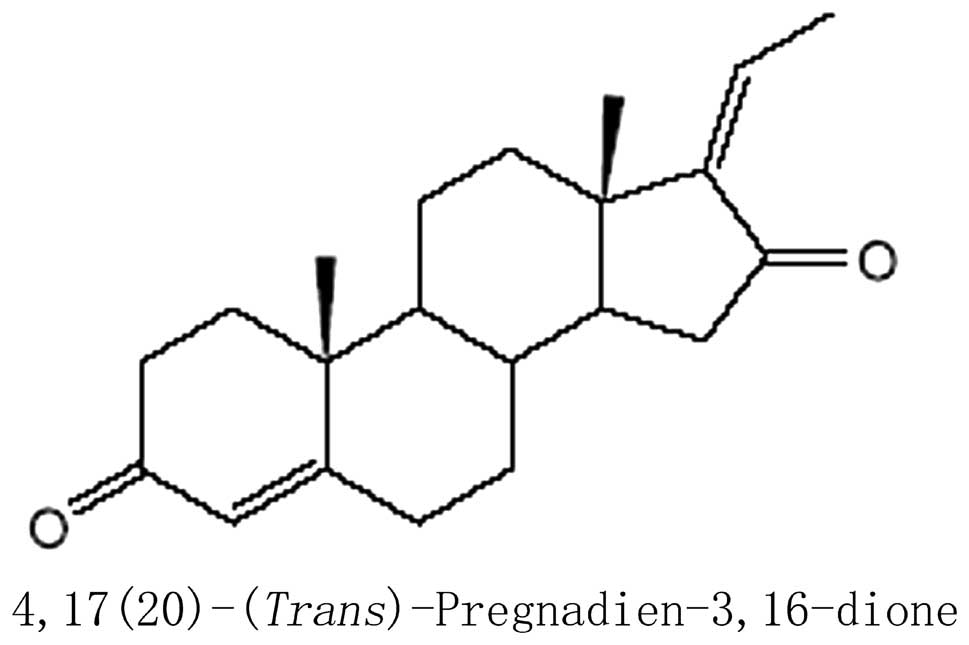
Z-guggulsterone (Fig.1) was purchased from Sigma-Aldrich (St.
Louis, MO, USA). The stock solution was prepared in dimethyl
sulfoxide (DMSO; Solar Biotechnology, Inc., Shanghai, China) and
then diluted with serum-free medium. RPMI-1640 and heat-inactivated
fetal calf serum were obtained from Gibco Industries (Tulsa, OK,
USA). The XTT proliferation kit and Hoechst 33258 were purchased
from Boehringer Ingelheim (Mannheim, Germany). The Caspase Activity
Colorimetric Assay kit, pan-caspase inhibitor z-VAD-fmk, capase-8
inhibitor z-IETD-fmk and caspase-9 inhibitor z-LEHD-fmk were
purchased from Assay Designs (Ann Arbor, MI, USA). The following
primary antibodies were also used: Rabbit anti-cleaved poly
(adenosine diphosphate-ribose) polymerase (PARP; cat no. G3411;
Promega, Madison, WI, USA); mouse anti-human survivin (cat no.
sc-65610; 1:300 dilution; Santa Cruz Biotechnology, Inc., Dallas,
TX, USA); mouse anti-human B-cell lymphoma 2 (Bcl-2; cat no.
sc-377576; 1:500 dilution; Santa Cruz Biotechnology, Inc.); and
goat anti-human Bcl-2-associated X protein (cat no. sc-20287;
1:1,100 dilution; Santa Cruz Biotechnology, Inc.). The rabbit
anti-mouse (cat no. sc-358961; 1:1,000 dilution), bovine
anti-rabbit (cat no. sc-362290; 1:1,000 dilution) and bovine
anti-goat (cat no. sc-362284; 1:1,000 dilution) secondary
antibodies were all obtained from Santa Cruz Biotechnology, Inc.
The present study was approved by the ethics committee of Anhui
Medical University (Hefei, China).
Cell lines culture
The human immortalized cholangiocarcinoma Sk-ChA-1
and Mz-ChA-1 cell lines were purchased from the Cell Bank of Type
Culture Collection of Chinese Academy of Sciences (Shanghai,
China). The cells were routinely cultured in RPMI-1640 supplemented
with 10% heat-inactivated fetal calf serum, 2 mmol/l L-glutamine
and 5 U/ml penicillin (Solar Biotechnology, Inc.)in a humidified
incubator with a 5% CO2 atmosphere at 37°C.
Cell viability assay
The effects of guggulsterone on the viability of
Sk-ChA-1 and Mz-ChA-1 cells were determined by the XTT assay. In
brief, 1×104 cells/well were seeded into a 96-well plate
and incubated for 24 h, and these cells were then treated with 0,
20, 40 and 60 µmol/l Z-guggulsterone. Cells treated with only DMSO
were used as the control. At the indicated time points, 50 µl of
XTT/phenazine methosulfate (PMS) mixture (Solar Biotechnology,
Inc.), consisting of 50 µmol/l PMS and 0.1% XTT in medium, was
added to the cell culture media and incubated continuously for 4
hours. The absorbance at 450 nm was then measured using a Synergy™
HT multi-mode microplate reader (Bio-Tek Instruments, Inc.,
Winooski, VT, USA).
Hoechst 33258 staining
The cholangiocarcinoma cells were seeded into a
six-well plate and routinely cultured overnight in RPMI-1640 medium
containing 10% fetal calf serum. These cells were then treated with
60 µmol/l Z-guggulsterone for 72 h. Subsequently, the cells were
fixed with 4% formaldehyde for 15 min at room temperature. The
cells were then washed twice with 1X phosphate-buffered saline
(PBS) and stained with 10 mg/l Hoechst 33258 for 1 h at room
temperature. Finally, the alterations in nuclear morphology were
observed under fluorescence microscopy (Nikon TE 2000-U; Nikon,
Tokyo, Japan).
DNA fragmentation assay
In total, 1×106 cells were harvested and
washed once in 1X Ca2+- and
Mg2+-free PBS. The cells were then suspended
in 400 µl cell lysis buffer consisting of 10 mM Tris-HCl (pH 7.4;
Solar Biotechnology, Inc.), 10 mM EDTA (pH 8.0) and 0.5% Triton
X-100 (Solar Biotechnology, Inc.). The suspension was incubated at
4°C for 15 min. Following centrifugation of the cell lysates at
15,000 × g for 20 min, the supernatants were collected and
incubated with 40 µg/ml proteinase K and 40 µg/ml RNase A (SBS
Genetech Co., Beijing, China) at 37°C for 3 h. The lysate was
isolated using 0.5 M NaCl and 50% 2-propanol and incubated at −20°C
overnight. Subsequent to centrifugation at 15000 × g for 20 min,
the pellets were suspended in Tris-EDTA buffer consisting of 10 mM
Tris-HCl (pH 7.4) and 1 mM EDTA (pH 8.0). The genomic DNA was
separated using a 2% agarose gel and stained with 0.1 µg/ml
ethidium bromide (19).
Flow cytometry
Flow cytometry analysis was performed as previously
described (18). In brief, the cells
treated with Z-guggulsterone or DMSO were harvested and fixed with
75% ethanol at −20°C. Subsequent to washing twice with cold PBS,
the fixed cells were then suspended in 100 µl RNase A solution (1
mg/ml) and incubated at 37°C for 1 h. Next, 400 µl propidium iodide
solution (50 µg/ml) was added to the cells, and the cells were
subsequently incubated for 30 min in the dark. Following
incubation, the apoptotic status of these cells was measured using
a FACScan flow cytometer (Becton-Dickinson, Franklin Lakes, NJ,
USA). The data were further analyzed using CellQuest Software
(Becton-Dickinson). In the caspase inhibitor assay, the cells were
treated with 60 µmol/l Z-guggulsterone for 72 h, with or without
2-h pre-treatment in 100 µM of the specific caspase inhibitors.
Cellular apoptosis was then measured as aforementioned.
Caspase activity assay
The activity of caspases-3, -8 and -9 was determined
using the colorimetric assay kit, according to the manufacturer's
instructions. In brief, the cells were lysed in buffer containing 5
mM DTT (Solar Biotechnology, Inc.), 10 µg/ml aprotinin (Santa Cruz
Biotechnology, Inc.), 20 mM EDTA (Solar Biotechnology, Inc.), 50 mM
HEPES (SBS Genetech Co.), 0.2% Triton X-100 (Santa Cruz
Biotechnology, Inc.), 150 mM NaCl and 1 mM phenylmethylsulfonyl
fluoride (Santa Cruz Biotechnology, Inc.) at 4°C for 10 min.
Following centrifugation at 10,000 × g for 10 min at 4°C, the
supernatant was collected and the protein concentration was
measured. Subsequently, 100 µg protein was incubated with 0.2 mM
DEVD-pNA, IETD-pNAu or LEHD-pNA (Santa Cruz Biotechnology, Inc.),
which are substrates of caspases-3, -8 and -9, respectively. The
caspase activity was determined from the absorbance at 405 nm using
the microplate reader.
Western blot analysis
Total protein was extracted using the RIPA lysis
buffer containing protease inhibitors. Equal amounts of protein
(20–50 µg) were then loaded and separated by 12% SDS-PAGE and
transferred onto the nitrocellulose membranes. For the blocking
procedure, the membranes were incubated in 5% non-fat milk for 1 h.
Appropriate primary antibodies were added to the membrane and the
membrane was then incubated at room temperature for 2 h. Next, the
membranes were incubated with the appropriate secondary antibodies
for 2 h at room temperature. The specific protein bands were then
developed using a commercial enhanced P1701 chemiluminescence kit
(Applygen Technologies Inc., Beijing, China). The staining density
was measured using the GS-710 Imaging Densitometer, which was
obtained from Bio-Rad Laboratories (Hercules, CA, USA).
Statistical analysis
All experiments were performed in triplicate and
repeated at least three times. The data were expressed as the mean
± standard deviation. Student's t-test was used to determine
the significance of the differences between groups. SPSS software
(version 15.0; SPSS, Inc., Chicago, IL, USA) was used to perform
all statistical analyses and P<0.01 was considered to indicate a
statistically significant difference.
Results
Z-guggulsterone suppressed the
proliferation of cholangiocarcinoma cells by enhancing cellular
apoptosis
To investigate the cytotoxicity of guggulsterone on
cholangiocarcinoma cells, the Sk-ChA-1 and Mz-ChA-1 cells were
incubated with Z-guggulsterone for 24, 48 and 72 h. As demonstrated
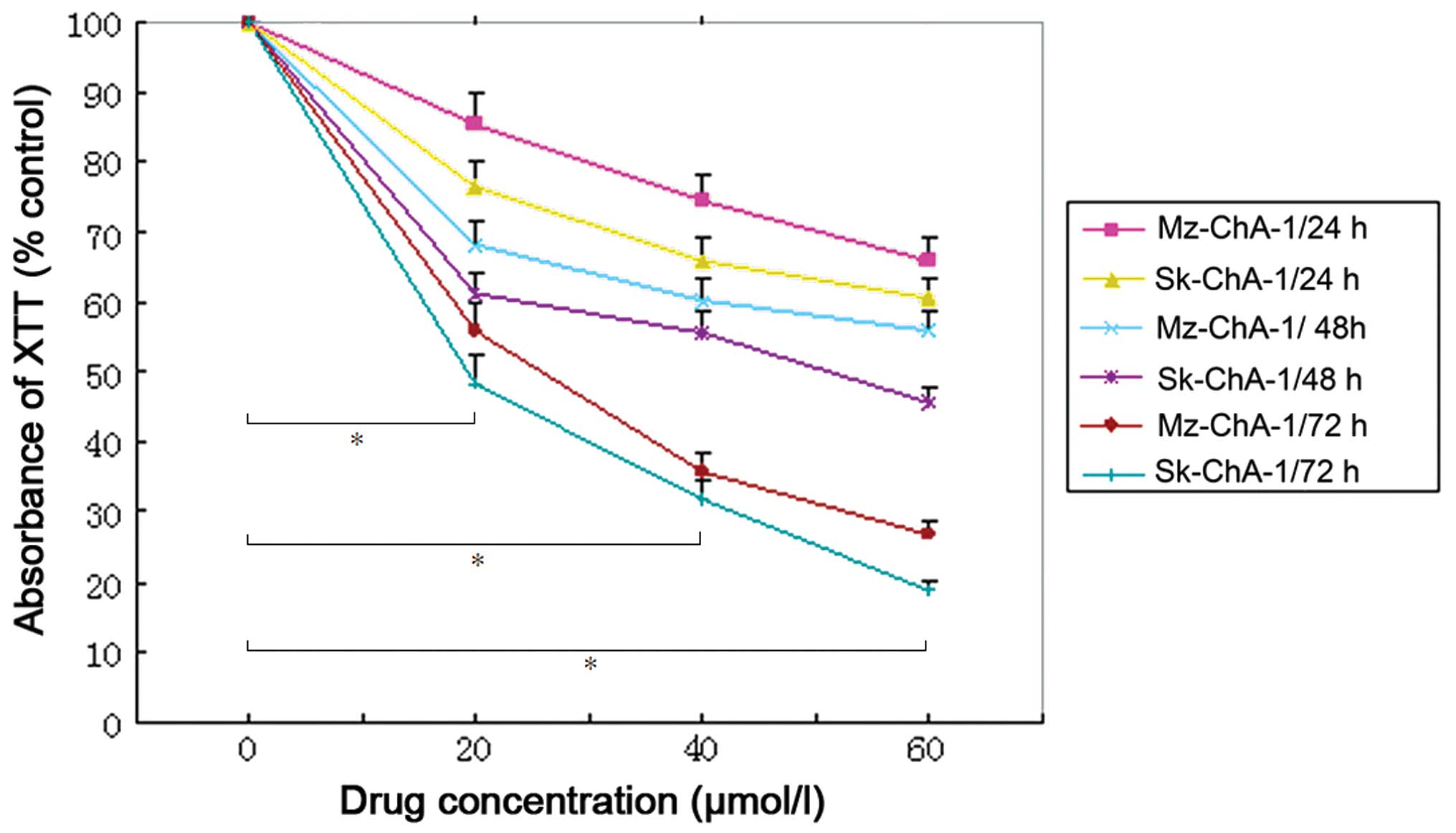
by the present data, treatment with Z-guggulsterone resulted in
significant growth suppression of the two cell lines in a dose- and
time-dependent manner (P<0.001 vs. control; Fig. 2). In addition, the Mz-ChA-1 and
Sk-ChA-1 cells were sensitive to treatment with Z-guggulsterone. In
the group of cells treated with 60 µmol/l Z-guggulsterone for 72 h,
the cell viability of Sk-ChA-1 and Mz-ChA-1 cells was 18.78 and
26.98%, respectively.
Hoechst 33258 staining and DNA fragmentation
analysis were then performed to investigate whether cytotoxicity
induced by guggulsterone resulted from cellular apoptosis.
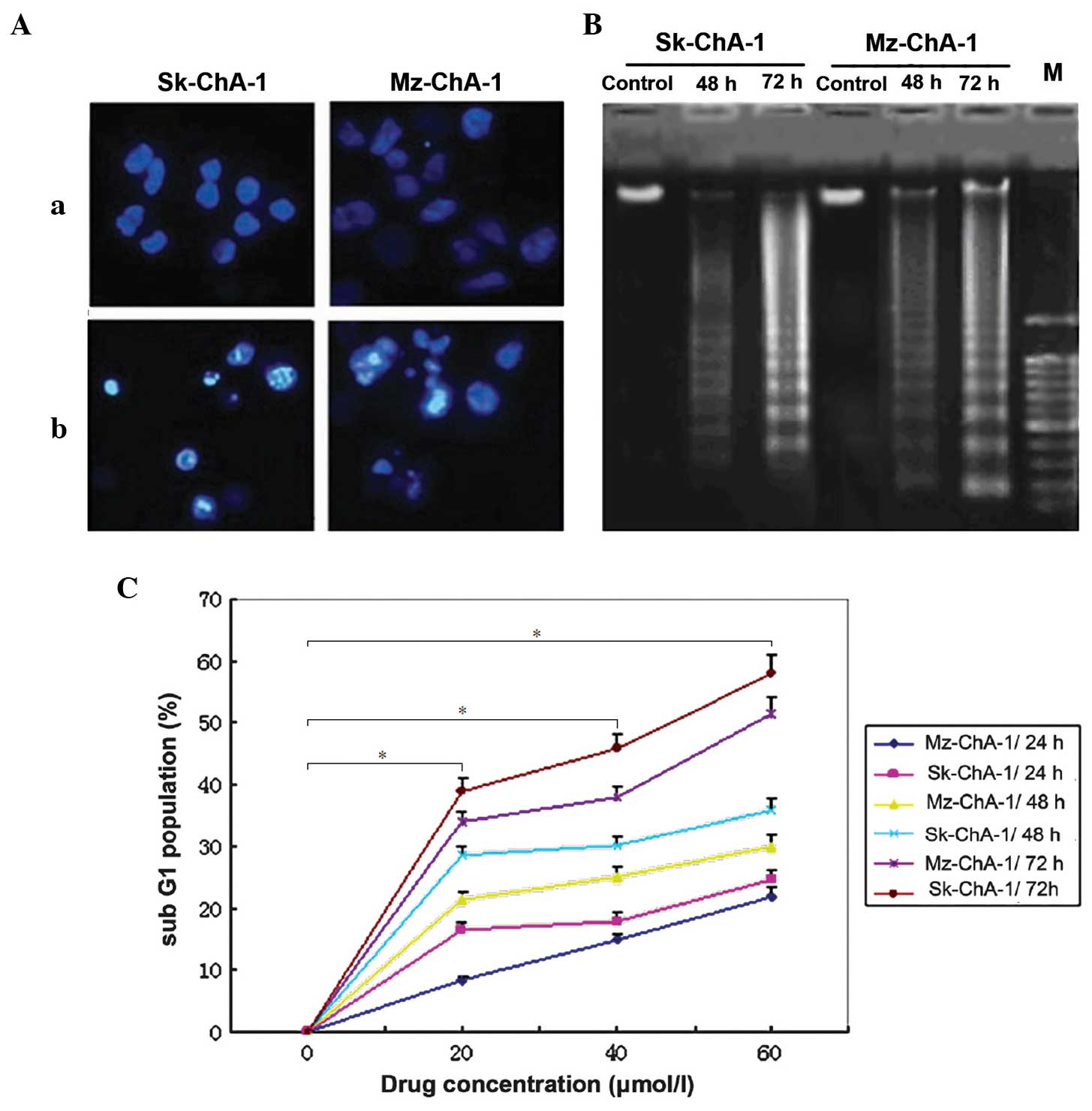
According to the results of Hoechst staining, 72-h treatment with
Z-guggulsterone led to notable morphological alterations, such as
chromatin condensation and nuclear fragmentation, which were
observed in each of the two cholangiocarcinoma cell lines (Fig. 3A). In the agarose gel electrophoresis,
a DNA fragment ladder was detected subsequent to 48-h treatment
with guggulsterone, which was more notable subsequent to 72-h
treatment in each of the two cell lines (Fig. 3B). These findings indicated that that
guggulsterone may lead to the death of cholangiocarcinoma cells by
inducing cellular apoptosis. In addition, flow cytometry was
performed to quantify the apoptosis induced by guggulsterone. As
revealed in Fig. 3C, there was a
significant dose- and time-dependent increase in the proportion of
Sk-ChA-1 and Mz-ChA-1 cells in the sub-G1 phase subsequent to
treatment with ≥20 µmol/l Z-guggulsterone (P<0.001 vs. control).
In addition to the increase of cell growth suppression, cellular
apoptosis was also upregulated gradually and the sub-G1 population
of the two cells was >50% when the cells were treated with 60
µmol/l Z-guggulsterone for 72 h.
Involvement of caspases in apoptosis
induced by Z-guggulsterone
Considering that caspases play critical roles in
cellular apoptosis, the effects of caspase inhibition on
Z-guggulsterone-induced apoptosis were then explored. Three caspase
inhibitors, consisting of pan-caspase, caspase-8 and caspase-9
inhibitors, were used in the present study. As shown in Fig. 4A, these inhibitors significantly
impaired the apoptosis induced by Z-guggulsterone in Sk-ChA-1
(P<0.01) and Mz-ChA-1 (P<0.001) cells, compared with the
cells treated with Z-guggulsterone.
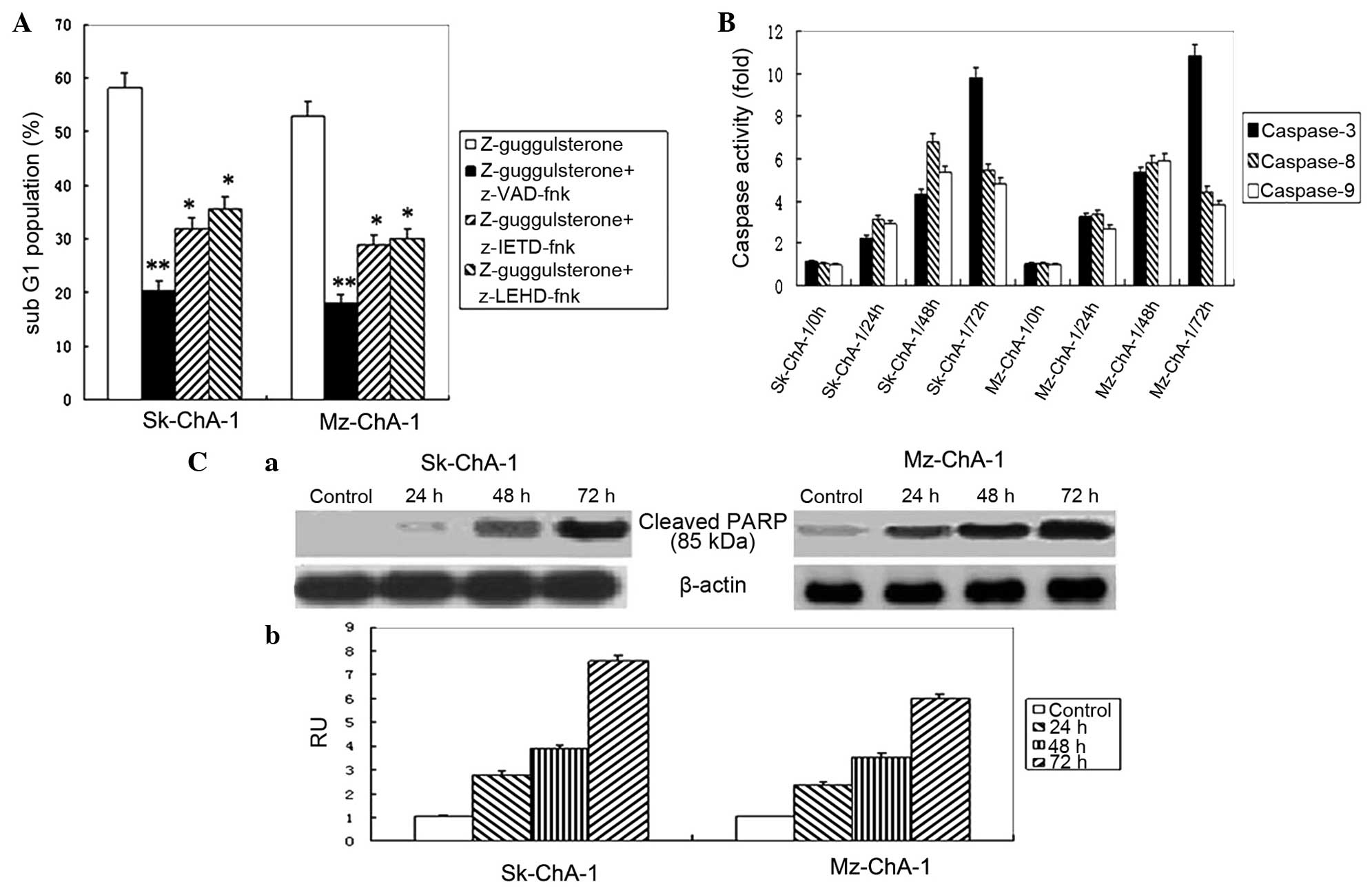 | Figure 4.Caspases involved in
guggulsterone-induced apoptosis. (A) Effect of caspase inhibitors
on guggulsterone-induced apoptosis. The Sk-ChA-1 and Mz-ChA-1 cells
were cultured in the absence or presence of 100 µM z-VAD-fmk,
z-LEHD-fmk or z-IETD-fmk for 2 h prior to the addition of 60 µmol/l
Z-guggulsterone, and the cells were then incubated for 72 h. The
data are expressed as the mean ± SD of three replicate experiments.
*P<0.01, **P<0.001 vs. Z-guggulsterone group. (B) Activity of
caspase-3, -8 and -9 in guggulsterone-treated cholangiocarcinoma
Sk-ChA-1 and Mz-ChA-1 cells. Subsequent to treatment with 60 µmol/l
Z-guggulsterone for the indicated times, the cytosolic fraction of
the cells was analyzed for changes in the activity of caspases-3,
-8 and -9 using a colorimetric assay. The data are expressed as the
mean ± SD of three independent experiments. (C) Cleavage of PARP in
guggulsterone treated cholangiocarcinoma Sk-ChA-1 and Mz-ChA-1
cells. The cells were treated with 60 µmol/l Z-guggulsterone for
the indicated times, and then whole-cell underwent western blotting
to assess the expression of the 85 kDa cleaved PARP. β-actin was
used as internal control to ensure that equal amounts of proteins
were loaded into each lane. (a) The western blotting results are
representative of three replicate experiments. (b) The densities of
the cleaved PARP bands were determined and the relative quantity of
the target protein was reported as RU. SD, standard deviation;
PARP, poly (adenosine diphosphate-ribose) polymerase; RU, relative
units. |
The activity of caspases-3, -8 and -9 following
treatment with 60 µmol/l Z-guggulsterone was then assessed using
colorimetric assay kits. In the Sk-ChA-1 and Mz-ChA-1 cell lines,
the highest activity of caspases-8 and -9 was detected at
subsequent to treatment for 48 h, which was followed by a further
enhancement of caspase-3 activity subsequent to 72 h of treatment
(Fig. 4B). In addition, the level of
cleaved PARP, a hallmark of apoptosis, significantly increased
following treatment with 60 µmol/l Z-guggulsterone in a
time-dependent manner (Fig. 4C).
Overall, the present findings indicated that guggulsterone may
induce cytotoxicity in cholangiocarcinoma cells through
caspase-dependent apoptosis.
Guggulsterone regulates
apoptosis-associated genes
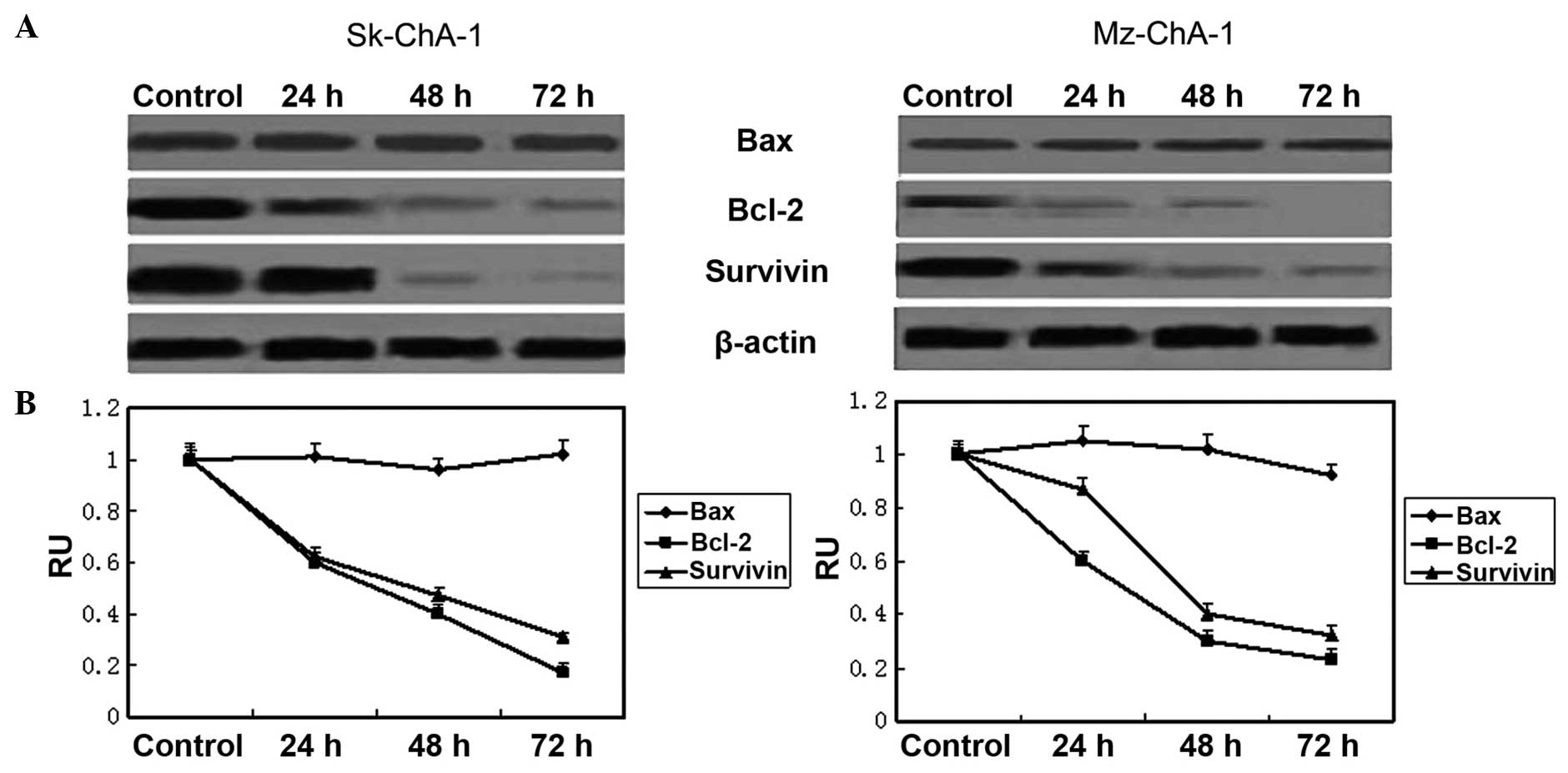
According to the results of western blot analysis,
the protein levels of survivin and Bcl-2 were markedly
downregulated in a time-dependent manner, while the expression of
Bax remained constant (Fig. 5),
indicating the important roles of survivin and Bcl-2 during the
apoptosis induced by guggulsterone.
Discussion
At present, anticancer drugs extracted from plants,
such as Adriamycin (20), paclitaxel
(21) and etoposide (22), are widely used in clinical settings.
The identification of novel anti-cancer agents from natural plants
is becoming a well-known strategy for studies (23).
In the present study, it was demonstrated that
guggulsterone was able to inhibit the proliferation of the
cholangiocarcinoma Sk-ChA-1 and Mz-ChA-1 cell lines, indicating
that guggulsterone may be a good candidate for additional
investigation as a chemotherapeutic agent for
cholangiocarcinoma.
A panel of experiments was then performed to explore
the underlying mechanisms of the effect of guggulsterone. Hoechst
33258 staining revealed that treatment with guggulsterone resulted
in notable morphological indications of apoptosis, including the
condensation of chromatin and nuclear fragmentation in
cholangiocarcinoma Sk-ChA-1 and Mz-ChA-1 cells. In addition,
typical DNA fragmentation, a hallmark of apoptosis, was also
observed subsequent to the cells being treated with 60 µmol/l
Z-guggulsterone for 48 and 72 h. Flow cytometry revealed that
Z-guggulsterone induced apoptosis in cholangiocarcinoma Sk-ChA-1
and Mz-ChA-1 cells in a time- and dose-dependent manner.
Consistently, the present findings were supported by previous
studies that identified similar effects of guggulsterone in other
types of cancer cells (13–16).
As previously reported, there are two major
signaling pathways involved in apoptosis, the extrinsic death
receptor pathway and intrinsic mitochondrial pathway (24–27). In
the extrinsic pathway, caspase-8 is first activated by the
death-inducing signaling complex. The active form of caspase-8 then
activates downstream executioner caspases, including caspase-3, and
also cleaves BH3 interacting domain death agonist (Bid) into
truncated Bid, which eventually leads to the activation of the
intrinsic pathway (28). In the
intrinsic pathway, several apoptogenic molecules, including
cytochrome c and apoptosis-inducing factor, are released
into the cytosol following the mitochondrial membrane permeability
transition (25). Cytochrome c
then interacts with apoptotic protease-activating factor 1 and
pro-caspase-9, and subsequently activates caspase-9 (23,24). The
active caspase-9 activates proteins, including pro-caspase-3 and
cleaved PARP, which initiates a cascade that finally leads to
cellular apoptosis (27). In
addition, there are also several other caspase-independent pathways
(29). For example,
apoptosis-inducing factor may translocate from the mitochondrial
intermembrane to the nucleus and result in chromatin condensation
and large-scale DNA fragmentation, leading to apoptotic cell death
(30).
In addition, pre-treatment with the caspase
inhibitors z-VAD-fmk, z-LEHD-fmk and z-IETD-fmk effectively
impaired guggulsterone-induced apoptosis, indicating that caspase
cascades play critical roles during the induction of apoptosis
caused by guggulsterone. The colorimetric assay revealed that
guggulsterone activated caspases-8, -9 and -3. As the main
substrate of caspases, the alteration in the expression of cleaved
PARP also supports this conclusion. Consistent with the present
results, several other studies have reported that caspase-dependent
apoptosis induced by guggulsterone also occurred in prostate and
colon cancer cells (13,16). Additional studies investigating the
possibility that guggulsterone triggers apoptosis through
caspase-independent pathways are required in the future, as the
caspase inhibitors used in the present study did not completely
abolish guggulsterone-induced apoptosis.
Survivin is a member of the inhibitor of apoptosis
protein family and is usually overexpressed in human cancers
(31). It has been found that cancer
cells demonstrating overexpression of survivin are resistant to
numerous apoptotic stimuli (32). By
contrast, the downregulation of survivin causes spontaneous
apoptosis or sensitizes cancer cells to these apoptotic stimuli
(32–34). It has previously been reported that
survivin is overexpressed in the majority of cholangiocarcinoma
cells, which was closely associated with poor outcomes, indicating
that survivin may be a promising target for the treatment of
cholangiocarcinoma (35–37).
The Bcl-2 family, which consists of ~20 homologues
of apoptosis-inhibiting and apoptosis-promoting Bcl-2 proteins,
plays key regulatory roles in mitochondria-mediated apoptosis
(38). Bcl-2 and Bax are major
apoptosis-inhibiting and -promoting proteins, respectively
(39). It has previously been
revealed that invalid therapeutic outcomes are mostly associated
with the overexpression of apoptosis-inhibiting proteins,
particularly Bcl-2, in numerous types of human cancer (40). By contrast, Bax has been revealed to
constitute a requisite gateway to the mitochondria-dependent
pathway of apoptosis (41).
Therefore, upregulating the level of Bax may also improve the
sensitivities to anti-cancer agents in these cancer cells (42). In addition, Bcl-2 and Bax may dimerize
with each other and mutually neutralize their biological functions,
resulting in the initiation of apoptosis (39). Thus, the Bcl-2/Bax ratio is an
important index for the determination of cellular susceptibility to
apoptosis (43).
The present study revealed that
guggulsterone-induced apoptosis is accompanied by a reduction in
the expression of survivin and Bcl-2. Despite survivin and Bcl-2
each acting as apoptosis inhibitors, these proteins execute
biological functions through various pathways that regulate
cellular apoptosis (44). Survivin
may directly interact with and suppress the terminal effector
cell-death proteases, such as caspase-3 and caspase-7 (40), while Bcl-2 mainly prevents the release
of cytochrome c and then blocks the progression of apoptosis
(34). The present study identified
that survivin and Bcl-2 were concurrently downregulated by
guggulsterone, which was consistent with the findings of previous
studies (41,42).
In summary, the present study demonstrated that
guggulsterone suppressed tumor proliferation by inducing cellular
apoptosis in human cholangiocarcinoma cells. The current study also
revealed that the apoptosis induced by guggulsterone was
caspase-dependent and accompanied by the downregulation of survivin
and Bcl-2. These findings indicate the therapeutic potential of
guggulsterone in treating cholangiocarcinoma.
Acknowledgements
This study was supported by the Scientific Research
Foundation of Anhui Medical University (grant no., 2012xkj049),
Youth Talents Plan of the First Affiliated Hospital of Anhui
Medical University (grant no., 2010KJ01) and Natural Science
Foundation of Higher Education of Anhui Province (grant no.,
KJ2012A163).
References
|
1
|
Lazaridis KN and Gores GJ:
Cholangiocarcinoma. Gastroenterology. 128:1655–1667. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
West J, Wood H, Logan RF, Quinn M and
Aithal GP: Trends in the incidence of primary liver and biliary
tract cancers in England and Wales 1971–2001. Br J Cancer.
94:1751–1758. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Anderson CD, Pinson CW, Berlin J and Chari
RS: Diagnosis and treatment of cholangiocarcinoma. Oncologist.
9:43–57. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sirica AE: Cholangiocarcinoma: Molecular
targeting strategies for chemoprevention and therapy. Hepatology.
41:5–15. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fulda S, Gorman AM, Hori O and Samali A:
Cellular stress responses: Cell survival and cell death. Int J Cell
Biol. 2010:2140742010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fabregat I, Roncero C and Fernández M:
Survival and apoptosis: A dysregulated balance in liver cancer.
Liver Int. 27:155–162. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ferreira CG, Epping M, Kruyt FA and
Giaccone G: Apoptosis: Target of cancer therapy. Clin Cancer Res.
8:2024–2034. 2002.PubMed/NCBI
|
|
8
|
Kim ND, Im E, Yoo YH and Choi YH:
Modulation of the cell cycle and induction of apoptosis in human
cancer cells by synthetic bile acids. Curr Cancer Drug Targets.
6:681–689. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gujral ML, Sareen K, Tangri KK, Amma MK
and Roy AK: Antiarthritic and anti-inflammatory activity of gum
guggul (Balsamodendron mukul Hook). Indian J Physiol
Pharmacol. 4:267–273. 1960.PubMed/NCBI
|
|
10
|
Sharma JN and Sharma JN: Comparison of the
anti-inflammatory activity of Commiphora mukul (an indigenous
drug) with those of phenylbutazone and ibuprofen in experimental
arthritis induced by mycobacterial adjuvant. Arzneimittelforschung.
27:1455–1457. 1977.PubMed/NCBI
|
|
11
|
Urizar NL and Moore DD: GUGULIPID: A
natural cholesterol-lowering agent. Annu Rev Nutr. 23:303–313.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Urizar NL, Liverman AB, Dodds DT, Silva
FV, Ordentlich P, Yan Y, Gonzalez FJ, Heyman RA, Mangelsdorf DJ and
Moore DD: A natural product that lowers cholesterol as an
antagonist ligand for FXR. Science. 296:1703–1706. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Singh SV, Choi S, Zeng Y, Hahm ER and Xiao
D: Guggulsterone-induced apoptosis in human prostate cancer cells
is caused by reactive oxygen intermediate dependent activation of
c-Jun NH2 -terminal kinase. Cancer Res. 67:7439–7449.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shishodia S, Sethi G, Ahn KS and Aggarwal
BB: Guggulsterone inhibits tumor cell proliferation, induces
S-phase arrest, and promotes apoptosis through activation of c-Jun
N-terminal kinase, suppression of Akt pathway, and downregulation
of antiapoptotic gene products. Biochem Pharmacol. 74:118–130.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Samudio I, Konopleva M, Safe S, McQueen T
and Andreeff M: Guggulsterones induce apoptosis and differentiation
in acute myeloid leukemia: Identification of isomer-specific
antileukemic activities of the pregnadienedione structure. Mol
Cancer Ther. 4:1982–1992. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
An MJ, Cheon JH, Kim SW, Kim ES, Kim TI
and Kim WH: Guggulsterone induces apoptosis in colon cancer cells
and inhibits tumor growth in murine colorectal cancer xenografts.
Cancer Letters. 279:93–100. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Macha MA, Rachagani S, Gupta S, Pai P,
Ponnusamy MP, Batra SK and Jain M: Guggulsterone decreases
proliferation and metastatic behavior of pancreatic cancer cells by
modulating JAK/STAT and Src/FAK signaling. Cancer Lett.
341:166–177. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Guan B, Li H, Yang Z, Hoque A and Xu X:
Inhibition of farnesoid X receptor controls esophageal cancer cell
growth in vitro and in nude mouse xenografts. Cancer.
119:1321–1329. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Herrmann M, Lorenz HM, Voll R, Grünke M,
Woith W and Kalden JR: A rapid and simple method for the isolation
of apoptotic DNA fragments. Nucleic Acids Res. 22:5506–5507. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kanjeekal S, Chambers A, Fung MF and Verma
S: Systemic therapy for advanced uterine sarcoma: A systematic
review of the literature. Gynecol Oncol. 97:624–637. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wiseman LR and Spencer CM: Paclitaxel. An
update of its use in the treatment of metastatic breast cancer and
ovarian and other gynaecological cancers. Drugs Aging. 12:305–334.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jiang J, Liang X, Zhou X, Huang L, Huang
R, Chu Z and Zhan Q: A meta-analysis of randomized controlled
trials comparing irinotecan/platinum with etoposide/platinum in
patients with previously untreated extensive-stage small cell lung
cancer. J Thorac Oncol. 5:867–873. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Karikas GA: Anticancer and chemopreventing
natural products: Some biochemical and therapeutic aspects. J BUON.
15:627–638. 2010.PubMed/NCBI
|
|
24
|
Reed JC: Apoptosis-regulating proteins as
targets for drug discovery. Trends Mol Med. 7:314–319. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Desagher S and Martinou JC: Mitochondria
as the central control point of apoptosis. Trends Cell Biol.
10:369–377. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Van Loo G, Saelens X, Van Gurp M,
MacFarlane M, Martin SJ and Vandenabeele P: The role of
mitochondrial factors in apoptosis: A Russian roulette with more
than one bullet. Cell Death Differ. 9:1031–1042. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Thornberry NA and Lazebnik Y: Caspases:
Enemies within. Science. 281:1312–1316. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fulda S: Targeting extrinsic apoptosis in
cancer: Challenges and opportunities. Semin Cell Dev Biol.
39:20–25. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liang Q, Li W and Zhou B:
Caspase-independent apoptosis in yeast. Biochim Biophys Acta.
1783:1311–1319. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Susin SA, Lorenzo HK, Zamzami N, Marzo I,
Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler
M, et al: Molecular characterization of mitochondrial
apoptosis-inducing factor. Nature. 397:441–446. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li F: Survivin study: What is the next
wave? J Cell Physiol. 197:8–29. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li F, Ambrosini G, Chu EY, Plescia J,
Tognin S, Marchisio PC and Altieri DC: Control of apoptosis and
mitotic spindle checkpoint by survivin. Nature. 396:580–584. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mirza A, McGuirk M, Hockenberry TN, Wu Q,
Ashar H, Black S, Wen SF, Wang L, Kirschmeier P, Bishop WR, et al:
Human survivin is negatively regulated by wild-type p53 and
participates in p53-dependent apoptotic pathway. Oncogene.
21:2613–2622. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Beltrami E, Plescia J, Wilkinson JC,
Duckett CS and Altieri DC: Acute ablation of survivin uncovers
p53-dependent mitotic checkpoint functions and control of
mitochondrial apoptosis. J Biol Chem. 279:2077–2084. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chang Q, Liu ZR, Wang DY, Kumar M, Chen YB
and Qin RY: Survivin expression induced by doxorubicin in
cholangiocarcinoma. World J Gastroenterol. 10:415–418.
2004.PubMed/NCBI
|
|
36
|
Javle MM, Tan D, Yu J, LeVea CM, Li F,
Kuvshinoff BW and Gibbs JF: Nuclear survivin expression predicts
poor outcome in cholangiocarcinoma. Hepatogastroenterol.
51:1653–1657. 2004.
|
|
37
|
Obama K, Ura K, Li M, Katagiri T, Tsunoda
T, Nomura A, Satoh S, Nakamura Y and Furukawa Y: Genome-wide
analysis of gene expression in human intrahepatic
cholangiocarcinoma. Hepatology. 41:1339–1348. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Antonsson B: Mitochondria and the Bcl-2
family proteins in apoptosis signaling pathways. Mol Cell Biochem.
256–257:141–155. 2004. View Article : Google Scholar
|
|
39
|
Nascimento Pde S, Ornellas AA, Campos MM,
Scheiner MA, Fiedler W and Alves G: Bax and bcl-2 imbalance and HPB
infection in penile tumors and adjacent tissues. Prog Urol.
14:353–359. 2004.(In French). PubMed/NCBI
|
|
40
|
Reed JC: Double identity for proteins of
the Bcl-2 family. Nature. 387:773–776. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wei MC, Zong WX, Cheng EH, Lindsten T,
Panoutsakopoulou V, Ross AJ, MacGregor GR, Thompson CB and
Korsmeyer SJ: Proapoptotic BAX and BAK: A requisite gateway to
mitochondrial dysfunction and death. Science. 292:727–730. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang CL, Wu LJ, Tashiro S, Onodera S and
Ikejima T: Oridonin induces apoptosis of HeLa cells via altering
expression of Bcl-2/Bax and activating caspase-3/ICAD pathway. Acta
Pharmacol Sin. 25:691–698. 2004.PubMed/NCBI
|
|
43
|
Manion MK and Hockenbery DM: Targeting
BCL-2-related proteins in cancer therapy. Cancer Biol Ther. 2((Sul
1)): S105–S114. 2003.PubMed/NCBI
|
|
44
|
Kanwar JR, Kamalapuram SK and Kanwar RK:
Targeting survivin in cancer: The cell-signalling perspective. Drug
Discov Today. 16:485–494. 2011. View Article : Google Scholar : PubMed/NCBI
|