Introduction
Acute myelocytic leukemia (AML) is a hematopoietic
disease, which results in excessive accumulation of myeloid
precursor cells in the bone marrow. It is the most common form of
adult leukemia and the survival rate is very low (1–3).
Epidemiological studies from the United States National Cancer
Institute reported that the overall incidence rate of AML in the
USA between 2001 and 2005 was 3.6/100,000 individuals, and the
mortality rate was 2.8/100,000 individuals (4). In China, the annual incidence rate of
AML was 2.57/100,000 individuals in 2009 (5). In the past 30 years, the treatment of
AML has made some progress. Although conventional chemotherapy has
improved, the treatment of elderly patients with AML and AML
patients with relapsed refractory remains a challenge (6–8). An
improved understanding of the molecular events underlying the
molecular mechanisms of AML may be helpful to identify therapeutic
targets and improve the treatment efficacy to prolong the survival
rates for patients, thus this area of research requires further
investigation. Nuclear factor-κB (NF-κB) is a family of proteins
comprising RelA (p65), RelB, c-Rel, NF-κB1 (p105/p50) and NF-κB2
(p100/p52), which form homo- and heterodimers (9). NF-κB family members are transcription
factors that may mediate survival pathways in a number of types of
tumor, including leukemia (10).
These proteins induce the expression of genes involved in cell
proliferation, angiogenesis, metastasis and serve important roles
in carcinogenesis and chemoresistance (11). Tumor necrosis factor α (TNF-α) is a
central regulator of inflammation. It has been demonstrated to
upregulate molecules involved in cell growth, proliferation via
NF-κB dependent and independent pathways in tumors (12). In the present study, the correlation
between TNF-α and p65 expression levels and their association with
AML were investigated.
Materials and methods
Patient samples
Bone marrow samples were obtained from 30 AML
patients and 10 control patients, who were enrolled at the
Hematology Department of the First Hospital of Lanzhou University
(Lanzhou, China) for the present study. Pathological diagnosis was
confirmed by two senior pathologists. The healthy individuals
exhibited no signs of infection with hepatitis B virus, hepatitis C
virus or human immunodeficiency virus. Abdominal ultrasonic,
routine blood tests, and biochemical examination findings were all
normal. Written informed consent was obtained from all patients and
the present study was approved by the Ethics Committee of the First
Hospital of Lanzhou University. The serum samples were collected
after obtaining informed consent and the clinical information of
the samples are included in Table I
(13).
 | Table I.Clinical characteristics of bone
marrow sample patients. |
Table I.
Clinical characteristics of bone
marrow sample patients.
|
| Patients (n=30) | Control (n=10) |
|---|
|
|
|
|
|---|
| Characteristic | n | % | n | % |
|---|
| Age |
|
|
|
|
| ﹤50 | 13 | 43.3 | 4 | 40 |
| ≥50 | 17 | 56.7 | 6 | 60 |
| Gender |
|
|
|
|
| Male | 15 | 50 | 5 | 50 |
|
Female | 15 | 50 | 5 | 50 |
Reagents
RPMI-1640 and fetal bovine serum (FBS) were
purchased from GE Healthcare Life Sciences (Logan, UT, USA); Rabbit
anti-TNF-α antibody was obtained from Abcam (Cambridge, MA, USA);
rabbit anti-p65 primary antibody was purchased from Cell Signaling
Technology, Inc. (Danvers, MA, USA); and the NF-κB inhibitor
(MG-132) was purchased from Beyotime Institute of Biotechnnology
(Jiangsu, China).
Cell culture and drug treatment
HL-60 human AML cells (Shanghai Institutes for
Biological Science, Chinese Academy of Sciences, Shanghai, China)
were grown in RPMI-1640 medium supplemented with 10% FBS, and
maintained in humidified 5% CO2 at 37°C. For treatment
with anti-TNF-α antibody and MG132, cells were seeded at
1.5×105 cells/well in 2 ml RM1640 in a 6-well plate.
After 24 h, the medium was changed and the anti-TNF-α antibody (10
ng/ml) and MG132 (3 µM) were added into the medium separately or
together. The cells were incubated at 37°C for 48 h and then used
for further experiments.
RNA isolation and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted by TRIzol method (Takara,
Dalian, China), according to the manufacturer's instructions, and
reverse transcribed using a SuperScript III First-strand synthesis
system (Invitrogen Life Technologies, Carlsbad, CA, USA) to
generate cDNA by following the manufacturer's instructions. qPCR
was performed using a LightCycler_ 480 SYBR Green I Master (Roche
Diagnostics, Welwyn Garden City, UK) according to the
manufacturer's instructions. The sequences of the primers used were
as follows: β-actin, F 5′-TGG CAC CCA GCA CAA TGAA-3′ and R 5′-CTA
AGT CAT AGT CCG CCT AGA AGC A-3′; TNF-α, F
5′-CCATCTATCTGGGAGGGGTCT-3′ and R 5′-CGT TTG GGA AGG TTG GAT GT-3′;
and p65, F 5′-TTC GTC CTC CTC CTC ACA CTCC-3′ and R 5′-CCA GCC TGC
TTC TCC AAC AACA-3′. After an initial denaturation step of 5 min at
95°C, 40 cycles of amplification for each primer pair were carried
out. Each cycle included a denaturation step; 10 s at 95°C, an
annealing step; 20 s at 60°C and an elongation step; 10 s at 72°C.
The final elongation temperature was 65°C for 1 min. Relative
levels of gene expression was measured using a LightCycler 480
(Roche Diagnostics) according to the manufacturer's instructions.
The relative changes in the expression levels of TNF-α and p65
genes were normalized against the level of β-actin gene expression
in each sample. Experiments were performed at least in duplicate
for each data point.
Western blot analysis
Total cells were lysated with the buffer (1% SDS, 10
mm tris-Cl, pH 7.6, 20 g/ml aprotinin, 20 g/ml leupeptin and 1 mm
AEBSF). The protein concentrations were determined using the
Bradford method (14). Protein (20
µg) was separated on 12% of SDS-PAGE gels and transferred to PVDF
membranes (Merck Millipore, Darmstadt, Germany). After blocking
with 10% non-fat milk, the membranes were incubated with the first
antibodies at 4°C overnight. After washing 3 times with
triethanolamine buffered saline solution (Sangon Biotech Co., Ltd.,
Shanghai, China), the membranes were incubated with goat anti-human
IgG horseradish peroxidase-conjugated secondary antibodies (1:200
dilution in 5% non-fat milk) at room temperature for 1 h. The
signals were developed with the ECL kit (Applygen Technologies,
Inc., Beijing, China) and using anti-β-actin antibody as an
internal control.
Statistical analysis
All statistical comparisons were performed using
SPSS software, version 16.0 (SPSS, Inc., Chicago, IL, USA).
Student's t-test was used to compare differences between the
2 groups or association. P<0.05 was considered to indicate a
statistically significant difference. Pearson's Correlation test
was used to show the correlation of TNF-α and p65 expression in AML
specimens. Receiver operating characteristic (ROC) curves were
plotted to determine the potential of TNF-α and p65 expression to
differentiate AML samples from non-leukemic samples (15).
Results
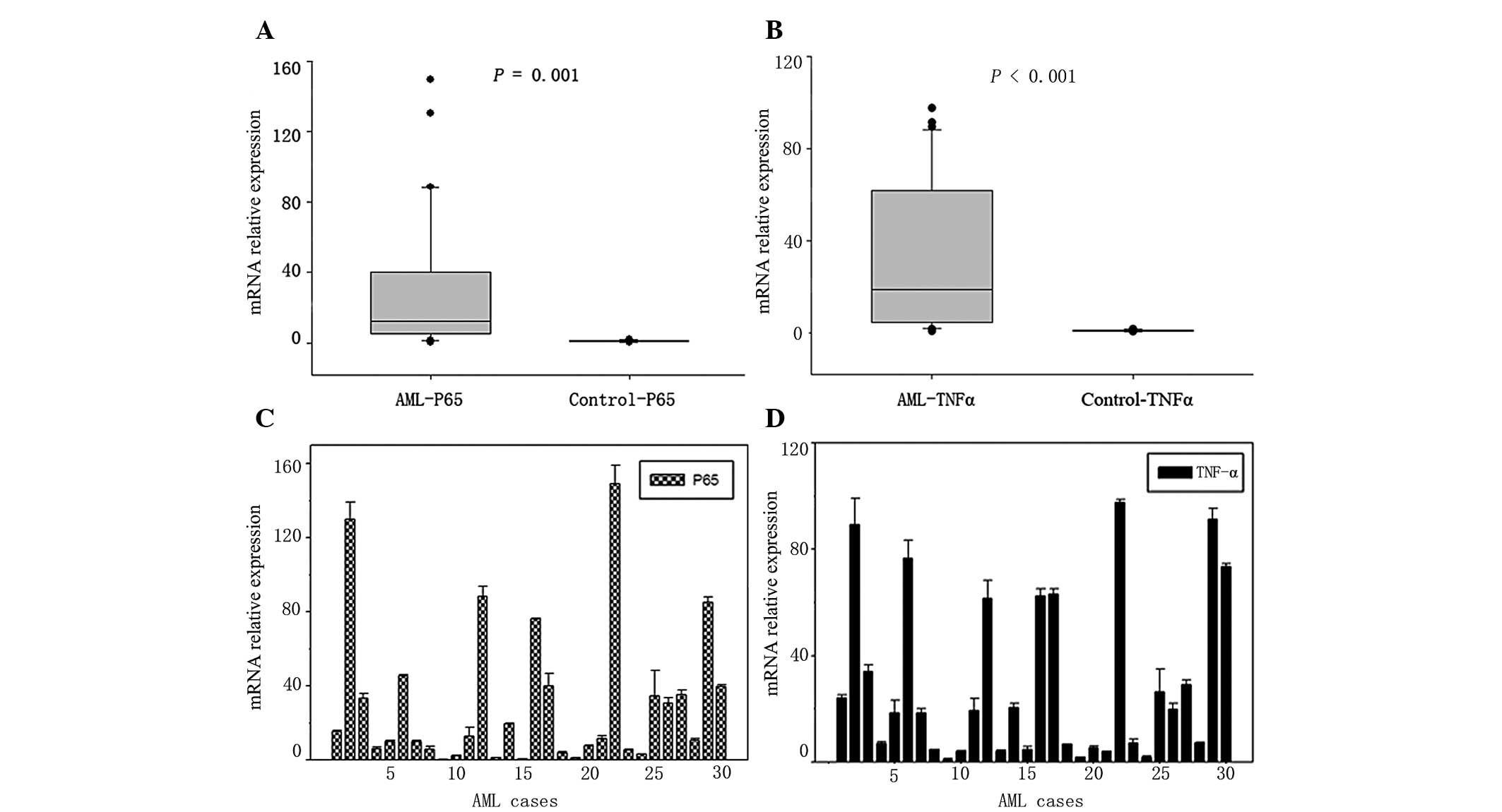
The mRNA expression levels of TNF-α
and p65 were increased in AML patient samples
A total of 40 samples were analyzed in the study:
The mRNA expression levels of TNF-α and p65 in bone marrow samples
from non-leukemia controls (n=10) and patients with AML (n=30) were
assessed by RT-qPCR. The results demonstrated that the expression
levels of p65 were significantly increased in AML patients compared
with non-leukemia control bone marrow samples (P=0.001) (Fig. 1A). The upregulation of TNF-α
(P<0.01) in AML patients was also confirmed (Fig. 1B). The mRNA expression levels of p65
and TNF-α in each AML case are presented in Fig. 1C and D, and demonstrate the variation
in mRNA expression levels between the patient samples.
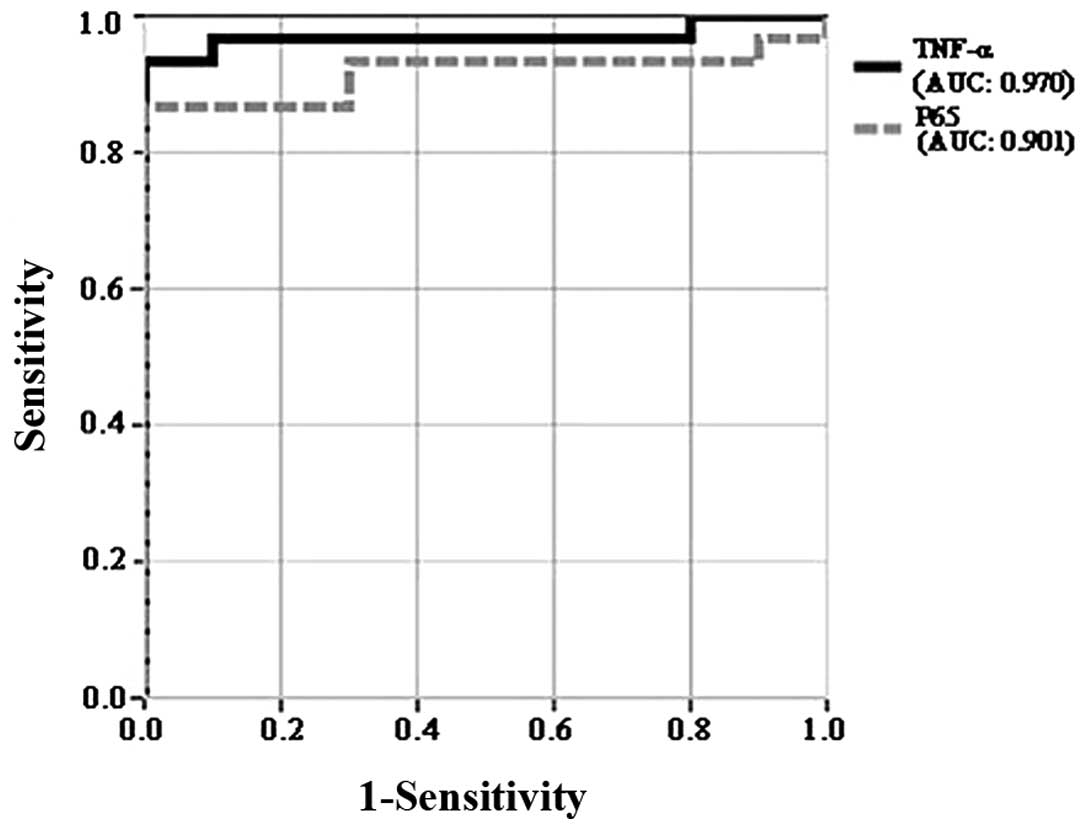
To further evaluate whether the expression levels of
TNF-α and p65 could distinguish AML patients from non-leukemia
control samples, receiver operating curves (ROC) were plotted. The
results demonstrated that the area under the curve (AUC) values of
TNF-α and p65 were 0.970 and 0.901 respectively, which demonstrated
that the 2 molecules exhibited sufficient power to distinguish AML
patients from non-leukemia control samples (Fig. 2).
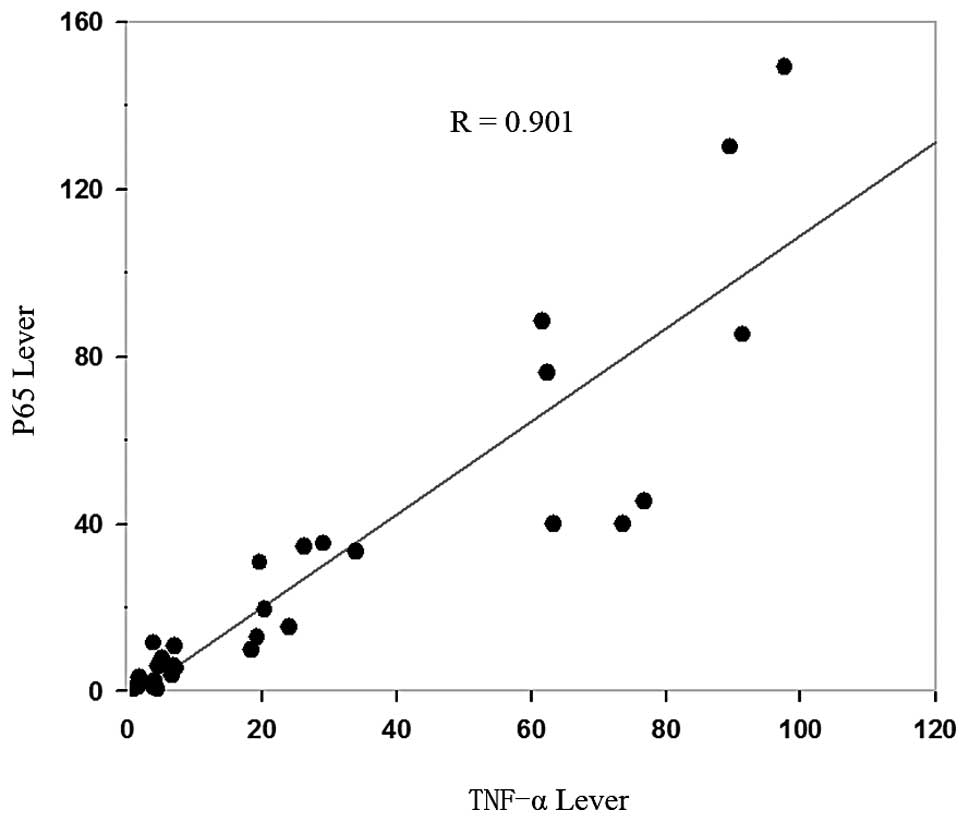
The expression of TNF-α was correlated
with p65 in AML patients
To assess the correlation between TNF-α and p65
expression in AML specimens, Pearson's correlation analysis was
used. The results demonstrated that TNF-α expression was strongly
correlated with p65 expression in AML bone marrow samples (Fig. 3, R=0.901).
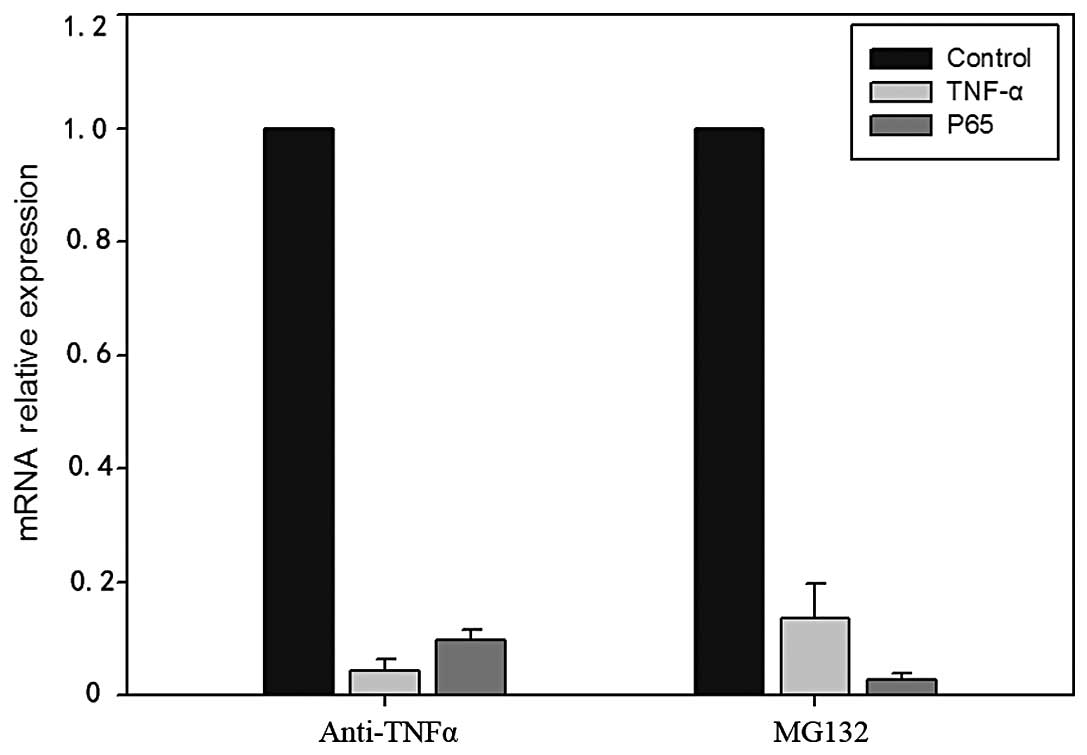
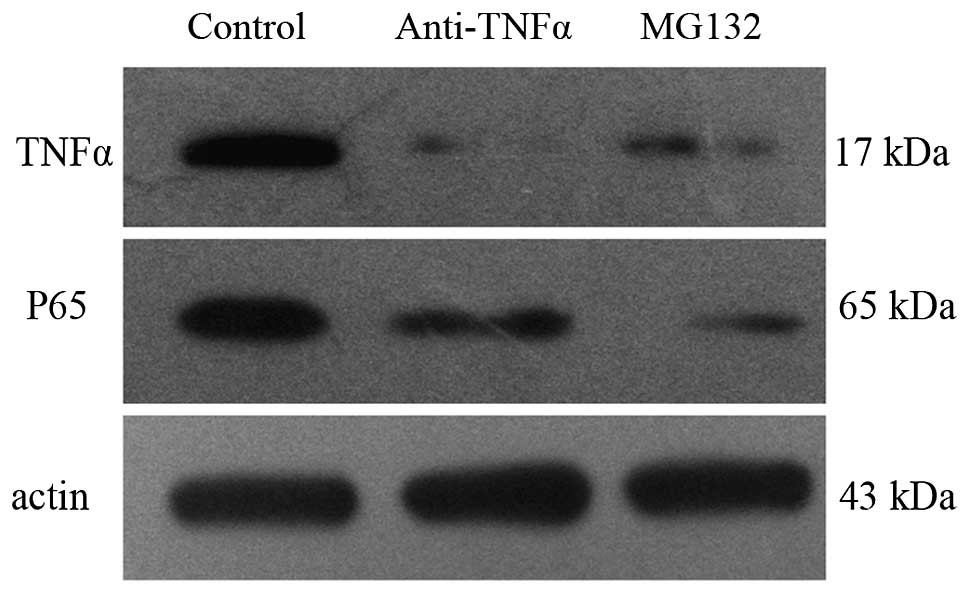
Inhibition of TNF-α reduced the
expression of p65 in HL-60 cells
HL-60 cells were treated with anti-TNF-α antibody
(TNF-α inhibitor), and the mRNA and protein expression levels of
TNF-α and p65 were detected by RT-qPCR and western blot analysis,
respectively. The results demonstrated that anti-TNF-α antibody
reduced the expression of TNF-α; the reduction in TNF-α expression
resulted in reduced expression levels of p65 mRNA and protein
levels (Figs. 4 and 5).
Inhibition of p65 reduced the
expression of TNF-α in HL-60 cells
HL-60 cells were treated with MG132 (an NF-κB
inhibitor) (16), and the mRNA and
protein expression levels of TNF-α and p65 were detected by RT-qPCR
and western blot analysis, respectively. The results demonstrated
that treatment with MG132 reduced the expression levels of p65; the
reduction in p65 expression resulted in concurrent reduced
expression of TNF-α at both the mRNA and protein level (Figs. 4 and 5).
Discussion
Nuclear factor-κB (NF-κB) are a group of
transcription factors that induces the expression of genes involved
in cell proliferation, angiogenesis and metastasis. It is a family
comprising RelA (p65), RelB, c-Rel, NF-κB1 (p105/p50) and NF-κB2
(p100/p52), which form homo- and heterodimers (10). NF-κB mediates survival pathways in a
number of types of tumor and serves important roles in
carcinogenesis and chemotherapy (11). Abdullah et al (17) detected the expression of p65 in
colorectal cancer cases and tumor-adjacent normal tissues from the
same subjects by immunohistochemical analysis, and demonstrated
that p65 was expressed at increased levels in colorectal cancer
case. A previous study demonstrated that the activity of NF-κB
could be detected in almost all leukemic cells, and its expression
was significantly increased in leukemic cells compared with normal
bone marrow cells (18).
Tumor necrosis factor α (TNF-α) is a central
regulator of inflammation (19). It
is also important for the development and progression of a number
of types of cancer. A previous study demonstrated that TNF-α
activated stromal COX-2 signalling and promoted the proliferative
and invasive potential of colon cancer epithelial cells (20). Another previous study demonstrated
that TNF-α acting on TNFR1 promotes breast cancer growth via
p42/P44 MAPK, JNK, Akt and NF-κB-dependent pathways (21).
The present study detected the expression levels of
p65 and TNF-α in bone marrow samples of AML patients and
non-leukemic controls by RT-qPCR. The mRNA expression levels of p65
and TNF-α were significantly increased in AML patients compared
with that of non-leukemic control bone marrow samples. NF-κB and
TNF-α may therefore active survival signaling pathways and serve
roles in AML development and progression. ROC curve analysis
revealed that these 2 molecules have potential as molecular markers
to distinguish AML patients from non-leukemic control samples and
thus act as a potential biomarker for AML.
TNF-α has been demonstrated to upregulate molecules
involved in cell growth and proliferation via NF-κB dependent or
independent pathways in tumors. TNF-α upregulates PTEN expression
via NF-κB signaling pathways in human leukemic cells (22). Positive feedback between NF-κB and
TNF-α promotes leukemia-initiating cell capacity (23). The present study also analyzed the
correlation between TNF-α and p65 expression in AML specimens.
Pearson's correlation analysis results demonstrated that TNF-α
expression was strongly correlated with p65 expression in AML bone
marrow samples. This correlation was also observed in an AML cell
line. Inhibition of TNF-α reduced the expression of p65 in HL-60
cells, and inhibition of p65 reduced the expression of TNF-α in
HL-60 cells.
In conclusion, the present study demonstrated that
p65 and TNF-α were expressed at high levels in AML patients, and
these 2 molecules were strongly correlated. p65 and TNF-α have
potential as molecular markers to distinguish AML patients from
non-leukemic control samples, and these 2 molecules may be useful
to predict prognostic factor for patients with AML.
Acknowledgements
The present study was supported by the Fundamental
Research Funds for the Central Universities (lzujbky-2013-150),
Science and Technology Plan Projects in Gansu Province
(1308RJYA071) and the Research Program of the First Hospital of
Lanzhou University (ldyyynlc201101).
References
|
1
|
Larkin K and Blum W: Novel therapies in
AML: Reason for hope or just hype? Am Soc Clin Oncol Educ Book.
e341–e351. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kohgo Y, Inamura J and Shindo M: Molecular
target drugs for AML-current state and prospects for the future.
Nihon Rinsho. 72:1063–1067. 2014.(In Japanese). PubMed/NCBI
|
|
3
|
Zeijlemaker W, Gratama JW and Schuurhuis
GJ: Tumor heterogeneity makes AML a ‘moving target’ for detection
of residual disease. Cytometry B Clin Cytom. 86:3–14. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Thomas R, Phuong J, McHale CM and Zhang L:
Using bioinformatic approaches to identify pathways targeted by
human leukemogens. Int J Environ Res Public Health. 9:2479–2503.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu YQ, Zhao FJ and Chen WQ: An analysis
of incidence and mortality of leukemia in China, 2009. Zhong Guo Ai
Zheng Yan Jiu. 22:528–534. 2013.(In Chinese).
|
|
6
|
Miyawaki S: Guideline for AML. Rinsho
Ketsueki. 54:1633–1642. 2013.(In Japanese). PubMed/NCBI
|
|
7
|
Levine RL: Molecular pathogenesis of AML:
Translating insights to the clinic. Best Pract Res Clin Haematol.
26:245–248. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Al-Ali HK, Jaekel N and Niederwieser D:
The role of hypomethylating agents in the treatment of elderly
patients with AML. J Geriatr Oncol. 5:89–105. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kamieńska E, Ociepa T, Wysocki M, Kurylak
A, Matysiak M, Urasiński T, Urasińska E and Domagała W: Activation
of NF-kB in leukemic cells in response to initial prednisone
therapy in children with acute lymphoblastic leukaemia: Relation to
other prognostic factors. Pol J Pathol. 62:5–11. 2011.PubMed/NCBI
|
|
10
|
Giuliani C, Napolitano G, Bucci I, Montani
V and Monaco F: Nf-kB transcription factor: Role in the
pathogenesis of inflammatory, autoimmune and neoplastic diseases
and therapy implications. Clin Ter. 152:249–253. 2001.(In Italian).
PubMed/NCBI
|
|
11
|
Baud V and Jacque E: The alternative NF-kB
activation pathway and cancer: friend or foe? Med Sci (Paris).
24:1083–1088. 2008.(In French). View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Schulz U, Munker R, Ertl B, Holler E and
Kolb HJ: Different types of human leukemias express the message for
TNF-alpha and interleukin-10. Eur J Med Res. 6:359–363.
2001.PubMed/NCBI
|
|
13
|
Sun Y, Mi W, Cai J, Ying W, Liu F, Lu H,
Qiao Y, Jia W, Bi X, Lu N, et al: Quantitative proteomic signature
of liver cancer cells: Tissue transglutaminase 2 could be a novel
protein candidate of human hepatocellular carcinoma. J Proteome
Res. 7:3847–3859. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zor T and Zvi S: Linearization of the
Bradford protein assay increases its sensitivity: Theoretical and
experimental studies. Anal Biochem. 236:302–308. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Karatas OF, Guzel E, Suer I, Ekici ID,
Caskurlu T, Creighton CJ, Ittmann M and Ozen M: miR-1 and miR-133b
are differentially expressed in patients with recurrent prostate
cancer. PLoS One. 9:e986752014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Morotti A, Cilloni D, Pautasso M, Messa F,
Arruga F, Defilippi I, Carturan S, Catalano R, Rosso V, Chiarenza
A, et al: NF-kB inhibition as a strategy to enhance
etoposide-induced apoptosis in K562 cell line. Am J Hematol.
81:938–945. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Abdullah M, Rani AA, Sudoyo AW, Makmun D,
Handjari DR and Hernowo BS: Expression of NF-kB and COX2 in
colorectal cancer among native Indonesians: The role of
inflammation in colorectal carcinogenesis. Acta Med Indones.
45:187–192. 2013.PubMed/NCBI
|
|
18
|
Guzman ML, Neering SJ, Upchurch D, Grimes
B, Howard DS, Rizzieri DA, Luger SM and Jordan CT: Nuclear
factor-kappaB is constitutively activated in primitive human acute
myelogenous leukemia cells. Blood. 98:2301–2307. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Choi S, Park YS, Koga T, Treloar A and Kim
KC: TNF-α is a key regulator of MUC1, an anti-inflammatory
molecule, during airway Pseudomonas aeruginosa infection. Am J
Respir Cell Mol Biol. 44:255–260. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhu M, Zhu Y and Lance P: TNF α-activated
stromal COX-2 signalling promotes proliferative and invasive
potential of colon cancer epithelial cells. Cell Prolif.
46:374–381. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rivas MA, Carnevale RP, Proietti CJ,
Rosemblit C, Beguelin W, Salatino M, Charreau EH, Frahm I, Sapia S,
Brouckaert P, et al: TNF alpha acting on TNFR1 promotes breast
cancer growth via p42/P44 MAPK, JNK, Akt and NF-kappa B-dependent
pathways. Exp Cell Res. 314:509–529. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee YR, Yu HN, Noh EM, Youn HJ, Song EK,
Han MK, Park CS, Kim BS, Park YS, Park BK, et al: TNF-alpha
upregulates PTEN via NF-kappaB signaling pathways in human leukemic
cells. Exp Mol Med. 39:121–127. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kagoya Y, Yoshimi A, Kataoka K, Nakagawa
M, Kumano K, Arai S, Kobayashi H, Saito T, Iwakura Y and Kurokawa
M: Positive feedback between NF-κB and TNF-α promotes
leukemia-initiating cell capacity. J Clin Invest. 124:528–542.
2014. View
Article : Google Scholar : PubMed/NCBI
|