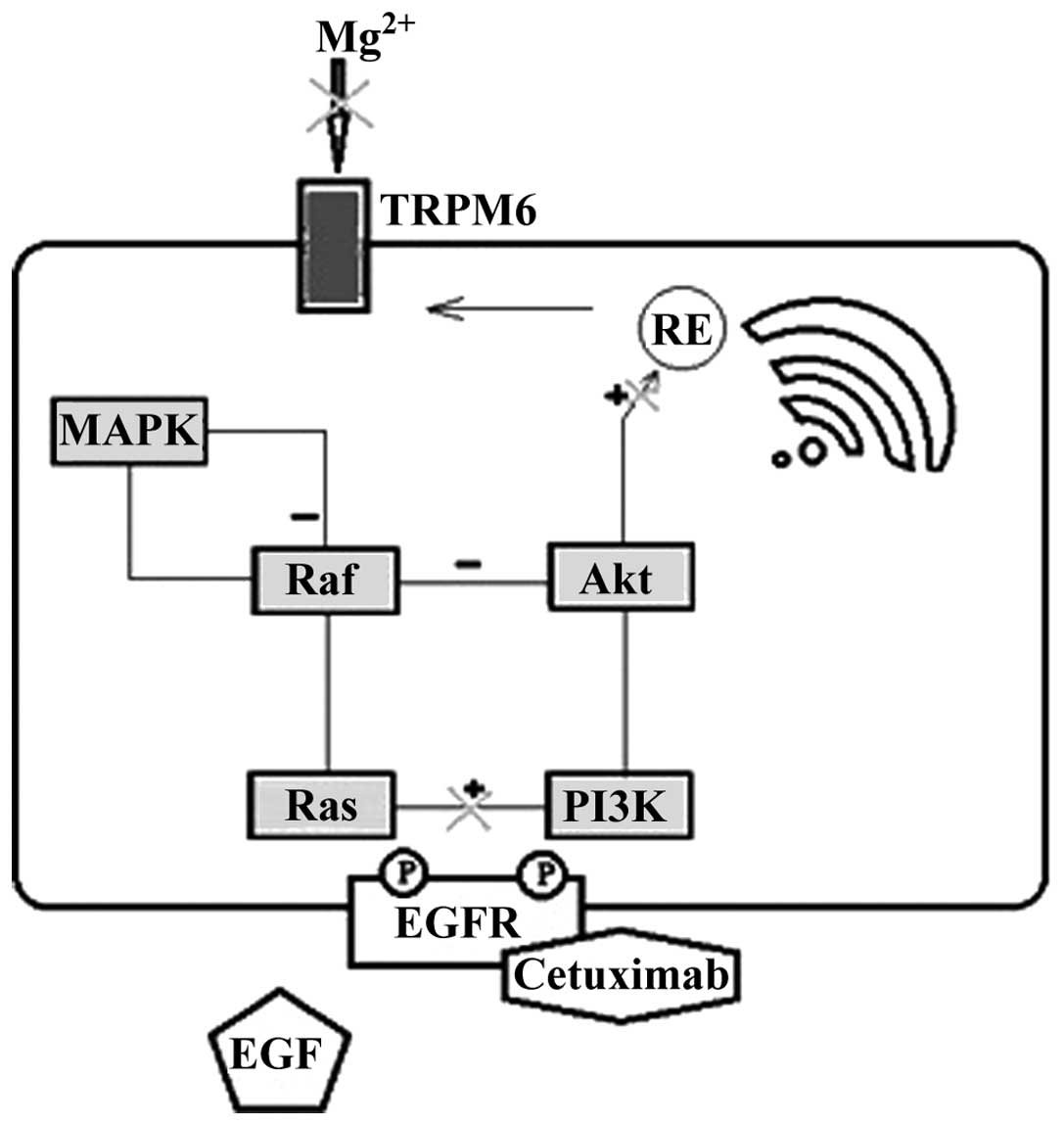
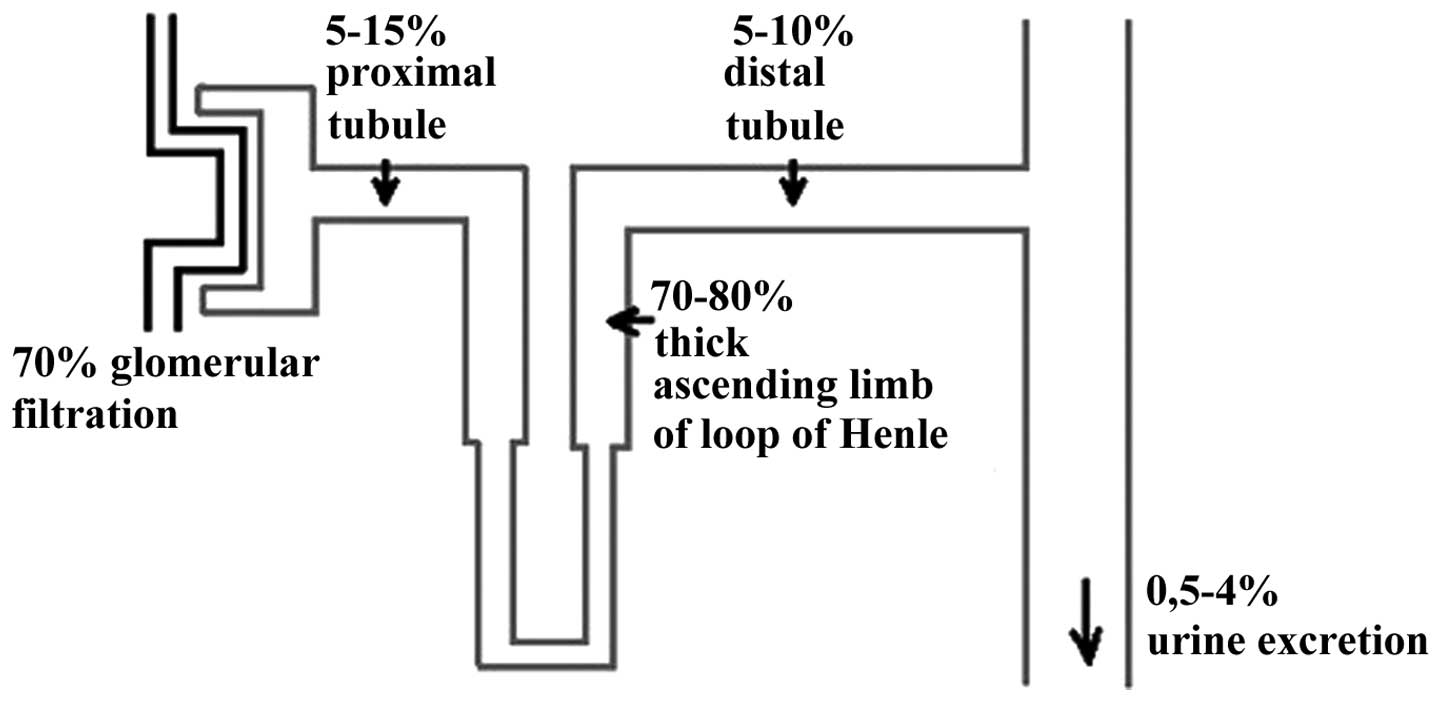
|
1
|
Fakih M and Vincent M: Adverse events
associated with anti-EGFR therapies for the treatment of metastatic
colorectal cancer. Curr Oncol. 17(Suppl 1): S18–S30.
2010.PubMed/NCBI
|
|
2
|
Boyle P and Langman JS: ABC of colorectal
cancer: Epidemiology. BMJ. 321:805–808. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Saltz LB, Meropol NJ, Loehrer PJ Sr,
Needle MN, Kopit J and Mayer RJ: Phase II trial of cetuximab in
patients with refractory colorectal cancer that expresses the
epidermal growth factor receptor. J Clin Oncol. 22:1201–1208. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cunningham D, Humblet Y, Siena S, Khayat
D, Bleiberg H, Santoro A, Bets D, Mueser M, Harstrick A, Verslype
C, et al: Cetuximab monotherapy and cetuximab plus irinotecan in
irinotecan-refractory metastatic colorectal cancer. N Engl J Med.
351:337–345. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jonker DJ, O'Callaghan CJ, Karapetis CS,
Zalcberg JR, Tu D, Au HJ, Berry SR, Krahn M, Price T, Simes RJ, et
al: Cetuximab for the treatment of colorectal cancer. N Engl J Med.
357:2040–2048. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bokemeyer C, Van Cutsem E, Rougier P,
Ciardiello F, Heeger S, Schlichting M, Celik I and Köhne CH:
Addition of cetuximab to chemotherapy as first-line treatment for
KRAS wild-type metastatic colorectal cancer: Pooled analysis of the
CRYSTAL and OPUS randomized clinical trials. Eur J Cancer.
48:1466–1475. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pacek A, Kozioł M, Püsküllüoğlu M,
Tomaszewski KA, Ochenduszko S, Zygulska AL and Krzemieniecki K:
Assessment of skin-related toxicity in patients with metastatic
colorectal cancer treated with cetuximab. Acta Dermatovenerol
Croat. 22:137–144. 2014.PubMed/NCBI
|
|
8
|
Martin KJ, González EA and Slatopolsky E:
Clinical consequences and management of hypomagnesemia. J Am Soc
Nephrol. 20:2291–2295. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Saif MW: Management of hypomagnesemia in
cancer patients receiving chemotherapy. J Support Oncol. 6:243–248.
2008.PubMed/NCBI
|
|
10
|
National Cancer Institute Common Toxicity
Criteria. Version 4.0. May 28–2009.http://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htmAccessed.
May 2–2014
|
|
11
|
Groenestege WM, Thébault S, van der Wijst
J, van den Berg D, Janssen R, Tejpar S, van den Heuvel LP, van
Cutsem E, Hoenderop JG, Knoers NV, et al: Impaired basolateral
sorting of pro-EGF causes isolated recessive renal hypomagnesemia.
J Clin Invest. 117:2260–2267. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tejpar S, Piessevaux H, Claes K, Piront P,
Hoenderop JG, Verslype C and Van Cutsem E: Magnesium wasting
associated with epidermal-growth-factor receptor-targeting
antibodies in colorectal cancer: A prospective study. Lancet Oncol.
8:387–394. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang XD, Jia C, Corvalan JR, Wang P and
Davis CG: Development of ABX-EGF, a fully human anti-EGF receptor
monoclonal antibody, for cancer therapy. Crit Rev Oncol Hematol.
38:17–23. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fakih M: Anty-EGFR monoclonal
antibody-induced hypomagnesaemia. Lancet Oncol. 8:366–367. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Edge S, Byrd DR, Compton CC, Fritz AG,
Greene FL and Trotti A: AJCC Cancer Staging Manual. (7th). (New
York, NY). Springer. 1432010.
|
|
16
|
Schrag D, Chung KY, Flombaum C and Saltz
L: Cetuximab therapy and symptomatic hypomagnesemia. J Natl Cancer
Inst. 97:1221–1224. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Erbitux-Summary of product
characteristics. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000558/WC500029119.pdfAccessed.
May 2–2014
|
|
18
|
Chen P, Wang L, Li H, Liu B and Zou Z:
Incidence and risk of hypomagnesemia in advanced cancer patients
treated with cetuximab: A meta-analysis. Oncol Lett. 5:1915–1920.
2013.PubMed/NCBI
|
|
19
|
Cao Y, Liao C, Tan A, Liu L and Gao F:
Meta-analysis of incidence and risk of hypomagnesemia with
cetuximab for advanced cancer. Chemotherapy. 56:459–465. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Price TJ, Peeters M, Kim TW, Li J, Cascinu
S, Ruff P, Suresh AS, Thomas A, Tjulandin S, Zhang K, et al:
Panitumumab versus cetuximab in patients with
chemotherapy-refractory wild-type KRAS exon 2 metastatic colorectal
cancer (ASPECCT): A randomised, multicentre, open-label,
non-inferiority phase 3 study. Lancet Oncol. 15:569–579. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lordick F, Kang YK, Chung HC, Salman P, Oh
SC, Bodoky G, Kurteva G, Volovat C, Moiseyenko VM, Gorbunova V, et
al: Capecitabine and cisplatin with or without cetuximab for
patients with previously untreated advanced gastric cancer
(EXPAND): A randomised, open-label phase 3 trial. Lancet Oncol.
14:490–499. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vickers MM, Karapetis CS, Tu D,
O'Callaghan CJ, Price TJ, Tebbutt NC, Van Hazel G, Shapiro JD,
Pavlakis N, Gibbs P, et al: Association of hypomagnesemia with
inferior survival in a phase III, randomized study of cetuximab
plus best supportive care versus best supportive care alone: NCIC
CTG/AGITG CO.17. Ann Oncol. 24:953–960. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Weickhardt AJ, Price TJ, Chong G, Gebski
V, Pavlakis N, Johns TG, Azad A, Skrinos E, Fluck K, Dobrovic A, et
al: Dual targeting of the epidermal growth factor receptor using
the combination of cetuximab and erlotinib: Preclinical evaluation
and results of the phase II DUX study in chemotherapy-refractory,
advanced colorectal cancer. J Clin Oncol. 30:1505–1512. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vincenzi B, Santini D, Galluzzo S, Russo
A, Fulfaro F, Silletta M, Battistoni F, Rocci L, Zobel BB, Adamo V,
et al: Early magnesium reduction in advanced colorectal cancer
patients treated with cetuximab plus irinotecan as predictive
factor of efficacy and outcome. Clin Cancer Res. 14:4219–4224.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Pazo-Oubiña Do F, Estefanell-Tejero A,
Riu-Viladoms G, Anglada-Martínez H, Molas-Ferrer G and Creus-Baró
N: Magnesium monitoring practice in monoclonal anti-epidermal
growth factor receptor antibodies therapy. J Clin Pharm Ther.
38:101–103. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Demizu M, Ueda H, Osawa M, Chihara S,
Igarashi T, Yano K, Kimura F, Tanaka N and Hiratsuka M: Effect of
magnesium supplementation on early-stage hypomagnesemia in patients
treated with cetuximab. Gan To Kagaku Ryoho. 40:897–900.
2013.PubMed/NCBI
|
|
27
|
Melichar B, Králíčková P, Hyšpler R,
Kalábová H, Cerman J Jr, Holečková P, Studentová H and Malířová E:
Hypomagnesaemia in patients with metastatic colorectal carcinoma
treated with cetuximab. Hepatogastroenterology. 59:366–371.
2012.PubMed/NCBI
|
|
28
|
Vincenzi B, Galluzzo S, Santini D, Rocci
L, Loupakis F, Correale P, Addeo R, Zoccoli A, Napolitano A,
Graziano F, et al: Early magnesium modifications as a surrogate
marker of efficacy of cetuximab-based anticancer treatment in KRAS
wild-type advanced colorectal cancer patients. Ann Oncol.
22:1141–1146. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Maliakal P and Ledford A: Electrolyte and
protein imbalance following anti-EGFR therapy in cancer patients: A
comparative study. Exp Ther Med. 1:307–311. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fakih MG, Wilding G and Lombardo J:
Cetuximab-induced hypomagnesemia in patients with colorectal
cancer. Clin Colorectal Cancer. 6:152–156. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Stintzing S, Fischhaber D, Mook C, Modest
DP, Giessen C, Schulz C, Haas M, Boeck S, Michl M, Stemmler J, et
al: Clinical relevance and utility of cetuximab-related changes in
magnesium and calcium serum levels. Anticancer Drugs. 24:969–974.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Thomas SE, Anderson S, Gordon KL, Oyama
TT, Shankland SJ and Johnson RJ: Tubulointerstitial disease in
aging: Evidence for underlying peritubular capillary damage, a
potential role for renal ischemia. J Am Soc Nephrol. 9:231–242.
1998.PubMed/NCBI
|
|
33
|
Wolf FI, Cittadini AR and Maier JA:
Magnesium and tumors: Ally or foe? Cancer Treat Rev. 35:378–382.
2009. View Article : Google Scholar : PubMed/NCBI
|