Introduction
B-cell acute lymphoblastic leukemia (B-ALL) is a
malignant neoplasm derived from B-cell progenitors. It is the most
commonly occurring malignancy in pediatric patients, accounting for
up to 80% of childhood leukemia cases. Thus, it is the primary
cause of cancer-associated mortality in children and young adults
(1,2).
Rearrangements involving the immunoglobulin (Ig)
heavy chain (IGH@) locus on chromosomal band 14q32.33 are
rare in B-ALL, occurring in <5% of childhood cases and detected
in ~10% of adult patients (3,4). IGH@ rearrangements occur more
often in adolescents and appear to be associated with a favorable
clinical outcome. This additionally applies to cases of B-ALL
associated with genetic aberrations, including deletion (del) in
9p21.3 [cyclin-dependent kinase inhibitor 2A/B
(CDKN2A/B)] and 9p13.3 (paired box-5) (5). In B-ALL, the most common IGH@
rearrangements involve translocation to partner genes such as
C-MYC in 8q24 by the well-characterized translocation
t(8;14)(q24.1;q32). An additional partner is the inhibitory
transcription factor ID4 in 6p22, which is cytogenetically
visible as translocation t(6;14)(q32;p22). Another translocation,
t(14;19)(q32;q13), leads to overexpression of the CCAAT-enhancer
binding protein (CEBP) gene family (6), whilst t(5;14)(q31;q32) involves
interleukin-3 in 5q31 (7), and the
translocations t(X;14)(p22;q32) and t(Y;14)(p11.2;q32) result in
dysregulated expression of cytokine receptor-like factor 2
(8). Translocations between
IGH@ and erythropoietin receptor in 19p13 have also been
reported together with other translocations appearing less
frequently (9–11).
In all of the aforementioned translocations, an
oncogene located close to the breakpoint of the translocation
partner is activated via juxtaposing to IGH@ regulatory
sequences (4). Notably, all
rearrangements involving IGH@ at 14q32.33 possess unique
biological characteristics and have been observed to correlate with
clinical, morphological and immunophenotypic characteristics.
Cryptic deletions in chromosomal band 9p21.3 involve
the CDKN2A gene, which encodes two transcripts
(p16Ink4A and p14ARF), as well
as the CDKN2B gene (p15Ink4B). The
functions of these transcripts in the cell cycle are to control the
transition from G1 to S phase. The sizes of 9p21.3 deletions in
patients with ALL appear to vary significantly; however, in the
majority of cases, CDKN2A is co-deleted with CDKN2B
and methylthioadenosine phosphorylase (12–14).
The present study reported a novel IGH@
rearrangement associated with a deletion in CDKN2A/B
in a young adult with B-ALL. In addition, the potential underlying
mechanism of chromosome 14 rearrangement is discussed.
Patient and methods
Clinical description
A 20-year-old female presented at the Hospital Maria
Sklodowska-Curie Memorial Cancer Centre and Institute (Warsaw,
Poland) in November 2008 with a white blood cell count of
3.7×109/l (normal range, 3.5–10×109/l),
hemoglobin of 11.0 g/dl (normal range, 12.0–16.0 g/dl) and
platelets of 334×109/l (normal range,
125–400×109/l). In the bone marrow, ~93% blast cells
(normal range, <5%) were observed. Immunophenotype was
characterized by the expression of a variety of B-cell-specific
antigens, with positivity for cluster of differentiation (CD)10,
CD19, CD22, CD34, CD38, CD45, CD52, CD79a, terminal
deoxynucleotidyl transferase and human leukocyte antigen-DR, and
negativity for CD2, CD15, CD20, CD33, CD56, CD66c and cIgM. These
results were consistent with common B-ALL.
The patient was treated with induction therapy,
which consisted of epirubicin, vincristine and PEG-L-asparaginase,
steroids, according to the Polish Adult Leukemia Group (PALG)
protocol (15), with two courses of
consolidation (consolidation I, vepesid, metrotrexate and
dexamethasone; consolidation II, cyclophosphamide, cytosar and
PEG-L-asparaginase) and maintenance treatment. From December 2011
to date, the patient has remained under the observation of an
outpatient clinic, and demonstrated complete remission with no
signs of minimal residual disease (MRD).
The present study was approved by the Ethical Board
at the Friedrich Schiller University (Jena, Germany; approval no.,
1105-04/03) and written informed consent was obtained from the
patient.
Cytogenetic results at diagnoses
Banding cytogenetic analyses were performed on
unstimulated bone marrow aspirate according to standard protocols
(16). A total of 25 metaphases were
available for cytogenetic evaluation, and were analyzed on a
banding level of 300 bands per haploid karyotype (17). GTG-banding revealed a normal female
karyotype of 46,XX.
Retrospective analyses
Molecular cytogenetics
Fluorescence in situ hybridization (FISH) was
performed according to standard procedures (18) and/or according to the manufacturer's
protocol.
Probes and probe sets were constructed as follows:
Bacterial artificial chromosome clones of interest were identified
using the Human Genome Browser Database of the Genome
Bioinformatics Group at the University of California at Santa Cruz
(Santa Cruz, CA, USA; http://genome.ucsc.edu/) and Ensembl Genome Data
Resources of the Sanger Institute Genome Database (http://www.ensembl.org/). DNA probes (Table I) obtained from the Resources Center
(Oakland, CA, USA) were labeled by polymerase chain reaction with
SpectrumGreen (Green-dUTP; catalog no., 02N32-050; Abbott
Molecular, Des Plaines, IL, USA), SpectrumOrange (Orange-dUTP;
catalog no., 02N33-050; Abbott Molecular) or TexasRed-dUTP
(ChromaTide-TexasRed-12-dUTP; catalog no., C-7631; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) and applied for two- or
three-color FISH approaches. The FISH-banding probe sets used were
as follows: Genome wide multitude multicolor banding (mMCB) and
chromosome-specific high-resolution array-proven multicolor-banding
(aMCB) (19–21).
 | Table I.Used probes, their location and
obtained results listed according to GRCh37/hg19. |
Table I.
Used probes, their location and
obtained results listed according to GRCh37/hg19.
| Cytoband | Location
(GRCh37/hg19) | Probe | Result |
|---|
| 3q26.32 | Chr3:
177,272,863–177,430,308 | RP11–114M1 | Deletion on
der(3) ish
3q26.3(RP11–114M1×1)[5] |
| 3q26.32 | Chr3:
177,488,843–177,646,481 | RP11–91K9 | Deletion on
der(3) ish 3q26.3(RP11–91K9)[5] |
| 9p21.3/ | Chr9:
21,967,751–21,975,132/ | SPEC p16/ | Deletion on
der(9) |
| 9p11.1q11 | Chr9:
47,300,001–50,700,000 | CEN9 | ish
9p21.3(p16×1)[8] nuc ish 9p21(p16×1)[147]/9p21(p16×2)[53] |
| 14q11.2 | Chr14:
20,814,125–20,814,672 | RP11–332N6 | 1 signal on
der(14) |
| 14q11.2 | Chr14:
20,940,682–21,103,092 | RP11–14J7 | 1 signal on
der(14) |
| 14q12 | Chr14:
29,511,827–29,698,386 | RP11–125A5 | 1 signal on
der(14) |
| 14q13.1 | Chr14:
32,299,162–32,460,130 | RP11–501E21 | 1 signal on
der(14) |
| 14q13.2 | Chr14:
35,335,072–35,521,841 | RP11–26M6 | 1 signal on
der(14) |
| 14q13.3 | Chr14:
36,683,813–36,704,814 | RP11–259K15 | 2 signals on
der(14) |
| 14q21.1 |
Chr14:39,897,747–40,060,823 | RP11–111A21 | 2 signals on
der(14) |
| 14q21.1 | Chr14:
40,408,068–40,537,355 | RP11–34O18 | 2 signals on
der(14) |
| 14q21.3 | Chr14:
49,809,988–49,981,102 | RP11–346L24 | 2 signals on
der(14) |
| 14q21.3 | Chr14:
50,148,020–50,148,604 | RP11–831F12 | 2 signals on
der(14) |
| 14q23.1 | Chr14:
59,967,413–60,142,554 | RP11–701B16 | 2 signals on
der(14) |
| 14q24.2 | Chr14:
70,701,212–70,701,81 | RP11–486O13 | 2 signals on
der(14) |
| 14q31.1 | Chr14:
80,030,106–80,193,689 | RP11–242P2 | 2 signals on
der(14) |
| 14q32.3 | Chr14:
106,053,226–106,518,932 | LSI IGH | Split signals on
der(14) |
| 14qter | Chr14:
107,038,129–107,238,316 | D14S1420 | 1 signal on
der(14) |
In addition, commercially available probes were
used: LSI IGH (14q32 break probe; Vysis; Abbott Molecular,
Inc., Des Plaines, IL, USA), POSEIDON p16 (9p21 and 9q21 control
probe; Kreatech Diagnostics, Amsterdam, Netherlands), SPEC
erythroblast transformation-specific (ETS)-related gene
(ERG)/TMPRSS2 TriCheck™ Probe (ERG in 21 q12.13-q22.3,
TMPRSS2 in 21q22.3; ZytoVision GmbH, Bremerhaven, Germany), and
subtelomeric probe for 14q (14q in D14S1420; Vysis; Abbott
Molecular, Inc.).
A total of 10–15 metaphase spreads were analyzed
with a fluorescence microscope (AxioImager Z1; Zeiss AG,
Oberkochen, Germany), which was equipped with appropriate filter
sets to discriminate between a maximum of five fluorochromes and
the counterstain 4′,6-diamidino-2-phenylindole. Image capturing and
processing was performed with the ISIS imaging system (MetaSystems
Hard & Software GmbH, Altlussheim, Germany).
DNA isolation
Genomic DNA was extracted from cells fixed in acetic
acid-methanol (1:3) using a Puregene DNA Purification kit (Gentra
Systems, Inc., Big Lake, MN, USA). DNA concentration was determined
using a spectrophotometer (NanoDrop Technologies; Thermo Fisher
Scientific, Inc.). The quality of DNA was assessed by agarose gel
electrophoresis. DNA samples extracted from fixed cells of two
healthy men and two healthy women by an identical method were used
as reference samples.
Multiplex ligation-dependent probe amplification
(MLPA)
P377-A1 Hematologic Malignancies probemix and SALSA
reagents were utilized for the present study (MRC Holland BV,
Amsterdam, Netherlands). Amplified probes and Genescan 500 ROX Size
Standard were separated using capillary electrophoresis using a
4-capillary ABI-PRISM 3130xl Genetic Analyzer (Applied Biosystems;
Thermo Fisher Scientific, Inc.). Sizing of peaks and quantification
of peak areas and heights was performed using GeneMarker software
version 1.9 (SoftGenetics, LLC., State College, PA, USA). A minimum
of 4 healthy control samples were included in each run.
High-resolution array-comparative genomic
hybridization (aCGH)
aCGH was performed with Agilent SurePrint G3 Human
Genome Microarray 180 kit (Agilent Technologies, Inc., Santa Clara,
CA, USA), and an oligonucleotide microarray containing ~180,000
probes 60-mer with a 17 kb average probe spacing. Genomic DNA of
the patient was co-hybridized with male control DNA (Agilent
Technologies, Inc.). Labeling was performed with a Genomic DNA
enzymatic labeling kit (Agilent Technologies, Inc.), according to
the manufacturer's protocol. Following hybridization, the aCGH
slide was scanned and processed by Feature Extraction software
version 10.7 (Agilent Technologies, Inc.), and the results were
analyzed with CytoGenomics software version 2.9.1.3, using
adrenomedullin 2 as an aberration algorithm.
Results
Molecular cytogenetics
G-banding at a low resolution did not reveal any
chromosomal aberrations. Retrospective application of mMCB revealed
only one gross chromosomal alteration, which was an inverted
duplication on chromosome 14. In order to characterize the
rearrangement in detail, additional FISH experiments, including
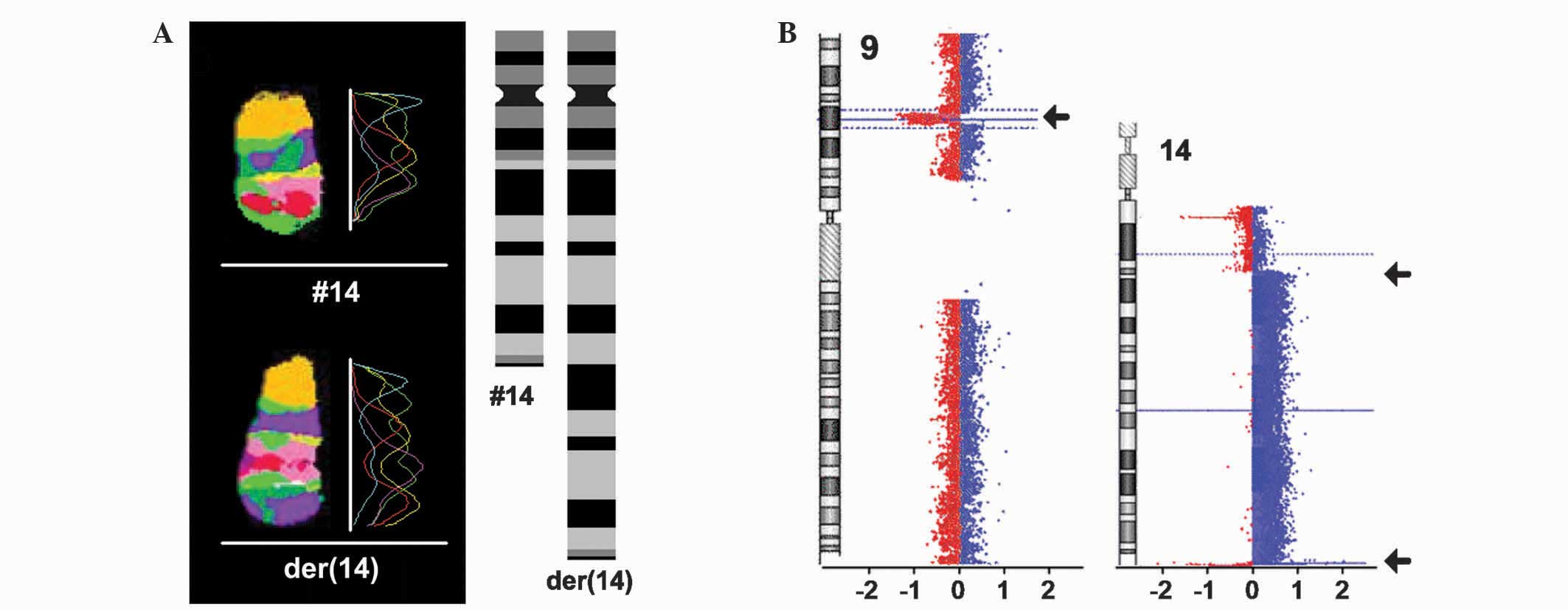
aMCB for chromosome 14 (Fig. 1A) and
locus-specific FISH probes (Table I),
were applied, revealing the derivative (der) chromosome
der(14) (pter-> q32.33 ::q32.33
-> q13 ::q32.33 -> qter).
Dual-color FISH, using a commercially available
break-apart rearrangement probe specific for IGH, was
performed. Interphase nuclei and metaphase studies revealed
splitting of the IGH variable region and 3′ flanking region,
located downstream of the IGH locus (data not shown). MLPA analysis
revealed heterozygous deletion of p16Ink4A,
p15Ink4B and p14ARF, and this
was confirmed by interphase FISH (data not shown).
aCGH
aCGH revealed two large genomic imbalances: A gain
of 70.6 Mb in the region of 14q13.2-q32.3 between the positions
(according to the Genome Reference Consortium Human 37/Human Genome
Issue 19; University of California Santa Cruz Genome
Bioinformatics, Santa Cruz, CA, USA; avaliable from https://genome-euro.ucsc.edu/index.html)
35,918,265 and 106,513,022, and loss of 3 Mb in the region of
9p21.3 between the positions 21,252,517 and 24,289,720. These
observations were compatible with the FISH and MLPA results
(Fig. 1B).
Furthermore, aCGH revealed five small genomic
imbalances with loss of copy number variants in the following: i)
3q26.32 between the positions 176,825,586 and 177,697,157 [one
Online Mendelian Inheritance in Man (OMIM; http://www.omim.org/) gene, transducin (β)-like 1
X-linked receptor 1 (TBL1XR1), is located in this position];
ii) 10p15.3 between the positions 1,491,986 and 1,582,072 [two OMIM
genes are located in this position: Adenosine deaminase,
RNA-specific, B2 (ADARB2), and NCRNA00168]; iii)
16q13 between the positions 57,275,940 and 57,331,381 [two OMIM
genes are located in this position: ADP-ribosylation factor-like 2
binding protein (ARL2BP) and plasmolipin (PLLP)]; iv)
21q22.2 between the positions 39,764,621 and 39,865,171 [one OMIM
gene is located in this position: ERG]; v) Xq13.3 between
the positions 47,330,212 and 47,335,227 [one OMIM gene in is
located in this position: Zinc finger protein 41 (ZNF41;
results not shown)].
Discussion
Copy number variants of specific target genes are
important in the development and progression of acute leukemia and
may serve as potential biomarkers for prognosis, as well as being
targets for molecular therapy. Gene amplification is a significant
mechanism of oncogene activation in acute leukemia. However, it is
difficult to identify or resolve genomic imbalances <10 Mb in
size by banding cytogenetics due to poor quality chromosomes, which
may not be well-spread and may appear with indistinct margins.
Molecular cytogenetic approaches, including FISH, MLPA, and aCGH,
have been demonstrated to be useful for detection of previously
hidden genomic imbalances (10,22). In
the present study, application of the aforementioned approaches
revealed a previously unreported genomic imbalance in a B-ALL case:
46, XX, del(9) (p21.3 p21.3),
der(14) (pter-> q32.33 ::q32.33
-> q13:: q32.33 -> qter). Characterization of this genomic
imbalance revealed that the involvement of the cancer-associated
oncogene IGH@ at 14q32.33 was critical in the leukemogenic
process (4).
Inversions (inv) within the long arm of chromosome
14 are common karyotypic abnormalities in T-cell lymphoid
malignancies, including T-chronic lymphocytic leukemia and adult
T-cell leukemia. By contrast, in B-cell-lineage ALL, inv(14)(q11q32) involving CEBP ε and IGH@
is a rare phenomenon that is associated with improved prognosis and
has been reported to be linked with complete remission (4,23,24). The positive outcome of the present
study supported this observation.
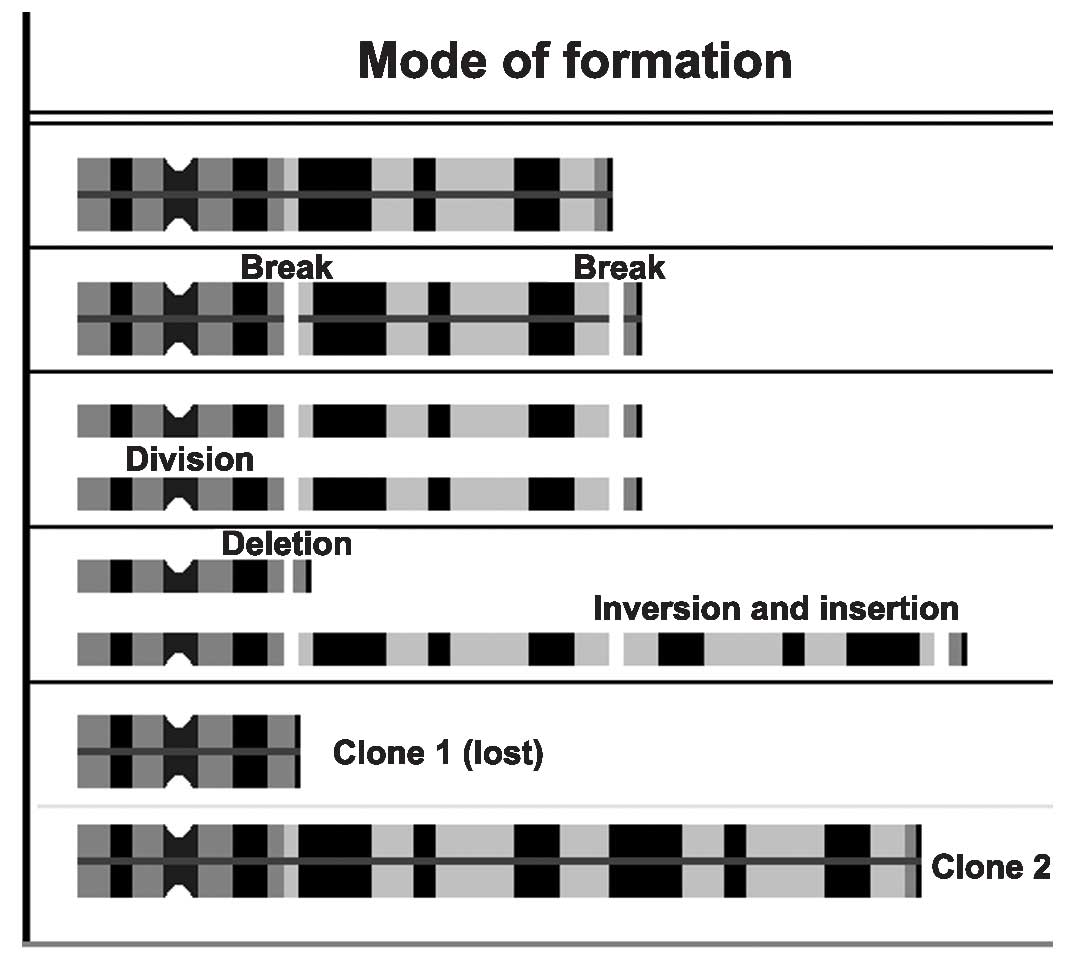
To the best of our knowledge, a derivative
chromosome 14, such as the one reported in the present study, has
not been previously observed in ALL. Fig.
2 depicts a suggestion as to how the genetic rearrangement may
have occurred. As it was a rearrangement involving an interstitial
part of the long arm of chromosome 14, U-type exchange mechanisms,
as reported in comparable cases from clinical genetics (25), may be discounted.
Homozygous deletions of the tumor suppressor genes
p16Ink4A, p15Ink4B and
p14ARF at 9p21 represent a marker of unfavorable
outcome. Thus, the heterozygote deletion observed in the present
case may be an indication of the requirement for careful follow-up
of the patient; particularly as there is evidence to suggest that
the prognosis is associated with, and depends upon, the treatment
received (9–13).
The present patient exhibited copy number changes in
five regions, the clinical significance of which remain to be
elucidated. The identification of a novel copy number change may
lead to the identification of functionally important genes in
leukemogenesis. Firstly, deletion of the TBL1XR1 gene on
3q26.32 has been detected in ETS variant 6-runt-related
transcription factor 1-positive ALL, primary central nervous system
lymphoma and diffuse B large cell lymphoma. Notably, TBL1XR1
is widely expressed in hematopoietic tissues and may have a
significant regulatory role in the nuclear factor-κB signaling
pathway, suggesting that TBL1XR1 may have a potential
biological role in ALL pathogenesis (26,27).
Secondly, ADARB2 at 10p15.3, which encodes a
member of the double-stranded RNA adenosine deaminase family of
RNA-editing enzymes, may possess a regulatory role in RNA editing,
as well as functioning as a tumor suppressor gene. Overall, a
reduction in RNA levels of ADARB2 due to a deletion may
favor cancer development and progression (28,29).
Thirdly, a recurrent deletion was identified on
21q22.22 that exclusively targets ERG. The ERG gene
is a transcription factor and a member of the ETS family. The ETS
family has a significant regulatory role in hematopoietic
differentiation during early T- and B-cell development.
Overexpression of the ERG gene was observed in acute myeloid
leukemia and T-cell ALL and was associated with poor prognosis.
Deletion of the ERG gene is considered to be associated with
a positive outcome in older children and young adults with B-cell
precursor-ALL, as was additionally observed in the present case in
the form of complete remission and no indication of MRD (30,31).
Finally, to the best of our knowledge,
submicroscopic losses of ARL2BP, PLLP and
ZNF41 genes were reported in the present study for the first
time in ALL. ARL2BP is part of the ADP ribosylation factor
family of RAS-associated GTPases, and has an essential role in
photoreceptor maintenance and function. Homozygous mutation in the
ARL2BP gene was identified in retinitis pigmentosa, with or
without situs inversus (32). In
addition, overexpression of the PLLP gene has been detected
in malignant pleural mesothelioma (33). Furthermore, mutations in ZNF41,
a transcription factor that is part of a cluster of human zinc
finger genes on chromosome Xp11.23, have been identified in
X-linked mental retardation (34).
Overall, the present study identified unbalanced
acquired gross and submicroscopic rearrangements in a case of
B-ALL, which had not been reported previously in this unique
combination, to the best of our knowledge. The clinical
consequences of the individual changes remain to be elucidated in
detail. However, it is notable that treatment according to the PALG
protocol achieved complete remission.
In conclusion, molecular cytogenetic approaches are
a useful tool for the identification of cryptic rearrangements and
potential novel target genes for prevention of leukemogenesis and
progression of disease, as well as for clinical outcome predictions
and selection of appropriate treatment options. The results of the
present study suggested that the detection of submicroscopic
alterations in B-ALL, including deletion of TBL1XR1,
CDKN2A/B and ERG genes, that are associated with
a positive outcome may be useful for diagnosis and risk
stratification, particularly for future protocols that include
B-ALL patients.
Acknowledgements
The present study was partially supported by the
German Academic Exchange Service (Bonn, Germany; grant no.,
A/10/98382).
References
|
1
|
Zhou Y, You MJ, Young KH, Lin P, Lu G,
Medeiros LJ and Bueso-Ramos CE: Advances in the molecular
pathobiology of B-lymphoblastic leukemia. Hum Pathol. 43:1347–1362.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zuckerman T and Rowe JM: Pathogenesis and
prognostication in acute lymphoblastic leukemia. F1000Prime Rep.
6:592014. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Woo JS, Alberti MO and Tirado CA:
Childhood B-acute lymphoblastic leukemia: A genetic update. Exp
Hematol Oncol. 3:162014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chapiro E, Russell LJ, Struski S, Cavé H,
Radford-Weiss I, Valle VD, Lachenaud J, Brousset P, Bernard OA,
Harrison CJ and Nguyen-Khac F: A new recurrent translocation
t(11;14)(q24;q32) involving IGH@ and miR-125b-1 in B-cell
progenitor acute lymphoblastic leukemia. Leukemia. 24:1362–1364.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Russell LJ, Enshaei A, Jones L, Erhorn A,
Masic D, Bentley H, Laczko KS, Fielding AK, Goldstone AH, Goulden
N, et al: IGH@ translocations are prevalent in teenagers and young
adults with acute lymphoblastic leukemia and are associated with a
poor outcome. J Clin Oncol. 32:1453–1462. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chapiro E, Russell L, Radford-Weiss I,
Bastard C, Lessard M, Struski S, Cave H, Fert-Ferrer S, Barin C,
Maarek O, et al: Overexpression of CEBPA resulting from the
translocation t(14;19)(q32;q13) of human precursor B acute
lymphoblastic leukemia. Blood. 108:3560–3563. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gallego M, Coccé M, Felice M, Rossi J,
Eandi S, Sciuccati G and Alonso C: A new case of t(5;14)(q31;q32)
in a pediatric acute lymphoblastic leukemia presenting with
hypereosinophilia. Atlas Genet Cytogenet Oncol Haematol.
16:183–184. 2012.
|
|
8
|
Russell LJ, De Castro DG, Griffiths M,
Telford N, Bernard O, Panzer-Grümayer R, Heidenreich O, Moorman AV
and Harrison CJ: A novel translocation, t(14;19)(q32;p13),
involving IGH@ and the cytokine receptor for erythropoietin.
Leukemia. 23:614–617. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Moorman AV, Schwab C, Ensor HM, Russell
LJ, Morrison H, Jones L, Masic D, Patel B, Rowe JM, Tallman M, et
al: IGH@ translocations, CRLF2 deregulation and microdeletions in
adolescents and adults with acute lymphoblastic leukemia. J Clin
Oncol. 30:3100–3108. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dyer MJ, Akasaka T, Capasso M, Dusanjh P,
Lee YF, Karran EL, Nagel I, Vater I, Cario G and Siebert R:
Immunoglobulin heavy chain locus chromosomal translocations in
B-cell precursor acute lymphoblastic leukemia: Rare clinical curios
or potent genetic drivers? Blood. 115:1490–1499. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Russell LJ, Akasaka T, Majid A, Sugimoto
KJ, Karran Loraine E, Nagel I, Harder L, Claviez A, Gesk S, Moorman
AV, et al: t(6;14)(p22;q32): A new recurrent IGH@ translocation
involving ID4 in B-cell precursor acute lymphoblastic leukemia
(BCP-ALL). Blood. 111:387–391. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim M, Yim SH, Cho NS, Kang SH, Ko DH, Oh
B, Kim TY, Min HJ, She CJ, Kang HJ, et al: Homozygous deletion of
CDKN2A (p16, p14) and CDKN2B (p15) genes is a poor prognostic
factor in adult but not in childhood B-lineage acute lymphoblastic
leukemia: A comparative deletion and hypermethylation study. Cancer
Genet Cytogenet. 195:59–65. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mullighan CG: The molecular genetic makeup
of acute lymphoblastic leukemia. Hematology Am Soc Hematol Educ
Program. 2012:389–396. 2012.PubMed/NCBI
|
|
14
|
Usvasalo A, Savola S, Räty R, Vettenranta
K, Harila-Saari A, Koistinen P, Savolainen ER, Elonen E,
Saarinen-Pihkala UM and Knuutila S: CDKN2A deletions in acute
lymphoblastic leukemia of adolescents and young adults: An array
CGH study. Leuk Res. 32:1228–1235. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Robak T, Szmigielska-Kapłon A,
Wrzesień-Kuś A, Wierzbowska A, Skotnicki AB, Piatkowska-Jakubas B,
Kuliczkowski K, Mazur G, Zduńczyk A, Stella-Hołowiecka B, et al:
Acute lymphoblastic leukemia in elderly: The Polish Adult Leukemia
Group (PALG) experience. Ann Hematol. 83:225–231. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Claussen U, Michel S, Mühlig P, Westermann
M, Grummt UW, Kromeyer-Hauschild K and Liehr T: Demystifying
chromosome preparation and the implications for the concept of
chromosome condensation during mitosis. Cytogenet Genome Res.
98:136–146. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shaffer LG, McGowan-Jordan J and Schmid M:
An International System for Human Cytogenetic Nomenclature (2013)
(1st). Basel: Karger Publishers. 2013.
|
|
18
|
Liehr T: Fluorescence In Situ
Hybridization (FISH) - Application Guide (1st). Berlin: Springer
Verlag Berlin Heidelberg. 2009. View Article : Google Scholar
|
|
19
|
Liehr T, Heller A, Starke H, Rubtsov N,
Trifonov V, Mrasek K, Weise A, Kuechler A and Claussen U:
Microdissection based high resolution multicolor banding for all 24
human chromosomes. Int J Mol Med. 9:335–339. 2002.PubMed/NCBI
|
|
20
|
Weise A, Heller A, Starke H, Mrasek K,
Kuechler A, Pool-Zobel BL, Claussen U and Liehr T: Multitude
multicolor chromosome banding (mMCB) - a comprehensive one-step
multicolor FISH banding method. Cytogenet Genome Res. 103:34–39.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Weise A, Mrasek K, Fickelscher I, Claussen
U, Cheung SW, Cai WW, Liehr T and Kosyakova N: Molecular definition
of high-resolution multicolor banding probes: First within the
human DNA sequence anchored FISH banding probe set. J Histochem
Cytochem. 56:487–493. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Othman MA, Melo JB, Carreira IM, Rincic M,
Alhourani E, Wilhelm K, Gruhn B, Glaser A and Liehr T: MLLT10 and
IL3 rearrangement together with a complex four-way translocation
and trisomy 4 in a patient with early T-cell precursor acute
lymphoblastic leukemia: A case report. Oncol Rep. 33:625–630.
2015.PubMed/NCBI
|
|
23
|
Han Y, Xue Y, Zhang J, Wu Y, Pan J, Wang
Y, Shen J, Dai H and Bai S: Translocation (14;14)(q11;q32) with
simultaneous involvement of the IGH and CEBPE genes in B-lineage
acute lymphoblastic leukemia. Cancer Genet Cytogenet. 187:125–129.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mitelman F, Johansson B and Mertens F:
Mitelman Database of Chromosome Aberrations and Gene Fusions in
Cancer (2014). http://cgap.nci.nih.gov/Chromosomes/MitelmanAccessed.
December. 2014
|
|
25
|
Sheth F, Ewers E, Kosyakova N, Weise A,
Sheth J, Patil S, Ziegler M and Liehr T: A neocentric isochromosome
Yp present as additional small supernumerary marker chromosome -
evidence against U-type exchange mechanism? Cytogenet Genome Res.
125:115–116. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Braggio E, McPhail ER, Macon W, Lopes MB,
Schiff D, Law M, Fink S, Sprau D, Giannini C, Dogan A, et al:
Primary central nervous system lymphomas: A validation study of
array-based comparative genomic hybridization in formalin-fixed
paraffin-embedded tumor specimens. Clin Cancer Res. 17:4245–4253.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Parker H, An Q, Barber K, Case M, Davies
T, Konn Z, Stewart A, Wright S, Griffiths M, Ross FM, et al: The
complex genomic profile of ETV6-RUNX1 positive acute lymphoblastic
leukemia highlights a recurrent deletion of TBL1XR1. Genes
Chromosomes Cancer. 47:1118–1125. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chan TH, Lin CH, Qi L, Fei J, Li Y, Yong
KJ, Liu M, Song Y, Chow RK, Ng VH, et al: A disrupted RNA editing
balance mediated by ADARs (Adenosine DeAminases that act on RNA) in
human hepatocellular carcinoma. Gut. 63:832–843. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Paz N, Levanon EY, Amariglio N, Heimberger
AB, Ram Z, Constantini S, Barbash ZS, Adamsky K, Safran M,
Hirschberg A, et al: Altered adenosine-to-inosine RNA editing in
human cancer. Genome Res. 17:1586–1595. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Clappier E, Auclerc MF, Rapion J, Bakkus
M, Caye A, Khemiri A, Giroux C, Hernandez L, Kabongo E, Savola S,
et al: An intragenic ERG deletion is a marker of an oncogenic
subtype of B-cell precursor acute lymphoblastic leukemia with a
favorable outcome despite frequent IKZF1 deletions. Leukemia.
28:70–77. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Marcucci G, Baldus CD, Ruppert AS,
Radmacher MD, Mrózek K, Whitman SP, Kolitz JE, Edwards CG, Vardiman
JW, Powell BL, et al: Overexpression of the ETS-related gene, ERG,
predicts a worse outcome in acute myeloid leukemia with normal
karyotype: A Cancer and Leukemia Group B study. J Clin Oncol.
23:9234–9242. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Davidson AE, Schwarz N, Zelinger L,
Stern-Schneider G, Shoemark A, Spitzbarth B, Gross M, Laxer U,
Sosna J, Sergouniotis PI, et al: Mutations in ARL2BP, encoding
ADP-ribosylation-factor-like 2 binding protein, cause
autosomal-recessive retinitis pigmentosa. Am J Hum Genet.
93:321–329. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pass HI, Liu Z, Wali A, Bueno R, Land S,
Lott D, Siddiq F, Lonardo F, Carbone M and Draghici S: Gene
expression profiles predict survival and progression of pleural
mesothelioma. Clin Cancer Res. 10:849–859. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Shoichet SA, Hoffmann K, Menzel C,
Trautmann U, Moser B, Hoeltzenbein M, Echenne B, Partington M, Van
Bokhoven H, Moraine C, et al: Mutations in the ZNF41 gene are
associated with cognitive deficits: Identification of a new
candidate for X-linked mental retardation. Am J Hum Genet.
73:1341–1354. 2003. View
Article : Google Scholar : PubMed/NCBI
|