Spandidos Publications style
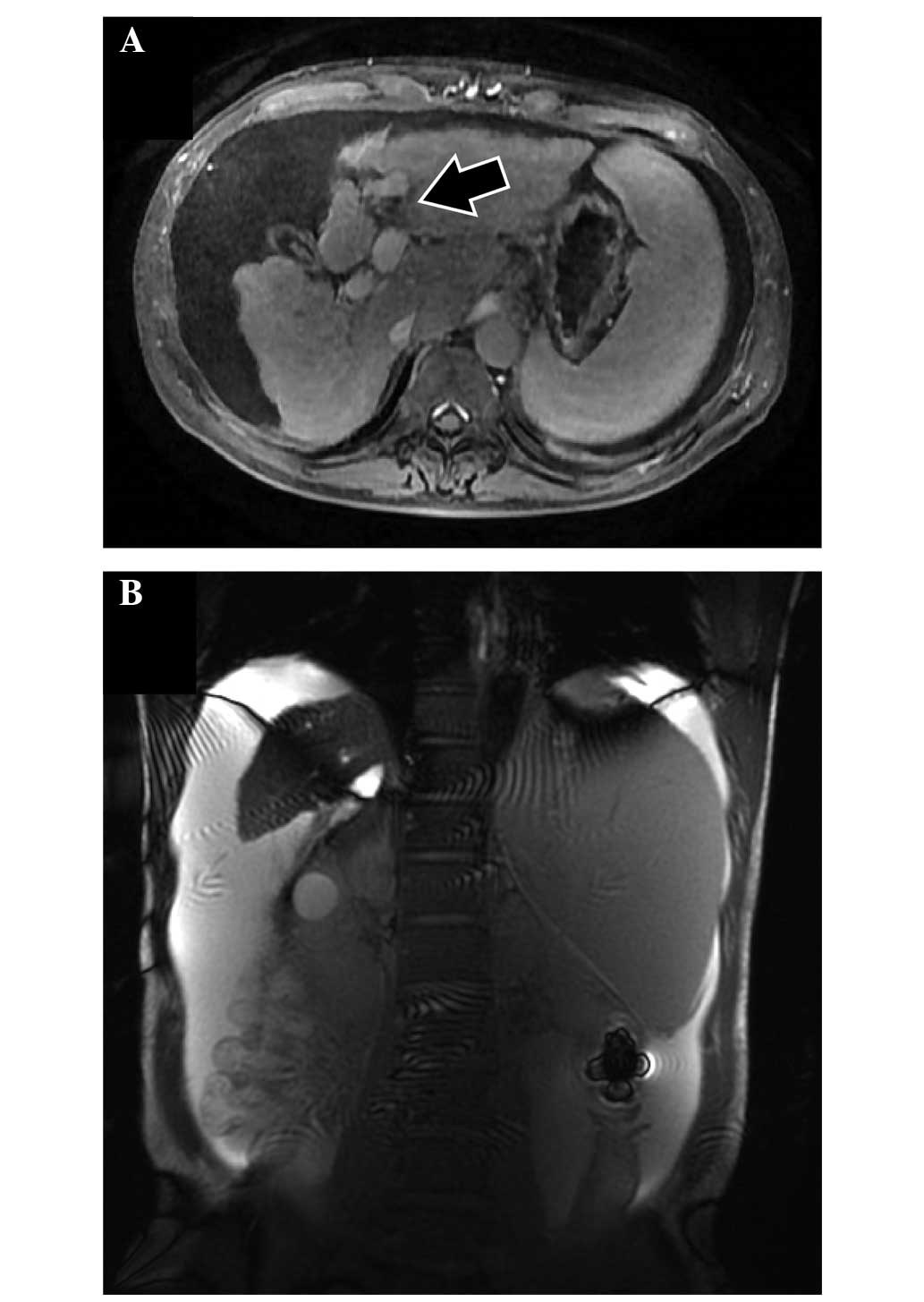
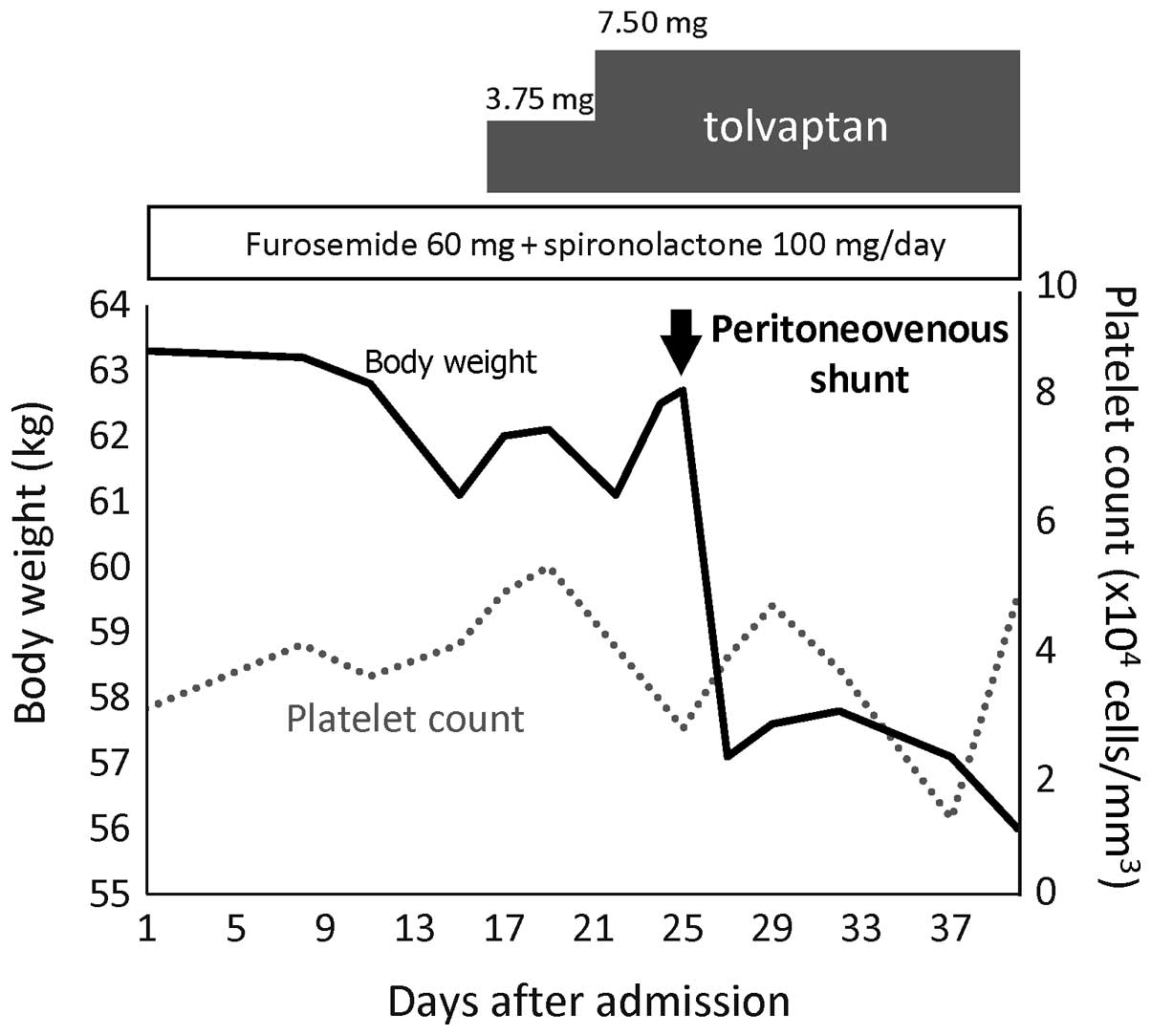
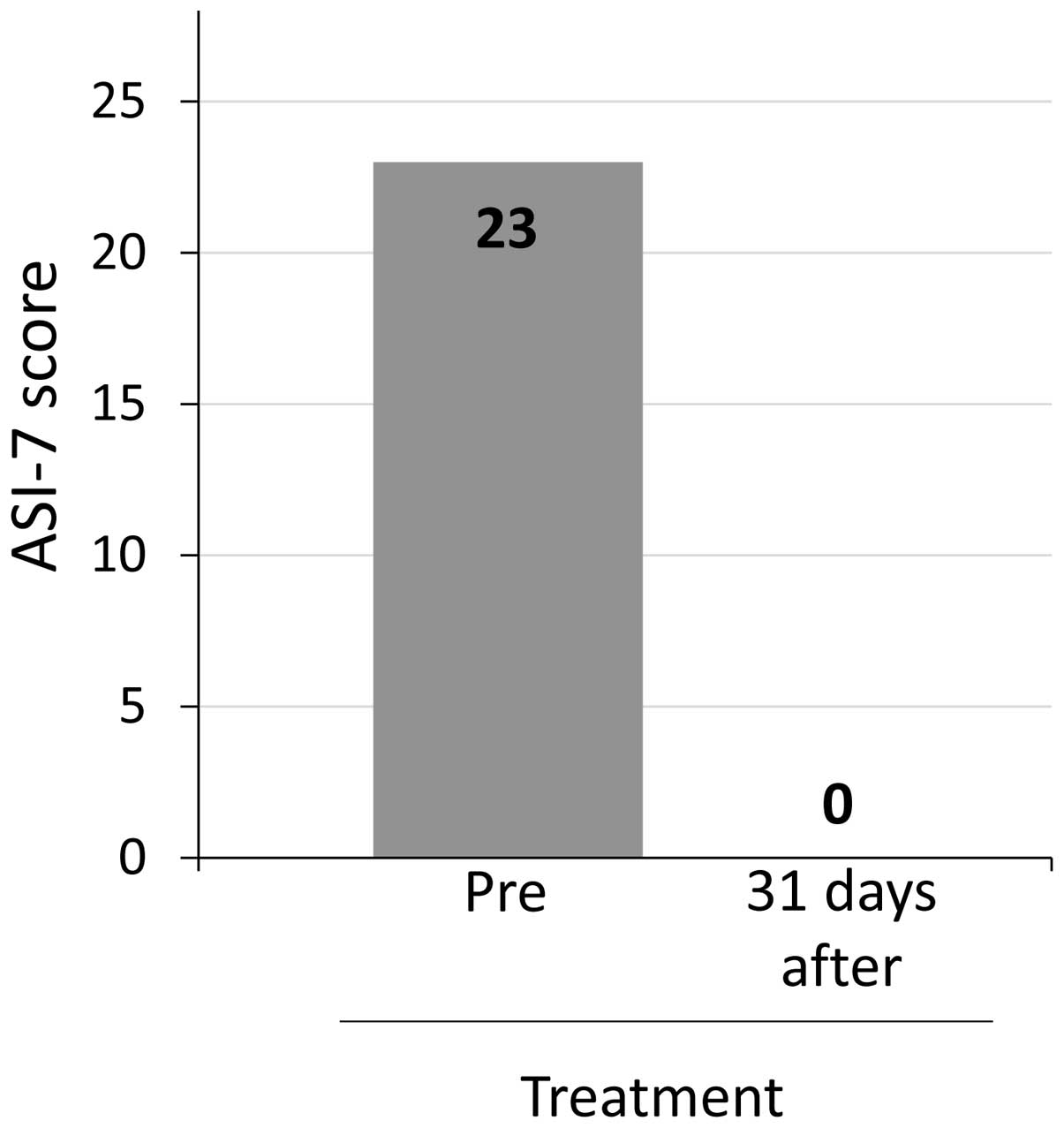
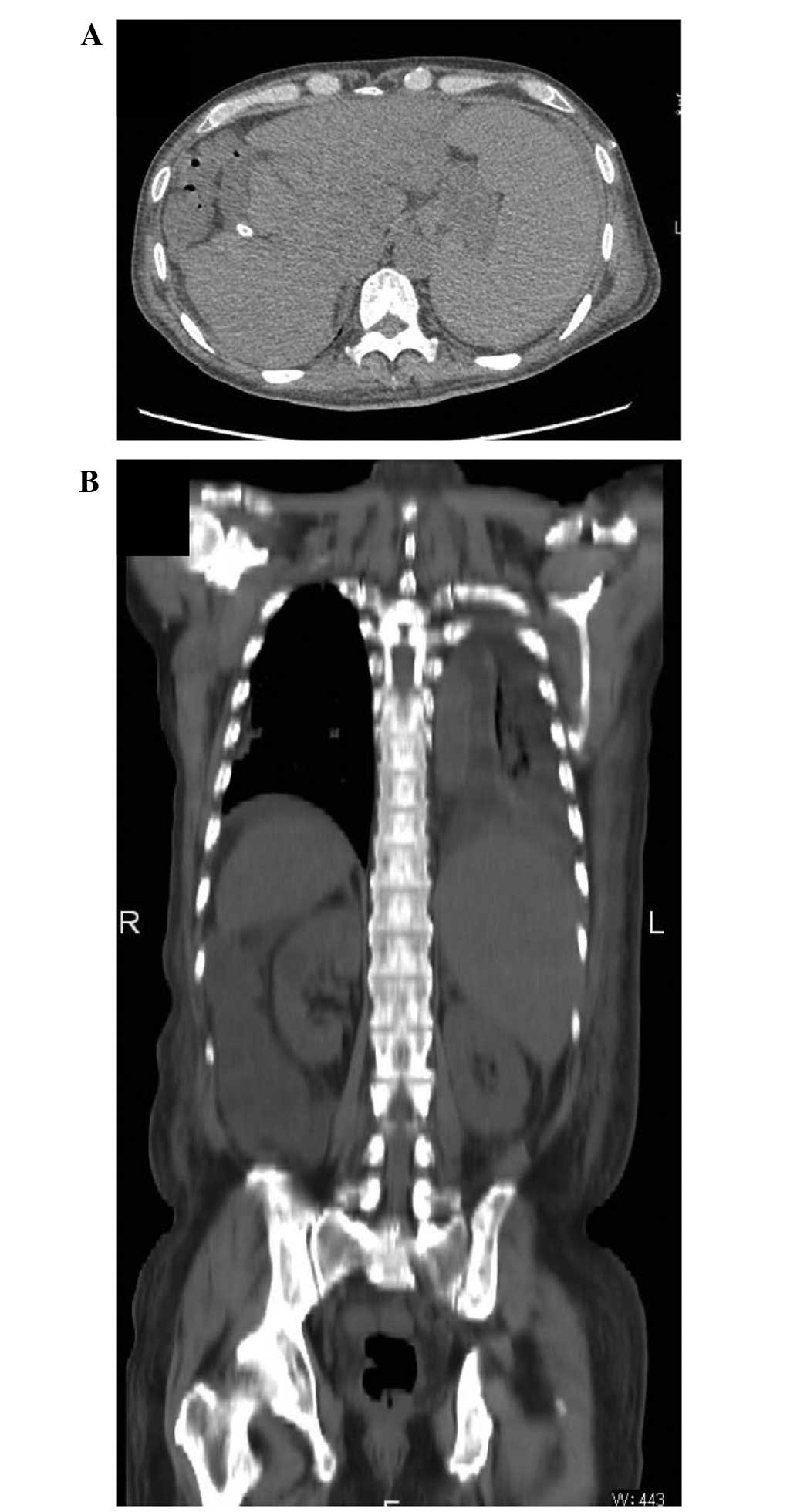
Shigeto K, Kawaguchi T, Niizeki T, Kunitake Y, Takedatsu H, Tonan T, Fujimoto K, Tanaka M, Abe T, Naito H, Naito H, et al: Efficacy of peritoneovenous shunt for treating tolvaptan-resistant refractory ascites in a cirrhotic patient with portal vein thrombosis: A case report. Oncol Lett 11: 3205-3209, 2016.
APA
Shigeto, K., Kawaguchi, T., Niizeki, T., Kunitake, Y., Takedatsu, H., Tonan, T. ... Torimura, T. (2016). Efficacy of peritoneovenous shunt for treating tolvaptan-resistant refractory ascites in a cirrhotic patient with portal vein thrombosis: A case report. Oncology Letters, 11, 3205-3209. https://doi.org/10.3892/ol.2016.4357
MLA
Shigeto, K., Kawaguchi, T., Niizeki, T., Kunitake, Y., Takedatsu, H., Tonan, T., Fujimoto, K., Tanaka, M., Abe, T., Naito, H., Torimura, T."Efficacy of peritoneovenous shunt for treating tolvaptan-resistant refractory ascites in a cirrhotic patient with portal vein thrombosis: A case report". Oncology Letters 11.5 (2016): 3205-3209.
Chicago
Shigeto, K., Kawaguchi, T., Niizeki, T., Kunitake, Y., Takedatsu, H., Tonan, T., Fujimoto, K., Tanaka, M., Abe, T., Naito, H., Torimura, T."Efficacy of peritoneovenous shunt for treating tolvaptan-resistant refractory ascites in a cirrhotic patient with portal vein thrombosis: A case report". Oncology Letters 11, no. 5 (2016): 3205-3209. https://doi.org/10.3892/ol.2016.4357