Introduction
Schwannomas are neurogenic tumors that originate
from Schwann cells, the cells of the peripheral nervous system
(1). Schwannomas have been shown to
occur in all ethnic groups and at all ages worldwide, with >90%
of the tumors being benign (2).
Schwannomas account for ~5% of benign soft-tissue neoplasms
(3). Schwannomas may develop in any
part of the body, but the most common sites include the head, neck
and flexor surfaces of the extremities (4). Generally, schwannomas have a single
place of origin, but 10% originate from multiple locations
(5). Surgery is the most common
treatment for schwannomas. Benign schwannomas rarely recur
subsequent to complete surgical resection. Long-term survival can
be expected in the majority of patients (4). Peripheral nerve sheath tumors account
for 2–6% of all gastrointestinal stromal tumors; however,
schwannomas occurring in the hepatoduodenal ligament are extremely
rare. To the best of our knowledge, only two such cases have been
reported in the literature (6,7). One
patient was female and the other patient was male, aged 62 and 29
years old, respectively (6,7). As schwannomas of the hepatoduodenal
ligament are normally asymptomatic and often discovered
incidentally, preoperative diagnosis is considerably challenging.
The two patients each accepted more than two imaging examinations
prior to treatment by laparotomy. The lesions were solitary and
found in the hepatoduodenal ligament in the two patients. Finally,
the patients were definitively diagnosed with schwannoma by
pathological examination (6,7). Following complete tumor excision,
patients with benign schwannomas generally have a good prognosis.
The present study describes the case of a hepatoduodenal ligament
schwannoma in a 50-year-old male patient, and presents a review of
the literature. To the best of our knowledge, this was the first
time laparoscopic surgery was used for the treatment of a benign
schwannoma of the hepatoduodenal ligament.
Case report
A 50-year-old male patient was referred to the
Department of General Surgery, The Third People's Hospital of
Haining (Jiaxing, Zhejiang, China), presenting with pain in the
right abdomen following trauma, on July 11, 2014. Physical
examination revealed a soft and flat abdomen, and no enlarged lymph
nodes were identified in the examinable sites. The patient's family
had no history of any specific disease; however, 3 years prior to
admission, a mass was incidentally detected in the hepatogastric
ligament of the patient by ultrasound (US; Philips IU22; Philips,
Amsterdam, Netherlands), during a health examination. As no
abnormal symptoms had presented at the time, the patient did not
receive any treatment. During the period of hospitalization in July
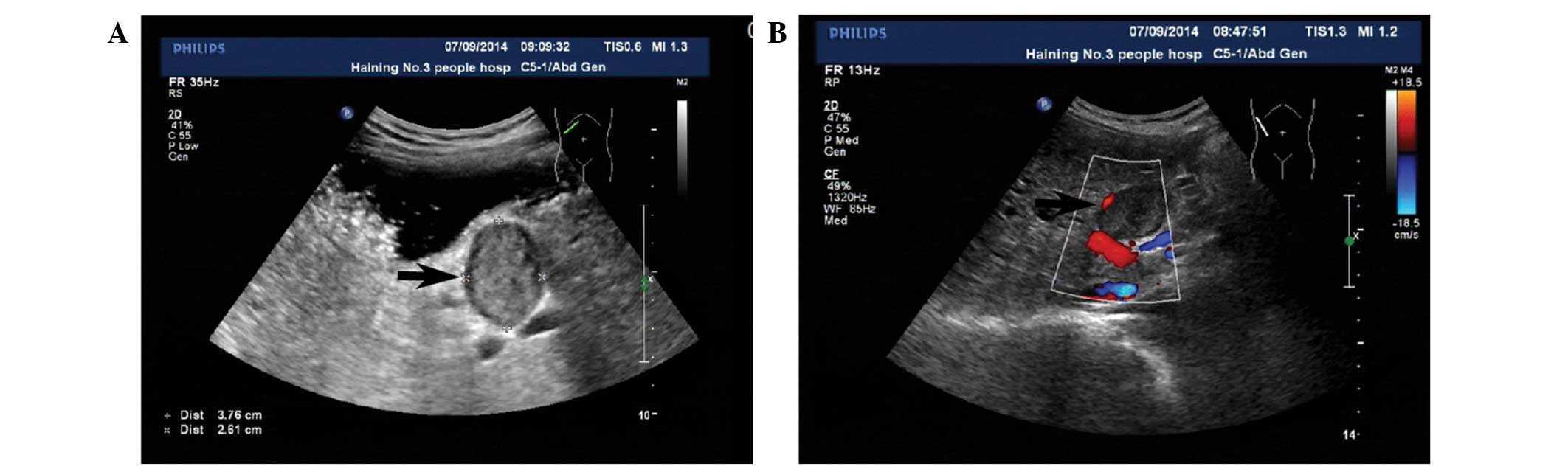
2014, an US revealed a well-defined hypodense lesion in the space
between the porta hepatis and the stomach, which measured 3.6×2.4
cm (Fig. 1A). Color Doppler US
(Philips IU22) showed no flow signals within the mass (Fig. 1B).
Subsequently, an abdominal computed tomography (CT)
scan (GE Brightspeed Elite; Hangwei Tongyong Electric Medicine
System Co., Ltd., Beijing, China) was performed to examine the
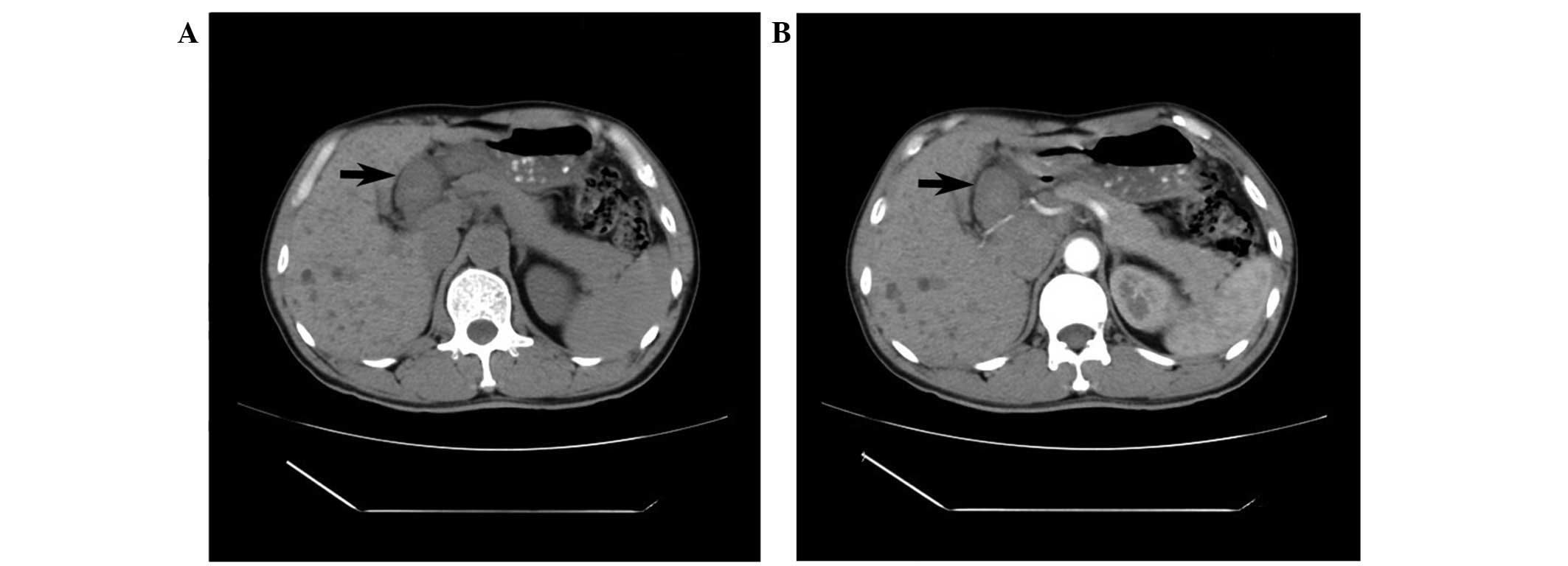
region. The non-enhanced CT scan showed a well-defined round
soft-tissue mass without internal calcification or liquefaction in
the space between the porta hepatis and the stomach. The mass
measured 3.8×3.0×2.5 cm, had a uniform density and was surrounded
by a clear tunica (Fig. 2A). On the
contrast-enhanced CT scan, the mass showed no evident enhancement
in the arterial phase (Fig. 2B).
Based on the findings of the CT scan, the diagnosis of gastric
stromal tumor was primarily considered. However, since the mass had
been growing slowly for 3 years and possessed a clear tunica, the
mass was then considered to be a benign tumor, which may be
removed.
Laboratory results were as follows: Red blood cells,
4.38×1012/l (normal values,
4.30–5.80×1012/l); white blood cells,
5.60×109/l (normal values, 4.00–10.00×109/l);
hemoglobin, 131 g/l (normal values, 130–175 g/l); lactate
dehydrogenase, 146 international units (IU)/l (normal values,
80–240 IU/l); total protein, 58.10 g/l (normal values, 65.00–85.00
g/l); total bilirubin, 10.80 µmol/l (normal values, 0.00–21.00
µmol/l); conjugated bilirubin, 5.70 µmol/l (normal values,
0.00–10.00 µmol/l); bile acids, 2.40 µmol/l (normal values,
0.00–12.00 µmol/l); aspartate aminotransferase, 18 IU/l (normal
values, 15–45 IU/l); alanine transaminase, 16 IU/l (normal values,
9–60 IU/l); γ-glutamyl transpeptidase, 34 U/l (normal values, 10–60
IU/l); alkaline phosphatase, 67 U/l (normal values, 45–125 IU/l);
amylase, 77 U/l (normal values, <100 IU/l); antimitochondrial
antibody, negative; α-fetoprotein, <20 ng/ml (normal values,
<20 ng/ml); carcinoembryonic antigen, 0.67 ng/ml (normal values,
<5.50 ng/ml); carbohydrate antigen 19–9, 4.93 U/ml (normal
values, <37.00 U/ml); cancer antigen 125, <4.00 U/ml (normal
values, <35.00 U/ml).
Subsequent to obtaining patient consent,
laparoscopic surgery was performed (Laparoscope operating system,
22202011V110; Karl Storz GmbH & Co. KG, Tuttlingen, Germany).
The patient was placed in a horizontal position. Following the
successful induction of anesthesia, the skin of the surgery field
was conventionally sterilized, and sterile drapes were whisked onto
the patient's body. Subsequently, a ~2.0 cm long incision was made
in the superior border of the umbilicus. A CO2
pneumoperitoneum was set up with 15 mmHg intra-abdominal pressure
using a veress needle (26120J; Karl Storz GmbH & Co. KG,
Tuttlingen, Germany), and a 1.1 cm Trocar puncture (30103MP; Karl
Storz GmbH & Co. KG) was made to insert a laparoscopic lens
(26003BA; Karl Storz GmbH & Co. KG). Under direct vision, 3
holes were made beneath the xiphoid process and left and right
upper abdomen, and the ultrasonic scalpel (GEN04; Johnson &
Johnson Medical (China) Ltd., Shanghai, China), dissecting forceps
(33321ML; Karl Storz GmbH & Co. KG), scratch-free grasping
forceps (33321R; Karl Storz GmbH & Co. KG) were inserted.
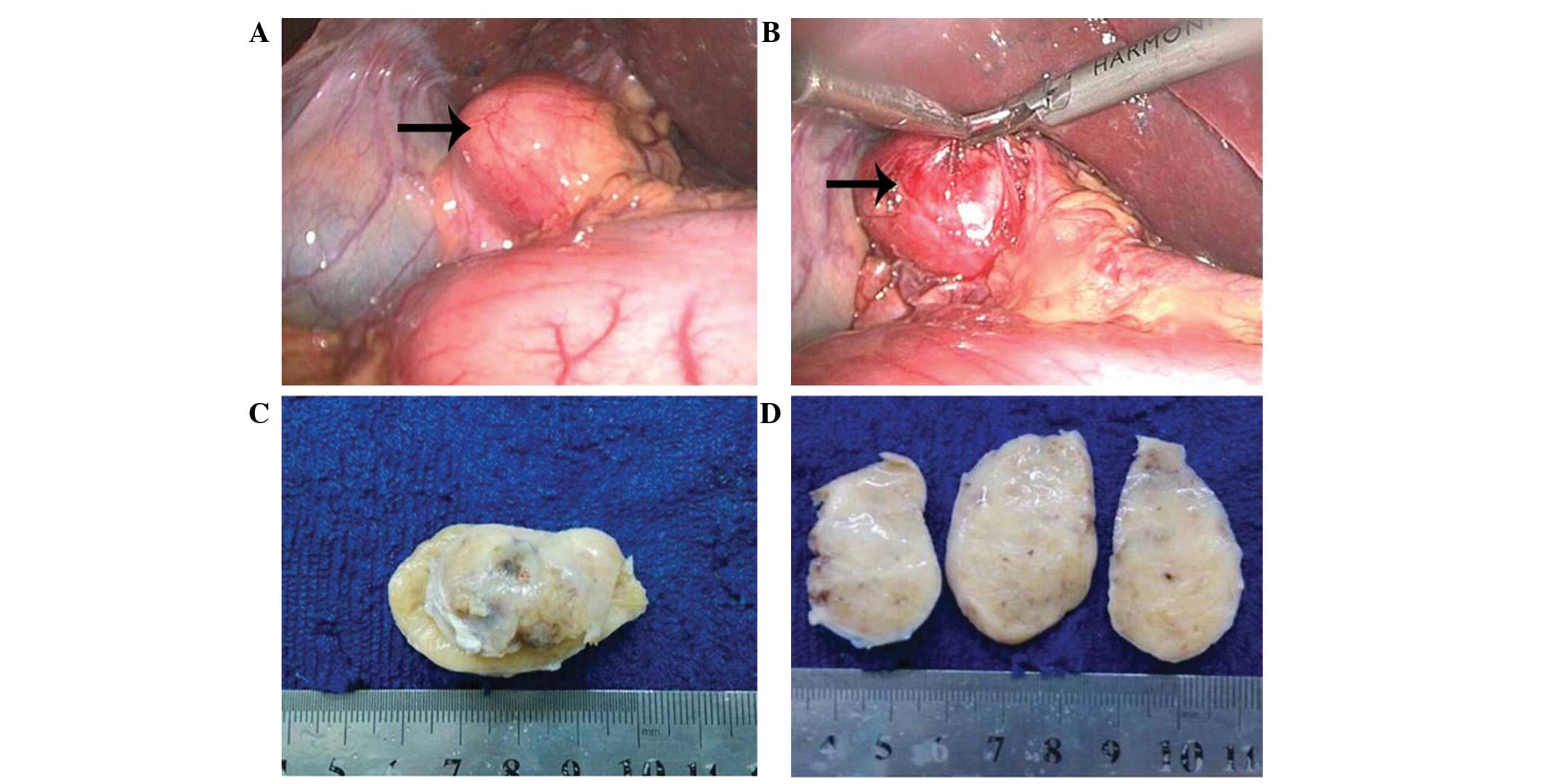
During laparoscopic exploration, a mass surrounded by a fibrous
capsule was located in the hepatoduodenal ligament, medial to the
gallbladder and near the upper antrum (Fig. 3A and B). No invasion of the
surrounding tissue was observed and the biliary ducts were not
dilated. The mass was completely resected. The capsule was then
removed and the gross specimen was described as a 4.5×2.5×2.5-cm
sized localized mass, yellowish-white in color (Fig. 3C and D). Following postoperative
pathological analysis of an intraoperative frozen section, the mass
was diagnosed as a schwannoma. Postoperatively, the patient had a
smooth recovery and left the hospital 3 days later.
The tissue section was analyzed at the Department of
Pathology, Zhejiang Provincial People's Hospital (Hangzhou,
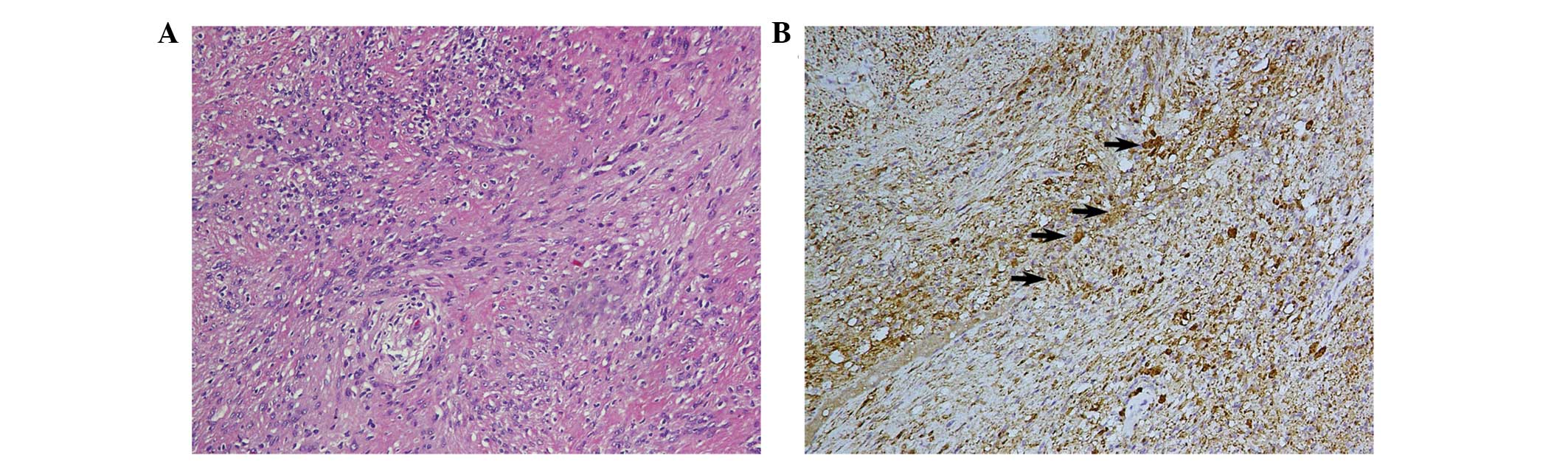
Zhejiang, China). Microscopically, the tumor mainly consisted of
spindle-shaped cells, and no atypical cells or signs of malignancy
were observed (Fig. 4A).
Immunohistochemical analysis showed that the tumor cells were
vimentin- and S-100 protein-positive (Fig. 4B), and epithelial membrane antigen-
and cluster of differentiation 34-negative. The final diagnosis of
the tumor was schwannoma. The patient was followed-up for 7 months
and, at the time of writing, was healthy and without any
complications.
The present study was approved by the Ethics
Committee of the Third People's Hospital of Haining (Jiaxing,
Zhejiang, China).
Discussion
Schwannomas originate from the sheaths of the
peripheral nerves and have been shown to occur in all ethnic groups
worldwide (2). Secondary degenerative
changes of schwannomas, including cyst formation, calcification,
hemorrhage, and hyalinization, sometimes appear (8). Schwannomas occur in most parts of the
body, but the head, neck and flexor surfaces of the extremities are
the most common sites (1). They
rarely occur in the retroperitoneum, accounting for 6% of all
primary retroperitoneal tumors (9,10). The
occurrence of schwannomas in the gallbladder (11), gastroduodenal ligament (12), pancreas (13), bowel mesentery (14) or colon (15) is extremely rare. To the best of our
knowledge, only two cases of schwannoma involving the
hepatoduodenal ligament have been reported (6,7). The
clinical characteristics of the aforementioned cases are listed in
Table I. These tumors are usually
asymptomatic and often incidentally discovered. Due to their low
incidence and, therefore, lack of adequate understanding of the
mechanism of this type of tumor, forming a preoperative diagnosis
is challenging.
 | Table I.Clinical characteristics of 3 patients
with benign schwannomas in the hepatoduodenal ligament. |
Table I.
Clinical characteristics of 3 patients
with benign schwannomas in the hepatoduodenal ligament.
| Author, year | Gender | Age, years | Symptoms | Imaging
technique | Location | Number | Size, cm | Preoperative
diagnosis | Treatment | Follow-up,
months | Status | Ref. |
|---|
| Nagafuchi et
al, 1988 | F | 62 | Asymp-tomatic | US, CT, CA MRI,
ERC | Hepato-duodenal
ligament | Solitary | 9×5×4.5 | NA | Laparotomy | 26 | Survived | (14) |
| Pinto et al,
2011 | M | 29 | Asymp-tomatic | US, MRI endoscopy,
biopsy | Hepato-duodenal
ligament | Solitary | 4.5×2.9 | Spindle cell
neoplasia or stromal tumor | Laparotomy | NA | NA | (15) |
| Present case | M | 50 | Right abdominal
pain | US, CT, CECT | Hepato-duodenal
ligament | Solitary | 4.5×2.5×2.5 | Stromal tumor | Laparoscopic
surgery | 7 | Survived |
|
Prior to treatment, comprehensive imaging
modalities, including US, CT and magnetic resonance imaging (MRI),
should be used to establish a probable diagnosis and determine the
lesion limits; however, these imaging techniques rarely provide a
definitive diagnosis, due to a lack of distinguishing features of
these tumors on imaging scans. Generally, schwannomas manifest as
well-defined, hypoattenuating masses on non-enhanced CT scans, and
on contrast-enhanced CT scans show peripheral enhancement with an
irregular pattern. Delayed peripheral enhancement until the late
venous phase reflects a fibrous capsule and an internal fibrillary
element on CT scan (11). MRI
comprises another useful tool for determining the nature of the
tumor. On MRI scans, schwannomas are usually presented as masses
with a low signal intensity on T1-weighted images, and
inhomogeneous high signal intensity on T2-weighted images (16).
Pathology is important for a definitive diagnosis.
Histologically, schwannomas consist of compact cellular lesions.
Microscopically, schwannomas are encapsulated tumors with a
biphasic growth pattern consisting of Antoni A (interlacing and
cellular fascicles) and Antoni B (less cellular and myxoid)
regions. In addition, typical Verocay bodies in the Antoni A areas
may be detected using hematoxylin and eosin staining. In
immunohistochemistry, positive uniform S-100 staining may be
detected, which is an indicator of schwannoma (17,18). In
the present case, the histological findings were predominantly
cellular Antoni A areas and immunohistochemistry showed a strong
positive staining of the tumor cells for S-100 protein, which
supported the benign nature of the schwannoma in the present
case.
In conclusion, surgical intervention may be the
optimal treatment for schwannomas, as surgery may be used to
determine the location and nature of the lesion and to successfully
treat it. In the present case, a large mass had developed; however,
no resultant symptoms were present prior to trauma. Definitively
determining the nature of the tumor preoperatively was challenging,
despite the use of imaging modalities. To the best of our
knowledge, this was the first case of benign schwannoma of the
hepatoduodenal ligament treated by laparoscopic surgery. Following
complete tumor excision, patients with benign schwannomas generally
have a good prognosis.
Acknowledgements
The authors would like to thank Dr Chen Yuan
(Department of Pathology, Zhejiang Provincial People's Hospital for
proofreading the pathological methods.
References
|
1
|
Das Gupta TK and Brasfield RD: Tumors of
peripheral nerve origin: Benign and malignant solitary schwannomas.
CA Cancer J Clin. 20:228–233. 1970. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ariel IM: Tumors of the peripheral nervous
system. CA Cancer J Clin. 33:282–299. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Pilavaki M, Chourmouzi D, Kiziridou A,
Skordalaki A, Zarampoukas T and Drevelengas A: Imaging of
peripheral nerve sheath tumors with pathologic correlation:
Pictorial review. Eur J Radiol. 52:229–239. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Le Guellec S: Nerve sheath tumours. Ann
Pathol. 35:54–70. 2015.(In French). View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fenoglio L, Severini S, Cena P, Migliore
E, Bracco C, Pomero F, Panzone S, Cavallero GB, Silvestri A, Brizio
R and Borghi F: Common bile duct schwannoma: A case report and
review of literature. World J Gastroenterol. 13:1275–1278. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Honjo Y, Kobayashi Y, Nakamura T, Takehira
Y, Kitagawa M, Ikematsu Y, Ozawa T and Nakamura H: Extrahepatic
biliary schwannoma. Dig Dis Sci. 48:2221–2226. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lane RH, Stephens DH and Reiman HM:
Primary retroperitoneal neoplasms: CT findings in 90 cases with
clinical and pathologic correlation. AJR Am J Roentgenol.
152:83–89. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li ZQ, Wang HY, Li J and Teng L: Recurrent
retroperitoneal Schwannomas displaying different differentiation
from primary tumor: Case report and literature review. World J Surg
Oncol. 8:662010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu LN, Xu HX, Zheng SG, Sun LP, Guo LH
and Wu J: Solitary schwannoma of the gallbladder: A case report and
literature review. World J Gastroenterol. 20:6685–6690. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bayraktutan U, Kantarci M, Ozgokce M,
Aydinli B, Atamanalp SS and Sipal S: Education and Imaging.
Gastrointestinal: Benign cystic schwannoma localized in the
gastroduodenal ligament; a rare case. J Gastroenterol Hepatol.
27:9852012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ciledag N, Arda K and Aksoy M: Pancreatic
schwannoma: A case report and review of the literature. Oncol Lett.
8:2741–2743. 2014.PubMed/NCBI
|
|
12
|
Tang SX, Sun YH, Zhou XR and Wang J: Bowel
mesentery (meso-appendix) microcystic/reticular schwannoma: Case
report and literature review. World J Gastroenterol. 20:1371–1376.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bugiantella W, Rondelli F, Mariani L,
Peppoloni L, Cristallini E and Mariani E: Schwannoma of the colon:
A case report. Oncol Lett. 8:2511–2512. 2014.PubMed/NCBI
|
|
14
|
Nagafuchi Y, Mitsuo H, Takeda S, Ohsato K,
Tsuneyoshi M and Enjoji M: Benign schwannoma in the hepatoduodenal
ligament: Report of a case. Surg Today. 23:68–72. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Pinto J, Afonso M, Veloso R, Tente D,
Fernandes S, Proença L, Carvalho J, Pontes JM and Fraga J: Benign
schwannoma of the hepatoduodenal ligament. Endoscopy. 43(Suppl 2):
E195–E196. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rha SE, Byun JY, Jung SE, Chun HJ, Lee HG
and Lee JM: Neurogenic tumors in the abdomen: Tumor types and
imaging characteristics. Radiographics. 23:29–43. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Das Gupta TK, Brasfield RD, Strong EW and
Hajdu SI: Benign solitary Schwannomas (neurilemomas). Cancer.
24:355–366. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Weiss SW, Langloss JM and Enzinger FM:
Value of S-100 protein in the diagnosis of soft tissue tumors with
particular reference to benign and malignant Schwann cell tumors.
Lab Invest. 49:299–308. 1983.PubMed/NCBI
|