Introduction
The Wnt/β-catenin signaling pathway is important in
embryogenesis and tumor development, and has been recognized as one
of the key signaling pathways during tumorigenesis (1,2). The
aberrant activation of the Wnt/β-catenin signaling pathway may
increase cell proliferation, which results in the generation of
abnormal proteins, thereby accelerating the formation of tumors and
carcinogenesis through the expression of downstream target genes
(3). A previous study demonstrated
that ~90% of cases of colon cancer were associated with excessive
activation of this pathway (4).
β-catenin is the key molecule of the Wnt/β-catenin signaling
pathway (5). Thus, silencing the
β-catenin gene may inhibit the development and invasion of colon
cancer. However, data regarding how to influence colon cancer cells
by silencing the β-catenin gene are limited. In the present study,
the expression of the β-catenin gene in the Wnt/β-catenin signaling
pathway was silenced by small interference (si)RNA to study its
effect on colon cancer cell proliferation, apoptosis and invasion,
and to explore the clinical value of blocking β-catenin gene
expression as a therapeutic molecular target.
Materials and methods
Materials
The human colon cancer cell line SW480 was purchased
from the China Center for Type Culture Collection of Wuhan
University (Wuhan, China). The liposomal transfection agent
Lipofectamine® 2000 was purchased from Invitrogen (Thermo Fisher
Scientific, Inc., Waltham, MA, USA). Reverse
transcription-polymerase chain reaction (RT-PCR) kit was purchased
from Fermentas (Thermo Fisher Scientific, Inc.). Matrigel was
purchased from BD Biosciences (Franklin Lakes, NJ, USA), while
Transwell chambers were purchased from Santa Cruz Biotechnology,
Inc. (Dallas, TX, USA).
Synthesis of β-catenin-siRNA
siRNA targeting the β-catenin gene was synthesized
by Shanghai GenePharma Co., Ltd. (Shanghai, China), and its
sequence was: Sense, 5′-CATGUGUTGGUAAGCUCUA-3′ and anti-sense,
5′-GCAACAGTTGCAGAGAGGU-3′. Upon synthesis, Basic Local Alignment
Search Tool analysis was performed in GeneBank (blast.ncbi.nlm.nih.gov/Blast.cgi)
excluding other homologous genes. The negative control siRNA used
in the present study was purchased from Shanghai GenePharma, Co.,
Ltd., and the sequence was as follows: Sense,
5′-AUGCUGATCAGUGUCGATU-3′ and anti-sense,
5′-CAGAGAGCTCGUGAGAGTA-3′.
Transfection of the human colon cancer
cell line SW480
SW480 cells were transfected with the Lipofectamine®
2000 transfection reagent, according to the manufacturer's
protocol. The human colon cancer cell line SW480 was conventionally
cultured in Dulbecco's modified Eagle's medium containing 10% fetal
bovine serum (FBS) (Shanghai Solarbio Bioscience & Technology
Co., Ltd., Shanghai, China) at 37°C in a 5% CO2 humidified
incubator. Cells in the logarithmic growth phase were selected for
transfection. Samples were divided into the following four groups:
i) Blank control group (non-transfected cells); ii) negative
control group (cells transfected with negative control-siRNA); iii)
liposome group (cells transfected only with the aforementioned
liposomal transfection agent); and iv) transfected group (cells
transfected with β-catenin-siRNA).
RT-PCR assay for the detection of
siRNA efficiency on β-catenin gene expression
Total RNA was extracted using TRIzol (Shanghai
GenePharma, Co., Ltd.), and next subjected to RT reaction. RT-PCR
experiments were performed using the AccessQuick RT-PCR System
(Promega Corporation, Madison, WI, USA) using 1 µg total RNA. The
primers were purchased from Shanghai GenePharma, Co., Ltd. and the
sequences were as follows: β-catenin, forward
5′-AAAGCGGCTGTTAGTCACTGG-3′ and reverse
5′-GACTTGGGAGGTATCCACATCC-3′; β-actin, forward
5′-GAAATCGTGCGTGACATTA-3′ and reverse 5′-ACTCATCGTACTCCTGCTTG-3′.
The expected lengths of the amplified products for β-catenin and
β-actin were 132 and 475 bp, respectively. The PCR was performed on
a thermal cycler (Mastercycler Nexus; Eppendorf China, Ltd.,
Shanghai, China) under the following conditions: 94°C for 2 min,
followed by incubation at 94°C for 45 sec, 55°C for 45 sec and 72°C
for 45 sec, for a total of 32 cycles, and a final step at 72°C for
5 min. The results were semiquantitatively analyzed using 3%
agarose gel electrophoresis (Submarine Electrophoresis Unit;
Hoefer™; Thermo Fisher Scientific, Inc.) and visualized using a
digital gel documentation system (GelDoc 2000 System/QuantityOne
software; Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Western blotting assay for the
detection of siRNA efficiency on β-catenin protein expression
Four groups of SW480 cells (Blank control; negative
control; liposome; transfected) were washed with phosphate-buffered
saline (PBS) and lysed with lysis buffer (150 mmol/l NaCl, 50
mmol/l Tris, 1% sodium deoxycholate, 0.1% sodium dodecylsulfate, 1%
Triton X-100, 5 mmol/l ethylene diaminetetra acetate; Shanghai
Solarbio Bioscience & Technology Co., Ltd.) for 20 min on ice
and then centrifuged (4,500 × g for 10 min). Protein levels were
determined using the Pierce BCA Protein Assay (Pierce™; Thermo
Fisher Scientific, Inc.), according to the manufacturer's protocol.
Next, equal aliquots of the cell lysates were run on 10% sodium
dodecyl sulfate-polyacrylamide gel and then electrotransferred onto
polyvinylidene fluoride membranes (Shanghai Solarbio Bioscience
& Technology Co., Ltd.), which were blocked with 5% skimmed
milk powder dissolved in Tris-buffered saline with Tween® 20 for 2
h at 4°C. The membranes were incubated with polyclonal rabbit
anti-β-catenin (catalog no. sc-7199) and mouse monoclonal
anti-β-actin (catalog no. sc-47778) antibodies (dilution, 1:500;
Santa Cruz Biotechnology Inc., Dallas, TX, USA) overnight at 4°C,
then with a secondary goat anti-rabbit IgG antibody (catalog no.
G21079; 1:1,000; Invitrogen™; Thermo Fisher Scientific, Inc.) for 2
h at room temperature. Chemiluminescence (Shanghai Solarbio
Bioscience & Technology Co., Ltd.) and optical density (OD)
analysis were used to detect the expression levels of β-catenin.
β-actin served as an internal reference.
Effect of siRNA on cell proliferation
by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
(MTT) assay
SW480 cells on logarithmic growth phase were
trypsinized (Shanghai Solarbio Bioscience & Technology Co.,
Ltd.) and seeded in 96-well plates (Shanghai Solarbio Bioscience
& Technology Co., Ltd.) at a density of 5×103
cells/ml. Upon 24-h incubation, cells were transfected for 24, 48,
72 or 96 h. Next, 200 µl MTT solution was added, and incubation
continued for additional 4 h. Subsequently, the cell supernatant
was discarded, and 100 µl dimethyl sulfoxide (Shanghai Solarbio
Bioscience & Technology Co., Ltd.) was added. Plates were
gently rocked at room temperature for 15 min, and the OD at 570 nm
was measured with a microplate reader (ELx800; BioTek, Winooski,
VT, USA). A growth curve was generated by plotting the incubation
time in the horizontal axis and the OD values is the vertical
axis.
Flow cytometry for the detection of
cell apoptosis
Apoptosis was detected using Annexin V/propidium
iodide double staining (Shanghai Solarbio Bioscience &
Technology Co., Ltd.). Cells in each group were collected, digested
and resuspended in PBS. Flow cytometry (BD FACSCalibur™; BD
Biosciences) was used to detect cell apoptosis, according the
protocol provided by the manufacturer of the kit. In the flow
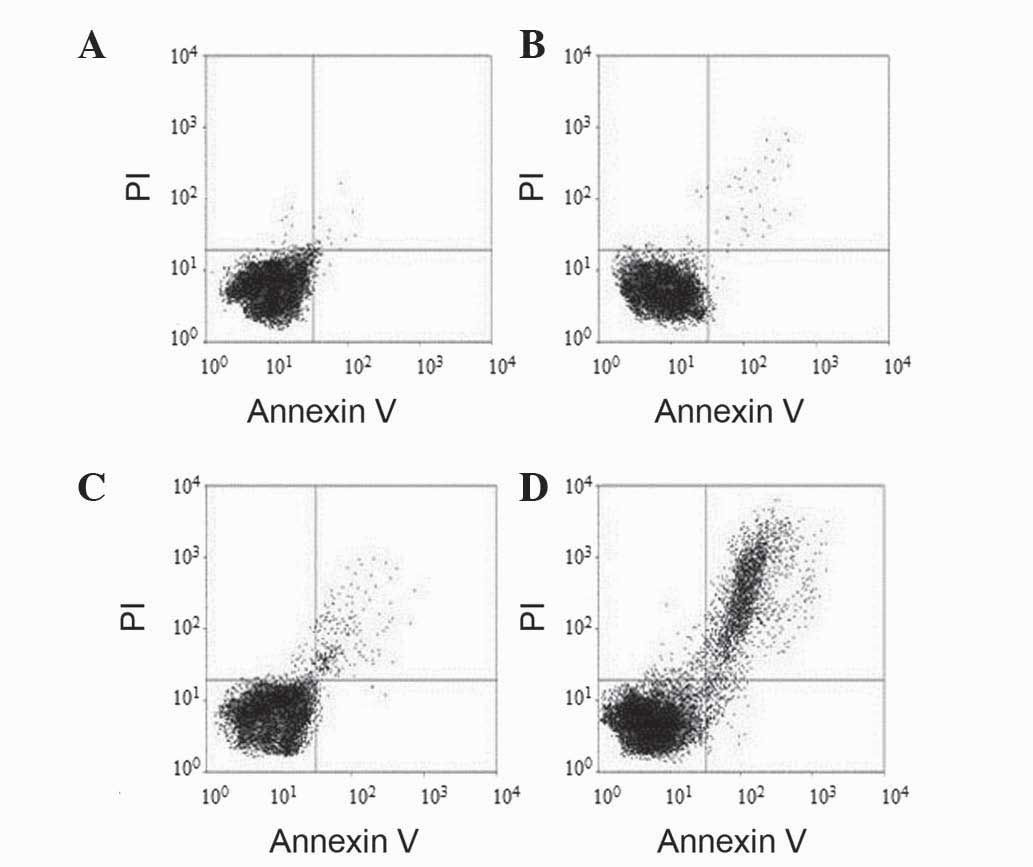
cytometry plots, the lower left quadrant represented cell debris,
while the lower and upper right quadrant represented early and late
apoptotic cells, respectively. The upper left quadrant corresponded
to dead cells. The apoptotic rate for each group was calculated as
the average of three independent measurements, according to the
following formula: Apoptosis rate (%) = (number of apoptotic
cells/number of total cells) × 100.
Matrigel transmembrane invasion assay
for the detection of cell invasiveness in siRNA-transfected SW480
cells
The Matrigel transmembrane invasion assay was
performed in a Transwell plate (Corning, Inc., Corning, NY, USA).
Complete medium was added to the upper chamber of Transwell plate
pretreated with Matrigel (BD Biosciences), and complete medium
containing 10% FBS was added to the lower chamber, following
incubation for 1 h at 37°C. The experimental and control groups
were suspended at a cell density of 1×105 cells/ml, and
200 µl of these suspensions were transferred to the upper chamber,
followed by incubation for 24 h. Subsequently, cells on the filter
membranes were harvested, and the number of cells was counted under
a microscope (IX71; Olympus Corporation, Tokyo, Japan).
Caspase-3 activity assay to detect the
influence of siRNA on the caspase-3 activity in SW480 cells
Caspase-3 activity was measured using a kit
(Shanghai GenePharma Co., Ltd.) that contained caspase-3
sequence-specific peptides conjugated to a chromophore. When the
substrate was cleaved by caspase-3, the chromophoric group was
released, which could be monitored by measuring the OD of the
sample at a wavelength of 405 nm on a microplate reader, thus
obtaining the relative activity of caspase-3. The experiments were
conducted according to the manufacturer's protocol.
Statistical analysis
Statistical analysis was performed using SPSS
version 17.0 software (SPSS Inc., Chicago, IL, USA). Data were
expressed as the mean ± standard deviation. Student's t-test
and one-way analysis of variance were used to compare the means
across different groups. P<0.05 was considered to indicate a
statistically significant difference.
Results
β-catenin-siRNA inhibits β-catenin
messenger (m)RNA expression
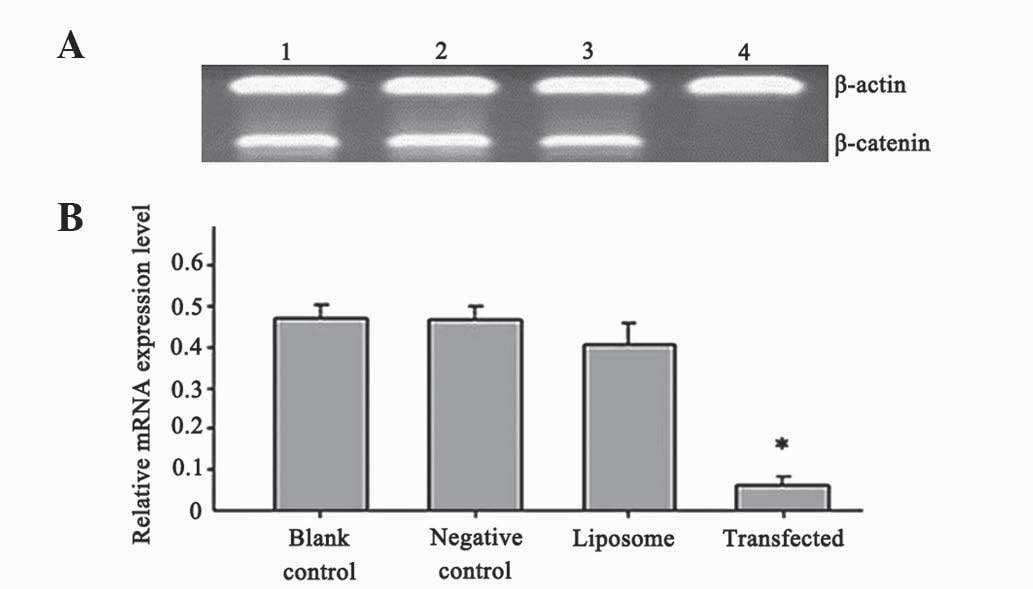
The results presented in Fig. 1 indicate that, following
β-catenin-siRNA transfection for 48 h, the relative mRNA expression
levels of β-catenin in β-catenin-siRNA transfected SW480 cells were
0.044±0.003, while in the blank control, negative control and
liposome groups, the relative mRNA expression levels of β-catenin
were 0.465±0.063, 0.468±0.037 and 0.417±0.066, respectively. These
results indicate that the expression of β-catenin was significantly
reduced in the β-catenin-siRNA experimental group (P=0.004),
compared with the blank control, negative control and liposome
groups. No significant differences were observed among the blank
control, negative control and liposome groups regarding the mRNA
expression levels of β-catenin (P=0.611). Therefore,
β-catenin-siRNA effectively inhibited the expression of the
β-catenin gene in SW480 cells.
β-catenin siRNA inhibits the protein
expression of β-catenin
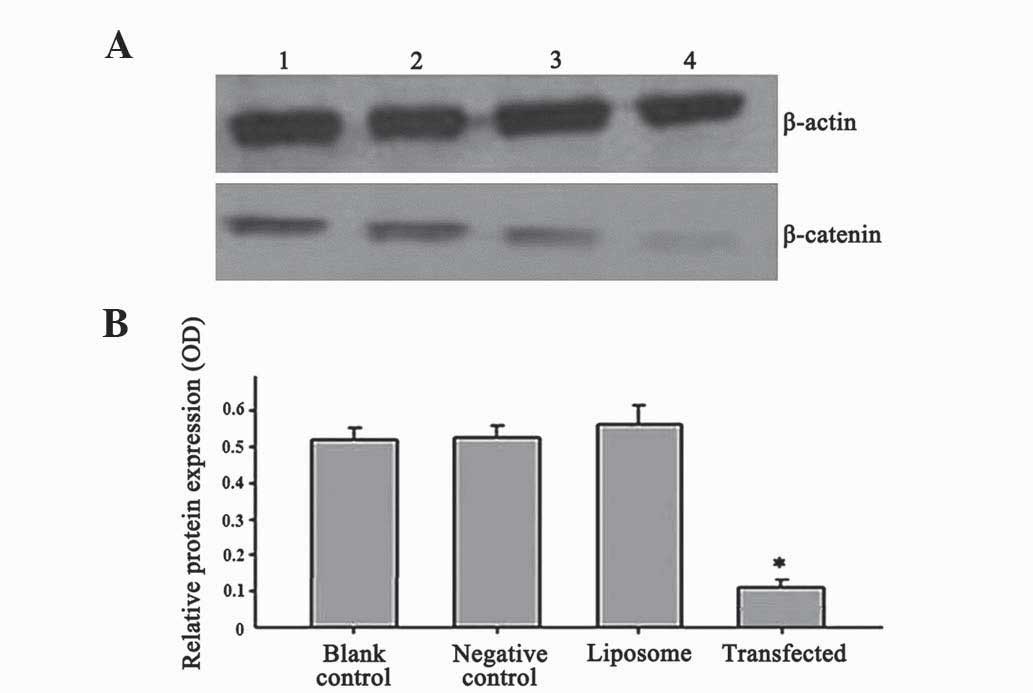
The results represented in Fig. 2 reveal that, following β-catenin-siRNA
transfection for 48 h, the OD value of β-catenin protein expression
in SW480 cells was 0.102±0.005, while in the blank control,
negative control and liposome groups the OD values were
0.512±0.035, 0.519±0.047 and 0.566±0.036, respectively. Thus, in
the transfected group, the protein expression levels of β-catenin
were significantly lower than in the other groups (P=0.014). No
significant differences were observed among the blank control,
negative control and liposome groups in terms of β-catenin protein
expression (P=0.237). Thus, β-catenin-siRNA effectively inhibited
the protein expression of β-catenin in SW480 cells.
β-catenin-siRNA inhibits the
proliferation of SW480 cells
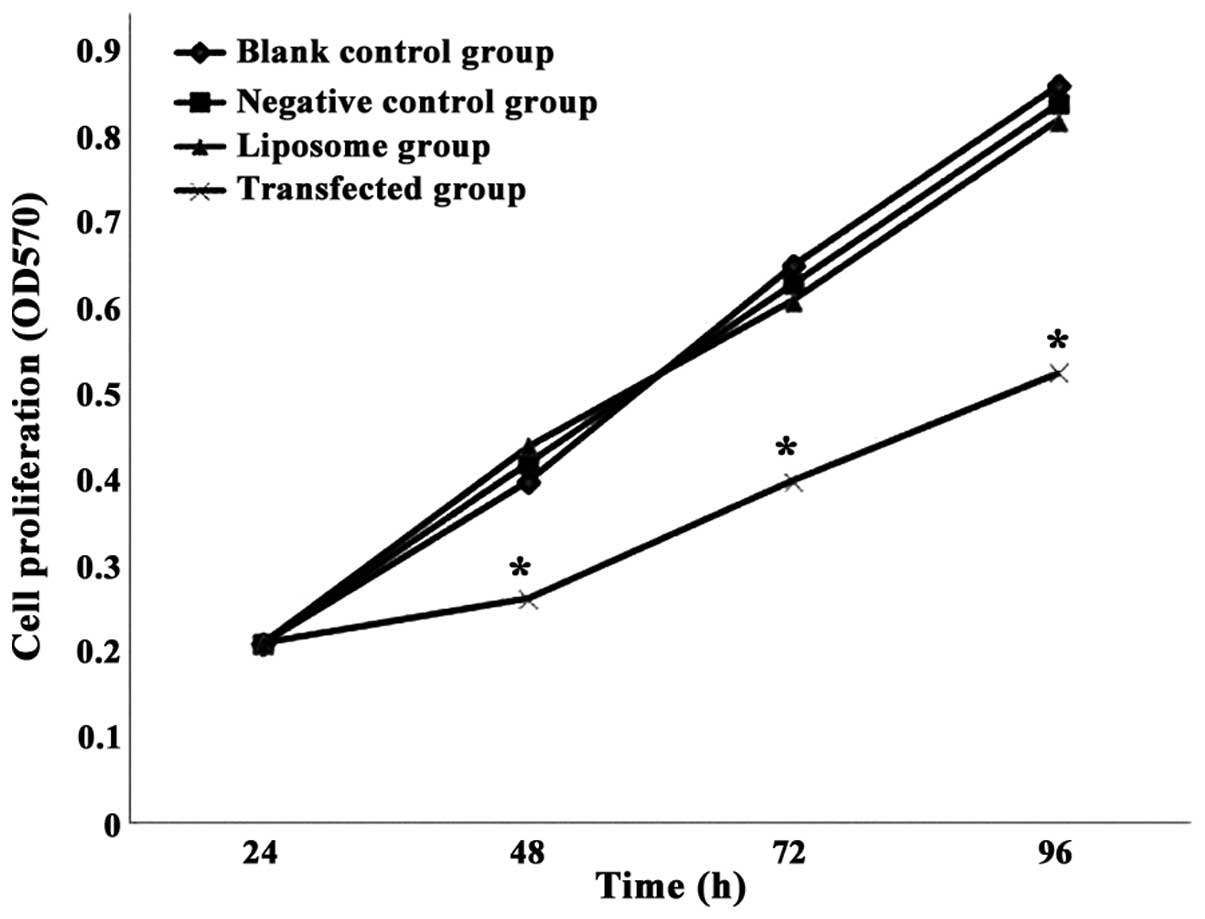
The results presented in Fig. 3 indicate that, following
β-catenin-siRNA transfection for 24 h, there was no significant
difference in SW480 cell proliferation in the
β-catenin-siRNA-transfected group, compared with the blank control,
negative control and liposome groups. However, upon transfection
for 48, 72 and 96 h, cell proliferation was significantly lower in
the β-catenin-siRNA-transfected group than in the other groups
(P=0.007). The blank control, negative control and liposome groups
exhibited no significant differences in cell proliferation
(P=0.255). Thus, β-catenin-siRNA-mediated silencing of β-catenin
gene expression was able to inhibit the proliferation of SW480
cells.
β-catenin-siRNA influences the
apoptosis of SW480 cells
The results presented in Fig. 4 and Table
I reveal that, upon silencing the β-catenin gene by
β-catenin-siRNA, the apoptosis rate of SW480 cells was
significantly higher in the β-catenin-siRNA-transfected group than
in the blank control, negative control and liposome groups
(18.76±2.79% vs. 5.22±1.13, 5.42±1.44 and 4.96±1.11%, respectively;
P=0.004). There was no significant difference in apoptosis among
the blank control, negative control and liposome groups (P=0.166).
Thus, β-catenin-siRNA transfection inhibited the apoptosis of SW480
cells.
 | Table I.Influence of small interference RNA
targeting β-catenin on the apoptosis rate and caspase-3 activity of
SW480 cells. |
Table I.
Influence of small interference RNA
targeting β-catenin on the apoptosis rate and caspase-3 activity of
SW480 cells.
| Group | Cell apoptosis rate,
% | Caspase-3 activity,
OD value |
|---|
| Blank control |
5.22±1.13 |
0.15±0.02 |
| Negative control |
5.42±1.44 |
0.16±0.02 |
| Liposome |
4.96±1.11 |
0.16±0.03 |
| Transfected |
18.76±2.79a |
0.48±0.05a |
β-catenin-siRNA promotes the
activation of caspase-3 in SW480 cells
The results in Table I
indicate that, following transfection with β-catenin-siRNA for 48
h, the levels of caspase-3 activity in the harvested SW480 cells
were 0.15±0.02, 0.16±0.02, 0.16±0.03 and 0.48±0.05 OD value
(P=0.005) in the blank control, negative control, liposome and
β-catenin-siRNA-transfected groups, respectively. Thus,
transfection with β-catenin-siRNA was able to increase caspase-3
activity in SW480 cells.
Influence of β-catenin-siRNA on SW480
cell invasion and metastasis
The number of SW480 cells passing through the
Matrigel membrane that was inhibited by transfection with
β-catenin-siRNA indicated the invasion ability of SW480 cells in
vitro. The results in Table II
show that the in vitro invasiveness of SW480 cells
transfected with β-catenin-siRNA for 24, 48 and 72 h, which were
subsequently seeded into the invasion chamber and observed 24 h
later, decreased following β-catenin-siRNA transfection (P=0.007),
while no significant differences were observed among the different
control groups (P=0.157). Following transfection with
β-catenin-siRNA for 48 and 72 h, the number of cells infiltrating
the membrane in the β-catenin-siRNA-transfected group was
significantly reduced, compared with that in the other control
groups (P=0.002), indicating that β-catenin-siRNA was able to
reduce the invasiveness of SW480 cells in vitro in a
time-dependent manner.
 | Table II.Influence of small interference RNA
targeting β-catenin on the invasive ability of SW480 cells in
vitro. |
Table II.
Influence of small interference RNA
targeting β-catenin on the invasive ability of SW480 cells in
vitro.
|
| Transfection, cell
number |
|---|
|
|
|
|---|
| Group | 24 h | 48 h | 72 h |
|---|
| Blank control |
88±12 |
89±10 |
84±15 |
| Negative control |
87±9 |
85±13 |
86±12 |
| Liposome |
84±14 |
82±11 |
83±6 |
| Transfected |
80±18 |
51±10a,b |
33±4a–c |
Discussion
Tumorigenesis, cancer progression and metastasis are
complex processes (6). In recent
years, the association between catenin and cancer has become well
known and the number of studies on the association between cell
adhesion molecules and malignant behavior has increased (7,8). β-catenin
is an important multifunctional cytoplasmic protein, which was
initially considered as an intracellular molecule on the cell
membrane that interacted with the calcium-dependent adhesion
molecule E-cadherin (9). However, the
isolation and cloning of β-catenin enabled to identify its dual
function as mediator of cell adhesion and participant in Wnt
signaling (10). β-catenin is located
in the activation center of the Wnt signaling pathway, and is
overexpressed in numerous tumors, including melanoma, leukemia, and
small cell, breast and head and neck cancer (11–13).
β-catenin is closely associated with various factors involved in
the formation of tumors, and participates in multiple aspects of
tumor development (14).
In the present study, siRNA technology was used, and
double-stranded siRNA targeting β-catenin was synthesized (15–17). The
experimental results indicated that specific siRNA targeting
β-catenin could inhibit the expression of the β-catenin gene.
RT-PCR and western blotting results demonstrated that, following
transfection with β-catenin-siRNA, the mRNA and protein expression
levels of β-catenin appeared to be significantly decreased,
suggesting that siRNA could effectively block the Wnt signaling
pathway.
The present study used RNA interference technology
to explore the impact of silencing the β-catenin gene on the
biological behavior of SW480 cells, and observed that the
chemically synthesized β-catenin-siRNA could effectively inhibit
the expression of β-catenin in SW480 cells. When the expression of
the β-catenin gene was suppressed, the proliferation of SW480 cells
was inhibited, and the apoptosis rate increased in these cells,
which may be explained by the inhibition of apoptosis and promotion
of proliferation mediated by β-catenin in SW480 cells. Thus, when
β-catenin expression was inhibited, the apoptosis rate of SW480
cells increased. The anti-apoptotic mechanism of β-catenin is not
yet entirely clear, but it may involve caspase-3, which is a major
effector caspase in downstream apoptotic signal transduction
pathways (18). In the present study,
β-catenin-siRNA was used to inhibit β-catenin gene expression,
which led to increased activity of caspase-3. Since β-catenin
inhibits the apoptosis of SW480 cells and promotes their
proliferation, the apoptosis of SW480 cells increases when the
expression of β-catenin is disrupted. Studies on β-catenin and
tumor invasiveness are limited. The present study has demonstrated
that, upon silencing β-catenin gene expression, the number of SW480
cells penetrating the membrane in an invasion assay was
significantly decreased, indicating that the invasive ability of
these cells was significantly decreased. Thus, β-catenin-siRNA
could effectively inhibit the expression of β-catenin in SW480
cells, and when β-catenin expression was suppressed, SW480 cell
proliferation and invasion capacity were decreased, while apoptosis
and caspase-3 activity were increased.
In the present study, SW480 cells with silenced
β-catenin gene in the experimental group were characterized by slow
proliferation in cell culture, low number of colonies formed in
soft agar and markedly inhibited cell growth. Considering the
experimental results obtained upon silencing the β-catenin gene and
colon cancer cell proliferation, it is possible to speculate that
in SW480 colon cancer cells specific siRNA targeting β-catenin may
silence the β-catenin gene and sequentially silence its downstream
genes. By silencing and inhibiting these upstream and downstream
genes, the proliferation of SW480 cells was inhibited in
vitro. Therefore, it can be proposed that, by interfering with
the Wnt signaling pathway, β-catenin may act as a potential target
gene for the treatment of colon cancer. However, further studies
are required to guarantee that β-catenin-siRNA technology is
stable, effective and secure to be used in clinical practice.
Acknowledgements
The present study was partly supported by grants
from the Natural Science Foundation of Hubei Province (Wuhan,
China; grant nos. 302-132139 and 302-131703).
References
|
1
|
Fodde R and Brabletz T: Wnt/beta-catenin
signaling in cancer stemness and malignant behavior. Curr Opin Cell
Biol. 19:150–158. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ring A, Kim YM and Kahn M: Wnt/catenin
signaling in adult stem cell physiology and disease. Stem Cell Rev.
10:512–525. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fan K, Li N, Qi J, Yin P, Zhao C, Wang L,
Li Z and Zha X: Wnt/β-catenin signaling induces the transcription
of cystathionine-γ-lyase, a stimulator of tumor in colon cancer.
Cell Signal. 26:2801–2808. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lazarova DL, Wong T, Chiaro C, Drago E and
Bordonaro M: p300 influences butyrate-mediated WNT hyperactivation
in colorectal cancer cells. J Cancer. 4:491–501. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kumawat K, Koopmans T and Gosens R:
β-catenin as a regulator and therapeutic target for asthmatic
airway remodeling. Expert Opin Ther Targets. 18:1023–1034. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Denais C and Lammerding J: Nuclear
mechanics in cancer. Adv Exp Med Biol. 773:435–470. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tan CW, Gardiner BS, Hirokawa Y, Layton
MJ, Smith DW and Burgess AW: Wnt signalling pathway parameters for
mammalian cells. PLoS One. 7:e318822012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Paul S and Dey A: Wnt signaling and cancer
development: Therapeutic implication. Neoplasma. 55:165–176.
2008.PubMed/NCBI
|
|
9
|
Lai T, Su C, Kuo W, Yeh Y, Kuo W, Tsai F,
Tsai C, Weng Y and Huang C: β-catenin plays a key role in
metastasis of human hepatocellular carcinoma. Oncology Reports.
26:415–422. 2011.PubMed/NCBI
|
|
10
|
Voronkov A and Krauss S: Wnt/beta-catenin
signaling and small molecule inhibitors. Curr Pharm Des.
19:634–664. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Clevers H and Nusse R: Wnt/β-catenin
signaling and disease. Cell. 149:1192–1205. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Schmeel LC, Schmeel FC, Kim Y, Endo T, Lu
D and Schmidt-Wolf IG: Targeting the Wnt/beta-catenin pathway in
multiple myeloma. Anticancer Res. 33:4719–4726. 2013.PubMed/NCBI
|
|
13
|
Yao H, Ashihara E and Maekawa T: Targeting
the Wnt/β-catenin signaling pathway in human cancers. Expert Opin
Ther Targets. 15:873–887. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Behrens J: Control of beta-catenin
signaling in tumor development. Ann N Y Acad Sci. 910:21–35. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Flemr M, Malik R, Franke V, Nejepinska J,
Sedlacek R, Vlahovicek K and Svoboda P: A retrotransposon-driven
dicer isoform directs endogenous small interfering RNA production
in mouse oocytes. Cell. 155:807–816. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dogini DB, Pascoal VD, Avansini SH, Vieira
AS, Pereira TC and Lopes-Cendes I: The new world of RNAs. Genet Mol
Biol. 37(Suppl 1): S285–S293. 2014. View Article : Google Scholar
|
|
17
|
Fellmann C and Lowe SW: Stable RNA
interference rules for silencing. Nat Cell Biol. 16:10–18. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ismail B, Ghezali L, Gueye R, Limami Y,
Pouget C, Leger DY, Martin F, Beneytout J, Duroux J, Diab-Assaf M,
et al: Novel methylsulfonyl chalcones as potential
antiproliferative drugs for human prostate cancer: Involvement of
the intrinsic pathway of apoptosis. Int J Oncol. 43:1160–1168.
2013.PubMed/NCBI
|