Introduction
The basis of preventive treatment for stent
thrombosis is the best percutaneous intervention (PCI) performance
including good expansion and apposition of the stent, otherwise,
dual anti-platelet therapy (DAPT) is the cornerstone of medical
therapy after PCI (1,2). Based on data from Platelet Inhibition
and Patient Outcomes (3), the newer
platelet inhibitors such as ticagrelor have been accepted in the
current American College of Cardiology guidelines (4). However, bleeding consituted an issue as
compared with clopidogrel (3).
In the present study, we describe a case of
spontaneous hematoma in a patient administered ticagrelor following
PCI.
Case report
The patient was a 69-year-old man who was admitted
to the Central Hospital of Xuzhou (Jiangsu, China) because a lump
was discovered, accompanied with pain under his right scapula. He
had a 12-year history of hypertension and 6-month history of
coronary heart disease, without history of trauma, diabetes,
stroke, or alimentary tract hemorrhage. Six months earlier, the
patient felt dyspnea and chest discomfort after certain activities.
After resting for a few minutes, such symptoms disappeared;
however, 2 months prior to admission, the symptoms became
aggravated, and the patient presented to the hospital for a
check-up. Coronary angiogram showed that the end of left main
coronary artery had 50% local stenosis, the opening to the proximal
segment of anterior descending branch had 70–90% stenosis, and the
opening to the proximal segment of circumflex artery had 80%
stenosis. A 3.5×24 mm resolute stent was implanted between the
proximal segment of the anterior descending branch and the end of
left main coronary artery. A 4.0×18 mm resolute stent was implanted
between the proximal segment of circumflex artery and the end of
left main coronary artery. The operation method used was
cullote.
After the operation, the patient was administered
100 mg aspirin (Bayer, Beijing, China) once a day, 90 mg ticagrelor
(AstraZeneca, Shanghai, China) twice a day, combined with normal
medication of rosuvastatin (AstraZeneca, Shanghai, China),
amlodipine besylate tablet (Pfizer, Dalian, China), and metoprolol
succinate sustained-release tablet (AstraZeneca, Shanghai, China).
At 5 h prior to admission, while the patient was sleeping, he
experienced sudden pain under the right scapula and considered that
the lump was getting larger gradually. Therefore, he was admitted
for emergency treatment. Two months prior to admission, ecchymosis
was found on all his limbs, waist and abdomen, but since he did not
experience any discomfort, he was not treated. In the course of
disease, no symptoms, such as chest pains, shortage of breath,
fever, or infection were evident. A physical examination on
admission indicated the following: temperature (T) 36.6°C, pulse
(P) 82 times/min, respiration (R) 18 times/min, blood pressure (BP)
135/85 mmHg, clear consciousness, no cyanosis, soft neck, no
distension of jugular vein, 8×10 cm enclosed mass under the right
scapula, normal complexion, obvious tenderness, clear breathing
sounds in both lungs, without dry or moist rale, medium heart
border under percussion, heart rate of 82 times/min, regular, no
murmur in auscultatory valve areas, a large bruise in the right
waist and abdomen, no obvious enclosed mass, no pressing pains,
soft abdomen, no pressing pains or rebound tenderness, no touch to
liver, spleen or subcostal, and no edema on the limbs.
Physiological reflection existed and pathologic reflex was not
drawn out.
The auxiliary examination included: blood routine
test showing red blood cells 3.23×1012/l [reference
value: (4.0–5.5)x1012/l], hemoglobin 98 g/l (reference
value: 120–160 g/l), hematocrit 30.2% (reference value: 40–50%),
and blood platelet 140×109/l [reference value:
(100–300)x109/l]. The coagulation function was:
prothrombin time (PT) 11.8 sec (reference value: 9–13 sec), active
partial PT (APTT) 22.5 sec (reference value: 21–34 sec), and PT
international normalized ratio (INR) 0.99 (reference value: 0.9–1.1
sec). Cardiac troponin I had a value of 0.02 (reference value:
0–0.08 ng/ml). Liver function and renal function were normal. The
patient underwent a thrombelastogram examination on the first day
of admission, and the following were indicated: R time 5.1 min
(reference value: 5–10 min), K time 1.9 min (reference value: 1–3
min), MA 62.5 mm (reference value: 50–70 mm), CI 0.7 (reference
value: −3-3), LY30 0.1% (reference value: 0–8%), AA inhibition rate
98.3%, and adenosine diphosphate (ADP) inhibition rate 88.8%.
Electrocardiogram and color Doppler ultrasound showed no
abnormality. The admitting diagnosis for the patient was
spontaneous edema under the right scapula.
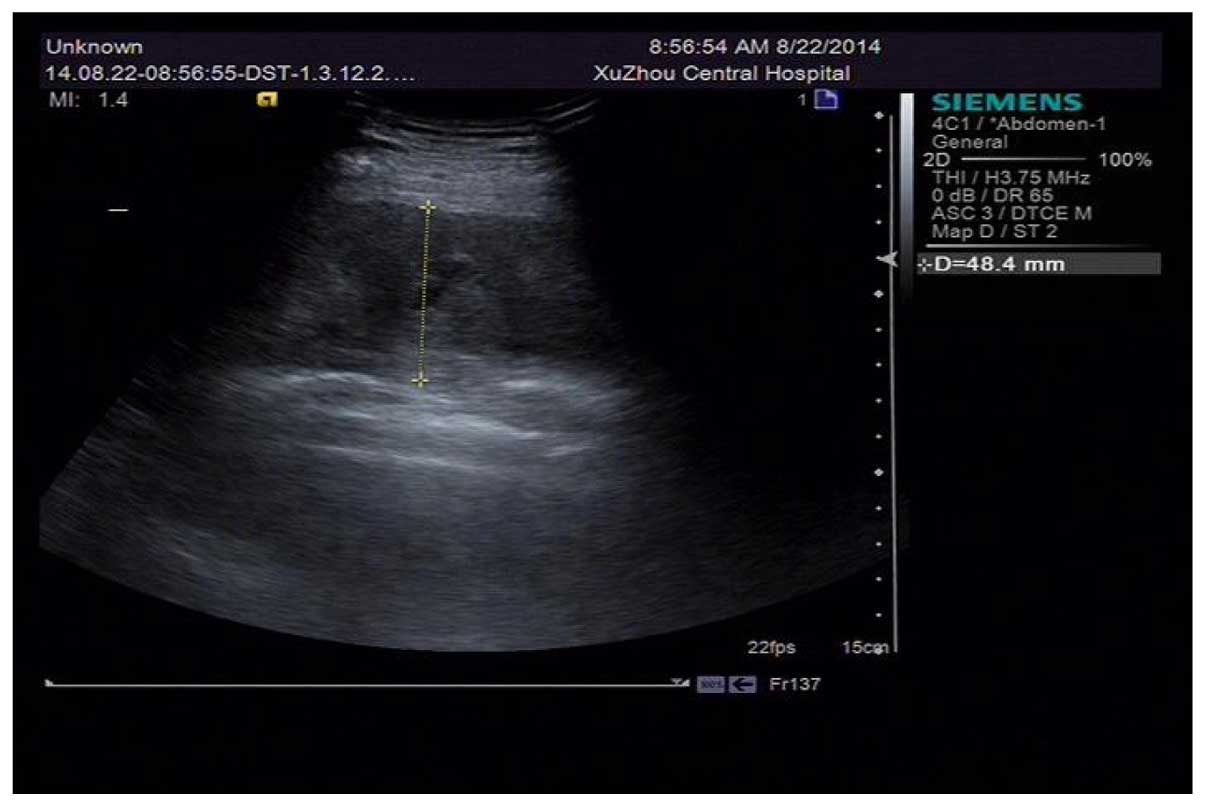
A B ultrasonic examination was conducted on the day
of admission indicating liquid anechoic area under right scapula, D
48.4 mm (Fig. 1).
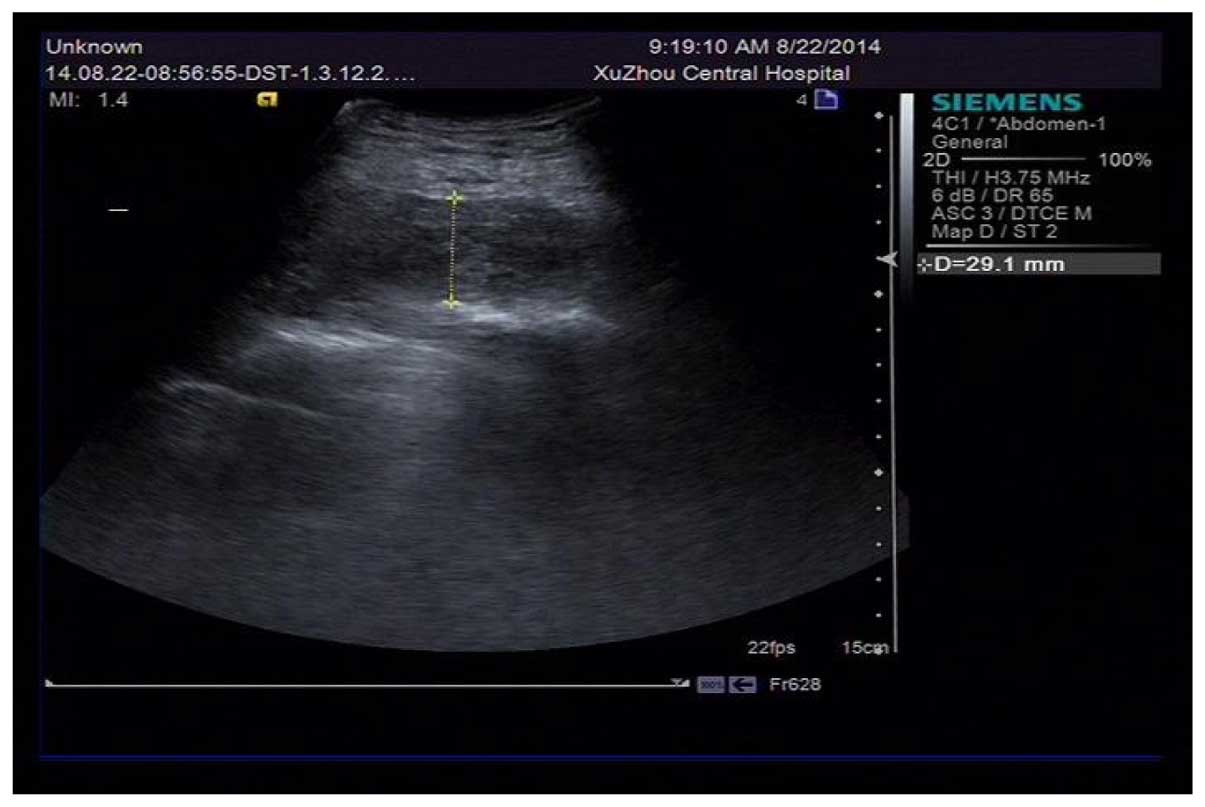
Puncture drainage by syringe was conducted on the
skin in an enclosed mass following disinfection, draining out 105
ml of incoagulable blood, and the liquid anechoic area turned to D
29.1 mm (Fig. 2). A pressure bandage
was subsequently applied.
Coagulation disorders and an abnormal quantity of
blood platelets were excluded according to the result of the
patient's PT, APTT, INR and thrombelastogram examination. The
thrombelastogram showed that the AA and ADP inhibition rate
increased significantly, indicating that the patient had a high
response towards dual antiplatelet drugs, and since the patient had
undergone implantation of double stents 2 months earlier due to
left main coronary artery lesion, anterior descending branch
lesion, and circumflex artery bifurcation lesion, and had used
aspirin 6 months earlier, no significant abnormality was evident.
Ticagrelor was used after the PCI operation and the patient denied
any trauma, thus it was deemed that spontaneous edema was relevant
to oral administration of ticagrelor.
Aspirin was continued, stopping ticagrelor for one
day and changing to clopidogrel 75 mg QD. Pressure bandaging,
combined with coronary artery-dilating medicine (isosorbide
mononitrate), lipid lowering to stabilize plaque (atorvastatin) and
depressurization (amlodipine besylate) treatment were applied.
After 6 days, thrombelastogram examination was again conducted and
the values identified were: R time 7.3 min (reference value: 5–10
min), K time 1.9 min (reference value: 1–3 min), MA 68.4 mm
(reference value: 50–70 mm), CI 0.1 (reference value: −3-3), LY30
0.1% (reference value: 0–8%), AA inhibition rate 74.3%, ADP
inhibition rate 81.3%, and hemoglobin 95 g/l. The enclosed mass in
the affected part wsa gradually absorbed. At 9 days after the onset
of disease, the patient had a hemoglobin of 102 g/l. The patient
was discharged from hospital. After DAPT with aspirin and
ticagrelor and a follow-up of 10 months, no hematoma recurred and
no skin ecchymosis was evident.
Discussion
Anti-platelet therapy is the cornerstone of
treatment for PCI. DAPT combined with clopidogrel (4) and aspirin has obvious limitations in
treatment, and patients have the risk of cardiovascular sudden
death and stent thrombosis. Patients in Asia, due to gene
polymorphism, have resistance or low reaction towards clopidogrel
(5). A high dose of clopidogrel is
unable to effectively avoid the risk of stent thrombosis on acute
coronary syndrome (ACS) patients after PCI treatment (3). Ticagrelor is a new type of oral
anti-platelet drug, which reversibly interacts with P2Y12 receptor
of platelet ADP, blocks signal transduction and platelet activation
(6). Compared with clopidogrel, it
does not need metabolic activation, and it may be quickly absorbed
by the human body, and takes effect faster, inhibiting platelets
significantly during the treatment period of maintenance dose.
After the last medication, the drug therapeutic effect decreases
significantly (7). Ticagrelor was the
first oral anti-platelet drug that was confirmed to be able to
reduce cardiovascular deaths and total deaths of ACS patients, and
it was also able to significantly reduce the risks of
cardiovascular events; thus, it has become widely used in the
clinic (8). In terms of the safety of
blood loss, the ticagrelor and clopidogrel groups had no
significant difference in terms of massive hemorrhage, fatal
massive hemorrhage, TIMI massive hemorrhage and the need of
infusion of erythrocyte. However, fatal intracranial hemorrhage and
non-coronary-artery bypass grafting-related hemorrhage in the
ticagrelor group increased (9).
Notably, the FDA, while authorizing ticagrelor stated that
ticagrelor, as in the case of other anti-platelet drugs,
occasionally results in fatal hemorrhage (10). Consequently, it could not be used in
patients with a history of active pathological or intracranial
hemorrhage (10). Particularly,
clopidogrel was deemed safer than ticagrelor on various hemorrhage
indexes (11). Most of the
subcutaneous hematomas on scapula were relevant to trauma and
diseases of the blood system. Only few spontaneous edemas were
relevant to dual anti-platelet drugs. Two months earlier, the
patient in our study was diagnosed with ACS. Radiography showed
that lesions appeared in the left main coronary artery, anterior
descending branch, and circumflex artery. Double stents were
implanted. In the Guideline for the Management of Patients with
Non-ST-Elevation Acute Coronary Syndromes (4) suggested using the more effective
anti-platelet drug ticagrelor on the basis of aspirin. Compared
with the independent use of aspirin, skin ecchymosis on the patient
increased significantly, and spontaneous edema appeared under the
right scapula, which under the laboratory examination, was
confirmed as the result of the patient's high response towards
ticagrelor. After changing ticagrelor for clopidogrel, such
symptoms disappeared and no recurrence was detected during the
follow-up period.
This case suggests that with the appearance of new
anti-platelet drugs, there is a stronger anti-thrombotic effect,
but also an increase in rare bleeding conditions, leading to new
issues that are to be resolved: i) for example, whether, as the
guideline suggests, all the ACS patients after operation should use
more effective anti-platelet drugs; ii) or whether certain blood
platelet detection methods, such as thrombelastogram, used to
verify the outcome, may be used as conventional auxiliary methods
to determine the type of anti-platelet drugs to be employed.
References
|
1
|
Lemesle G, Delhaye C, Bonello L, de
Labriolle A, Waksman R and Pichard A: Stent thrombosis in 2008:
definition, predictors, prognosis and treatment. Arch Cardiovasc
Dis. 101:769–777. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kurz DJ and Eberli FR: Medical therapy of
coronary artery disease after percutaneous intervention. Curr Opin
Pharmacol. 13:287–293. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wallentin L, Becker RC, Budaj A, et al:
Ticagrelor versus clopidogrel in patients with acute coronary
syndromes. N Eng J Med. 361:1045–1057. 2009. View Article : Google Scholar
|
|
4
|
Amsterdam EA, Wenger NK, Brindis RG, Casey
DE Jr, Ganiats TG, Holmes DR Jr, Jaffe AS, Jneid H, Kelly RF,
Kontos MC, et al: American Association for Clinical Chemistry:
22014 AHA/ACC Guideline for the Management of Patients with
Non-ST-Elevation Acute Coronary Syndromes: a report of the American
College of Cardiology/American Heart Association Task Force on
Practice Guidelines. J Am Coll Cardiol. 64:e139–e228. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kubica AI, Kozinski M, Grzesk G, Fabiszak
T, Navarese EP and Goch A: Genetic determinants of platelet
response to clopidogrel. J Thromb Thrombolysis. 32:459–466. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Husted S and van Giezen JJ: Ticagrelor:
the first reversibly binding oral P2Y12 receptor antagonist.
Cardiovasc Ther. 27:259–274. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gurbel PA, Bliden KP, Butler K, Tantry US,
Gesheff T, Wei C, Teng R, Antonino MJ, Patil SB, Karunakaran A, et
al: Randomized double-blind assessment of the ONSET and OFFSET of
the antiplatelet effects of ticagrelor versus clopidogrel in
patients with stable coronary artery disease: The ONSET/OFFSET
study. Circulation. 120:2577–2585. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Juneja S, Gupta K and Kaushal S:
Ticagrelor: An emerging oral antiplatelet agent. J Pharmacol
Pharmacother. 4:78–80. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wallentin L, Becker RC, Budaj A, Cannon
CP, Emanuelsson H, Held C, Horrow J, Husted S, James S, Katus H, et
al: PLATO Investigators: Ticagrelor versus clopidogrel in patients
with acute coronary syndromes. N Engl J Med. 361:1045–1057. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gaglia MA Jr and Waksman R: Overview of
the 2010 Food and Drug Administration Cardiovascular and Renal
Drugs Advisory Committee meeting regarding ticagrelor. Circulation.
123:451–456. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
DiNicolantonio JJ, D'Ascenzo F, Tomek A,
Chatterjee S, Niazi AK and Biondi-Zoccai G: Clopidogrel is safer
than ticagrelor in regard to bleeds: a closer look at the PLATO
trial. Int J Cardiol. 168:1739–1744. 2013. View Article : Google Scholar : PubMed/NCBI
|