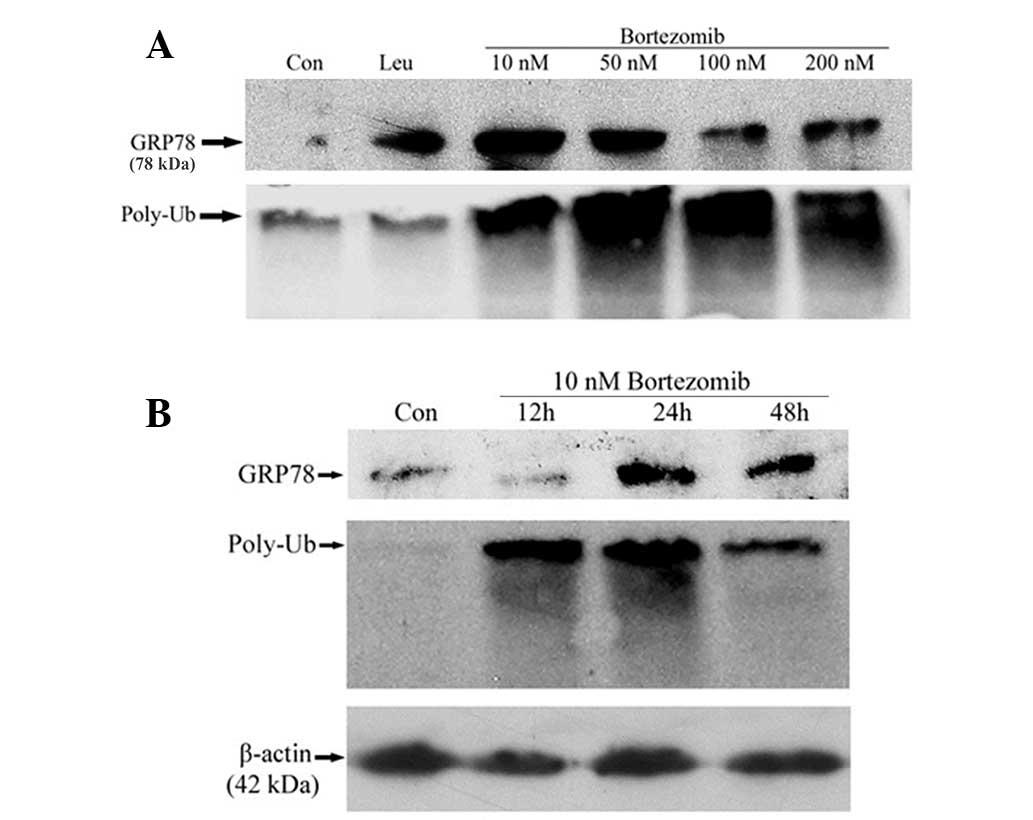
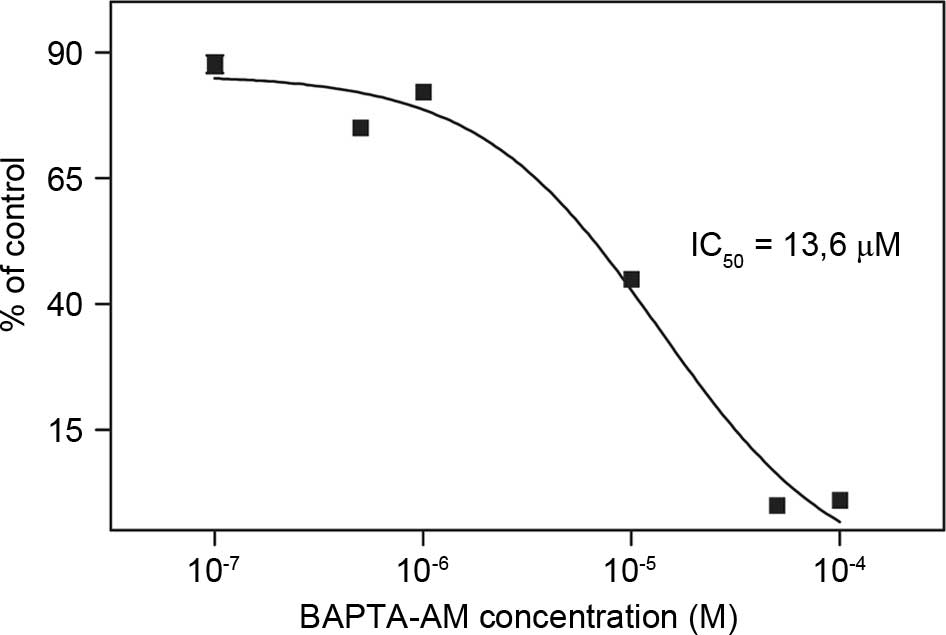
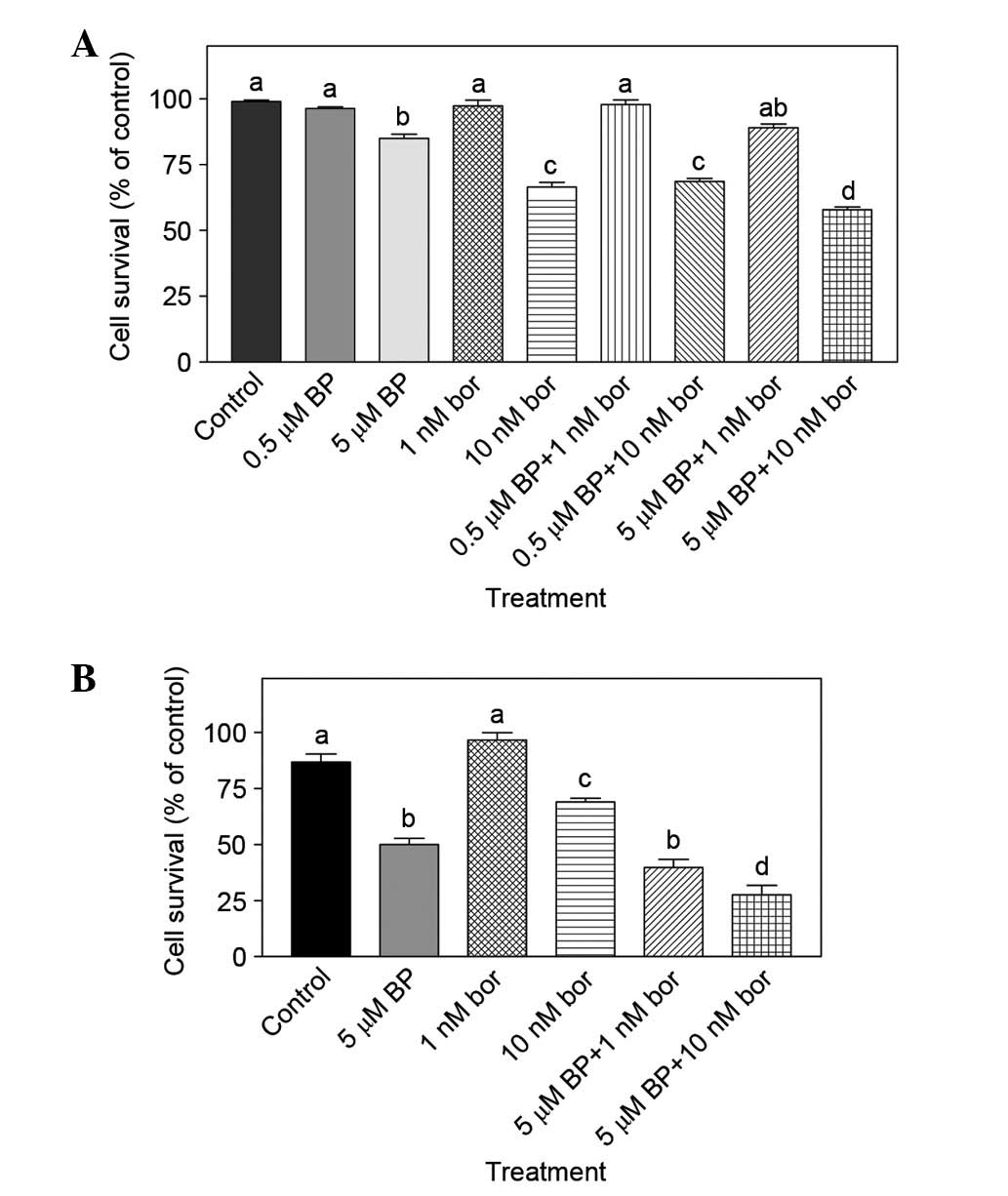
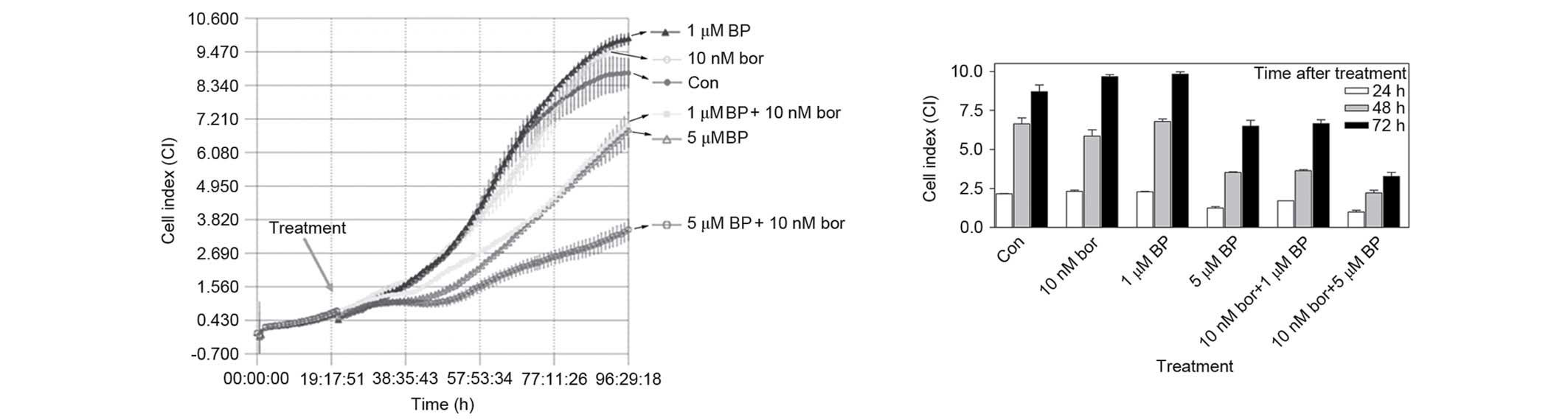
|
1
|
Hendershot LM: The ER function BiP is a
master regulator of ER function. Mt Sinai J Med. 71:289–297.
2004.PubMed/NCBI
|
|
2
|
Hendershot LM, Valentine VA, Lee AS,
Morris SW and Shapiro DN: Localization of the gene encoding human
BiP/GRP78, the endoplasmic reticulum cognate of the HSP70 family,
to chromosome 9q34. Genomics. 20:281–284. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Murphy ME: The HSP70 family and cancer.
Carcinogenesis. 34:1181–1188. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Roller C and Maddalo D: The molecular
chaperone GRP78/BiP in the development of chemoresistance:
Mechanism and possible treatment. Front Pharmacol. 4:102013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Qiu W, Kohen-Avramoglu R, Mhapsekar S,
Tsai J, Austin RC and Adeli K: Glucosamine-induced endoplasmic
reticulum stress promotes ApoB100 degradation: Evidence for
Grp78-mediated targeting to proteasomal degradation. Arterioscler
Thromb Vasc Biol. 25:571–577. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhang Y, Tseng CC, Tsai YL, Fu X, Schiff R
and Lee AS: Cancer cells resistant to therapy promote cell surface
relocalization of GRP78 which complexes with PI3K and enhances
PI(3,4,5)P3 production. PLoS One. 8:e800712013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li B, Cheng XL, Yang YP and Li ZQ: GRP78
mediates radiation resistance of a stem cell-like subpopulation
within the MCF-7 breast cancer cell line. Oncol Rep. 30:2119–2126.
2013.PubMed/NCBI
|
|
8
|
Dong D, Ko B, Baumeister P, Swenson S,
Costa F, Markland F, Stiles C, Patterson JB, Bates SE and Lee AS:
Vascular targeting and antiangiogenesis agents induce drug
resistance effector GRP78 within the tumor microenvironment. Cancer
Res. 65:5785–5791. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang J, Jiang Y, Jia Z, Li Q, Gong W,
Wang L, Wei D, Yao J, Fang S and Xie K: Association of elevated
GRP78 expression with increased lymph node metastasis and poor
prognosis in patients with gastric cancer. Clin Exp Metastasis.
23:401–410. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang HQ, Du ZX, Zhang HY and Gao DX:
Different induction of GRP78 and CHOP as a predictor of sensitivity
to proteasome inhibitors in thyroid cancer cells. Endocrinology.
148:3258–3270. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mozos A, Roué G, López-Guillermo A, Jares
P, Campo E, Colomer D and Martinez A: The expression of the
endoplasmic reticulum stress sensor BiP/GRP78 predicts response to
chemotherapy and determines the efficacy of proteasome inhibitors
in diffuse large b-cell lymphoma. Am J Pathol. 179:2601–2610. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen LY, Chiang AS, Hung JJ, Hung HI and
Lai YK: Thapsigargin-induced grp78 expression is mediated by the
increase of cytosolic free calcium in 9l rat brain tumor cells. J
Cell Biochem. 78:404–416. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Freshney RI: Culture of Animal Cells: A
Manual of Basic Technique. Wiley-Liss. Hoboken, NJ: 2005.
View Article : Google Scholar
|
|
14
|
Savran B, Yerlikaya A, Erdoğan E and Genç
O: Anticancer agent ukrain and bortezomib combination is
synergistic in 4T1 breast cancer cells. Anticancer Agents Med Chem.
14:466–472. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yerlikaya A and Erin N: Differential
sensitivity of breast cancer and melanoma cells to proteasome
inhibitor velcade. Int J Mol Med. 22:817–823. 2008.PubMed/NCBI
|
|
16
|
Oerlemans R, Franke NE, Assaraf YG, Cloos
J, van Zantwijk I, Berkers CR, Scheffer GL, Debipersad K, Vojtekova
K, Lemos C, et al: Molecular basis of bortezomib resistance:
Proteasome subunit beta5 (PSMB5) gene mutation and overexpression
of PSMB5 protein. Blood. 112:2489–2499. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chauhan D, Li G, Shringarpure R, Podar K,
Ohtake Y, Hideshima T and Anderson KC: Blockade of Hsp27 overcomes
bortezomib/proteasome inhibitor PS-341 resistance in lymphoma
cells. Cancer Res. 63:6174–6177. 2003.PubMed/NCBI
|
|
18
|
Ni M, Zhang Y and Lee AS: Beyond the
endoplasmic reticulum: Atypical GRP78 in cell viability, signalling
and therapeutic targeting. Biochem J. 434:181–188. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang Y, Wang W, Wang S, Wang J, Shao S and
Wang Q: Down-regulation of GRP78 is associated with the sensitivity
of chemotherapy to VP-16 in small cell lung cancer NCI-H446 cells.
BMC Cancer. 8:3722008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dhanasekaran DN and Reddy EP: JNK
signaling in apoptosis. Oncogene. 27:6245–6251. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dai Y, Rahmani M and Grant S: Proteasome
inhibitors potentiate leukemic cell apoptosis induced by the
cyclin-dependent kinase inhibitor flavopiridol through a SAPK/JNK-
and NF-kappaB-dependent process. Oncogene. 22:7108–7122. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dai Y, Rahmani M, Pei XY, Dent P and Grant
S: Bortezomib and flavopiridol interact synergistically to induce
apoptosis in chronic myeloid leukemia cells resistant to imatinib
mesylate through both Bcr/Abl-dependent and -independent
mechanisms. Blood. 104:509–518. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Roué G, Pérez-Galan P, Mozos A,
López-Guerra M, Xargay-Torrent S, Rosich L, Saborit-Villarroya I,
Normant E, Campo E and Colomer D: The Hsp90 inhibitor IPI-504
overcomes bortezomib resistance in mantle cell lymphoma in vitro
and in vivo by down-regulation of the prosurvival ER chaperone
BiP/Grp78. Blood. 117:1270–1279. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kern J, Untergasser G, Zenzmaier C, Sarg
B, Gastl G, Gunsilius E and Steurer M: GRP-78 secreted by tumor
cells blocks the antiangiogenic activity of bortezomib. Blood.
114:3960–3967. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kardosh A, Golden EB, Pyrko P, Uddin J,
Hofman FM, Chen TC, Louie SG, Petasis NA and Schönthal AH:
Aggravated endoplasmic reticulum stress as a basis for enhanced
glioblastoma cell killing by bortezomib in combination with
celecoxib or its non-coxib analogue, 2,5-dimethyl-celecoxib. Cancer
Res. 68:843–851. 2008. View Article : Google Scholar : PubMed/NCBI
|