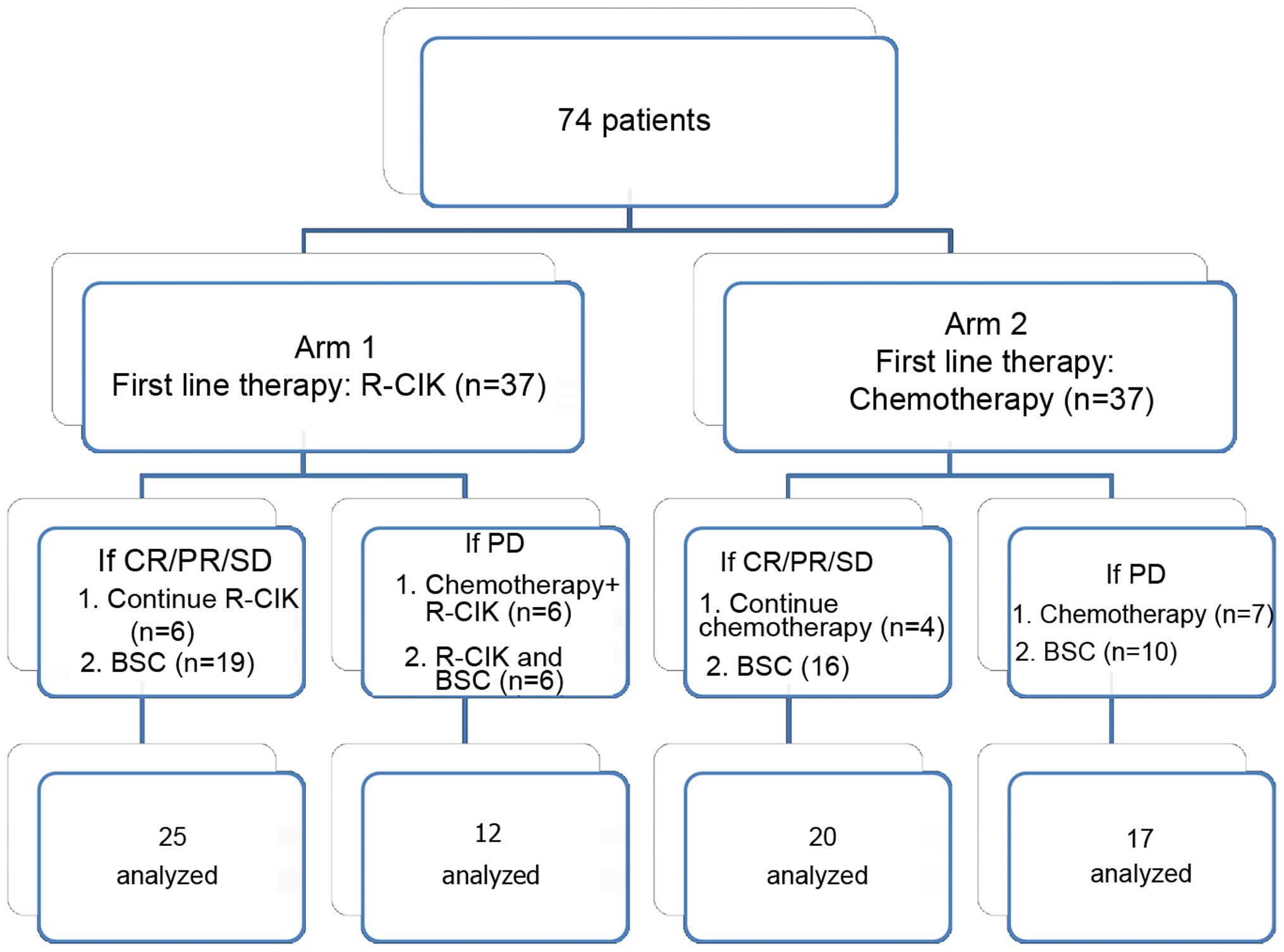
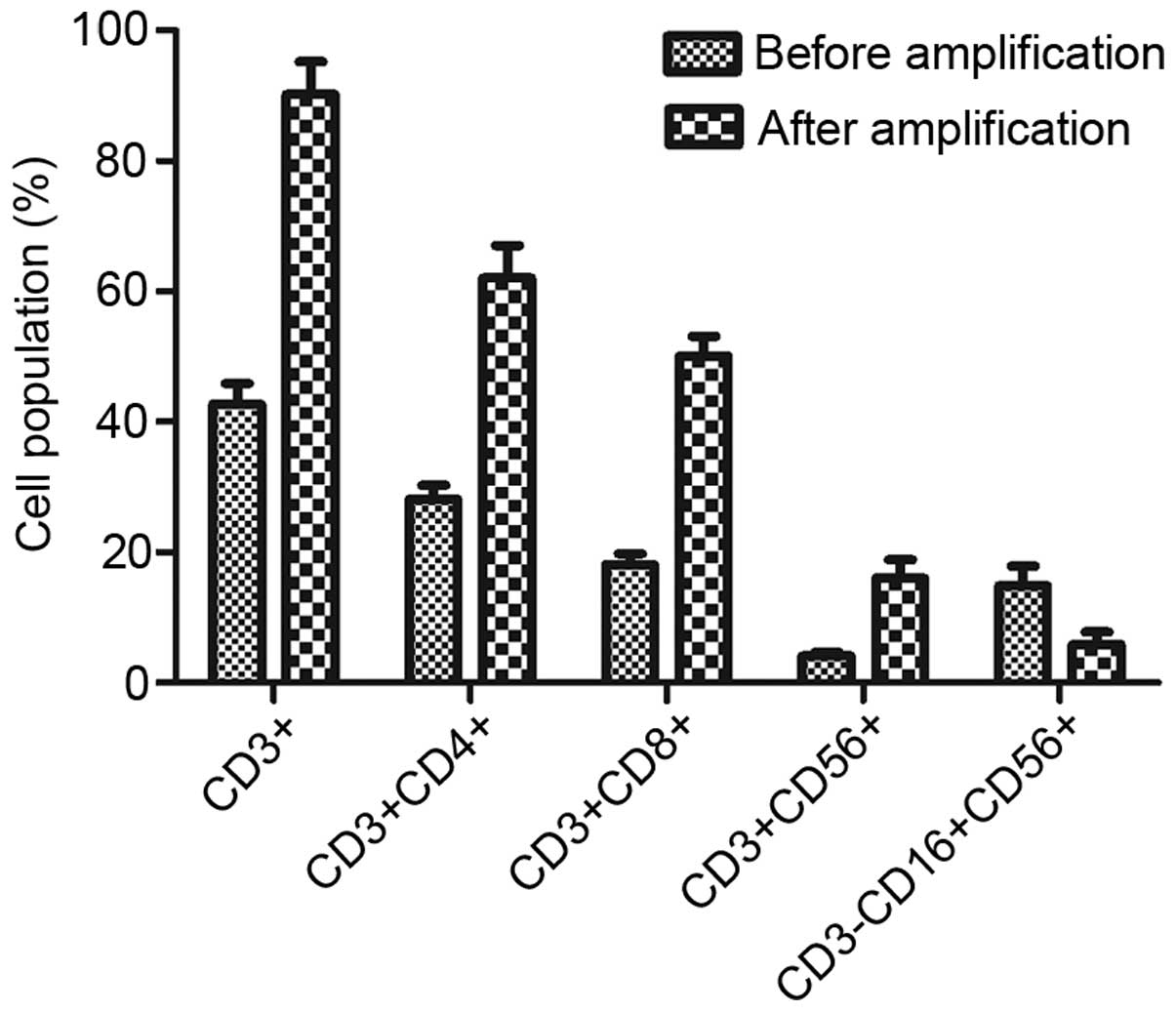
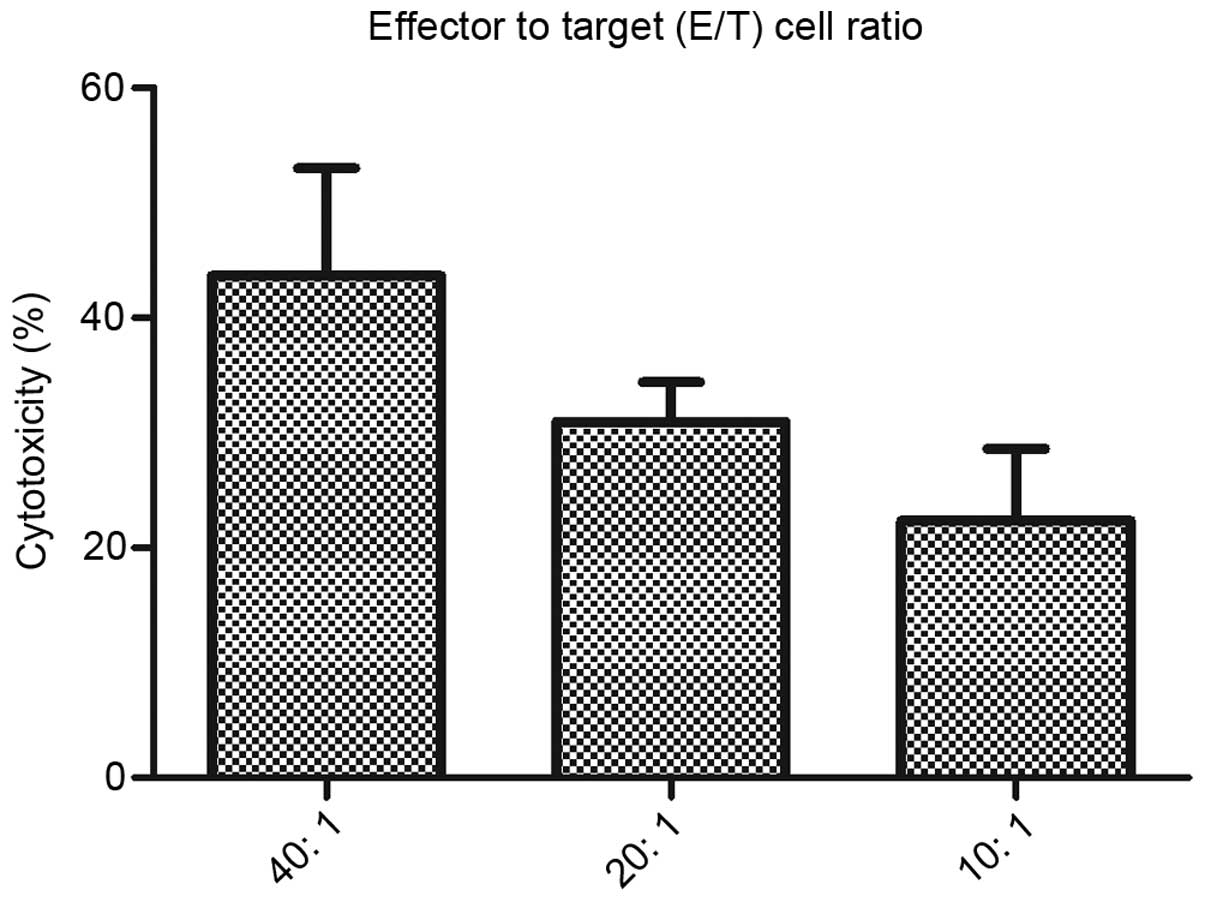
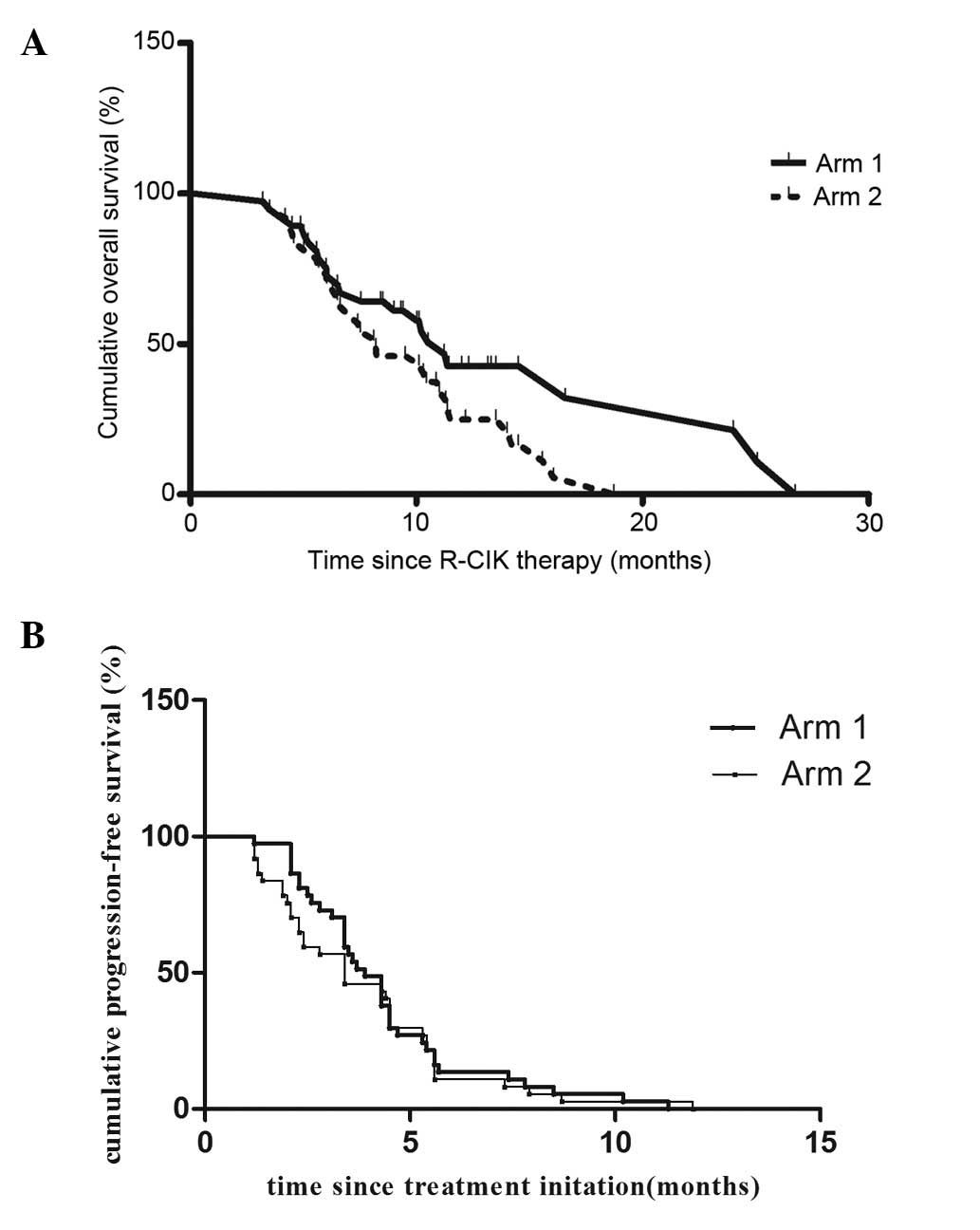
|
1
|
Qiang L, Huikai L, Butt K, Wang PP and Hao
X: Factors associated with disease survival after surgical
resection in Chinese patients with hepatocellular carcinoma. World
J Surg. 30:439–445. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bosch FX, Ribes J and Borràs J:
Epidemiology of primary liver cancer. Semin Liver Dis. 19:271–285.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
El-Serag HB and Mason AC: Rising incidence
of hepatocellular carcinoma in the United States. N Engl J Med.
340:745–750. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Taylor-Robinson SD, Foster GR, Arora S,
Hargreaves S and Thomas HC: Increase in primary liver cancer in the
UK, 1979–94. Lancet. 350:1142–1143. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jarnagin WR: Management of small
hepatocellular carcinoma: A review of transplantation, resection,
and ablation. Ann Surg Oncol. 17:1226–1233. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Page AJ, Cosgrove DC, Philosophe B and
Pawlik TM: Hepatocellular carcinoma: Diagnosis, management, and
prognosis. Surg Oncol Clin N Am. 23:289–311. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Truty MJ and Vauthey JN: Surgical
resection of high-risk hepatocellular carcinoma: Patient selection,
preoperative considerations, and operative technique. Ann Surg
Oncol. 17:1219–1225. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Asham EH, Kaseb A and Ghobrial RM:
Management of hepatocellular carcinoma. Surg Clin North Am.
93:1423–1450. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chok KS, Ng KK, Poon RT, Lo CM and Fan ST:
Impact of postoperative complications on long-term outcome of
curative resection for hepatocellular carcinoma. Br J Surg.
96:81–87. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Seo DD, Lee HC, Jang MK, Min HJ, Kim KM,
Lim YS, Chung YH, Lee YS, Suh DJ, Ko GY, et al: Preoperative portal
vein embolization and surgical resection in patients with
hepatocellular carcinoma and small future liver remnant volume:
Comparison with transarterial chemoembolization. Ann Surg Oncol.
14:3501–3509. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ikai I, Itai Y, Okita K, Omata M, Kojiro
M, Kobayashi K, Nakanuma Y, Futagawa S, Makuuchi M and Yamaoka Y:
Report of the 15th follow-up survey of primary liver cancer.
Hepatol Res. 28:21–29. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Deans C and Leslie P: Hepatocellular
carcinoma. Lancet. 354:253–254. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ribero D, Curley SA, Imamura H, Madoff DC,
Nagorney DM, Ng KK, Donadon M, Vilgrain V, Torzilli G, Roh M and
Vauthey JN: Selection for resection of hepatocellular carcinoma and
surgical strategy: Indications for resection, evaluation of liver
function, portal vein embolization, and resection. Ann Surg Oncol.
15:986–992. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Vivarelli M, Guglielmi A, Ruzzenente A,
Cucchetti A, Bellusci R, Cordiano C and Cavallari A: Surgical
resection versus percutaneous radiofrequency ablation in the
treatment of hepatocellular carcinoma on cirrhotic liver. Ann Surg.
240:102–107. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bruix J, Raoul JL, Sherman M, Mazzaferro
V, Bolondi L, Craxi A, Galle PR, Santoro A, Beaugrand M,
Sangiovanni A, et al: Efficacy and safety of sorafenib in patients
with advanced hepatocellular carcinoma: Subanalyses of a phase III
trial. J Hepatol. 57:821–829. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S,
Kim JS, Luo R, Feng J, Ye S, Yang TS, et al: Efficacy and safety of
sorafenib in patients in the Asia-Pacific region with advanced
hepatocellular carcinoma: A phase III randomised, double-blind,
placebo-controlled trial. Lancet Oncol. 10:25–34. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Qin S, Bai Y, Lim HY, Thongprasert S, Chao
Y, Fan J, Yang TS, Bhudhisawasdi V, Kang WK, Zhou Y, et al:
Randomized, multicenter, open-label study of oxaliplatin plus
fluorouracil/leucovorin versus doxorubicin as palliative
chemotherapy in patients with advanced hepatocellular carcinoma
from Asia. J Clin Oncol. 31:3501–3508. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
von Delius S, Lersch C, Mayr M, Stock K,
Schulte-Frohlinde E, Schmid RM and Eckel F: Capecitabine for
treatment of advanced hepatocellular carcinoma.
Hepatogastroenterology. 54:2310–2314. 2007.PubMed/NCBI
|
|
19
|
Zaanan A, Williet N, Hebbar M, Dabakuyo
TS, Fartoux L, Mansourbakht T, Dubreuil O, Rosmorduc O, Cattan S,
Bonnetain F, et al: Gemcitabine plus oxaliplatin in advanced
hepatocellular carcinoma: A large multicenter AGEO study. J
Hepatol. 58:81–88. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schmidt-Wolf IG, Negrin RS, Kiem HP, Blume
KG and Weissman IL: Use of a SCID mouse/human lymphoma model to
evaluate cytokine-induced killer cells with potent antitumor cell
activity. J Exp Med. 174:139–149. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ma H, Zhang Y, Wang Q, Li Y, He J, Wang H,
Sun J, Pan K, Chen M and Xia J: Therapeutic safety and effects of
adjuvant autologous RetroNectin activated killer cell immunotherapy
for patients with primary hepatocellular carcinoma after
radiofrequency ablation. Cancer Biol Ther. 9:903–907. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee HJ, Lee YS, Kim HS, Kim YK, Kim JH,
Jeon SH, Lee HW, Kim S, Miyoshi H, Chung HM and Kim DK: Retronectin
enhances lentivirus-mediated gene delivery into hematopoietic
progenitor cells. Biologicals. 37:203–209. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lamers CH, van Elzakker P, van Steenbergen
SC, Sleijfer S, Debets R and Gratama JW: Retronectin-assisted
retroviral transduction of primary human T lymphocytes under good
manufacturing practice conditions: Tissue culture bag critically
determines cell yield. Cytotherapy. 10:406–416. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yu SS, Nukaya I, Enoki T, Chatani E, Kato
A, Goto Y, Dan K, Sasaki M, Tomita K, Tanabe M, et al: In vivo
persistence of genetically modified T cells generated ex vivo using
the fibronectin CH296 stimulation method. Cancer Gene Ther.
15:508–516. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang Z, Zhang Y, Liu Y, Wang L, Zhao L,
Yang T, He C, Song Y and Gao Q: Association of myeloid-derived
suppressor cells and efficacy of cytokine-induced killer cell
immunotherapy in metastatic renal cell carcinoma patients. J
Immunother. 37:43–50. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Watanabe H, Okada M, Kaji Y, Satouchi M,
Sato Y, Yamabe Y, Onaya H, Endo M, Sone M and Arai Y: New response
evaluation criteria in solid tumours-revised RECIST guideline
(version 1.1). Gan To Kagaku Ryoho. 36:2495–2501. 2009.(In
Japanese). PubMed/NCBI
|
|
27
|
Trotti A, Colevas AD, Setser A, Rusch V,
Jaques D, Budach V, Langer C, Murphy B, Cumberlin R, Coleman CN and
Rubin P: CTCAE v3.0: Development of a comprehensive grading system
for the adverse effects of cancer treatment. Semin Radiat Oncol.
13:176–181. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu L, Zhang W, Qi X, Li H, Yu J, Wei S,
Hao X and Ren X: Randomized study of autologous cytokine-induced
killer cell immunotherapy in metastatic renal carcinoma. Clin
Cancer Res. 18:1751–1759. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
DeVita VJ Jr and Rosenberg SA: Two hundred
years of cancer research. N Engl J Med. 366:2207–2214. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hontscha C, Borck Y, Zhou H, Messmer D and
Schmidt-Wolf IG: Clinical trials on CIK cells: First report of the
international registry on CIK cells (IRCC). J Cancer Res Clin
Oncol. 137:305–310. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yu X, Zhao H, Liu L, Cao S, Ren B, Zhang
N, An X, Yu J, Li H and Ren X: A randomized phase II study of
autologous cytokine-induced killer cells in treatment of
hepatocelluar carcinoma. J Clin Immunol. 34:194–203. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hui D, Qiang L, Jian W, Ti Z and Da-Lu K:
A randomized, controlled trial of postoperative adjuvant
cytokine-induced killer cells immunotherapy after radical resection
of hepatocellular carcinoma. Dig Liver Dis. 41:36–41. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shi M, Zhang B, Tang ZR, Lei ZY, Wang HF,
Feng YY, Fan ZP, Xu DP and Wang FS: Autologous cytokine-induced
killer cell therapy in clinical trial phase I is safe in patients
with primary hepatocellular carcinoma. World J Gastroenterol.
10:1146–1151. 2004.PubMed/NCBI
|
|
34
|
Pan K, Li YQ, Wang W, Xu L, Zhang YJ,
Zheng HX, Zhao JJ, Qiu HJ, Weng DS, Li JJ, et al: The efficacy of
cytokine-induced killer cell infusion as an adjuvant therapy for
postoperative hepatocellular carcinoma patients. Ann Surg Oncol.
20:4305–4311. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lee JH, Lee JH, Lim YS, Yeon JE, Song TJ,
Yu SJ, Gwak GY, Kim KM, Kim YJ, Lee JW and Yoon JH: Adjuvant
immunotherapy with autologous cytokine-induced killer cells for
hepatocellular carcinoma. Gastroenterology. 148:1383–1391.e6. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lu PH and Negrin RS: A novel population of
expanded human CD3+CD56+ cells derived from T cells with potent in
vivo antitumor activity in mice with severe combined
immunodeficiency. J Immunol. 153:1687–1696. 1994.PubMed/NCBI
|
|
37
|
Shablak A, Hawkins RE, Rothwell DG and
Elkord E: T cell-based immunotherapy of metastatic renal cell
carcinoma: Modest success and future perspective. Clin Cancer Res.
15:6503–6510. 2009. View Article : Google Scholar : PubMed/NCBI
|