Introduction
Despite improvements in treatment strategies for
oral squamous cell carcinoma (OSCC), it remains one of the most
devastating malignancies. OSCC is the sixth most common type of
cancer in the world and accounts for almost 3% of all cancer cases
(1–3).
Although significant advancements have been made in the prevention,
diagnostics and treatment strategies for OSCC, only modest progress
has been made in improving the survival time in patients with
progression or metastatic disease over the last 20 years (4,5). In 2012,
~300,400 new cases of oral cavity cancer, including lip cancer, and
~145,400 associated mortalities occurred worldwide (6). Promising markers are the basis for
improving the early detection and accurate survival evaluation of
patients with OSCC.
Cluster of differentiation (CD)73, also known as
5′-nucleotidase (5′-NT) or ecto-5′-NT, is a 70-kD,
glycosyl-phosphatidylinositol anchored cell surface enzyme that is
encoded by the 5′-nucleotidase ecto gene. CD73 was originally
defined as a lymphocyte differentiation antigen, and has both
enzymatic and non-enzymatic functions (7). CD73 catalyzes the dephosphorylation of
extracellular adenosine monophosphate to adenosine, promoting its
suppressive effects on the immune system in the tumor
microenvironment, invasion and metastasis of cancer (8). In addition to its enzymatic function,
CD73 is also an adhesive and signaling molecule that mediates
cancer invasion and metastasis (9).
Thus, both the enzymatic and non-enzymatic functions of CD73 are
involved in the processes of cancer occurrence and development.
Increased expression of CD73 expression has been
observed in several types of malignancy (10,11), such
as breast cancer, prostate cancer, bladder cancer and malignant
melanoma. In addition, it has prognostic value for patients with
colon cancer and has been suggested as a diagnostic factor in
papillary thyroid carcinoma (12).
However, there is almost no information available regarding the
survival influence of CD73 expression on tumor cells in patients
with SCC. The current study analyzed the association between CD73
expression and clinicopathological characteristics, including
disease-free survival (DFS) and overall survival (OS) time, in
patients with OSCC.
Materials and methods
Enrolled patients
Patients with OSCC who underwent surgery (combined
primary tumor resection and neck dissection/rescontruction) at the
Department of Oral and Maxillofacial-Head and Neck Oncology, Ninth
People's Hospital, Shanghai Jiao Tong University School of Medicine
(Shanghai, China) were included in the present study. The inclusion
criteria were as follows: i) Patients with OSCC (preoperative
pathological diagnosis); ii) patients that underwent primary
therapy in the Department of Oral and Maxillofacial-Head and Neck
Oncology; iii) patients that did not receive preoperative
chemotherapy, radiation therapy or any other treatment prior to
surgery; and iv) patients diagnosed between January 2007 and
December 2008. Cancerous and adjacent tumor samples were collected
immediately after surgery. The distance between the tumor and the
adjacent samples was >2 cm. According to the aforementioned
criteria, 113 patients were included in the present study. The
study was approved by the Ethics Committee of the Ninth People's
Hospital, Shanghai Jiao Tong University School of Medicine.
Informed consent was obtained from each patient.
Definition of OS and DFS
OS time was calculated from the date of initial
surgery to the date mortality from OSCC. If the patient did not
succumb to OSCC, the end point was defined as mortality from any
cause or the last date of follow-up. DFS was defined as the time
from the date of initial surgery to the date of local or distant
progression. If progression did not occur, the end point was
defined as mortality from any cause or as the fifth year after
surgery.
Immunohistochemical analysis
Sections (4 µm) were cut from formalin-fixed
(Mingsheng Disinfectant Co., Ltd., Chengdu, China) and
paraffin-embedded (Shanghai Hualing Recovery Appliance Factory,
Shanghai, China) tissues blocks of OSCC, and placed on positively
charged glass slides (Citotest Labware Manufacturing Co., Ltd.,
Haimen, China). Following paraffin removal with xylene (Sangon
Biotech Co., Ltd., Shanghai, China) and dehydration with ethanol
absolute (Ling Feng Chemical Reagent Co., Ltd., Shanghai, China),
the slides were steamed with 10 mmol/l citrate buffer (pH 6.0;
DakoCytomation, Carpinteria, CA, USA) for 20 min for antigen
retrieval. After cooling to room temperature, the sections were
incubated overnight at 4°C (15 h) with primary mouse monoclonal
anti-CD73 antibody (1:50 dilution; cat no. sc-32299; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA). After several washes with
phosphate-buffered saline, the sections were incubated with
horseradish peroxidase-labeled goat anti-mouse or ant-rabbit
secondary antibody (cat no. GK500705; Gene Tech, Shanghai, China)
for 45 min at 37°C, prior to adding diaminobenzidine (Dako,
Glostrup, Denmark) for 3.5 min at room temperature.
Using an Axio Scope.A1 microscope (Carl Zeiss AG,
Oberkocken, Germany), CD73 staining was independently evaluated by
an expert pathologist who was blinded to the clinical information.
The evaluation was analyzed according to the intensity of staining
and the percentage of stained cells. Scoring estimations were
stratified into four categories, as follows: +++, 20–49% moderately
to intensely stained cells or ≥50% positively stained cells; ++,
10–19% intensively stained cells or 20–49% weakly stained cells; +,
10–19% weakly to moderately stained cells; ±, <10% positively
stained cells; and -, 0% positively stained cells. For statistical
analysis, + and above were recorded as positive; - and ± were
ranked as negative (13).
Statistical analysis
Comparisons of variables between the groups were
based on the χ2 goodness-of-fit test or Fisher's exact
test. DFS and OS times were estimated by the Kaplan-Meier
life-table method. Univariate and multivariate parameters [gender,
age, smoking, alcohol consumption, tumor stage (T stage) (14), lymph node metastasis, clinical stage
(15), degree of differentiation]
were analyzed with respect to DFS and OS using the Cox regression
hazard model. Each experiment was repeated 3 times All analyses
were performed using SPSS software (version 13; SPSS Inc., Chicago,
IL, USA). P<0.05 was considered to indicate a statistically
significant difference.
Results
CD73 expression in primary OSCCs and
its correlation with clinicopathological characteristics
The characteristics of enrolled patients are
indicated in Table I. Positive
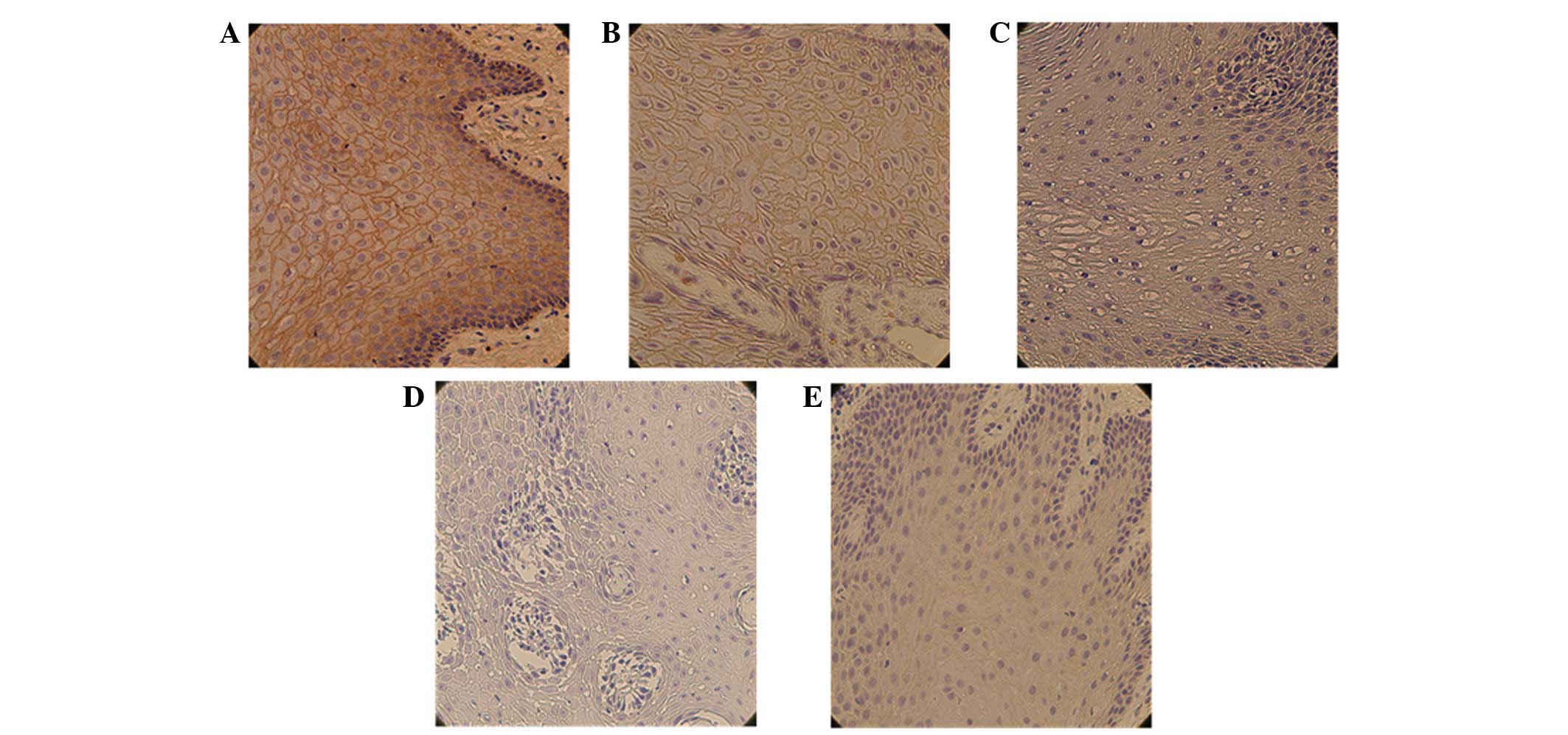
staining was noted in 58.4% (66/113) of OSCC samples. CD73 staining
was predominantly localized in the cytomembrane with a small amount
of staining in the cytoplasm. The OSCC samples showed marked
overexpression of CD73 compared with matched adjacent noncancerous
mucosa (Fig. 1). The association
between CD73 expression and clinicopathological variables are
indicated in Table II. Statistical
analysis of the association between CD73 expression and
clinicopathological characteristics identified that CD73 has a
direct association with T stage (P=0.021) and clinical stage
(P=0.047). CD73 expression was not significantly associated with
other characteristics.
 | Table I.Characteristics of the patients
enrolled in this study (n=113). |
Table I.
Characteristics of the patients
enrolled in this study (n=113).
| Characteristic | Value |
|---|
| Age, years |
|
| Median
(range) | 59.5 (26–83) |
| Gender, n (%) |
| Male | 71 (62.8) |
|
Female | 42 (37.2) |
| T stage, n (%) |
|
| T1 | 44 (38.9) |
| T2 | 45 (39.8) |
| T3 | 15 (13.3) |
| T4 | 9 (8.0) |
| Clinical stage, n
(%) |
|
| I | 23 (20.4) |
| II | 23 (20.4) |
| III | 31 (27.4) |
| IV | 36 (31.8) |
| Differentiation, n
(%) |
|
| Well | 24 (21.2) |
|
Moderate | 80 (70.8) |
| Poor | 9 (8.0) |
| CD73 cytology, n
(%) |
|
|
Positive | 66 (58.4) |
|
Negative | 47 (41.6) |
| Metastatic lymph
nodes, n (%) |
|
|
Positive | 36 (31.9) |
|
Negative | 77 (68.1) |
 | Table II.Association between
clinicopathological characteristics and CD73 expression in 113
patients with oral squamous cell carcinoma. |
Table II.
Association between
clinicopathological characteristics and CD73 expression in 113
patients with oral squamous cell carcinoma.
|
|
| CD73 expression,
% |
|
|
|---|
|
|
|
|
|
|
|---|
| Clinicopathological
feature | No. | Negative | Positive | χ2 | P-value |
|---|
| Gender |
|
|
| 0.999 | 0.331 |
| Male | 71 | 27 | 44 |
|
|
|
Female | 42 | 20 | 22 |
|
|
| Age, years |
|
|
| 2.004 | 0.184 |
|
<60 | 56 | 27 | 29 |
|
|
| ≥60 | 57 | 20 | 37 |
|
|
| Smoking |
|
|
| 0.065 | 0.844 |
| Yes | 40 | 16 | 24 |
|
|
| No | 73 | 31 | 42 |
|
|
| Drinking |
|
|
| 0.656 | 0.522 |
| Yes | 31 | 11 | 20 |
|
|
| No | 82 | 36 | 46 |
|
|
| T stage |
|
|
| 4.358 | 0.021 |
|
T3/T4 | 24 | 5 | 19 |
|
|
|
T1/T2 | 89 | 42 | 47 |
|
|
| Lymph node
metastasis |
|
|
| 0.159 | 0.838 |
| + | 36 | 14 | 22 |
|
|
| – | 77 | 33 | 44 |
|
|
| Clinical stage |
|
|
| 3.941 | 0.047 |
|
III/IV | 46 | 14 | 32 |
|
|
|
I/II | 67 | 33 | 34 |
|
|
| Histological
type |
|
|
| 0.778 | 0.378 |
|
Poor | 9 | 5 | 4 |
|
|
|
Well/moderate | 104 | 42 | 62 |
|
|
Association between CD73 expression,
and DFS and OS times among patients with OSCC
Using follow-up data from the 113 patients with
OSCC, the present study analyzed whether CD73 expression affected
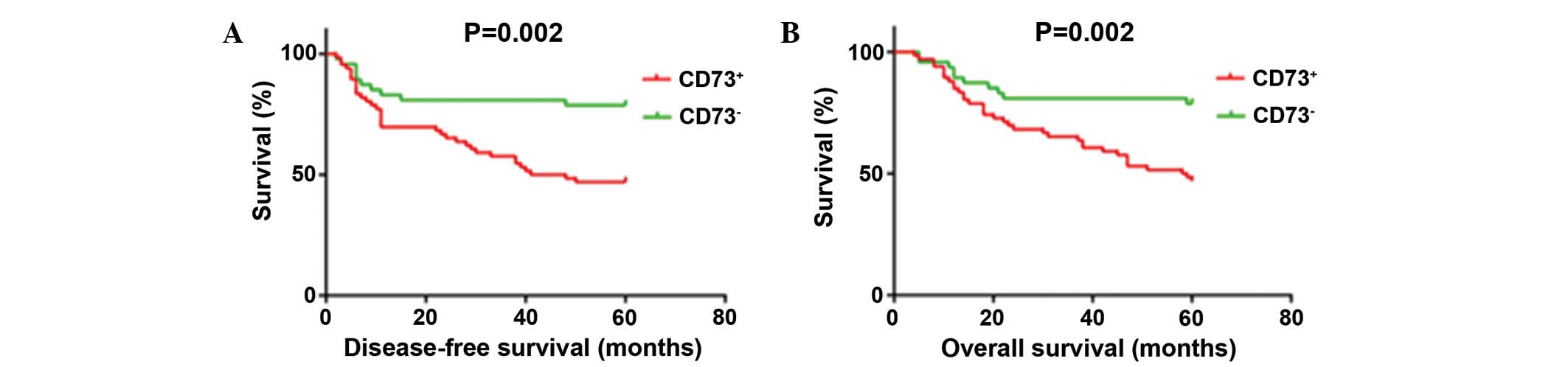
DFS and OS. Kaplan-Meier survival curves showed that CD73-positive
expression cases had significantly poorer DFS (P=0.002) and OS
(P=0.002) times compared with CD73-negative cases. The 5-year DFS
rate for positive CD73 expression was 47.0 compared with 78.7% for
patients that were negative for CD73; similarly, the 5-year OS
rates for CD73-positive and -negative expression were 50.0 vs.
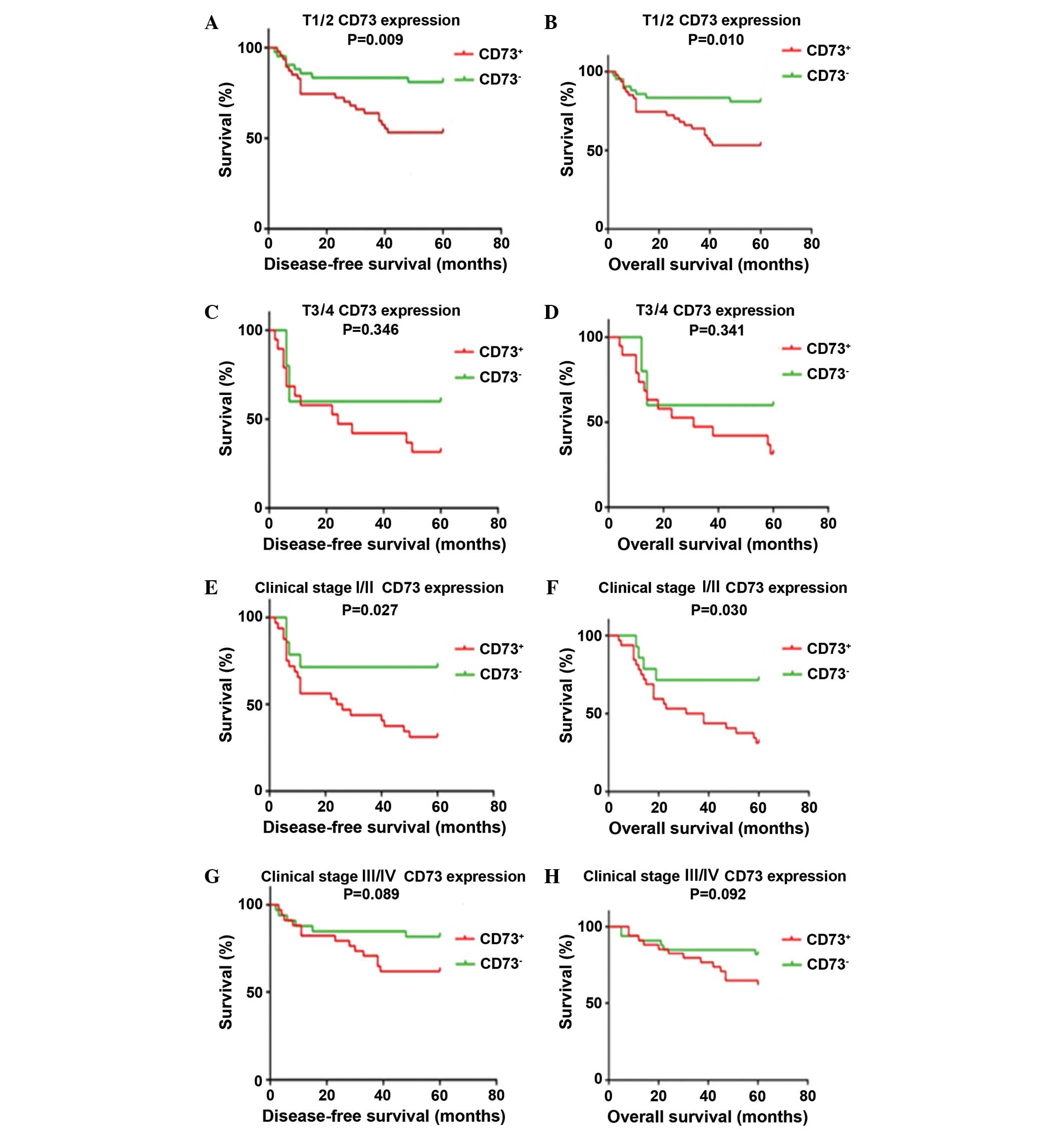
78.7%, respectively (Fig. 2). After
patients were stratified by T stage, cases with positive CD73
expression showed significantly poorer DFS and OS compared with
CD73-negative cases among those classified as T1/T2 or clinical
stage I/II. This trend was not observed in T3/T4 and clinical stage
III/IV patients with OSCC (Fig.
3).
CD73 is an independent poor prognostic
marker for DFS and OS times among patients with OSCC
Univariate Cox regression analysis showed that CD73
expression was significantly associated with poor DFS (HR, 2.926;
95% CI, 1.447–5.917; P=0.003) and OS (HR, 2.936; 95% CI,
1.452–5.936; P=0.003) times of patients with OSCC (Table III). Multivariate analysis using the
Cox regression hazard model confirmed that CD73 expression was an
independent prognostic factor for poor DFS (HR, 2.417; 95% CI,
1.162–5.028; P=0.018) and OS (HR, 2.355; 95% CI, 1.137–4.878;
P=0.021; Table IV) among patients
with OSCC. Multivariate analysis also indicated that T stage,
clinical stage, degree of differentiation and lymph node metastasis
have independent prognostic value in OSCC (Table IV).
 | Table III.Statistical analysis of
clinicopathological variables associated with DFS and OS in
patients with oral squamous cell carcinoma using the univariate Cox
proportional hazards model. |
Table III.
Statistical analysis of
clinicopathological variables associated with DFS and OS in
patients with oral squamous cell carcinoma using the univariate Cox
proportional hazards model.
|
| DFS | OS |
|---|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| CD73
expression |
|
| 0.003 |
|
| 0.003 |
|
Positive vs. negative | 2.926 | 1.447–5.917 |
| 2.936 | 1.452–5.936 |
| Gender |
|
| 0.584 |
|
| 0.567 |
| Male
vs. female | 1.189 | 0.640–2.210 |
| 1.199 | 0.645–2.228 |
| Age, years |
|
| 0.060 |
|
| 0.057 |
| <60
vs. ≥60 | 1.027 | 0.999–1.055 |
| 1.027 | 0.999–1.056 |
| Smoking |
|
| 0.751 |
|
| 0.751 |
| Yes vs.
no | 0.904 | 0.487–1.681 |
| 0.904 | 0.487–1.681 |
|
Drinking |
|
| 0.905 |
|
| 0.913 |
| Yes vs.
no | 0.961 | 0.496–1.860 |
| 0.964 | 0.498–1.866 |
| T stage |
|
| 0.000 |
|
| 0.000 |
| T3/T4
vs. T1/T2 | 1.797 | 1.342–2.408 |
| 1.816 | 1.356–2.432 |
| Lymph node
metastasis |
|
| 0.000 |
|
| 0.000 |
| + vs.
− | 1.897 | 1.344–2.687 |
| 1.915 | 1.358–2.701 |
| Clinical stage |
|
| 0.000 |
|
| 0.000 |
| III/IV
vs. I/II | 1.677 | 1.282–2.194 |
| 1.698 | 1.298–2.220 |
| Histological
type |
|
| 0.008 |
|
| 0.009 |
| Poor
vs. well-moderate | 2.174 | 1.227–3.853 |
| 2.134 | 1.213–3.754 |
 | Table IV.Statistical analysis of
clinicopathological variables associated with DFS and OS in
patients with oral squamous cell carcinoma using the multivariate
Cox proportional hazards model. |
Table IV.
Statistical analysis of
clinicopathological variables associated with DFS and OS in
patients with oral squamous cell carcinoma using the multivariate
Cox proportional hazards model.
|
| DFS |
| OS |
|
|---|
|
|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| CD73
expression |
|
| 0.018 |
|
| 0.021 |
|
Positive vs. negative | 2.417 | 1.162–5.028 |
| 2.355 | 1.137–4.878 |
| Gender |
|
| 0.947 |
|
| 0.897 |
| Male
vs. female | 1.025 | 0.486–2.165 |
| 1.050 | 0.498–2.214 |
| Age, years |
|
| 0.087 |
|
| 0.070 |
| <60
vs. ≥60 | 1.028 | 0.996–1.061 |
| 1.030 | 0.998–1.063 |
| Smoking |
|
| 0.757 |
|
| 0.715 |
| Yes vs.
no | 0.852 | 0.308–2.353 |
| 0.823 | 0.289–2.343 |
| Drinking |
|
| 0.681 |
|
| 0.715 |
| Yes vs.
no | 0.795 | 0.267–2.369 |
| 0.810 | 0.262–2.505 |
| T stage |
|
| 0.002 |
|
| 0.003 |
| T3/T4
vs. T1/T2 | 2.735 | 1.428–5.236 |
| 2.598 | 1.369–4.932 |
| Lymph node
metastasis |
|
| 0.002 |
|
| 0.003 |
| + vs.
− | 2.775 | 1.443–5.337 |
| 2.633 | 1.376–5.039 |
| Clinical stage |
|
| 0.040 |
|
| 0.060 |
| I/II
vs. III/IV | 0.464 | 0.223–0.964 |
| 0.500 | 0.243–1.030 |
| Histological
type |
|
| 0.025 |
|
| 0.045 |
| Poor
vs. well/moderate | 2.569 | 1.127–5.857 |
| 2.269 | 1.019–5.049 |
|
Discussion
To the best of our knowledge, the current study is
the first to evaluate the expression status of CD73 in patients
with OSCC. The associations between CD73 expression and
clinicopathological characteristics were evaluated in patients with
OSCC. The findings indicated that positive CD73 expression may be a
novel prognosticator of adverse clinical outcome in patients with
OSCC. In addition, the present results identified that CD73
expression status was statistically associated with the T stage,
clinical stage, degree of differentiation and lymph node
metastasis. According to these findings, we propose that CD73 may
be a novel molecular prognostic marker in the evaluation of OSCC
patient survival. The current findings may be the beginning of a
new era of research into the role of CD73 in SCC.
Overexpression of CD73 has been observed in broad
types of cancer (16) and a growing
body of literature has revealed the function of CD73 in cancer
progression. For example, overexpression of CD73 was significantly
associated with a worse prognosis in patients with triple negative
breast cancer (17). By contrast, a
number of retrospective studies reported that overexpression of
CD73 was strongly correlated with improved clinical outcome in
patients with breast cancer (18,19).
Furthermore, Lu et al (20)
investigated the expression status of CD73 in gastric cancer, and
showed that CD73 expression was positively associated with cancer
stage, depth of invasion and metastasis, with low OS observed in
the patients with overexpression of CD73. In addition, numerous
studies have revealed that high levels of CD73 are statistically
associated with a poor prognosis in colorectal cancer and
gallbladder cancer (21,22). Zhao et al (23) reported the CD73 status in various
leukemia subtypes, and found the CD73 status was correlated with
leukemia subtype and differentiation. In addition, a retrospective
study reported that CD73 overexpression was positively correlated
with lymph node metastasis in prostate cancer, and was more
frequently observed in epithelial ovarian cancer patients with
better prognosis, lower stage and better differentiation (19,24).
Similar results were observed in malignant melanoma (25). Taken together, the aforementioned
findings indicate that CD73 is a significant molecular
prognosticator in various types of cancer. However, there is almost
no information regarding CD73 expression in the tumor cells of
patients with SCC. Therefore, the current study aimed to conduct
research in this field.
T stage and clinical stage are the most important
prognostic markers in the evaluation of OSCC patient survival
(26,27). The current findings confirmed the
validity of the T stage and clinical stage at the molecular level.
Furthermore, CD73 may be a crucial molecular prognostic marker in
early T stage (T1/T2) or early clinical stage (I/II) OSCC.
The limitations of the present study include the
retrospective nature of the study and the limited number of
enrolled patients. The findings of the study should be replicated
in a randomized prospective study that includes a large sample
size. Additional molecular studies are required to confirm whether
CD73 has an important role in OSCC progression. The current authors
are currently performing a further molecular study to confirm
whether CD73 has an important role in OSCC.
In conclusion, the results of the present study
revealed upregulation of CD73 in clinical OSCC tissues. In
addition, it was identified that the grading of CD73 expression by
immunohistochemical staining was able to significantly predict OSCC
prognosis in a multivariate analysis. Furthermore, the CD73
expression status was significantly associated with T stage and
clinical stage, and overexpression of CD73 was inversely correlated
with the DFS and OS times of patients with OSCC. Thus, the results
of the present study consistently suggest that CD73 is likely to be
a novel prognostic marker of OSCC and holds promise for future
tailored treatment strategies.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Warnakulasuriya S: Global epidemiology of
oral and oropharyngeal cancer. Oral Oncol. 45:309–316. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ren Z and Wu H: Advances in radical
surgery of tongue cancer. J Prac Stomatology. 30:110–114. 2014.(In
Chinese).
|
|
4
|
Silverman S Jr: Demographics and
occurrence of oral and pharyngeal cancers. The outcomes, the
trends, the challenge. J Am Dent Assoc. 132(Suppl): S7–S11. 2001.
View Article : Google Scholar
|
|
5
|
Chandler K, Vance C, Budnick S and Muller
S: Muscle invasion in oral tongue squamous cell carcinoma as
predictor of nodal status and local recurrence: Just as effective
as depth of invasion? Head Neck Pathol. 5:359–363. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Stagg J and Smyth MJ: Extracellular
adenosine triphosphate and adenosine in cancer. Oncogene.
29:5346–5358. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ghiringhelli F, Bruchard M, Chalmin F and
Rébé C: Production of adenosine by ectonucleotidases: A key factor
in tumor immunoescape. J Biomed Biotechnol. 2012:4737122012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhi X, Chen S, Zhou P, Shao Z, Wang L, Ou
Z and Yin L: RNA interference of ecto-5′-nucleotidase (CD73)
inhibits human breast cancer cell growth and invasion. Clin Exp
Metastasis. 24:439–448. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang L, Tang S, Wang Y, Xu S, Yu J, Zhi X,
Ou Z, Yang J, Zhou P and Shao Z: Ecto-5′-nucleotidase (CD73)
promotes tumor angiogenesis. Clin Exp Metastasis. 30:671–680. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Arozarena I, Sanchez-Laorden B, Packer L,
Hidalgo-Carcedo C, Hayward R, Viros A, Sahai E and Marais R:
Oncogenic BRAF induces melanoma cell invasion by downregulating the
cGMP-specific phosphodiesterase PDE5A. Cancer Cell. 19:45–57. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang B: CD73 promotes tumor growth and
metastasis. Oncoimmunology. 1:67–70. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Xing XF, Li H, Zhong XY, Zhang LH, Wang
XH, Liu YQ, Jia SQ, Shi T, Niu ZJ, Peng Y, et al: Phospholipase A2
group IIA expression correlates with prolonged survival in gastric
cancer. Histopathology. 59:198–206. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Patel SG and Shah JP: TNM staging of
cancers of the head and neck: striving for uniformity among
diversity. CA Cancer J Clin. 55:242–258; quiz 261–262, 264. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Greene FL and Sobin LH: The staging of
cancer: A retrospective and prospective appraisal. CA Cancer J
Clin. 58:180–190. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gao ZW, Dong K and Zhang HZ: The roles of
CD73 in cancer. Biomed Res Int. 2014:4606542014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Loi S, Pommey S, Haibe-Kains B, Beavis PA,
Darcy PK, Smyth MJ and Stagg J: CD73 promotes anthracycline
resistance and poor prognosis in triple negative breast cancer.
Proc Natl Acad Sci USA. 110:11091–11096. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Supernat A, Markiewicz A,
Welnicka-Jaskiewicz M, Seroczynska B, Skokowski J, Sejda A, Szade
J, Czapiewski P, Biernat W and Zaczek A: CD73 expression as a
potential marker of good prognosis in breast carcinoma. Appl
Immunohistochem Mol Morphol. 20:103–107. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Oh HK, Sin JI, Choi J, Park SH, Lee TS and
Choi YS: Overexpression of CD73 in epithelial ovarian carcinoma is
associated with better prognosis, lower stage, better
differentiation and lower regulatory T cell infiltration. J Gynecol
Oncol. 23:274–281. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lu XX, Chen YT, Feng B, Mao XB, Yu B and
Chu XY: Expression and clinical significance of CD73 and
hypoxia-inducible factor-1α in gastric carcinoma. World J
Gastroenterol. 19:1912–1918. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu XR, He XS, Chen YF, Yuan RX, Zeng Y,
Lian L, Zou YF, Lan N, Wu XJ and Lan P: High expression of CD73 as
a poor prognostic biomarker in human colorectal cancer. J Surg
Oncol. 106:130–137. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xiong L, Wen Y, Miao X and Yang Z: NT5E
and FcGBP as key regulators of TGF-1-induced epithelial-mesenchymal
transition (EMT) are associated with tumor progression and survival
of patients with gallbladder cancer. Cell Tissue Res. 355:365–374.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhao SX, Zhang HM, Dong SX, Liu JH, Zhou
Z, Wang HJ, Zhu XF, Mi YC and Ru YX: Characteristics and clinical
significance of CD73 expression in subtypes of leukemia. Zhongguo
Shi Yan Xue Ye Xue Za Zhi. 19:1141–1144. 2011.(In Chinese).
PubMed/NCBI
|
|
24
|
Yang Q, Du J and Zu L: Overexpression of
CD73 in prostate cancer is associated with lymph node metastasis.
Pathol Oncol Res. 19:811–814. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang H, Lee S, Nigro CL, Lattanzio L,
Merlano M, Monteverde M, Matin R, Purdie K, Mladkova N, Bergamaschi
D, et al: NT5E (CD73) is epigenetically regulated in malignant
melanoma and associated with metastatic site specificity. Br J
Cancer. 106:1446–1452. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jardim JF, Francisco AL, Gondak R,
Damascena A and Kowalski LP: Prognostic impact of perineural
invasion and lymphovascular invasion in advanced stage oral
squamous cell carcinoma. Int J Oral Maxillofac Surg. 44:23–28.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lawaetz M and Homøe P: Risk factors for
and consequences of inadequate surgical margins in oral squamous
cell carcinoma. Oral Surg Oral Med Oral Pathol Oral Radiol.
118:642–646. 2014. View Article : Google Scholar : PubMed/NCBI
|