Introduction
DNA polymerase β (polβ) is a highly conservative
gene that is expressed in a low level and constant manner in
mammalian animal cell cycles (1). DNA
polβ is not a major DNA polymerase in the DNA synthesis process, as
it lacks intrinsic nuclease activities; however, the specific role
of DNA polβ is to participate in DNA base excision repair (BER) for
maintaining genomic stability (2,3). DNA polβ
is highly expressed when genomic DNA is attacked by alkylating
agents, ultraviolet radiation or endogenous factors (4–6). However,
overexpressed polβ can also participate in DNA synthesis, which is
an important mechanism of genetic instability due to the inaccuracy
of its base pairing (7). Accumulating
evidence also indicates that overexpressed polβ is associated with
cell transformation, tumor genesis and chemotherapeutic resistance
(8–10). The overexpression of polβ was
identified in various human cancers, including esophageal
carcinoma, gastric carcinoma and ovarian cancer (8,9,11). Although the overexpression of polβ is
commonly observed in human cancers, the regulation of polβ and its
association with the signaling pathway remains elusive. Answers to
these questions may benefit our understanding of tumorigenesis and
provide information regarding novel ways for chemoprevention or
chemotherapy.
A previous study showed that alternariol (AOH), a
source of DNA damage, could induce DNA polβ expression through the
protein kinase A (PKA) signal transduction pathway, and that the
PKA inhibitor, H89, can partly block polβ expression (12). In addition, mitogen-activated protein
kinase 14 (p38) mitogen-activated protein kinase (MAPK) was
activated by AOH in NIH3T3 cells (12). Activating transcription factor 2
(ATF2) is one of the downstream molecules of the MAPK signal
pathway and can be activated by p38MAPK and mitogen-activated
protein kinase 8 (JNK) (13,14). ATF2 is a member of ATF or
cAMP-responsive element binding protein (CREB) family, which
contains the basic/leucine zipper motifs (bZIP) and can bind to
cAMP-responsive elements (CREs) (15,16). The
DNA polβ promoter also contains the CRE sequence (17). Therefore the MAPK-ATF2 signaling
pathway may be involved in polβ expression. In the present study,
DNA damage was found to be associated with polβ expression induced
by alternariol (AOH), and p38MAPK and ATF2 activation was detected.
When cells were pretreated with the p38MAPK inhibitor SB203580, the
level of phosphorylation of p38MAPK and ATF2 was decreased, and the
expression of polβ was reduced. This finding is consistent with the
result that polβ expression decreased in NIH3T3 cells with p38MAPK
knockdown.
Materials and methods
Chemicals and reagents
AOH and dimethyl sulfoxide (DMSO) were purchased
from Sigma-Aldrich, Inc. (St. Louis, MO, USA). Anti-phosphorylated
(p-)p38MAPK [rabbit polyclonal immunoglobulin G (IgG); catalog no.,
sc-17852-R], anti-DNA polβ (goat polyclonal IgG; catalog no.,
sc-5927), anti-β-actin (mouse monoclonal; catalog no., sc-47778)
and anti-α-tubulin (mouse monoclonal; catalog no., sc-23948)
primary antibodies and goat anti-rabbit IgG-horseradish peroxidase
(HRP) (catalog no., sc-2004), rabbit anti-mouse IgG-HRP (catalog
no., sc-358914) and donkey anti-goat IgG-HRP (sc-2020) secondary
antibodies were obtained from Santa Cruz Biotechnology, Inc.
(Dallas, TX, USA). Anti-p-ATF2 (rabbit polyclonal; catalog no.,
CST9225) was obtained from Cell Signaling Technology, Inc.
(Danvers, MA, USA). RPMI-1640 and fetal bovine serum (FBS) was
purchased from Thermo Fisher Scientific, Inc. (Waltham, MA, USA).
SB203580 and the Enhanced Chemiluminescence (ECL) Plus Detection
kit were purchased from Beyotime Institute of Biotechnology
(Haimen, China). Reagents for protein concentration analysis were
obtained from Bio-Rad Laboratories, Inc. (Hercules, CA, USA).
Cell culture
The NIH3T3 cell line was obtained from China Center
for Type Culture Collection (Wuhan, China). The cells were cultured
in RPMI-1640 with 10% FBS, 100 units (U)/ml penicillin and 100
µg/ml streptomycin at 37°C and 5% CO2 in a humidified
incubator.
Comet assay
To detect the DNA damage of NIH3T3 cells induced by
various concentrations of AOH, an alkaline comet assay was
performed. Cells (2×106) were cultured in 10-cm dishes
for ~48 h. When the cells reached 80% confluence, they were treated
with 2, 10 or 20 µM AOH or DMSO for 2 h. Fully frosted slides were
then coated with 1% normal melting agarose at 45°C as a base layer,
and were solidificated at 4°C for 10 min. The cells were suspended
in 0.8% low melting agarose at 37°C. This suspension was pipetted
over the base layer, which was covered with a cover slip
immediately. The slides were placed at 4°C for 10 min, and then the
cover slips were removed. The slides were kept in the lysis buffer
(pH 10) at 4°C for 1 h and transferred into electrophoretic running
buffer (pH 13) for unwinding for 30 min. Electrophoresis was
performed in the same buffer for 20 min at 0.8 v/cm. Subsequently,
the slides were washed with neutralizing buffer and stained with
ethidium bromide (20 µg/ml) for 5 min, and washed with cold saline.
Finally, the glycerine buffer was placed on the slides in a
dropwise manner and cover slips were placed on top. The slides were
visualized using a fluorescence microscope (Olympus, Tokyo,
Japan).
Western blotting
Cells (2×106) were cultured in 10-cm
dishes for ~48 h. When the cells reached 80% confluence, they were
treated with various concentrations (2, 10 or 20 µM) of AOH, cells
were lysed in radioimmunoprecipitation assay buffer [50 mm
Tris/HCl, pH 7.5, 150 mM NaCl, 1 mM ethylenediaminetetraacetic
acid, 1 mM Na3VO4, 0.1% sodium dodecyl
sulfate (SDS), 1% (v/v) NP-40, 1 mM NaF, containing complete
protease and phosphatase inhibitor cocktail (Roche Diagnostics,
Indianapolis, IN, USA)], and supernatants were collected by
centrifugation at 21,341 × g for 20 min. Cell lysate (50 µg) was
separated on a 10% SDS-polyacrylamide gel and transferred to a
polyvinylidene fluoride membrane. After blocking with 5% nonfat
dried milk for 1 h, the membrane was incubated with a primary
antibody at 4°C overnight (anti-p-p38MAPK, 1:200 dilution;
anti-p-ATF2, 1:1,000 dilution; anti-DNA polβ, 1:200 dilution;
anti-α-tubulin and anti-β-actin, 1:1,000 dilution). IgG-HRP
secondary antibodies were incubated for 2 h at room temperature.
Protein bands were visualized using the ECL Plus Detection kit. The
bands were subjected to densitometry for quantitative analysis
using Quantity One software (Bio-Rad Laboratories, Inc.).
p38MAPK-small hairpin RNA (shRNA)
lentiviral vector
The pLKO.1, pCMV-dR8.2 and pCMV-VSVG vectors were
kindly provided by Dr Zigang Dong (The Hormel Institute, Austin,
MN, USA). The lentivirus-based shRNA-expressing vectors was a
pLKO.1 back bone with the human U6 promoter driving RNA polymerase
III transcription. The puromycin resistance marker encoded in
pLKO.1 allows for convenient stable selection. The shRNA sequence
of p38MAPK (5′-ATACATTCGGCTGACATAATT-3′) was cloned into the
lentiviral shRNA-expressing vectors at the AgeI and EcoRI
restriction sites (New England Biolabs Ltd., Beijing, China), and
were confirmed by direct DNA sequencing (Genewiz Inc., Beijing,
China).
The lentiviral vector containing the p38MAPK-shRNA,
a packaging vector (pCMV-dR8.2) and an envelope vector (pCMV-VSVG)
were co-transfected into 293T cells using Lipofectamine 2000
(Invitrogen; Thermo Fisher Scientific, Inc.). After 24 h and 48 h,
the viral supernatants were harvested and added to the NIH3T3
cells. Subsequently, 2 mg/ml puromycin was used to select the
stably infected cells.
Statistics
All results were presented as mean ± standard
deviation of the mean. Statistics analysis was performed using
analysis of variance in SPSS 11.0 software (SPSS, Inc., Chicago,
IL, USA). P<0.05 was considered to indicate statistical
significance.
Results
AOH-induced DNA damage
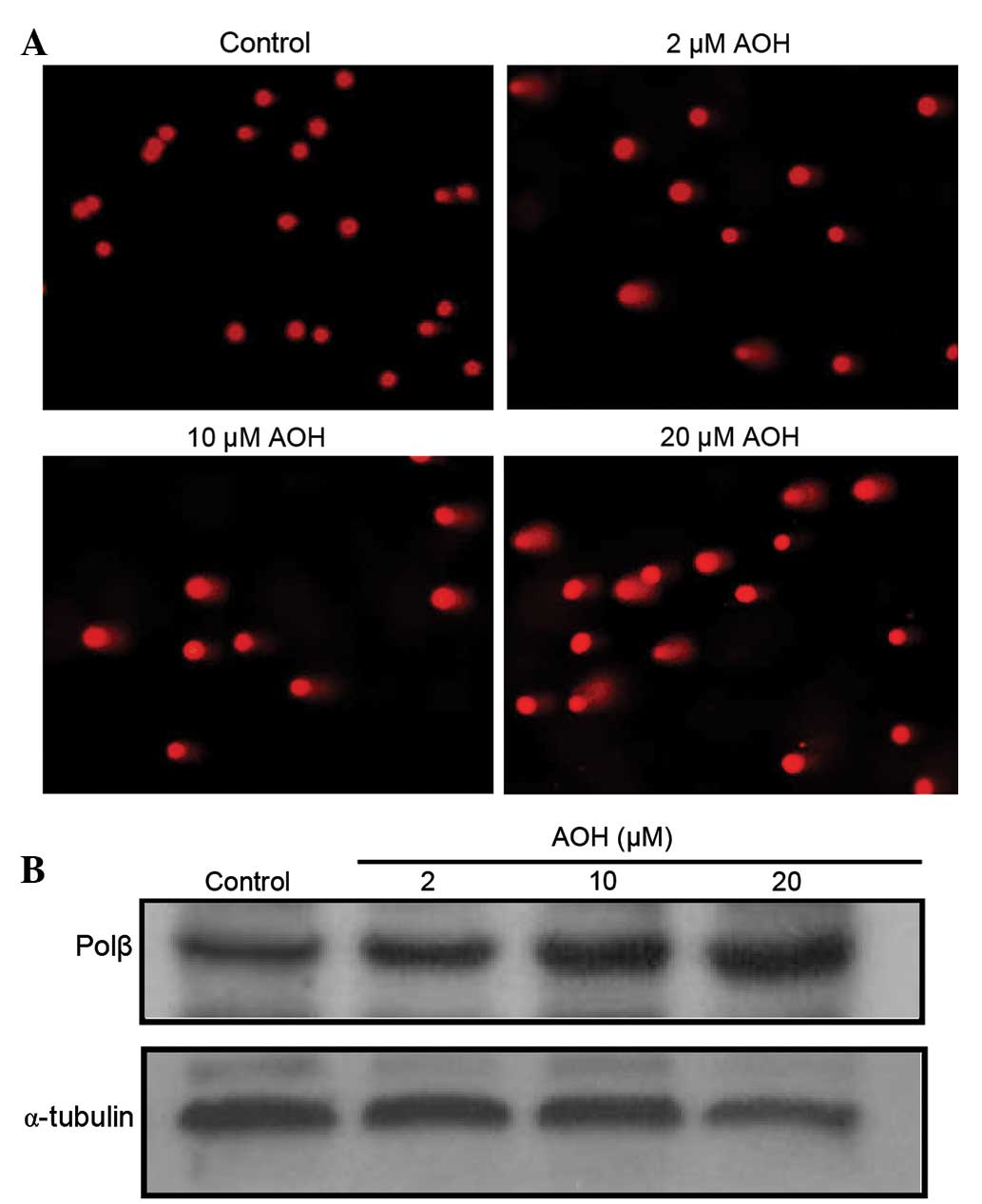
To investigate the DNA toxic effect of AOH on NIH3T3
cells, a comet assay was conducted. NIH3T3 cells were treated with
DMSO or 2, 10 or 20 µM AOH for 2 h. The results of single cell gel
electrophoresis showed that 10 and 20 µM AOH increased the length
and area of comet tails, a phenotype of DNA damage (Fig. 1A). DNA polβ is a polymerase that
participates in the repair of DNA strand breaks. Thus, the present
study investigated whether or not DNA polβ was activated in DNA
damage induced by AOH. NIH3T3 cells were treated with AOH. Western
blotting was performed to determine polβ protein level with various
doses of AOH treatment. The results showed that the protein level
of polβ protein increased in a dose-dependent manner with AOH
treatment compared with the control (Fig.
1B).
AOH-induced activation of p38MAPK and
ATF2
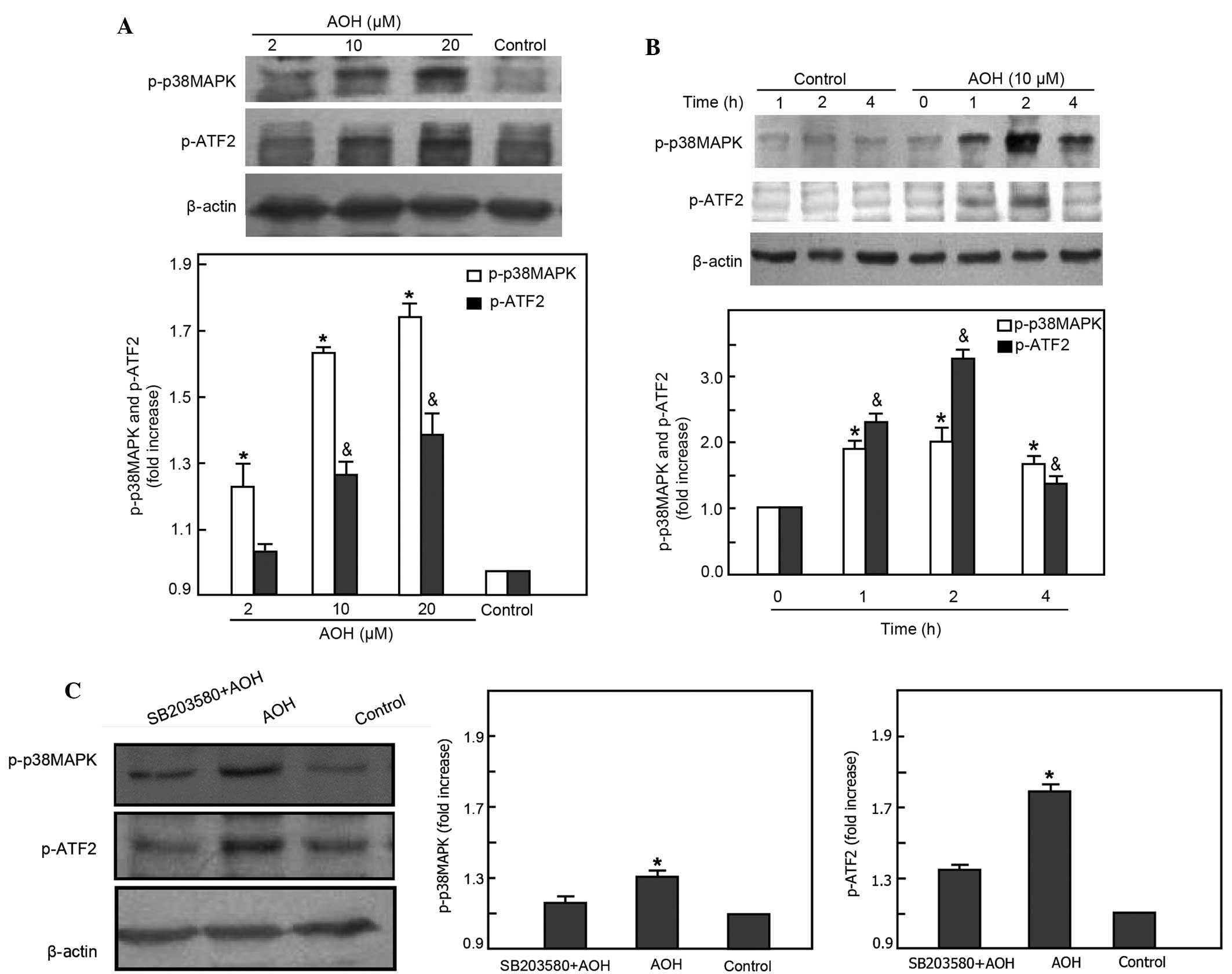
Western blotting was used to determine the level of
p38MAPK phosphorylation after AOH treatment at various doses and
time points. Results showed that the activation of p38MAPK
increased in a dose-dependent manner (2 µM AOH group, P=0.002 vs.
control group; 10 µM AOH group, P<0.001 vs. control group; 20 µM
AOH group, P<0.001 vs. control group) (Fig. 2A). The level of p38MAPK
phosphorylation was highest when the NIH3T3 cells were treated with
10 µM AOH at 2 h (1 h group, P<0.001 vs. control group; 2 h
group, P<0.001 vs. control group; 4 h group, P<0.001 vs.
control group) (Fig. 2B). SB203580,
an inhibitor of p38MAPK, can effectively block the phosphorylation
of p38MAPK. To determine whether AOH induces the phosphorylation of
p38MAPK, the NIH3T3 cells were pretreated with 10 µM SB203580 for 1
h, then treated with 10 µM AOH. Western blot analysis showed a
significant decrease of p38MAPK phosphorylation in SB203580
pretreatment group compared with the AOH-only treated group
(SB203580+AOH group, P=0.013 vs. control group; AOH group,
P<0.001 vs. control group) (Fig.
2C).
ATF2 is a transcription factor that can be activated
by p38MAPK. Activated p38MAPK can phosphorylate ATF2 in the cell
stress response process. To further identify the activation of ATF2
evoked by phosphorylated p38MAPK with AOH treatment, western
blotting was performed to determine the level of ATF2
phosphorylation when NIH3T3 cells were treated with various doses
of AOH at different time points. The results were consistent with
the change of p38MAPK phosphorylation, which revealed that
phosphorylation occurred in a dose-dependent manner and that the
peak activation of ATF2 occurred at the same time as p38MPAK (in
dose groups, 2 µM AOH group, P=0.384 vs. control group; 10 µM AOH
group, P=0.001 vs. control group; 20 µM AOH group, P<0.001 vs.
control group; in time groups, 1 h group, P<0.001 vs. control
group; 2 h group, P<0.001 vs. control group; 4 h group, P=0.001
vs. control group) (Fig. 2A and B).
Furthermore, the level of ATF2 phosphorylation decreased in the
SB203580 pretreatment group compared with the AOH-only treated
group (SB203580+AOH group, P=0.057 vs. control group; AOH group,
P<0.001 vs. control group) (Fig.
2C).
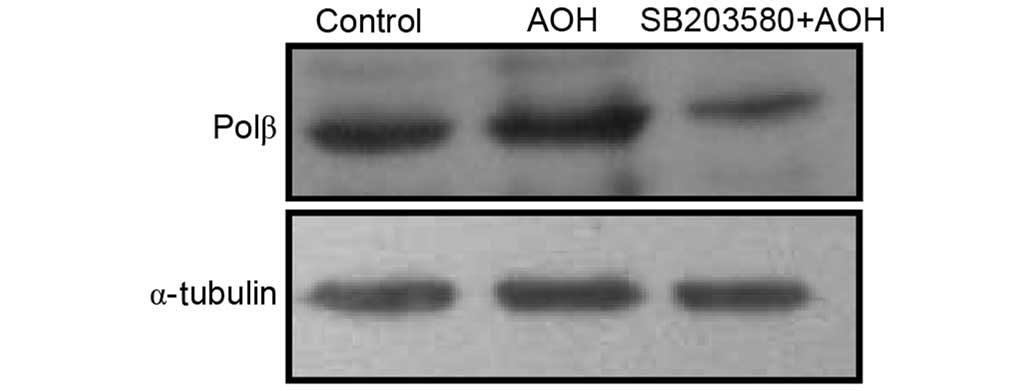
Subsequently, the AOH-induced phosphorylation of
p38MAPK and ATF2 was assessed for associations with the expression
level of polβ. Polβ expression was observed in western blotting
assays when cells were pretreated with SB203580, an inhibitor of
p38MAPK. The expression level of polβ was found to decrease in the
pretreatment group compared with the AOH stimulation group
(Fig. 3).
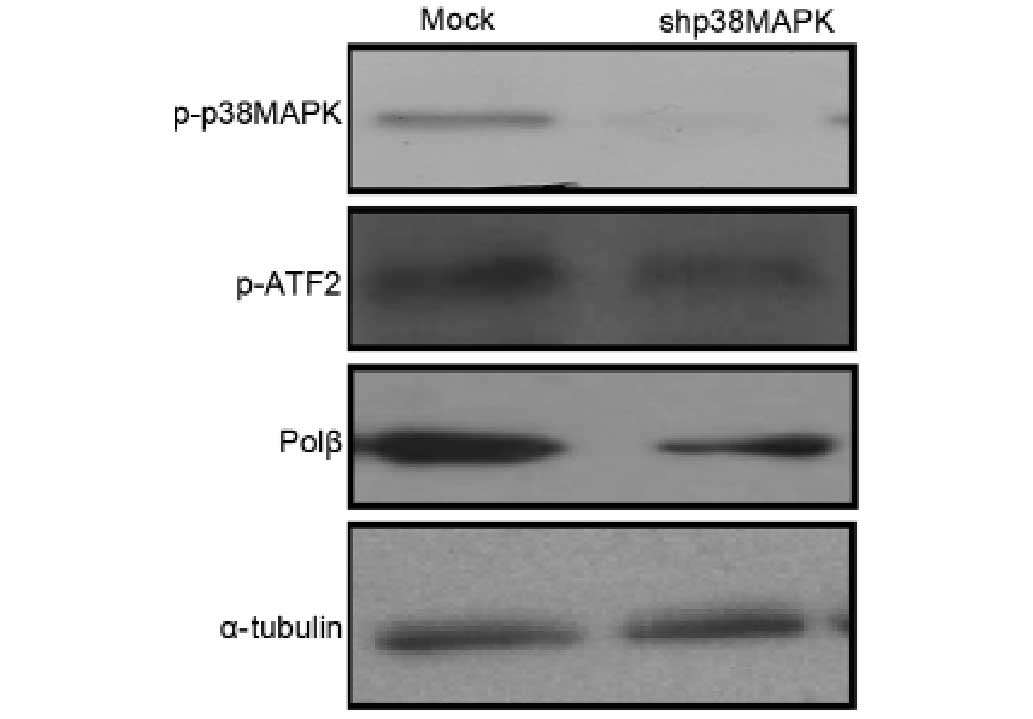
To further verify the role of the p38MAPK-ATF2
pathway in polβ expression, p38MAPK knockdown was performed by
lentiviral shRNA transfection. The results of the western blotting
showed that p38MAPK knockdown was associated with the reduced
phosphorylation of ATF2 and the decreased expression of polβ
(Fig. 4).
Discussion
The present study investigated the role of the
signaling transduction pathway of p38MAPK and ATF2 in the
expression of the DNA polβ gene, induced by AOH. When this
signaling pathway was blocked, the level of expression of DNA polβ
decreased. In a previous study, AOH was indicated to induce the
overexpression of DNA polβ through the activation of the PKA-CREB
pathway in NIH3T3 cells (12);
however, when H89 (18), a inhibitor
of PKA, was used to pretreat the cells, DNA polβ expression was
observed to partly decrease, rather than entirely. The results of
the present study suggest that p38MAPK-ATF2 may also be involved in
DNA polβ expression when NIH3T3 cells are stimulated by AOH.
MAPK family members can respond to diverse stimuli,
and p38MAPK, one of four subgroups in the MAPK family, is important
for cellular essential activity (19). p38MAPK can phosphorase numerous
transcription factors and regulate gene expression. ATF2 is one of
the downstream molecules of p38MAPK that belongs to the ATF/CREB
transcription factor family and can recognize and bind to the CRE
sequence as dimers in the promoters of certain genes (15). The DNA polβ gene promoter includes a
CRE site with the 8 bp palindromic sequence TGA CGT CA. In the
study by Kreideweiss et al (20), Ca2+-mediated intracellular
signaling pathways events in human peripheral blood mononuclear
cells (PBMCs) activated MAPK cascades and phosphorylated
transcription factor ATF2. The study found that ATF2 and CREB can
bind to the CRE site of the DNA polβ promoter and ATF2 contributes
to the expression of the polβ gene (20). However, p38 was not the kinase
mediating ATF2 phosphorylation upon stimulation of primary PBMCs
with Ca2+, as the induction of ATF2 phosphorylation was
not affected by the presence of the p38a inhibitor SB203580 at 10
µM concentrations (20). This result
is in contrast to the results of the present study. In the current
study, AOH stimulated ATF2 activation, followed by p38
phosphorylation. Chyan et al (21) identified a novel ATF2 isoform, which
encodes a 60-kDa protein with an incomplete N-terminal domain, and
a novel C-terminal region, but an intact bZIP domain, which binds
to the human polβ CRE and functions as a repressor of the cloned
human polβ promoter in 293 cells. Interestingly, the results of the
study by Faumont et al (22)
indicated that nuclear factor-κB activation participates in the
overexpression of DNA polβ in Epstein-Barr virus-immortalized B
cells. Therefore, the data additionally indicated that the
expression of DNA polβ is regulated by multiple factors and
pathways in cells, and that that expression is stimuli, cell type
and context dependent (22). This
mobile regulation of DNA polβ also implies that its overexpression
is important for working against DNA damage. The expression of DNA
polβ is easily adjusted when the intracellular or extracellar
environment changes through a corresponding pathway, which helps to
maintain genomic stability.
In summary, the present study indicates that p38MAPK
phosphorylation and the activation of its downstream molecule,
ATF2, is important for mediating DNA polβ expression following
treatment with AOH in NIH3T3 cells. However, it may be worthwhile
to additionally study a suitable method that could be used to
regulate the balance of DNA polβ expression for genomic
stability.
Acknowledgements
The present study was supported by grants from
National Science Foundation in China, Beijing, China (grant nos.
81372269 and 81472324), and the Natural Science Foundation of the
Henan province of China, Zhengzhou, China (grant no.
14A310011).
References
|
1
|
Zmudzka BZ, Fornace A, Collins J and
Wilson SH: Characterization of DNA polymerase beta mRNA: Cell-cycle
and growth response in cultured human cells. Nucleic Acids Res.
16:9587–9596. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kim YJ and Wilson DM III: Overview of base
excision repair biochemistry. Curr Mol Pharmacol. 5:3–13. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kidane D, Dalal S, Keh A, Liu Y, Zelterman
D and Sweasy JB: DNA polymerase beta is critical for genomic
stability of sperm cells. DNA Repair (Amst). 10:390–397. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cabelof DC, Raffoul JJ, Yanamadala S, Guo
Z and Heydari AR: Induction of DNA polymerase beta-dependent base
excision repair in response to oxidative stress in vivo.
Carcinogenesis. 23:1419–1425. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Horton JK, Baker A, Berg BJ, Sobol RW and
Wilson SH: Involvement of DNA polymerase beta in protection against
the cytotoxicity of oxidative DNA damage. DNA Repair (Amst).
1:317–333. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Servant L, Cazaux C, Bieth A, Iwai S,
Hanaoka F and Hoffmann JS: A role for DNA polymerase beta in
mutagenic UV lesion bypass. J Biol Chem. 277:50046–50053. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chan K, Houlbrook S, Zhang QM, Harrison M,
Hickson ID and Dianov GL: Overexpression of DNA polymerase beta
results in an increased rate of frameshift mutations during base
excision repair. Mutagenesis. 22:183–188. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Albertella MR, Lau A and O'Connor MJ: The
overexpression of specialized DNA polymerases in cancer. DNA Repair
(Amst). 4:583–593. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tan XH, Zhao M, Pan KF, Dong Y, Dong B,
Feng GJ, Jia G and Lu YY: Frequent mutation related with
overexpression of DNA polymerase beta in primary tumors and
precancerous lesions of human stomach. Cancer Lett. 220:101–114.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang J, Parsons J, Nicolay NH, Caporali S,
Harrington CF, Singh R, Finch D, D'Atri S, Farmer PB, Johnston PG,
et al: Cells deficient in the base excision repair protein, DNA
polymerase beta, are hypersensitive to oxaliplatin chemotherapy.
Oncogene. 29:463–468. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dong ZM, Zheng NG, Wu JL, Li SK and Wang
YL: Difference in expression level and localization of DNA
polymerase beta among human esophageal cancer focus, adjacent and
corresponding normal tissues. Dis Esophagus. 19:172–176. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao J, Liu K, Lu J, Ma J, Zhang X, Jiang
Y, Yang H, Jin G, Zhao G, Zhao M and Dong Z: Alternariol induces
DNA polymerase β expression through the PKA-CREB signaling pathway.
Int J Oncol. 40:1923–1928. 2012.PubMed/NCBI
|
|
13
|
Vlahopoulos SA, Logotheti S, Mikas D,
Giarika A, Gorgoulis V and Zoumpourlis V: The role of ATF-2 in
oncogenesis. Bioessays. 30:314–327. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gupta S, Campbell D, Dérijard B and Davis
RJ: Transcription factor ATF2 regulation by the JNK signal
transduction pathway. Science. 267:389–393. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
De Cesare D and Sassone-Corsi P:
Transcriptional regulation by cyclic AMP-responsive factors. Prog
Nucleic Acid Res Mol Biol. 64:343–369. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Green TA, Alibhai IN, Unterberg S, Neve
RL, Ghose S, Tamminga CA and Nestler EJ: Induction of activating
transcription factors (ATFs) ATF2, ATF3, and ATF4 in the nucleus
accumbens and their regulation of emotional behavior. J Neurosci.
28:2025–2032. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Widen SG and Wilson SH: Mammalian
beta-polymerase promoter: Large-scale purification and properties
of ATF/CREB palindrome binding protein from bovine testes.
Biochemistry. 30:6296–6305. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chijiwa T, Mishima A, Hagiwara M, Sano M,
Hayashi K, Inoue T, Naito K, Toshioka T and Hidaka H: Inhibition of
forskolin-induced neurite outgrowth and protein phosphorylation by
a newly synthesized selective inhibitor of cyclic AMP-dependent
protein kinase,
N-[2-(p-bromocinnamylamino)ethyl]-5-isoquinolinesulfonamide (H-89),
of PC12D pheochromocytoma cells. J Biol Chem. 265:5267–5272.
1990.PubMed/NCBI
|
|
19
|
Yang SH, Sharrocks AD and Whitmarsh AJ:
MAP kinase signalling cascades and transcriptional regulation.
Gene. 513:1–13. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kreideweiss S, Ahlers C, Nordheim A and
Rühlmann A: Ca2+-induced p38/SAPK signalling inhibited by the
immunosuppressant cyclosporin A in human peripheral blood
mononuclear cells. Eur J Biochem. 265:1075–1084. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chyan YJ, Rawson TY and Wilson SH: Cloning
and characterization of a novel member of the human ATF/CREB
family: ATF2 deletion, a potential regulator of the human DNA
polymerase beta promoter. Gene. 312:117–124. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Faumont N, Le Clorennec C, Teira P,
Goormachtigh G, Coll J, Canitrot Y, Cazaux C, Hoffmann JS, Brousset
P, Delsol G, et al: Regulation of DNA polymerase beta by the LMP1
oncoprotein of EBV through the nuclear factor-kappaB pathway.
Cancer Res. 69:5177–5185. 2009. View Article : Google Scholar : PubMed/NCBI
|