Introduction
Adult T-cell leukemia/lymphoma (ATLL) is one
histological type of aggressive lymphoma that is endemic in Japan
(southwestern region), the Caribbean basin and Central Africa
(1). ATLL is caused by human T-cell
leukemia virus type I (HTLV-1) infection, and the HTLV-1 p40 tax
protein and HTLV-1 basic leucine zipper factor are considered to be
important for oncogenesis (2). Since
ATLL cells are generally resistant to chemotherapy, the median
survival time of acute and lymphomatous ATLL is reported to be ~1
year (3). It is therefore a matter of
great urgency to understand the resistance of ATLL cells to
chemotherapy at a molecular level in order to uncover useful
targets for the therapy of ATLL.
T cell Ig and mucin domain-containing molecule-3
(TIM-3), also known as hepatitis A virus cellular receptor 2, is
widely expressed on immune cells such as monocyte/macrophages,
dendritic cells, natural killer cells and T cells (4,5).
Galectin-9 and high mobility group box 1 are known to be the
ligands of TIM-3, and TIM-3 signaling is generally involved in the
regulation of immune responses by negatively regulating interferon
secretion (6). Recently, TIM-3 has
also been found to be expressed in melanoma, liver cancer and lung
cancer, and higher expression of TIM-3 has been shown to be
associated with a poor clinical prognosis in these cancers
(7–10).
We previously demonstrated that macrophages induced
ATLL cell proliferation and that a higher density of
tumor-associated macrophages (TAMs) in ATLL tissues was closely
associated with a poor clinical prognosis (11). It was also found that direct contact
with macrophages induced ATLL cell activation, although the
detailed mechanisms have not yet been elucidated (11).
In the present study, direct co-culture with
macrophages induced chemoresistance and TIM-3 overexpression in
ATLL cells, and the correlation between TIM-3 expression and
chemoresistance was investigated.
Materials and methods
Cell lines and cell culture
The human ATLL cell lines (ATN-1, TL-Mor, ED and
ATL-2s) (12) and a T-cell lymphoma
cell line without HTLV-1 infection (MOLT-4) were maintained in RPMI
(Wako Pure Chemical Industries, Ltd., Tokyo, Japan) supplemented
with 10% fetal bovine serum. The ATN-1, TL-Mor and MOLT-4 cells
were purchased from the RIKEN Cell Bank (Wako, Japan). The ED and
ATL-2 cell lines were previously established by Professor Masao
Matsuoka (Institute for Virus Research, Kyoto University, Kyoto,
Japan). The mycoplasma test was performed by using a polymerase
chain reaction (PCR) detection kit purchased from Takara Bio Inc.
(Otsu, Japan). Adriamycin (ADR) and carboplatin (CBDCA) were
obtained from Wako Pure Chemical Industries, Ltd. The number of
ATLL cells following 2-day culture with ADR (50 or 100 µM) and
CBDCA (0.3 or 1.0 µM) was tested using the WST assay kit (Dojindo
Molecular Technologies Inc., Kumamoto, Japan).
Macrophage culture
Peripheral blood mononuclear cells were obtained
from healthy volunteer donors, and all donors supplied written
informed consent for experimental use of these cells. Cluster of
differentiation (CD)14-positive monocytes were isolated using
CD14-microbeads (Miltenyi Biotec, Bergisch Gladbach, Germany).
These monocytes were plated in 24-well plates
(5×104/well) and cultured with granulocyte-macrophage
colony-stimulating factor (GM-CSF; 5 ng/ml) and macrophage CSF
(M-CSF; 100 ng/ml) (Wako) for 7 days at 37°C.
Flow cytometry (fluorescence-activated
cell sorting)
Phycoerythrin-labeled anti-TIM-3 antibody (mouse
monoclonal; clone F382E2; #345006; dilution, 1:50), fluorescein
isothiocyanate-labeled anti-CD14 antibody (mouse monoclonal; clone
HCD14; #325604; dilution, 1:50) and isotype matched control
antibodies (mouse IgG1 kappa; clone MOPC-21; #400112 and #400110)
were purchased from BioLegend Inc. (San Diego, CA). The surface
expression of these molecules on stained cell samples was analyzed
using a FACSverse flow cytometer (BD Biosciences, Franklin Lakes,
NJ, USA) with FACSuite (BD Biosciences) software.
TIM-3-expressing ATLL cell lines
A plasmid encoding the TIM-3 gene (pDsRed-TIM-3) and
a control pDsRed plasmid (Takara Bio Inc., Otsu, Japan) were
transfected into ATN-1 and ED cells using HilyMax (Dojindo
Molecular Technologies Inc.). Following selection with neomycin,
single cell cloning was performed. Cell clones were selected and
cell surface TIM-3 expression was tested using flow cytometry.
Tissue samples
Paraffin-embedded tumor samples (thickness, 3 µm)
from the lymph nodes of 58 patients with ATLL (lymphomatous and
acute type) were examined. Patients diagnosed between January 1993
and December 2003 were treated with conventional chemotherapy and
no patient was treated with bone marrow transplantation. A total of
29 patients showed leukemic change. Written informed consent was
obtained from all patients in accordance with protocols approved by
the Fukuoka University and Kurume University Review Board (Kurume,
Japan). The experimental procedure was approved by the Kumamoto
University Review Board (Kumamoto, Japan). We previously published
characteristics of this same set of ATLL specimens, and thus, the
numbers of CD68- and CD163-positive cells published in that
previous report (11) were used in
this study.
Immunohistochemistry
Rabbit polyclonal anti-Iba-1 (macrophage marker;
#019-19741; Wako Pure Chemical Industries, Ltd.) and goat
polyclonal anti-TIM-3 (#LS-C55594; LifeSpan Biosciences Inc.,
Seattle, WA, USA; dilution, 1:2,000) were used as primary
antibodies. Normal goat immunoglobulin (Santa Cruz, Dallas, Texas)
was used as a negative control and no signal was observed in
negative control-stained sections. Each immunostaining was
performed at the same time, and the reactions were visualized using
the diaminobenzidine substrate system (Nichirei Bioscience, Tokyo,
Japan). The data were evaluated by two investigators who were
blinded to any information regarding the samples. For
double-immunostaining, TIM-3 was visualized using the
diaminobenzidine substrate system in the first step. After washes
in glycine buffer (pH 2.2), sections were then stained with the
anti-Iba-1 antibody and visualized using HistoGreen solution
(Linaris Biologische Produkte, Wertheim-Bettingen, Germany).
Statistical analysis
Statistical analysis of in vitro and in
vivo data was performed using JMP 10 software (SAS Institute,
Chicago, IL, USA). All values from in vitro studies
represent results of 2 or 3 independent experiments. Data are
expressed as the mean ± standard deviation. Student's t-test was
used for comparisons of two groups in in vitro studies.
P<0.05 was considered to indicate a statistically significant
difference.
Results
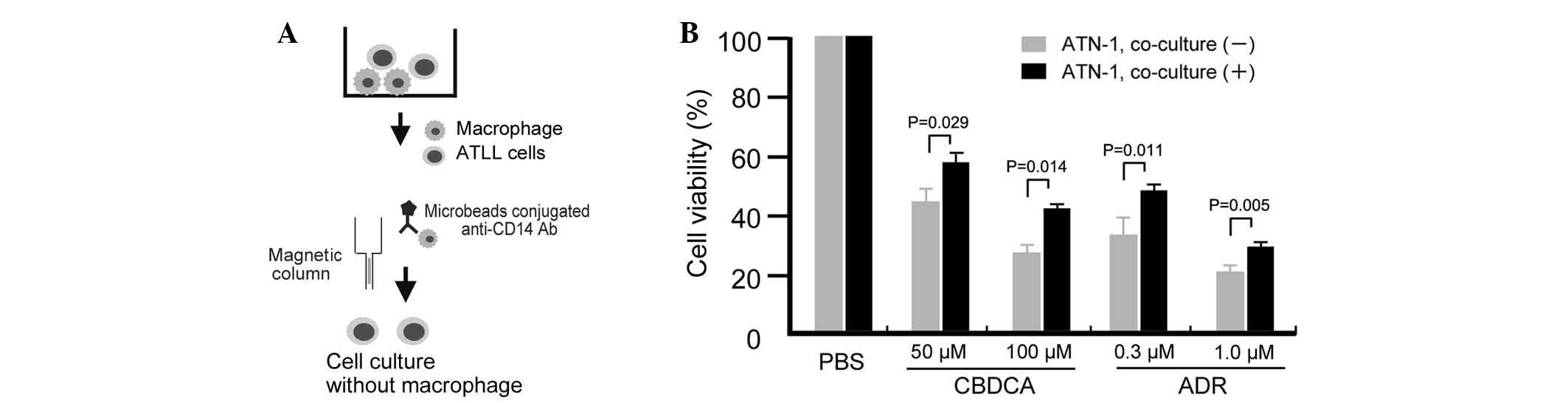
Long-term co-culture with macrophages
induces chemoresistance in ATN-1 cells
The study first tested whether the sensitivity of
ATLL cell lines to anticancer compounds may change following their
co-culture with macrophages through use of an in vitro
co-culture assay. In this direct co-culture system, cells from the
ATLL ATN-1, TL-Mor, ED, ATL-2s or MOLT-4 cell lines were
co-cultured with macrophages for 1, 2 or 3 weeks, following which
the co-cultured cells were depleted of macrophages by using
microbeads conjugated to an anti-CD14 antibody and a magnetic
column (Fig. 1A). Contamination of
the lymphoma cells with macrophages was <2% following this
depletion procedure (data not shown). The sensitivity of the
co-cultured ATLL cells to the anticancer drugs ADR or CBDCA was
then assayed by evaluation of cell viability using a WST assay. The
sensitivity of ATN-1 cells to ADR and CBDCA was significantly
decreased by prior co-culture with macrophages for 3 weeks (all
P<0.05; Fig. 1B). Resistance of
ATN-1 cells to ADR and CBDCA was also induced by 2 weeks of prior
co-culture with macrophages; however, the differences in anticancer
drug sensitivities between cells cultured with or without
macrophages were smaller than those of the 3-week co-cultured cells
(data not shown). Indirect co-culture using Transwells did not
impact the sensitivity of the ATN-1 cells to ADR or CBDCA (data not
shown), suggesting that direct contact between the macrophages and
ATN-1 cells was required for the observed effect.
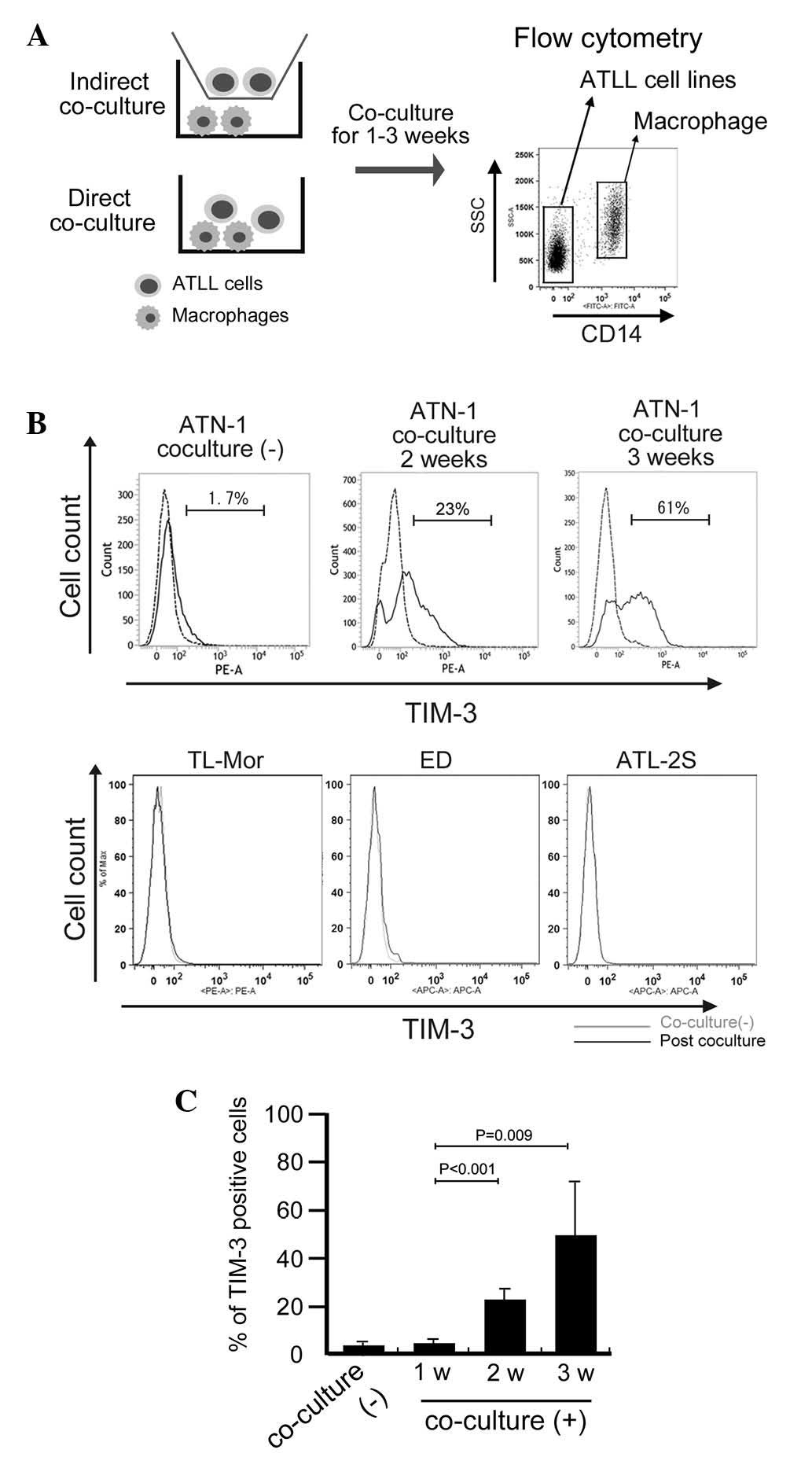
TIM-3 expression on ATN-1 cells is
induced by long-term direct co-culture with macrophages
Based on preliminary cDNA microarray data (data not
shown), we suspected that TIM-3 expression in ATN-1 cells was
upregulated by co-culture with macrophages. To confirm this
possibility, the effect of co-culture with macrophages on TIM-3
expression by ATN-1 cells was analyzed using flow cytometry. ATN-1
cells and macrophages were distinguished from each other using the
anti-CD14 antibody as a macrophage marker (Fig. 2A). This flow cytometric analysis
showed that, although little TIM-3 expression was detected in the
control ATN-1 cells or in the ATN-1 cells co-cultured with
macrophages for 1 week, TIM-3 expression was significantly induced
in the ATN-1 cells by 2 and 3 weeks of co-culture with macrophages
(P<0.05; Fig. 2B and C). By
contrast, TIM-3 expression was not detected in, and was not induced
by co-culture with macrophages in other cell lines (Fig. 2B). The induction of TIM-3
overexpression in ATLL cell lines by co-culture with macrophages
was not observed in the indirect co-culture system (data not
shown).
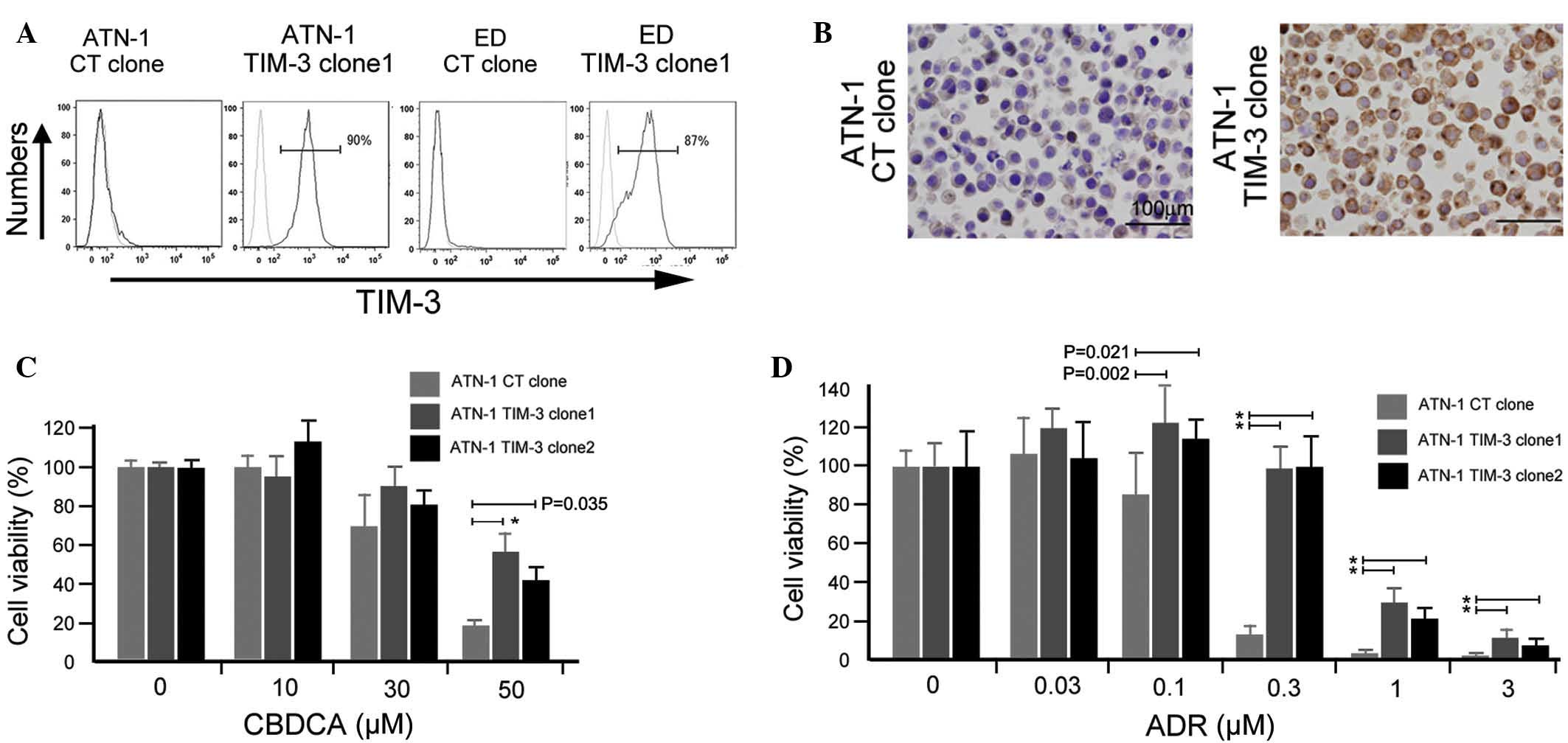
TIM-3 overexpression is involved in
the chemoresistance of ATN-1 cells
Next, an in vitro experiment was performed
using ATLL cell lines to confirm the involvement of TIM-3 in their
chemoresistance. A TIM-3 gene construct was transfected into ATN-1
and ED cells, and TIM-3-expressing ATN-1 and ED cells were
established (Fig. 3A). TIM-3
expression in established ATN-1 and ED clones was confirmed using
flow cytometry (Fig. 3A) and its
expression in ATN-1 clones was further confirmed by immunostaining
(Fig. 3B). Two TIM-3-positive ATN-1
clones showed increased chemoresistance to ADR and CBDCA compared
with control ATN-1 cells (P<0.05; Fig.
3C and D), whereas the growth rate of TIM-3-positive ATN-1
cells did not differ from that of control ATN-1 cells (data not
shown). Notably, TIM-3 overexpression did not affect either the
sensitivity of the ED cells to ADR and CBDCA or the growth rate of
the ED cells (data not shown).
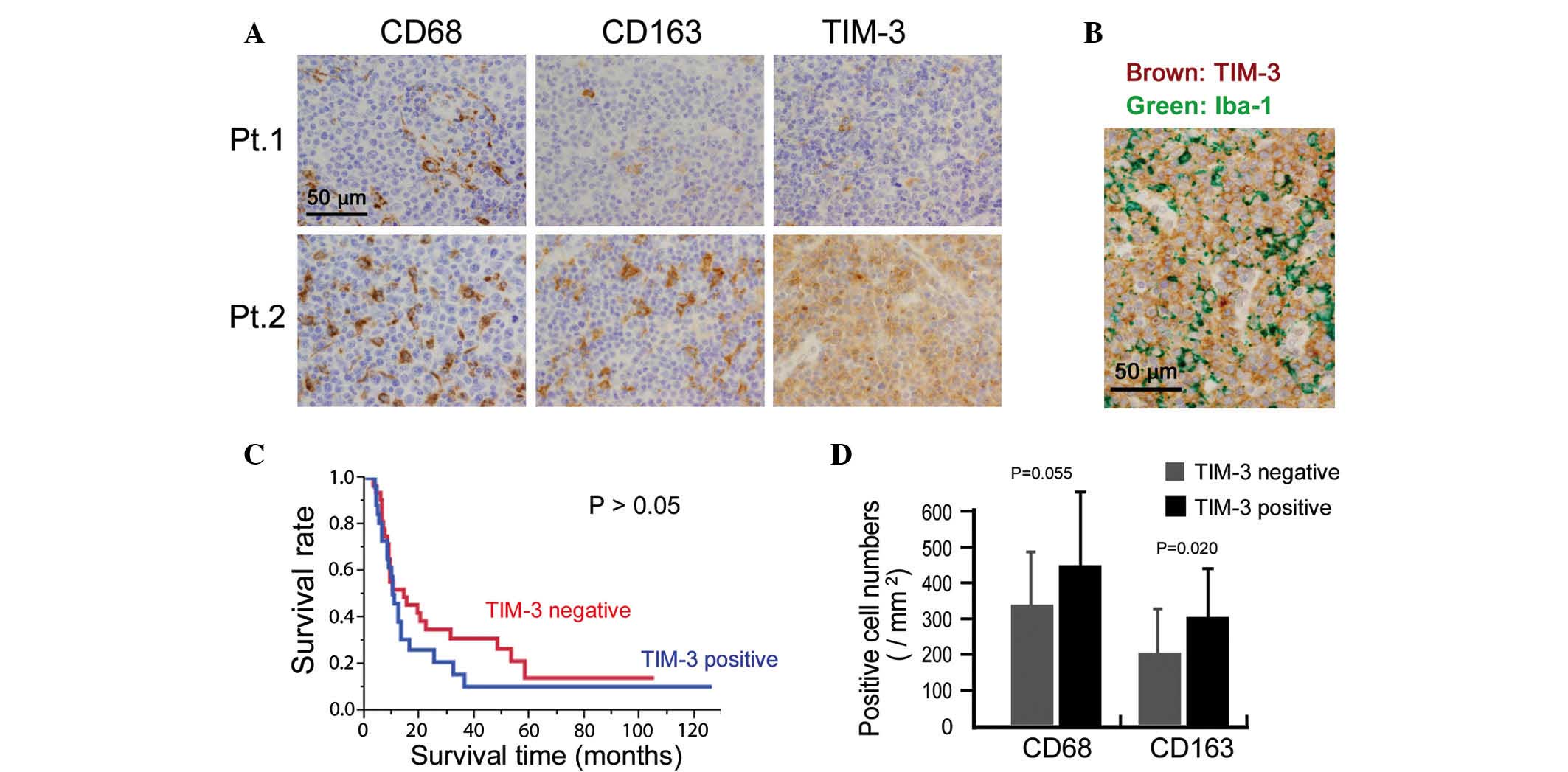
TIM-3 expression in lymphoma cells is
correlated with chemoresistance in patients with ATLL
Finally, the potential clinical significance of
TIM-3 expression was investigated by immunostaining for TIM-3
expression in 58 ATLL tissue specimens. Cases in which >50% of
the lymphoma cells were positive for TIM-3 were classified as
TIM-3-positive. This analysis indicated that 25 out of the total 58
cases were TIM-3-positive (Fig. 4A).
Since certain macrophages also stained for TIM-3, TIM-3 expression
on lymphoma cells was confirmed by means of double-immunostaining.
Thus, TIM-3 expression was detected in lymphoma cells that were
negative for the macrophage marker Iba-1 (Fig. 4B). The association between TIM-3
expression and clinicopathological factors was then analyzed, and
TIM-3-positive cases were found to be resistant to chemotherapy
(P=0.015; Table I). However, TIM-3
expression was not associated with overall survival or other
clinical factors (all P>0.05; Fig.
4C and Table I).
 | Table I.TIM-3 expression and
clinicopathological factors. |
Table I.
TIM-3 expression and
clinicopathological factors.
|
|
| TIM-3, n |
|
|---|
|
|
|
|
|
|---|
| Factor | n | Negative | Positive | P-value |
|---|
| Age, years |
|
|
| 0.79 |
| ≥65 | 29 | 17 | 12 |
|
|
<65 | 29 | 16 | 13 |
|
| Gender |
|
|
| 0.15 |
| Male | 34 | 22 | 12 |
|
|
Female | 24 | 11 | 13 |
|
| Stagea |
|
|
| 0.66 |
| I, II,
III | 24 | 15 | 9 |
|
| IV | 30 | 17 | 13 |
|
| Response to
chemotherapya |
|
|
| 0.015 |
|
Partial/complete | 38 | 24 | 14 |
|
| No
response | 10 | 2 | 8 |
|
| Leukemic
changea |
|
|
| 0.46 |
| Yes | 29 | 15 | 14 |
|
| No | 26 | 16 | 10 |
|
The correlation between macrophage infiltration and
TIM-3 expression in the lymphoma cells was also evaluated and a
higher density of CD163-positive macrophages was detected in the
TIM-3-positive cases (P=0.020; Fig.
4D). The density of CD68-positive macrophages appeared to be
higher in the TIM-3-positive cases, however, no significant
association was found (P=0.055; Fig.
4D).
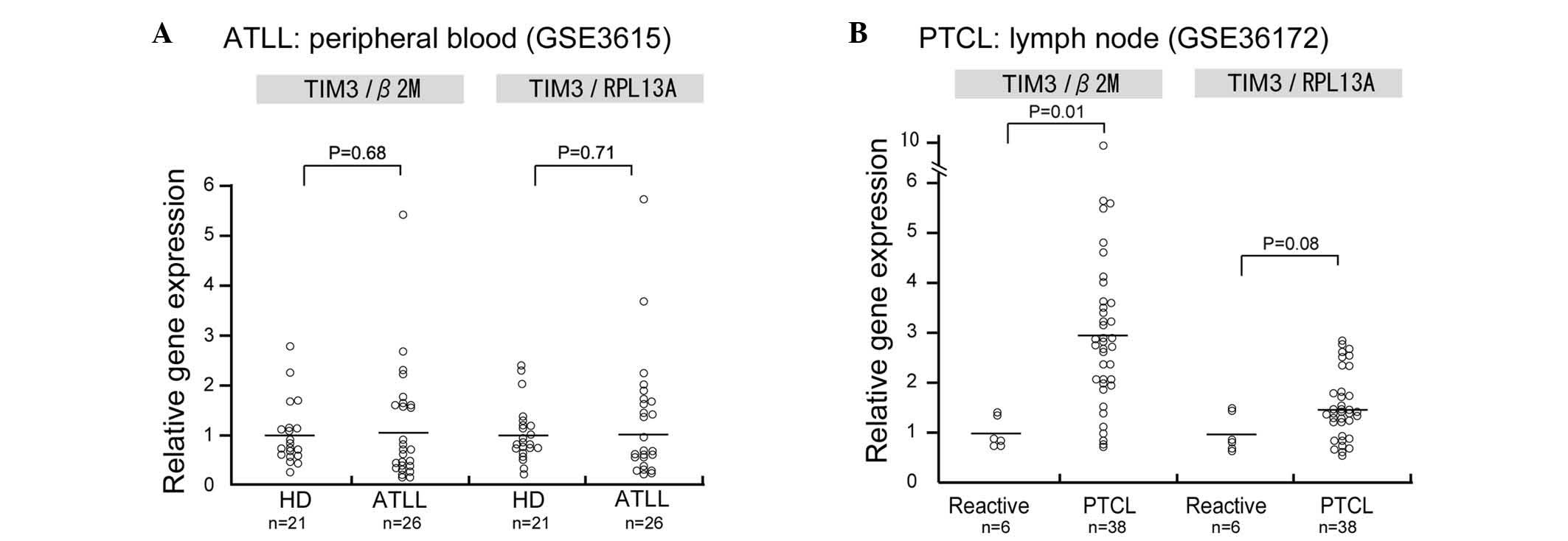
Since the mRNA expression of TIM-3 in human ATLL
samples was not evaluated in the present study, data analysis using
the PubMed central database was performed to determine TIM-3 mRNA
expression in ATLL. In this analysis, TIM-3 mRNA expression was
observed in CD4-positive lymphocytes of patients with acute ATLL;
however, no significant differences in TIM-3 mRNA expression were
observed between the CD4-positive lymphocytes of healthy donors and
those of patients with acute ATLL (Fig.
5A). Since no microarray dataset analysis of lymph node
biopsies could be found in the database, TIM-3 expression was
analyzed in a dataset of peripheral T-cell lymphoma (PTCL) samples
and TIM-3 was found to be upregulated in the PTCL samples compared
with the control reactive lymphadenitis samples (P=0.01; Fig. 5B).
Discussion
In our previous study, a large number of TAMs were
detected in cells from lymph node biopsy samples of almost all ATLL
cases and a notably higher percentage of CD163-positive TAMs were
found to be significantly linked to a worse clinical course
(11). In vitro analysis
showed that direct contact between ATLL cells and TAMs induced ATLL
cell proliferation via several TAM-derived growth factors,
including IL-6 and tumor necrosis factor-α (11). Based on these observations, we next
considered that long-term co-culture with macrophages may change
the characteristics of ATLL cells. In the present study, it was
found that direct co-culture with macrophages induced
chemoresistance in ATLL cells. This phenomenon was not observed
when the ATLL cells were indirectly co-cultured with macrophages;
therefore, direct contact with macrophages is suggested to play an
important role in macrophage-induced chemoresistance in ATLL cells.
This effect may be caused by several cytokines derived from
macrophages that are strongly activated by direct contact with ATLL
cells. The expression of intracellular adhesion molecule-1
(ICAM-1), membrane type M-CSF, P-selectin glycoprotein ligand-1 and
unknown CD163 ligands on tumor cells is considered to be associated
with macrophage activation in certain types of tumors (13,14). Zheng
et al demonstrated that an ICAM-1-blocking antibody reduced
macrophage-induced chemoresistance in myeloma cells (15). Since ICAM-1 is also expressed in ATLL
cells (16), ICAM-1 may be associated
with macrophage-induced chemoresistance.
In the present study, the expression of TIM-3 in
ATLL cells was induced by long-term co-culture with macrophages;
however, the detailed molecular mechanisms by which this occurs
remain unclear. Galectin-9, high mobility group protein B1 (HMGB-1)
and phosphatidylserine are known to be ligands of TIM-3 (4–6), and our
unpublished data that HMGB-1 is highly expressed in ATLL cells
suggest that TIM-3 activation is induced by HMGB-1 that is derived
from ATLL cells in an autocrine manner. Since no TIM-3 expression
was observed in any of the lymphoma cell lines used in the present
study in the absence of macrophage co-culture, unknown factors
derived from activated macrophages or epigenetic change induced by
direct contact with macrophages are considered to be necessary for
the induction of TIM-3 in ATLL cells.
The present study also showed that TIM-3 expression
is potentially associated with chemoresistance in in vivo
and in vitro studies. However, a TIM-3 neutralizing antibody
failed to affect the macrophage-induced chemoresistance in ATLL
cells in an in vitro study (Horlad et al, unpublished
data). This observation indicates the complexity of TIM-3
signaling. TIM-3 overexpression is also observed in activated
lymphocytes and, as with cytotoxic T lymphocyte antigen 4 and
programmed death 1, it is associated with tolerance and exhaustion
(4–6).
Recently, Huang et al demonstrated that carcinoembryonic
antigen cell adhesion molecule 1 (CEACAM1) mediates TIM-3
activation and that blocking of the two molecules was required to
induce lymphocyte activation (17).
Thus, blocking of CEACAM1 and TIM-3 together may be necessary to
impair the chemoresistance of TIM-3-positive ATLL cells.
By means of database analysis, in the present study,
TIM-3 mRNA overexpression was observed in the samples of lymph node
biopsies, but not in the peripheral CD4-positive lymphocytes of
patients, as shown in Fig. 5. We
suspect that direct contact between TAMs and lymphoma cells is
observed only in lymph node samples of ATLL and that long-term
direct contact with TAMs induced TIM-3 overexpression in the ATLL
cells.
In conclusion, in an in vitro experiment, the
present study found that direct contact with TAMs induced
chemoresistance and TIM-3 expression in ATLL cells. It was also
found that the TIM-3 expression found in the lymphoma cells of the
ATLL biopsy samples was closely associated with the resistance to
chemotherapy. In vitro study suggested that long-term direct
contact with TAMs caused TIM-3 expression. Although TIM-3
overexpression induced the chemoresistance of ATLL cells,
antibody-mediated neutralization of TIM-3 failed to improve the
macrophage-induced ATLL chemoresistance. Although other unknown
mechanisms are considered to be involved in macrophage-induced
chemoresistance, TIM-3-related signals may be a useful target
therapy for patients with ATLL. In addition, TIM-3 expression in
biopsy samples of ATLL may be a good predictor of the effectiveness
of chemotherapy.
Acknowledgements
The authors would like to thank Ms. Emi Kiyota, Mr.
Osamu Nakamura, Ms. Yui Hayashida and Mr. Takenobu Nakagawa of the
Department of Cell Pathology of Kumamoto University for their
technical assistance. This study was supported by grants from the
Ministry of Education, Culture, Sports, Science and Technology of
Japan (no. 23790407 to Dr Yoshihiro Komohara, and no. 20390113 to
Professor Motohiro Takeya).
References
|
1
|
Shirono K, Hattori T, Hata H, Nishimura H
and Takatsuki K: Profiles of expression of activated cell antigens
on peripheral blood and lymph node cells from different clinical
stages of adult T-cell leukemia. Blood. 73:1664–1671.
1989.PubMed/NCBI
|
|
2
|
Ohshima K: Pathological features of
diseases associated with human T-cell leukemia virus type I. Cancer
Sci. 98:772–778. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shimoyama M: Diagnostic criteria and
classification of clinical subtypes of adult T-cell
leukaemia-lymphoma. A report from the Lymphoma Study Group
(1984–87). Br J Haematol. 79:428–437. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Monney L, Sabatos CA, Gaglia JL, Ryu A,
Waldner H, Chernova T, Manning S, Greenfield EA, Coyle AJ, Sobel
RA, et al: Th1-specific cell surface protein Tim-3 regulates
macrophage activation and severity of an autoimmune disease.
Nature. 415:536–541. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chiba S, Baghdadi M, Akiba H, Yoshiyama H,
Kinoshita I, Dosaka-Akita H, Fujioka Y, Ohba Y, Gorman JV, Colgan
JD, et al: Tumor-infiltrating DCs suppress nucleic acid-mediated
innate immune responses through interactions between the receptor
TIM-3 and the alarmin HMGB1. Nat Immunol. 13:832–842. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tang D and Lotze MT: Tumor immunity times
out: TIM-3 and HMGB1. Nat Immunol. 13:808–810. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wiener Z, Kohalmi B, Pocza P, Jeager J,
Tolgyesi G, Toth S, Gorbe E, Papp Z and Falus A: TIM-3 is expressed
in melanoma cells and is upregulated in TGF-beta stimulated mast
cells. J Invest Dermatol. 127:906–914. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li H, Wu K, Tao K, Chen L, Zheng Q, Lu X,
Liu J, Shi L, Liu C, Wang G and Zou W: Tim-3/galectin-9 signaling
pathway mediates T-cell dysfunction and predicts poor prognosis in
patients with hepatitis B virus-associated hepatocellular
carcinoma. Hepatology. 56:1342–1351. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhuang X, Zhang X, Xia X, Zhang C, Liang
X, Gao L, Zhang X and Ma C: Ectopic expression of TIM-3 in lung
cancers: A potential independent prognostic factor for patients
with NSCLC. Am J Clin Pathol. 137:978–985. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Komohara Y, Morita T, Annan DA, Horlad H,
Ohnishi K, Yamada S, Nakayama T, Kitada S, Suzu S, Kinoshita I, et
al: The coordinated actions of TIM-3 on cancer and myeloid cells in
the regulation of tumorigenicity and clinical prognosis in clear
cell renal cell carcinomas. Cancer Immunol Res. 3:999–1007. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Komohara Y, Niino D, Saito Y, Ohnishi K,
Horlad H, Ohshima K and Takeya M: Clinical significance of CD163*
tumor-associated macrophages in patients with adult T-cell
leukemia/lymphoma. Cancer Sci. 104:945–951. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Takeda S, Maeda M, Morikawa S, Taniguchi
Y, Yasunaga J, Nosaka K, Tanaka Y and Matsuoka M: Genetic and
epigenetic inactivation of tax gene in adult T-cell leukemia cells.
Int J Cancer. 109:559–567. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Komohara Y, Jinushi M and Takeya M:
Clinical significance of macrophage heterogeneity in human
malignant tumors. Cancer Sci. 105:1–8. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Komohara Y, Niino D, Ohnishi K, Ohshima K
and Takeya M: Role of tumor-associated macrophages in hematological
malignancies. Pathol Int. 65:170–176. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zheng Y, Yang J, Qian J, Qiu P, Hanabuchi
S, Lu Y, Wang Z, Liu Z, Li H, He J, et al: PSGL-1/selectin and
ICAM-1/CD18 interactions are involved in macrophage-induced drug
resistance in myeloma. Leukemia. 27:702–710. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tanaka Y, Fukudome K, Hayashi M, Takagi S
and Yoshie O: Induction of ICAM-1 and LFA-3 by Tax1 of human T-cell
leukemia virus type 1 and mechanism of down-regulation of ICAM-1 or
LFA-1 in adult-T-cell-leukemia cell lines. Int J Cancer.
60:554–561. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Huang YH, Zhu C, Kondo Y, Anderson AC,
Gandhi A, Russell A, Dougan SK, Petersen BS, Melum E, Pertel T, et
al: CEACAM1 regulates TIM-3-mediated tolerance and exhaustion.
Nature. 517:386–390. 2015. View Article : Google Scholar : PubMed/NCBI
|