Introduction
Multiple myeloma (MM) is a plasma cell (PC) disorder
that induces anemia, skeletal destruction, renal failure and
hypercalcemia (1). It is the second
most common hematological malignancy, and it is characterized by
the presence of a monoclonal immunoglobulin (Ig) expressed and
secreted in the bone marrow, where there is a collection of
abnormal PCs that interfere with the production of normal blood
cells (2).
IgD MM constitutes ≤2% of all MM cases, displays
generally an aggressive phenotype (often with renal failure) and is
usually characterized by poor prognosis (3). IgD-κ occurs only in 3–4% of all IgD MM
cases, and is also associated with the difficulty of obtaining a
reliable diagnosis and of conducting a constant and precise
follow-up (4).
The diagnostic panel for IgD-κ MM should include PC
flow cytometry characterization, fluorescence in situ
hybridization (FISH), serum protein electrophoresis (SPE), serum
immunofixation electrophoresis (sIFE), measurement of serum free
light chains (sFLCs) and total heavy chain IgD, and urinary IFE for
Bence Jones (BJ) protein (5).
Diagnostically, with novel agent therapy (including
thalidomide, bortezomib and lenalidomide) specifically integrated
with autologous stem cells transplantation (ASCT) when feasible,
the survival for IgD MM is improved; however, the outcomes remain
inferior to those achieved in patients with other myeloma isotypes,
thus highlighting the requirement for better and more innovative
approaches in treatment and monitoring (6).
The present report describes the follow-up of a case
of an IgD-K MM patient, who often refused to undergo a bone marrow
aspirate, even in certain critical phases of the disease. Thus,
given the occasional inability to obtain bone marrow aspirate
samples, at times when a relapse was suspected, minimal residual
disease (MRD) was alternatively monitored uniquely by serological
evaluation of FLC and total heavy chain IgD levels (7,8).
The current report presents the case of a long
survival patient monitored almost exclusively by sFLC and IgD
measurements as an essential, non-invasive marker. Written informed
consent was obtained from the patient [medical records no. 4249 on
June 10, 2013 at Hematology and Stem Cell Trasplantation Unit,
Italian National Cancer Institute ‘Regina Elena’ (Rome,
Italy)].
Case report
In March 2007, a 51 year-old woman presented for the
first time at the Hematology and Stem Cell Trasplantation Unit of
the Italian National Cancer Institute ‘Regina Elena’ with multiple
osteolytic lesions. PC flow cytometry characterization (FACSCanto™;
BD Biosciences, Franklin Lakes, NJ, USA) identified an infiltration
(23% of bone marrow population) of cluster of differentiation
(CD)38+ CD138+ CD28+
CD56+ CD117+ CD19−
CD45− tumor PCs, with κ-sFLC restriction, as illustrated
by flow cytometric analysis at diagnosis (Fig. 1A). Bone marrow examination by FISH
revealed no abnormalities.
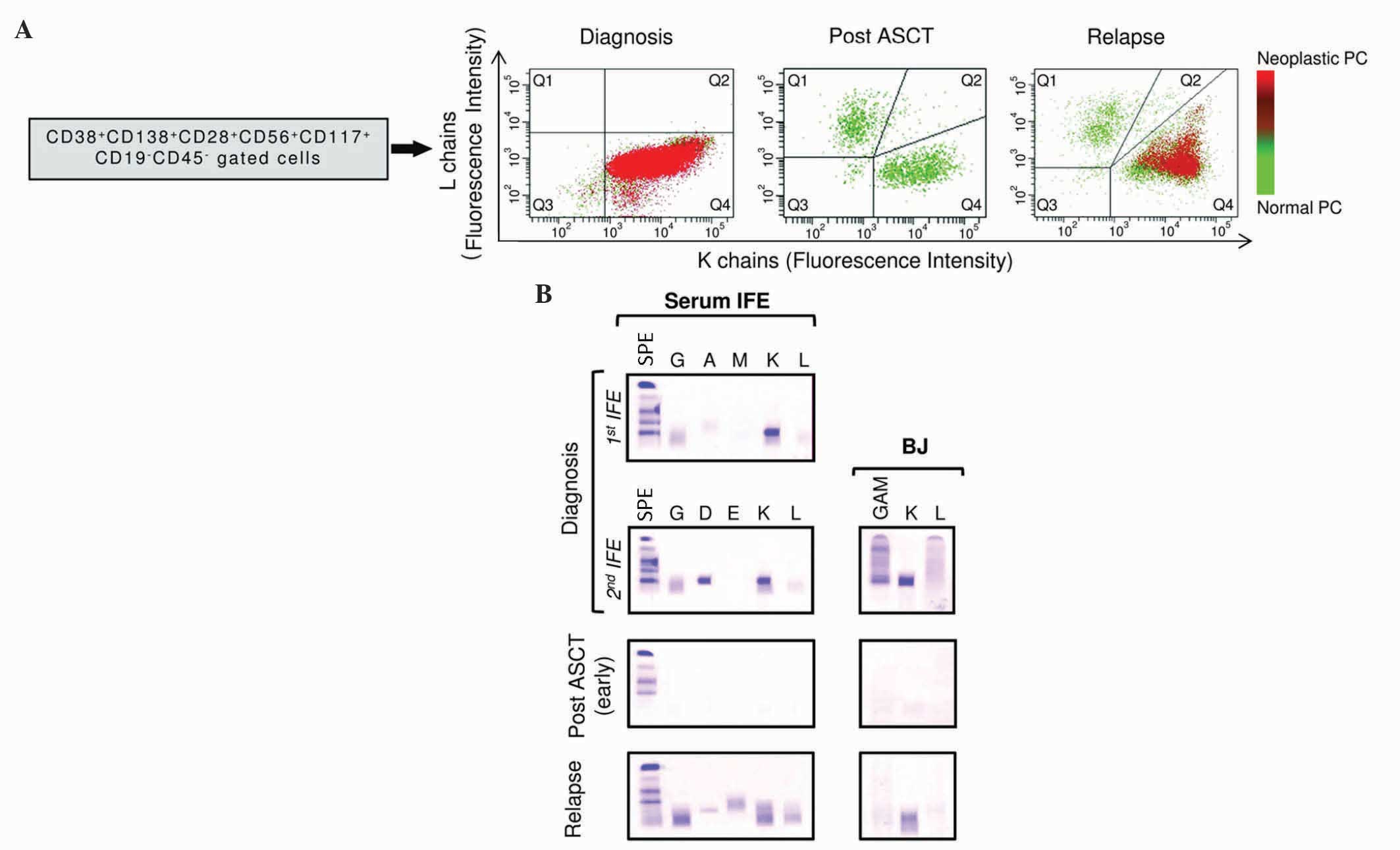 | Figure 1.Flow cytometric analysis and IFE
detection during patient monitoring. (A) Flow cytometric evaluation
of the expression of κ and λ chains in normal vs. malignant PCs at
diagnosis, upon ASCT and at relapse. Q1-Q4 represent the distinct
regions analyzed by flow cytometry, where Q1 comprises λ-positive
PCs and Q4 contains κ-positive ones. The green color in the plots
represents normal PCs, whereas the red color depicts the presence
of neoplastic PCs. These bone marrow aspirates indicate the
presence of neoplastic cells at diagnosis, which disappear
following ASCT, while they are still present at the time of
relapse. Their progress was coherent with the values of serum free
light chains tested (B) IFE and Bence Jones protein at diagnosis,
pre/post ASCT and during relapse. The term ‘early’ inside
parentheses refers to the first post-ASCT timepoint. IFE was
performed with the immunoglobulin antisera indicated above each
lane. IFE, immunofixation electrophoresis; CD, cluster of
differentiation; PC, plasma cell; ASCT, autologous stem cells
transplantation; BJ, Bence Jones; GAM, mixed antisera against
immunoglobulins G, A and M; SPE, serum protein electrophoresis. |
sIFE and BJ protein IFE on urine evidenced the
presence of an IgD-κ monoclonal component and κ light chains,
respectively (Fig. 1B). In addition,
sFLC quantification (The Binding Site Group, Ltd., Birmingham, UK)
revealed a marked increase in κ-sFLC with an abnormal FLC κ/λ ratio
(Table I). Total heavy chain IgD
quantification (The Binding Site Group, Ltd.) confirmed the
presence of elevated IgD levels (Fig.
1B and Table I).
 | Table I.FLC/IgD values in the course of
monitoring with BMD chemotherapy and ASCT. |
Table I.
FLC/IgD values in the course of
monitoring with BMD chemotherapy and ASCT.
|
|
|
| FLCsa |
|
|---|
|
|
|
|
|
|
|---|
| Therapy | Therapy schedule | Date | κ (3.3–19.4) | λ (5.7–26.3) | κ/λ ratio
(0.26–1.65) | Heavy chain total IgD
(7.7–132.1) |
|---|
| Pre-BMD | – | Oct 10 2007 | 5,889.00 | 8.30 | 709.00 | 8,678.00 |
| BMD | 1st cycle, basal | Oct 31 2007 | 4,812.00 | 3.44 | 1,398.00 | ND |
|
| 1st cycle, 5
days | Nov 16 2007 | 3,518.00 | 2.61 | 1,348.00 | ND |
|
| 2nd cycle, basal | Dec 12 2007 | 3,352.00 | 2.82 | 1,188.00 | 5,028.00 |
|
| 2nd cycle, 5
days | Dec 14 2007 | 2,539.00 | 2.09 | 1,215.00 | ND |
|
| 2nd cycle, 15
days | Dec 24 2007 | 2,484.00 | 2.74 | 906.00 | 3,862.00 |
|
| 2nd cycle, 28
days | Jan 7 2008 | 3,432.00 | 2.93 | 1,171.00 | 4,966.00 |
|
| 3rd cycle, 5
days | Jan 11 2008 | 2,651.00 | 2.26 | 1,173.00 | ND |
|
| 4th cycle, basal | Feb 4 2008 | 3,460.00 | 8.13 | 425.00 | 3,957.00 |
|
| 4th cycle, 7
days | Feb 11 2008 | 2,627.00 | 8.00 | 328.00 | 4,050.00 |
|
| 4th cycle, 15
days | Feb 19 2008 | 3,580.00 | 8.13 | 440.00 | 3,503.00 |
| Pre-ASCT | – | Apr 10 2008 | 5,149.00 | 8.00 | 643.00 | 8,406.00 |
| Post-ASCT | 15 days | May 9 2008 | 42.09 | 14.10 | 2.91 | ND |
|
| 2 months | Jul 23 2008 | 19.92 | 71.40 | 0.28 | 6.44 |
|
| 4 months | Sep 3 2008 | 27.41 | 59.32 | 0.46 | ND |
|
| 6 months | Nov 3 2008 | 30.08 | 28.91 | 1.04 | 6.44 |
|
| 17 months | Sep 17 2009 | 25.83 | 17.77 | 1.45 | ND |
|
| 21 months | Jan 20 2010 | 24.26 | 26.43 | 0.92 | 8.65 |
|
| 24 months | Apr 19 2010 | 27.95 | 21.80 | 1.28 | ND |
|
| 43 months | Feb 27 2012 | 26.97 | 17.66 | 1.53 | 12.50 |
|
| 48
monthsb | Jul 2 2012 | 37.02 | 16.62 | 2.23 | 208.00 |
|
| 56
monthsb | Mar 6 2013 | 262.00 | 14.78 | 14.78 | 190.00 |
|
| 59
monthsb | Jun 10 2013 | 692.00 | 11.27 | 61.40 | 748.00 |
The average and standard deviation values of
β2-microglobulin (β2 M) levels were 7.47 and 3.77 mg/l,
respectively (normal range, 0.80–2.20 mg/l). Other classical
serological parameters [calcium (Ca)-renal-anemia-bone criteria]
were altered. The diagnosis was an IgD-κ MM stage IIIA.
According to the current clinical practice at the
Italian National Cancer Institute ‘Regina Elena’, the patient
underwent chemotherapy with two cycles of vincristine, doxorubicin
(Adriblastina®) and dexamethasone (VAD regimen) from
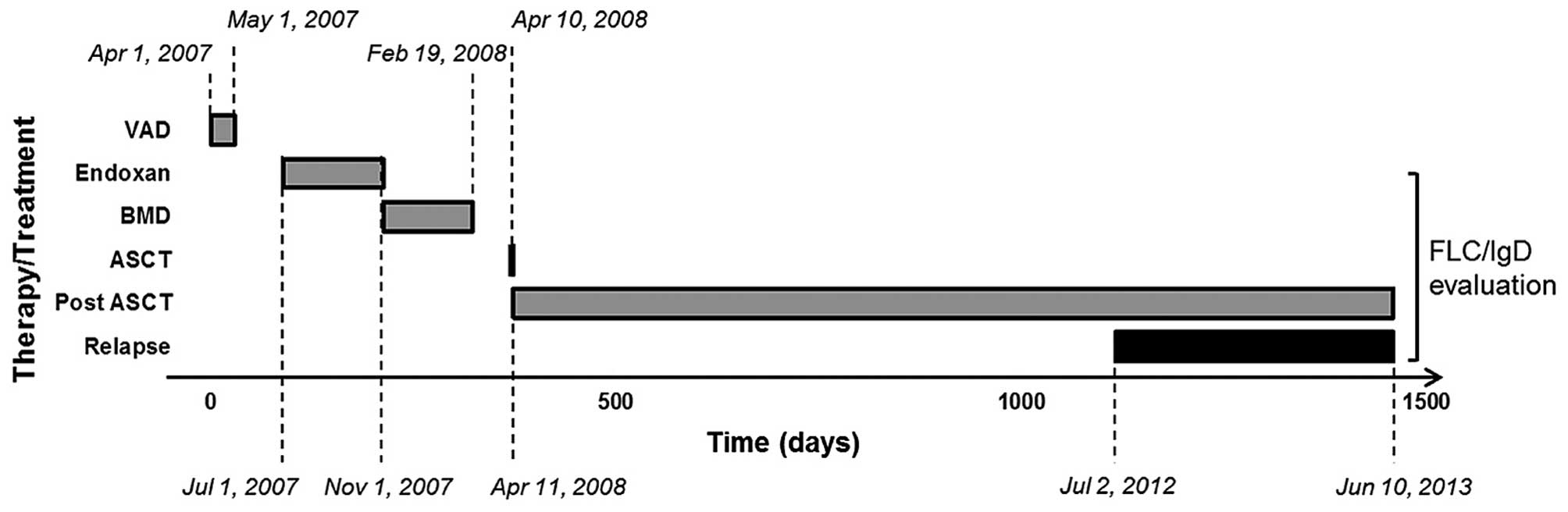
April until May 2007 (9). The patient
was then treated with two cycles of cyclophosphamide (Endoxan
Baxter®) from July until November 2007, and four
subsequent cycles of bortezomib (Velcade®),
Myocet® and dexamethasone (BMD regimen) from November
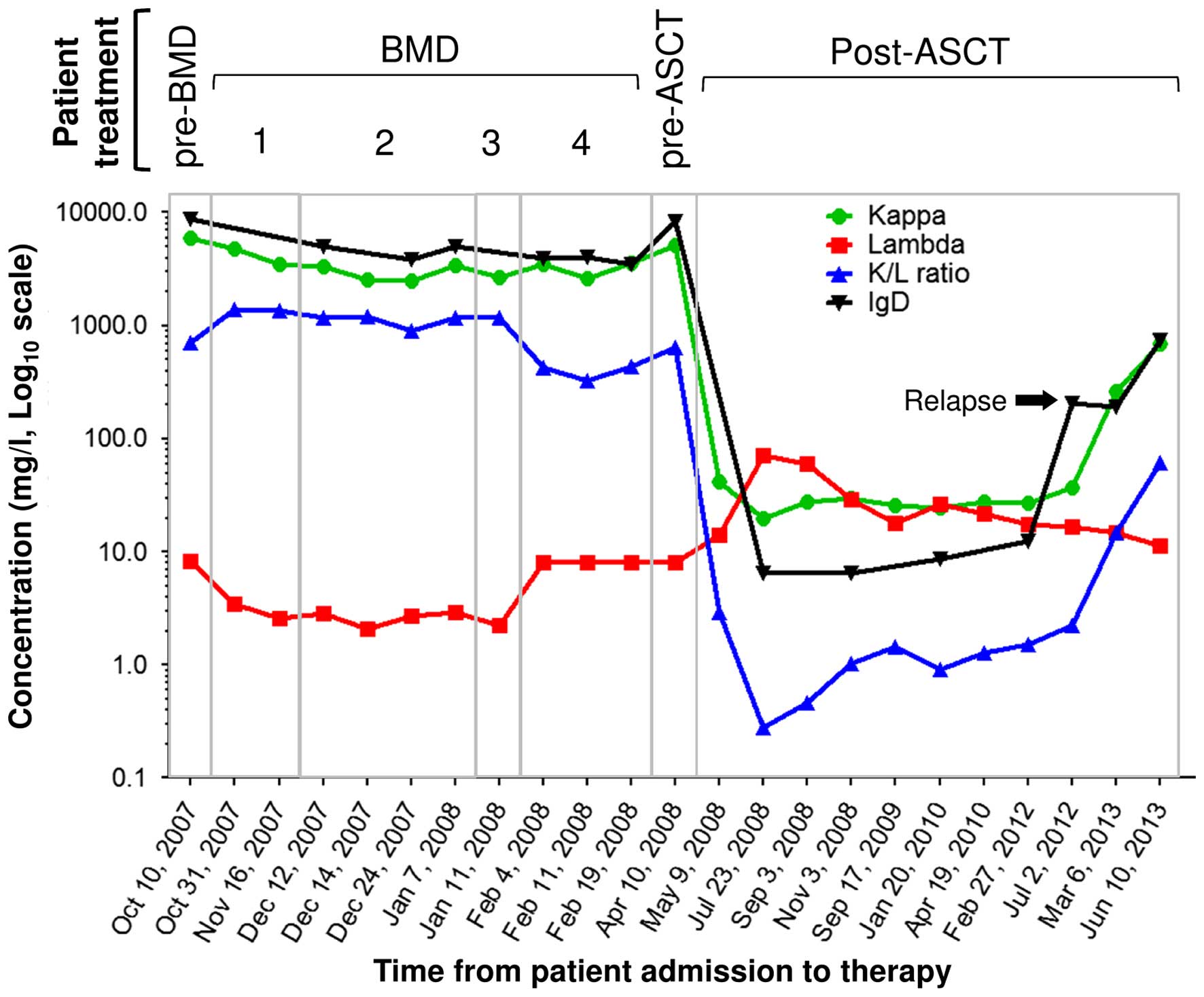
2007 until February 2008 (Fig. 2 and
Table I). The FLC measurements and
total IgD evaluations commenced upon ASCT (10) and were extended until the relapse
phase (Fig. 2). Following the first
VAD cycle, bone marrow stem cells were collected.
The patient was classified as ‘non responder’ to the
different chemotherapeutic agents; however, the patient was
selected for ASCT in April 10, 2008 (Fig.
2). Following ASCT, severe complications occurred, including
pneumonia with Morganella morganii infection and then
sepsis. Based on these observations, the hematological asset of the
patient was re-evaluated upon ASCT, and bone barrow
immunophenotyping revealed a 0.1% of PC population in the
lymphocytes region. As displayed by post-ASCT flow cytometric
analysis, the sFLC κ/λ ratio decreased, and no presence of
neoplastic PCs was detected (Fig.
1A). In parallel, sIFE appeared without a monoclonal component,
and the level of BJ protein was less pronounced overtime (Fig. 1B). Furthermore, these parameters were
associated with a marked reduction in κ-sFLC (Fig. 3), suggesting that the patient was
effectively responding to the treatment. However, osteolytic
lesions were still present but did not progress overtime.
In November 2008, the patient was diagnosed with
Guillain-Barré syndrome. Due to the several complications occurred
during the course of this syndrome, the patient refused a second
ASCT and other bone marrow aspirates. Therefore, the patient was
regularly monitored with sFLC and total IgD measurements in order
to assess the response to the first ASCT performed in April 2008
(Fig. 2), in addition to evaluation
of other classical serological parameters, including β2 M, lactate
dehydrogenase, Ca and hemoglobin). FLC/IgD parameters were also
evaluated for the whole duration of post-ASCT and disease relapse
(Fig. 2 and Table I).
In July 2012, after 4 years of ASCT, the levels of
IgD first exhibited a substantial increase above the normal range
(Fig. 3 and Table I), and 8 months later, in March 2013,
the levels of κ-sFLC exhibited a further significant increase
(Fig. 3). Due to this κ-sFLC
increase, the presence of an IgD-κ MM was further confirmed by sIFE
and urine IFE (Fig. 1B).
Accordingly, in view of a possible relapse, the
patient agreed to undergo a bone marrow aspirate in March 2013,
evidencing a PC population (4% of bone marrow cells) with the
typical phenotypic hallmarks of CD38+ CD138+
CD28+ CD56+ CD117+
CD19− CD45− cells, including the presence of
neoplastic PCs with κ light chains restriction, as indicated by the
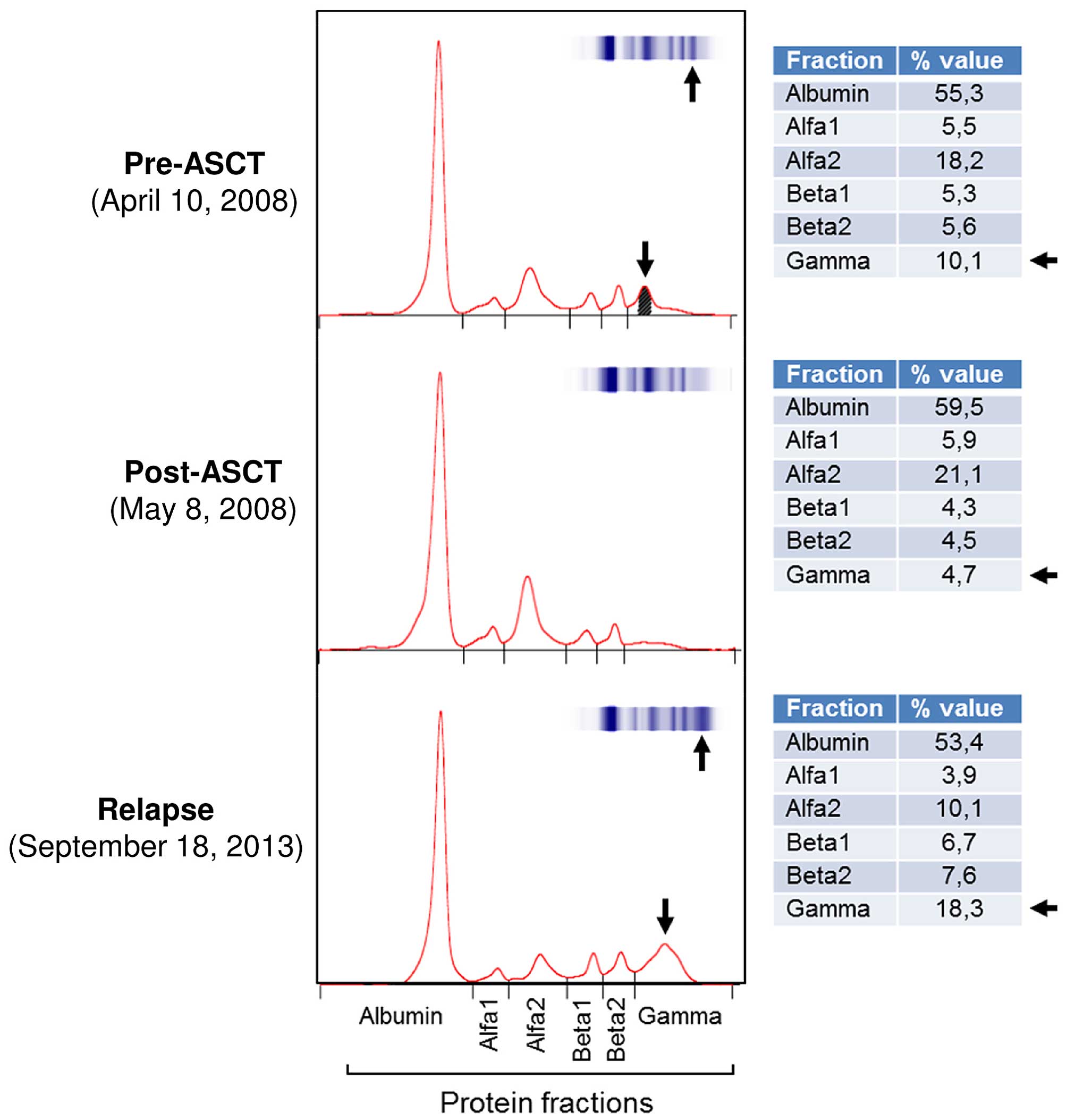
flow cytometric analysis performed at relapse (Fig. 1A). In addition, SPE was performed,
which further supported the recurrence of a monoclonal peak inside
the γ region. This recurrence was also confirmed by the percentage
values of the γ regions from the SPE and IFE profiles (Fig. 4). Indeed, the high γ fraction value
depicted in the pre-ASCT phase (10.1%, Fig. 4) reflects the values observed at
relapse (18.3%, Fig. 4); there are
similarly high values compared with post-ASCT (4.7%, Fig. 4).
Upon another chemotherapy line, the patient
underwent a second ASCT, and is currently monitored by measurement
of sFLC and total heavy chain IgD.
Discussion
The present study reports the follow-up of a patient
with IgD-κ MM that requires a careful approach and monitoring.
The patient refused bone marrow aspirate in various
occasions, even under suspicion of relapse; thus, the patient had
to be followed up by means of sFLC and total heavy chain IgD
measurements for monitoring MRD. The sFLC values over the four BMD
cycles were generally constant, indicating the unresponsiveness of
the patient. Compared with pre-ASCT levels, the total IgD and light
chain values displayed a post-ASCT decrease, including the FLC
levels. This was further confirmed by the disappearance of the
κ-sFLC in the post-ASCT IFE. In addition, the total heavy chain IgD
and κ-sFLC values observed in July 2012 suggested a potential
relapse of the disease, since a consequent relapse in March 2013
supported by the relative sIFE values was observed. These findings
were also confirmed by the flow cytometric analysis performed at
relapse, which demonstrated unbalanced expression levels of κ/λ
chains, also displaying the presence of neoplastic PCs. Lastly, the
SPE profiles of the patient appeared to be compatible with the
overall diagnostic evaluation of the disease evolution, and
provided further confirmation to the reliability of this unusual
IgD-κ MM patient follow-up.
These data clearly suggest a coherent outcome of
heavy chain and FLC levels alongside the diagnostic profile, and
demonstrate its effectiveness as an early marker for diagnosis by
monitoring the MRD and relapse of IgD-κ MM. Furthermore, in the
present and peculiar case study, the availability of a rapid,
quantitative and non-invasive test has been revealed of high
significance in the monitoring of IgD-κ MM patients, particularly
when bone marrow aspirate analysis is not available. In addition,
evaluation of serum total heavy chain IgD could represent another
useful clinical tool when employed in specialized centers where IgD
MM cases could be better characterized and monitored (9,11).
Finally, the present conclusions appears to be in line with the
recent International Myeloma Working Group statements for MM
patients that are not eligible for ASCT (12).
Acknowledgements
The authors are grateful to Dr Gaia Illuminati and
Miss Marianna Attanasio (Clinical Pathology Unit, Italian National
Cancer Institute ‘Regina Elena’) for their technical support on
sample bio-banking and analytical procedures.
References
|
1
|
Geng C, Liu N, Yang G, Liu A, Leng Y, Wang
H, Li L, Wu Y, Li Y and Chen W: Retrospective analysis of 264
multiple myeloma patients. Oncol Lett. 5:707–713. 2013.PubMed/NCBI
|
|
2
|
Raab MS, Podar K, Breitkreutz I,
Richardson PG and Anderson KC: Multiple myeloma. Lancet.
374:324–339. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Pandey S and Kyle RA: Unusual myelomas: A
review of IgD and IgE variants. Oncology (Williston Park).
27:798–803. 2013.PubMed/NCBI
|
|
4
|
Paiva B, Martinez-Lopez J, Vidriales MB,
Mateos MV, Montalban MA, Fernandez-Redondo E, Alonso L, Oriol A,
Teruel AI, de Paz R, et al: Comparison of immunofixation, serum
free light chain and immunophenotyping for response evaluation and
prognostication in multiple myeloma. J Clin Oncol. 29:1627–1633.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sinclair D: IgD myeloma: Clinical,
biological and laboratory features. Clin Lab. 48:617–622.
2002.PubMed/NCBI
|
|
6
|
Richardson PG, Laubach J, Paba-Prada C and
Anderson KC: IgD and IgE variants of myeloma: Valuable insights and
therapeutic opportunities. Oncology (Williston Park). 27:803–804.
2013.PubMed/NCBI
|
|
7
|
Ferrero S, Drandi D, Mantoan B, Ghione P,
Omede P and Ladetto M: Minimal residual disease detection in
lymphoma and multiple myeloma: Impact on therapeutic paradigms.
Hematol Oncol. 29:167–176. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ozaki S, Harada T, Saitoh T, Shimazaki C,
Itagaki M, Asaoku H, Kuroda Y, Chou T, Yoshiki Y, Suzuki K, et al:
Survival of multiple myeloma patients aged 65–70 years in the era
of novel agents and autologous stem cell transplantation. A
multicenter retrospective collaborative study of the japanese
society of myeloma and the european myeloma network. Acta Haematol.
132:211–219. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pisani F, Petrucci MT, Giannarelli D,
Bongarzoni V, Montanaro M, De Stefano V, La Verde G, Gentilini F,
Levi A, Za T, et al: IgD multiple myeloma a descriptive report of
17 cases: Survival and response to therapy. J Exp Clin Cancer Res.
31:172012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fu C, Wang J, Xin X, Liu H, Xue S, Ma X,
Jin Z, Sun A, Qiu H and Wu D: Therapeutic effects of autologous
hematopoietic stem cell transplantation in multiple myeloma
patients. Exp Ther Med. 6:977–982. 2013.PubMed/NCBI
|
|
11
|
Morabito F, Gentile M, Ciolli S, Petrucci
MT, Galimberti S, Mele G, Casulli AF, Mannina D, Piro E, Pinotti G,
et al: Safety and efficacy of bortezomib-based regimens for
multiple myeloma patients with renal impairment: A retrospective
study of Italian Myeloma Network GIMEMA. Eur J Haematol.
84:223–228. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Palumbo A, Rajkumar SV, San Miguel JF,
Larocca A, Niesvizky R, Morgan G, Landgren O, Hajek R, Einsele H,
Anderson KC, et al: International Myeloma Working Group consensus
statement for the management, treatment, and supportive care of
patients with myeloma not eligible for standard autologous
stem-cell transplantation. J Clin Oncol. 32:587–600. 2014.
View Article : Google Scholar : PubMed/NCBI
|