Introduction
Invasive meningioma is a common neoplasm of central
nervous system, which can infiltrate adjacent tissues (dura mater,
arachnoid membrane, vascular space and skull) without atypical
hyperplasia (1,2). Meningioma comprises of ~24–30% of
primary intracranial neoplasm and commonly affects females during
middle or later adult life (3,4). Primary
intracranial meningioma usually arises in proximity to meninges,
particularly in the parasagittal meninges, falx cerebri and
sphenoid sphenoid ridges (3).
Currently, various methods are employed for diagnosis and
evaluation of meningioma (5,6). Computed tomography (CT) and magnetic
resonance imaging (MRI) have become useful diagnostic tools for
accurately definition. In addition, CT and MRI imaging can also be
used to determine whether meningioma is invasive or not. Invasive
growth substantially increases the risk of recurrence (7–9).
In order to provide an effective way to prevent or
substantially delay the recurrence of invasive meningioma, and
improve the curative effect of surgical treatment, we collected and
analyzed the clinical manifestations, pathological features,
preoperative imaging characteristics as well the data obtained
during the surgical treatment of invasive meningioma. In most
cases, postoperative radiotherapy was needed to delay, or even
prevent the recurrence.
Materials and methods
General materials
From February 2014 to February 2016, 59 patients
with invasive meningioma were enrolled in this study. Presence of
invasive meningioma was confirmed by operation. There were 43 males
and 16 females (gender ratio, 2.3:1), and the age range was from 45
to 75 years (average, 55.3 years). The course of disease ranged
from 15 days to 7 years (average, 13.2 months). There were 4 cases
with multiple symptoms and 3 cases suffered from recurrence.
Clinical data and methods
We used World Health Organization (WHO) criteria for
classification of meningioma in the nervous system tumors as our
reference (10). There are 3 grades
in WHO classification: Benign (grade I), atypical (grade II), and
anaplastic (grade III). All situations during the operation were
recorded and scope of surgical resection was determined according
to Simpson classification (11).
The selection criteria
The selection basis was as follows: i) During the
operation, the tumor surrounding tissues were visibly invaded by
cancer cells; ii) pathological examination was performed on the
tumor surrounding tissues, and presence of tumor cell infiltration
was confirmed; and iii) results obtained from imaging examinations
revealed that tumor cells were invading the adjacent blood vessels
and adjacent nerves. Patients who complied with (i) and/or (ii)
were selected and (iii) was regarded as the standard. This study
was approved by the Ethics Committee of Weifang People's Hospital.
Signed written informed consents were obtained from all
participants before the study.
Clinical manifestations
Ten patients suffered from cranial nerve dysfunction
with symptoms such as ocular motility disorders, visual disturbance
and facial numbness. Twenty-nine patients suffered from
intracranial hypertension with symptoms such as dizziness, headache
and vomiting. Eleven patients suffered from epilepsy and paroxysmal
tic. Nine patients were found, for first time, to have invasive
meningioma after examination or postoperative re-examination.
Imaging examinations
All patients agreed to undergo skull MRI and/or CT
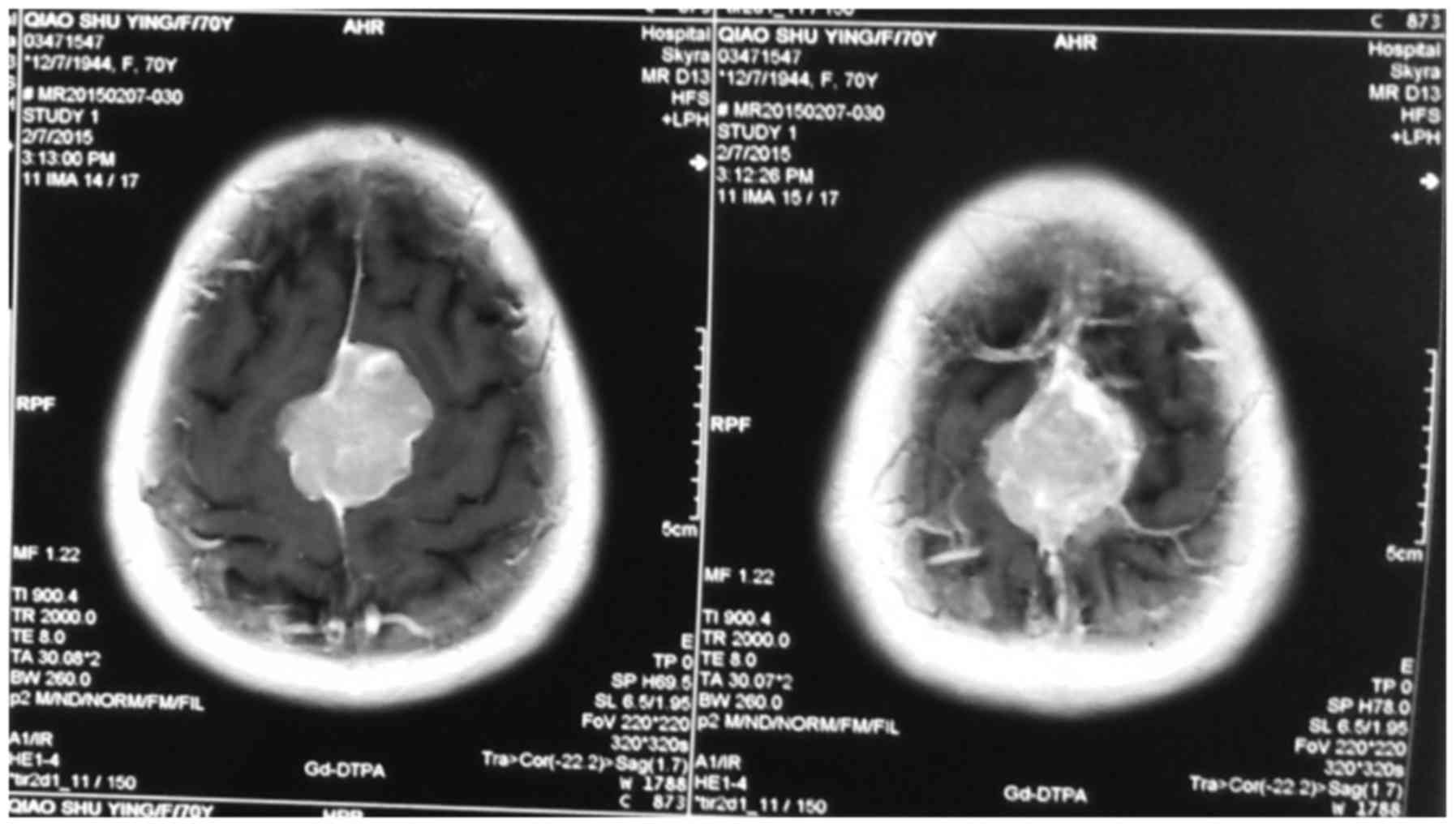
scan examinations. MRI results showed that, on T1 weighted images,
meningioma was mainly manifested as the equal signal (the majority)
and low signal (the minority). High signal revealed that, on T2
weighted images, ‘pseudocapsule sign’ was seen around the tumor,
significantly intensified and dural tail sign appeared after
enhancement (Figs. 1–3).
Contrast-enhanced MRI scan results showed that the
phenomenon of dural tail sign appeared in 41 cases. The results of
cranial CT scan showed middle-density masses or slight
hyper-density masses in all the patients, low-density masses with
different sizes were found in some patients, significant and
enhanced intensification appeared after imaging enhancement. Edema
area was detected around the meningioma.
Before surgery, all patients underwent imaging
examination. The invasion of the organizational structure around
the tumor that was revealed by MRI and/or CT scan were used as the
basis of imaging invasiveness. The sign of invasiveness is the
presence of invasion in exterior structure and the wrapping of
important blood vessels including large blood vessels (Table I). Meningioma was observed in a
variety of areas such as cerebral convexity (18 cases),
parasagittal region (17 cases), tentorium of cerebellum (10 cases),
cerebellar hemisphere (5 cases), CPA (3 cases), petroclival region
(2 cases), olfactory sulcus (1 case) and sphenoid ridge (1
case).
 | Table I.The classification and location of
invasive meningioma. |
Table I.
The classification and location of
invasive meningioma.
|
|
| Pathological
classification |
|---|
|
|
|
|
|---|
|
|
| WHO | WHO | WHO |
|---|
| The area of
tumors | Cases | grade I | grade II | grade III |
|---|
| Cerebral
convexity | 18 | 18 | 0 | 0 |
| Surrounding area of
parasagittal region of cerebral falx | 17 | 17 | 0 | 0 |
| Surrounding area of
transverse sinus and sigmoid sinus | 8 | 8 | 0 | 0 |
| Large blood and
vessels | 4 | 2 | 2 | 0 |
| Skull | 6 | 6 | 0 | 0 |
| Cavernous sinus | 4 | 4 | 0 | 0 |
| Periorbital
region | 1 | 1 | 0 | 0 |
| The infiltration of
brain | 1 | 0 | 0 | 1 |
| In total | 59 | 56 | 2 | 1 |
Results
Pathological examination
Pathological examination found 56 cases in WHO grade
I (10). There were 20 cases of
meningioma of mixed cell type, 11 cases of meningioma of
meningothelial type, 9 cases of fibroblastic meningioma, 5 cases of
angiomatous meningioma. We had 2 cases of WHO grade II with 1 case
of meningioma of clear cell type and 1 case of meningioma of
atypical meningioma. Also we had one case of anaplastic meningioma
(WHO grade III).
The invasive region of meningioma
During the surgeries we found out that meningioma
was invading other regions, such as cerebral convexity (18 cases),
area surrounding the parasagittal region (17 cases), area
surrounding transverse sinus and sigmoid sinus (8 cases), skull (6
cases), large blood vessels (4 cases), cavernous sinus (4 cases)
and periorbital region (1 case). Brain tissues around the tumor
were collected for pathological examination and results showed that
there was 1 case of the infiltration of brain. According to the
region invaded by the tumor, patients were divided into 3 grades:
i) Simpson grade I (the invasive region was cerebral convexity);
ii) Simpson grade II (the invasive region was venous sinus, and
tumor invaded large blood vessels and cavernous sinus); and iii)
Simpson grade III (Table II).
 | Table II.The relationship between the invasive
region and surgical resection of meningioma. |
Table II.
The relationship between the invasive
region and surgical resection of meningioma.
|
|
| Surgical
classification |
|---|
|
|
|
|
|---|
| The invasive region
of meningioma | Cases | Simpson grade I | Simpson grade II | Simpson grade
III |
|---|
| Cerebral
convexity | 18 | 18 | 0 | 0 |
| Surrounding area of
parasagittal region of cerebral falx | 17 | 4 | 12 | 1 |
| Surrounding area of
transverse sinus and sigmoid sinus | 8 | 2 | 6 | 0 |
| Skull | 6 | 3 | 3 | 0 |
| Cavernous sinus | 4 | 0 | 2 | 2 |
| Large blood and
vessels | 4 | 0 | 1 | 3 |
| Periorbital
region | 1 | 0 | 1 | 0 |
| The infiltration of
brain | 1 | 1 | 0 | 0 |
| In total | 59 | 56 | 25 | 6 |
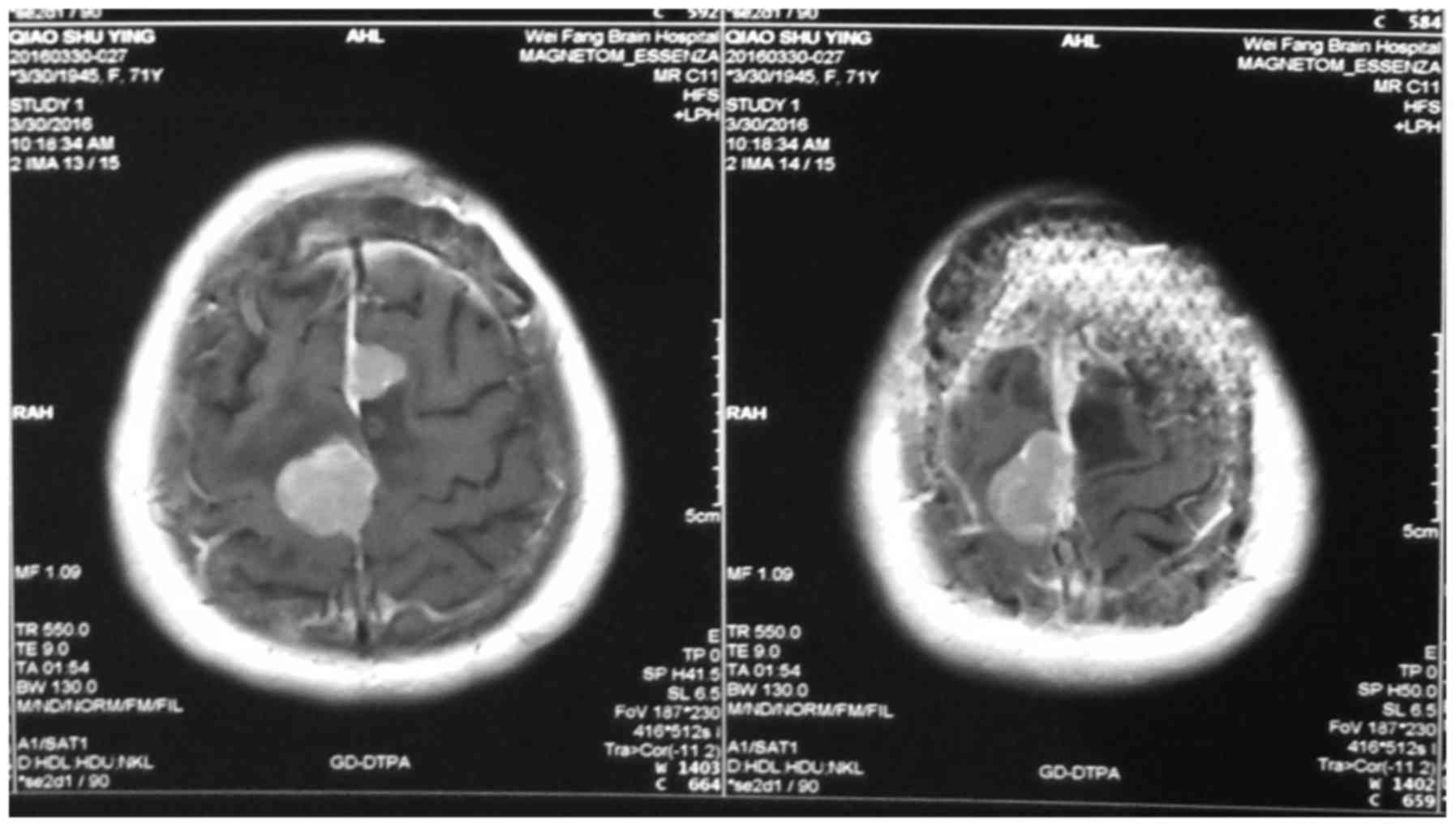
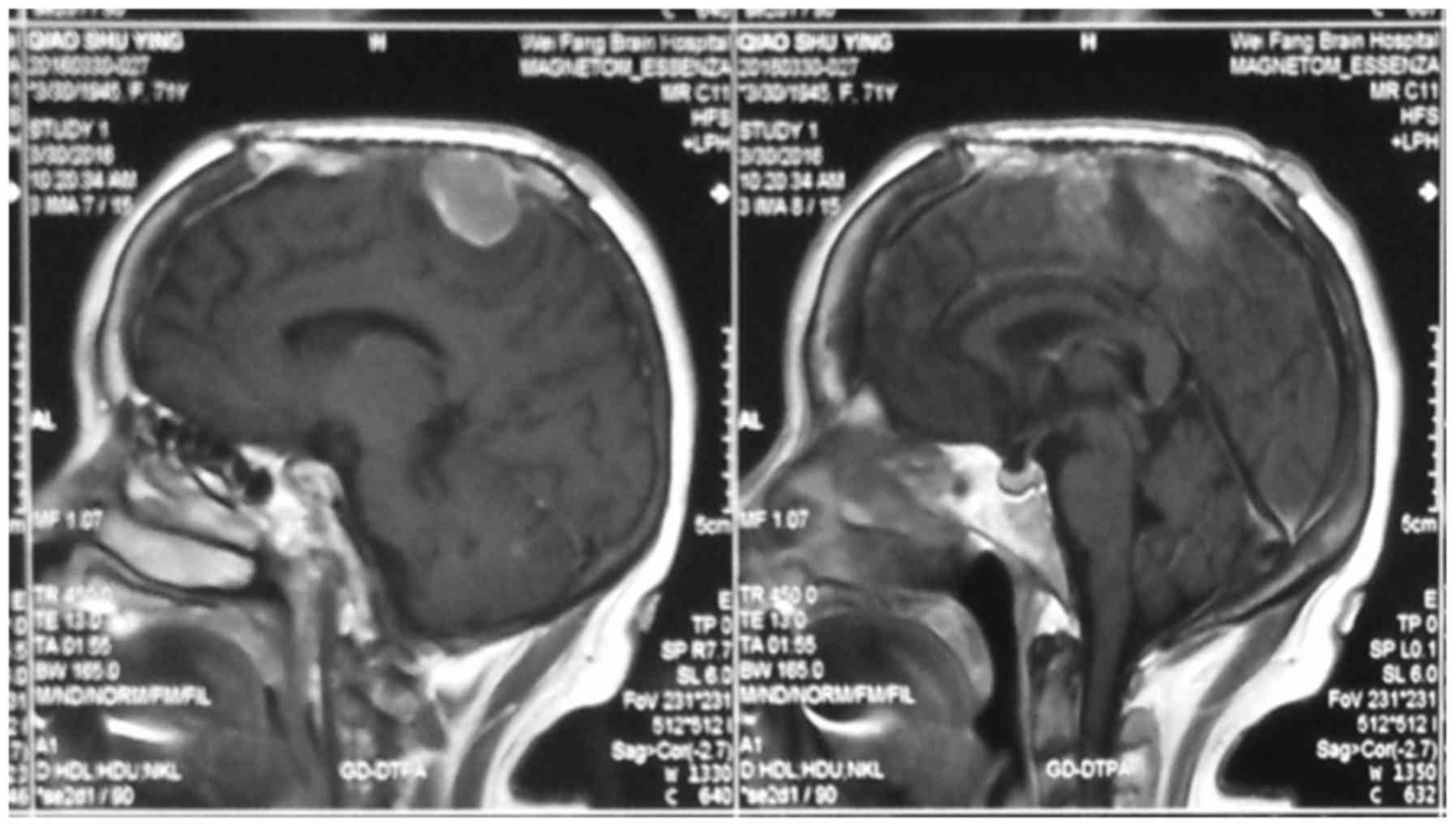
Imaging examination
During the CT and MRI examinations, we clearly
observed organizational structures around the tumor; structures
such as edema around meningioma (47 cases), venous sinus invasion
(17 cases), changes in bone structure (11 cases), brain tumor
interfaces (16 cases), blood vessels invasion (4 cases) and
exterior structure invasion (1 case) (Figs. 1–4).
Other imaging characteristics such as nodular tumors
(15 cases), tumors with cystic necrotic area (8 cases) and tumors
with unclear border (16 cases) were also observed (Tables III and IV).
 | Table III.The relationship between imaging
characteristics and pathological classification. |
Table III.
The relationship between imaging
characteristics and pathological classification.
|
| Pathological
classification |
|---|
|
|
|
|---|
| Imaging | WHO | WHO | WHO |
|---|
| characteristics | grade I | grade II | grade III |
|---|
| The edema around
meningioma | 34 | 6 | 7 |
| The invasion of
venous sinus | 17 | 0 | 0 |
| The change of
bone | 9 | 0 | 2 |
| The brain tumor
interfaces | 4 | 6 | 6 |
| The invasion of blood
and vessels | 0 | 2 | 2 |
| The invasion of
exterior structure | 0 | 1 | 0 |
 | Table IV.The relationship between imaging
manifestations and surgical resection. |
Table IV.
The relationship between imaging
manifestations and surgical resection.
|
|
| Surgical
classification |
|
|---|
|
|
|
|
|
|---|
| The invasive region
of meningioma | Cases | Simpson grade
I | Simpson grade
II | Simpson grade
III | The invasive cases
shown by imageological examination |
|---|
| Cerebral
convexity | 18 | 18 | 0 | 0 | 8 |
| Surrounding area of
parasagittal region of cerebral falx | 17 | 4 | 12 | 1 | 11 |
| Surrounding area of
transverse sinus and sigmoid sinus | 8 | 2 | 6 | 0 | 6 |
| Skull | 6 | 3 | 3 | 0 | 3 |
| Large blood and
vessels | 4 | 0 | 1 | 2 | 4 |
| Cavernous
sinus | 4 | 0 | 2 | 3 | 4 |
| Periorbital
region | 1 | 0 | 1 | 0 | 1 |
| The infiltration of
brain | 1 | 1 | 0 | 0 | 0 |
| In total | 59 | 56 | 25 | 6 | 37 |
Discussion
Brain tumors are divided into primary and secondary
tumors. The most common sites for primary tumors are cranial nerve
and meninx. Several tissues have been reported as the origin of
brain tumor and among them, the most common benign brain tumor is
meningioma which accounts for ~20% of all brain tumors (12,13).
Invasive meningioma can invade neighboring tissues. Brain tumors
are slow growing and have a prolonged latency. The most common
malignant brain tumor is glioma with characteristics such as fast
growth, easy recurrence and high mortality rate (14,15).
Clinical manifestations of invasive meningioma are
similar to those of benign meningioma, the main symptoms are
intracranial hypertension and chronic and progressive symptoms of
brain damage (16,17). Our results demonstrated that the most
common symptoms among our patients were intracranial hypertension
(29 cases), cranial nerve dysfunction (10 cases) and epilepsy (11
cases).
In most cases, the invasive meningiomas were located
in supratentorial region. The most common invasion area was
cerebral convexity and the area surrounding parasagittal region (35
cases or 59.3%). The secondary invasive area were the areas
surrounding the transverse sinus and sigmoid sinus (8 cases),
followed by internal carotid artery, optic nerve and sponge antrum
(4 cases), and finally the basilar artery and posterior cranial
fossa cranial nerve (4 cases) (Tables
I and II). These results were
consistent with the results obtained in previous studies (18).
Prior studies revealed that pathological
characteristics and classification of meningioma was closely
related to the risk of tumor recurrence (19). The biological mechanisms underlying
brain-invasive growth in meningioma are not completely understood.
The invasive mechanism of invasive meningioma may be linked to
long-term compression atrophy of cerebral cells. The confirmed
diagnosis of invasive meningioma should rely on the common decision
of imaging examinations, pathological examinations and
intraoperative observation. Morphological diagnosis of meningioma
relies on MRI and/or CT scan examinations (20).
Results obtained from prior studies showed that
usual manifestation of meningioma included uniform density/signal,
clear boundary, occasional hemorrhage, necrosis or cystic
degeneration and calcification (21).
Because of the uneven rate of tumor growth, one of the main
characteristics of high invasive meningioma is the mushroom
syndrome (22). Another
characteristic of high invasive meningioma is the uneven density or
signal and heterogeneous enhancement after contrast-enhanced scan.
Due to the rapid proliferation rate, the central area of the tumor
usually suffers from inadequate blood circulation, which can lead
to ischemic necrosis or cystic degeneration. The necrotic area in
more invasive tumors is usually larger.
Surgical resection is the preferred treatment for
invasive meningioma, however it was previously reported that in
17–50% of meningioma cases, complete resection cannot be achieved.
Therefore, the recurrence rate of meningioma is relatively high
(23). Positive correlation among the
recurrence rate of meningioma, the degree of malignancy of
meningioma and the invasiveness of meningioma has been reported
(24). Principle factors affecting
the recurrence of meningioma were the biological characteristics of
tumor and surgical resection extent (25) (Table
IV).
In this study, 18 cases of meningioma with the
invasive area of cerebral convexity had Simpson grade I resection.
Also, we detected Simpson grade I resection in 4 cases of
meningioma with invasion to venous sinus area. In 12 cases, we had
Simpson grade II resection. In this study, meningioma invading
vascular, nerve and cavernous sinus were identified as WHO grade I,
there were 3 cases with Simpson grade II resection and 5 cases with
Simpson grade III resection, while we had no case with Simpson
grade I resection (Figs. 3 and
4).
In general, during surgeries we did not proceed to
complete resection, because in most cases some key structures were
invaded and meningioma was very deep and any attempt for total
resection could easily lead to significant damage to these
structures. In one case we had Simpson grade I resection, however,
because tumor cells were already infiltrating the brain tissue, the
surgery could not assure avoiding future recurrence. Therefore,
surgery could not achieve a full remedy in the case of meningioma
with high invasiveness, and postoperative radiotherapy was needed
to delay, or prevent the recurrence (26,27).
In conclusion, WHO grade I invasive meningioma was
the most common case among our patients, and the course of disease
was relatively short. Imaging examinations performed prior to the
surgery, played an important role in judging the position and
invasiveness of meningioma and the formulation of our surgical
plan. Surgical intervention was the preferred method of treatment
for invasive meningioma. During the surgery, the comprehensive
treatment of the total resection of tumor and invasive dura was
conducted, and surgical treatment was combined with postoperative
radiotherapy and postoperative follow-up in order to manage the
recurrence.
References
|
1
|
Gelabert-González M and Serramito-García
R: Intracranial meningiomas: I. Epidemiology, aetiology,
pathogenesis and prognostic factors. Rev Neurol. 53:165–172.
2011.(In Spanish). PubMed/NCBI
|
|
2
|
Bondy M and Ligon BL: Epidemiology and
etiology of intracranial meningiomas: a review. J Neurooncol.
29:197–205. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hanft S, Canoll P and Bruce JN: A review
of malignant meningiomas: diagnosis, characteristics, and
treatment. J Neurooncol. 99:433–443. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rockhill J, Mrugala M and Chamberlain MC:
Intracranial meningiomas: an overview of diagnosis and treatment.
Neurosurg Focus. 23:E12007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang S, Meng H, Gui Y, Chen L, Zhao T,
Zhang X, Li G and Yang G: On diagnostics of malignant meningioma
and invasive meningioma. Hua Xi Yi Ke Da Xue Xue Bao. 27:192–195.
1996.(In Chinese). PubMed/NCBI
|
|
6
|
Yamazaki T, Takahashi S, Ishii K,
Matsumoto K, Ishibashi T, Sakamoto K and Ogawa A: Meningioma in the
pineal region: preoperative diagnosis with CT, MRI, and
angiography. Radiat Med. 9:22–25. 1991.PubMed/NCBI
|
|
7
|
Chen XR: CT diagnosis of meningioma
(analysis of 215 cases). Zhonghua Fang She Xue Za Zhi. 20:292–295.
1986.(In Chinese). PubMed/NCBI
|
|
8
|
Freeman AC, Platt SR, Kent M and
Hofmeister E: What is the evidence? Diagnosis of an intracranial
lesion as a meningioma on the basis of MRI characteristics. J Am
Vet Med Assoc. 239:60–62. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Huang SQ, Liang BL, Xie BK, Yuan JP, Zhong
JL and Ye RX: MRI performance and diagnosis of meningioma - a
report of 126 cases. Ai Zheng. 23:1329–1333. 2004.PubMed/NCBI
|
|
10
|
Jain D, Ebrahimi KB, Miller NR and
Eberhart CG: Intraorbital meningiomas: a pathologic review using
current World Health Organization criteria. Arch Pathol Lab Med.
134:766–770. 2010.PubMed/NCBI
|
|
11
|
Nanda A, Bir SC, Maiti TK, Konar SK,
Missios S and Guthikonda B: Relevance of Simpson grading system and
recurrence-free survival after surgery for World Health
Organization Grade I meningioma. J Neurosurg. 126:201–211. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fisher JL, Schwartzbaum JA, Wrensch M and
Wiemels JL: Epidemiology of brain tumors. Neurol Clin. 25:867–890.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wrensch M, Minn Y, Chew T, Bondy M and
Berger MS: Epidemiology of primary brain tumors: current concepts
and review of the literature. Neuro Oncol. 4:278–299. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Huairong L, Peng S and Yaping Z: Detection
for single nucleotide polymorphisms. Hereditas. 23:471–476.
2001.
|
|
15
|
Chunxia Z, Xianzhe S and Shen L: Recent
advances in single nucleotide polymorphism of human genome
research. Chinese J Chromatog. 21:110–114. 2003.(In Chinese).
|
|
16
|
Younis GA, Sawaya R, DeMonte F, Hess KR,
Albrecht S and Bruner JM: Aggressive meningeal tumors: review of a
series. J Neurosurg. 82:17–27. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sołtys J, Pietniczka-Załeska M,
Młyńczyk-Budzynowska K and Majkowski J: Neoplasms of paranasal
sinuses in material of ENT Department MSS Hospital in Warsaw
between 2006–2007. Otolaryngol Pol. 62:451–454. 2008.(In Polish).
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Iaconetta G, Santella A, Friscia M, Abbate
V and Califano L: Extracranial primary and secondary meningiomas.
Int J Oral Maxillofac Surg. 41:211–217. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yamashima T, Kida S and Yamamoto S:
Ultrastructural comparison of arachnoid villi and meningiomas in
man. Mod Pathol. 1:224–234. 1988.PubMed/NCBI
|
|
20
|
Daneshi A, Asghari A and Bahramy E:
Primary meningioma of the ethmoid sinus: a case report. Ear Nose
Throat J. 82:310–311. 2003.PubMed/NCBI
|
|
21
|
Preston DL, Ron E, Yonehara S, Kobuke T,
Fujii H, Kishikawa M, Tokunaga M, Tokuoka S and Mabuchi K: Tumors
of the nervous system and pituitary gland associated with atomic
bomb radiation exposure. J Natl Cancer Inst. 94:1555–1563. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ron E, Modan B, Boice JD Jr, Alfandary E,
Stovall M, Chetrit A and Katz L: Tumors of the brain and nervous
system after radiotherapy in childhood. N Engl J Med.
319:1033–1039. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yamada SM, Yamada S, Takahashi H, Teramoto
A and Matsumoto K: Extracranially extended meningothelial
meningiomas with a high MIB-1 index: a report of two cases.
Neuropathology. 24:66–71. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lombardi D, Tomenzoli D, Buttà L, Bizzoni
A, Farina D, Sberze F, Karligkiotis A, Castelnuovo P and Nicolai P:
Limitations and complications of endoscopic surgery for treatment
for sinonasal inverted papilloma: a reassessment after 212 cases.
Head Neck. 33:1154–1161. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Busquets JM and Hwang PH: Endoscopic
resection of sinonasal inverted papilloma: a meta-analysis.
Otolaryngol Head Neck Surg. 134:476–482. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gotlib T, Osuch-Wójcikiewicz E,
Held-Ziółkowska M, Kużmińska M and Niemczyk K: Endoscopic
transnasal management of sinonasal malignancies - our initial
experience. Wideochir Inne Tech Malo Inwazyjne. 9:131–137.
2014.PubMed/NCBI
|
|
27
|
Lund VJ, Stammberger H, Nicolai P,
Castelnuovo P, Beal T, Beham A, Bernal-Sprekelsen M, Braun H,
Cappabianca P, Carrau R, et al: European Rhinologic Society
Advisory Board on Endoscopic Techniques in the Management of Nose,
Paranasal Sinus and Skull Base Tumours: European position paper on
endoscopic management of tumours of the nose, paranasal sinuses and
skull base. Rhinol. 22:(Suppl). 1–143. 2010.
|