Introduction
Adenocarcinoma of the esophagogastric junction (AEG)
is a type of adenocarcinoma at the junction between the esophagus
and stomach. As classified by Siewert and Stein (1), the prototypical AEG is located within
the region 1 cm above and 2 cm below the esophagogastric junction.
Due to its unique epidemiological, genetic and prognostic
characteristics, AEG has been regarded as a disease that is
distinct from other types of gastric cancer (2). Previous studies have demonstrated that
the incidence of AEG has increased in Western countries during last
decade (3,4). Due to the lack of diagnostic molecular
biomarkers, >80% of AEG cases are diagnosed at the advanced
stage, resulting in a poor prognosis with <30% of patients
having 5-year overall survival (OS) (5).
SGK1 (serum- and glucocorticoid-induced protein
kinase-1) is a ubiquitous serine/threonine kinase belonging to the
protein kinase A, G and C family, which is able to be activated by
multiple stimuli, such as serum, follicle stimulating hormone,
osmotic shock, ischemia, glucocorticoids and other cytokines
(6,7).
As a downstream molecule in the phosphatidylinositol-3 kinase
(PI3K) signaling pathway, SGK1 regulates numerous target genes,
influencing many physiological processes, such as proliferation,
differentiation and apoptosis (8,9). Previous
studies have demonstrated that the expression of SGK1 is
significantly increased in many types of cancer, including
colorectal, breast and lung cancer (10–12).
Inhibition of SGK1 or SGK1 gene knockdown was revealed to inhibit
the growth of tumor cells and increase the resistance of cells to
chemically-mediated carcinogenesis (10,13).
However, several studies reported conflicting results, whereby SGK1
expression was demonstrated to be decreased in cancerous tissue
compared with adjacent control groups in prostate cancer, ovarian
cancer, and adrenocortical tumor (14–16).
Although these results suggest aberrant expression of SGK1 occurs
in various cancerous tissue, which may affect the progression and
prognosis of different types of cancer, the expression of SGK1 in
AEG, particularly the association with the clinicopathological
characteristics and prognosis of patients with AEG, remains to be
established.
In the present study, the expression of SGK1 was
determined in cancerous tissue samples of patients with AEG.
Furthermore, the association between SGK1 expression and various
clinicopathological characteristics were assessed in order to
determine whether SGK1 is a potential molecular marker for the
diagnosis, interference therapy and prognosis of AEG.
Materials and methods
Patient tissue samples
A total of 60 cases of postoperative pathologically
diagnosed AEG and 20 healthy controls between 2012 and 2014 from
The First Affiliated Hospital of the University of Henan University
of Science and Technology (HAUST; Luoyang, China) and Anyang Tumor
Hospital (Anyang, China were investigated. The tissue samples
(cancerous and adjacent healthy) resected from patients were
retrieved from the pathology departments of the two hospitals. The
specimens were fixed in 10% formalin at 37°C for 24 h and embedded
in paraffin wax. The paraffin embedded tissue blocks were freshly
cut into 4-µm thick slices for subsequent immunohistochemistry
analysis. Adjacent tissue samples were obtained 3 cm away from the
cancerous tissue. Healthy control samples were selected from
esophagogastric junction biopsy tissues obtained during endoscopic
examinations of 20 age- and gender-matched participants. All
samples, including healthy controls, were confirmed histologically
by two pathologists. Demographics (gender and age) and
clinicopathological characteristics (differentiation status,
lymphatic invasion, lymph node metastasis and tumor-node-metastasis
stage) were obtained from patient medical records. OS rates were
determined over 60 months. Up to the date of follow-up, 2 were
unavailable, 45 succumbed and 13 were alive by the end of the
follow-up period. The present study was approved by the
Institutional Review Board of HAUST. Ethical permission was
received from the Regional Ethical Board of the First Affiliated
Hospital of HAUST and Anyang Tumor Hospital, and the written
informed consent was obtained from all patients.
Immunohistochemistry analysis
The slides were dewaxed and rehydrated in a
descending series of alcohol, immunohistochemical detection was
performed using an indirect immunoperoxidase technique (17) following high-temperature (100°C)
antigen retrieval in 10 mM citric acid monohydrate buffer (pH 6.5;
Sigma; Merck KGaA, Darmstadt, Germany) for 13 min. Blocking of
endogenous peroxidase and non-specific staining was performed.
Samples were incubated at 4°C overnight with primary rabbit
monoclonal antibody directed against SGK1 (cat. no. ab32374;
dilution, 1:200; Abcam, Cambridge, MA, USA) and PBS was used as a
negative control for the primary antibody. Signal amplification was
achieved using biotin-labeled anti-rabbit/rat and chain mildew
avidin-peroxidase conjugated secondary antibody (cat. no. ZS-9001;
OriGene Technologies, Inc., Beijing, China) at room temperature for
1 h. The Streptavidin-Biotin DAB kit (cat. no. ZLI-9019; OriGene
Technologies, Inc.) was used to stain sections for between 2 and 5
min, nuclei were counterstained with hematoxylin. SGK1 was
exclusively expressed in the cytoplasm. Immunostaining scores were
calculated using light microscopy (Eclipse 80i; Nikon Corporation,
Tokyo, Japan) by two independent experienced pathologists who were
unaware of the patients' clinical details. Whenever discrepancies
occurred, the results were jointly assessed by the two
investigators and the final score was formed by consensus. The
staining intensity of SGK1 expression was classified using the
following numerical scale: <10% stained cells, negative (grade
0); 10–30%, weak (grade 1); 30–60%, moderate (grade 2); and ≥60%,
strong (grade 3). A grade of ≥2 was considered positively-stained
for the SGK1 antibody.
Statistical analysis
The continuous data are presented as the mean ±
standard deviation. The χ2 test was used to analyze the
differences in the distribution of clinicopathological
characteristics and to analyze differences between categorical
variables. OS was defined as the time between the date of primary
diagnosis and mortality. Kaplan-Meier estimator analysis and the
log-rank test were used to estimate differences in OS in strata
according to high and low SGK1 expression. All tests were
two-tailed. The corresponding 95% confidence interval (CI) was
calculated, and the univariate and multivariate analysis were
performed using SPSS software (version 21.0; IBM SPSS, Armonk, NY,
USA). P<0.05 was considered to indicate a statistically
significant difference.
Results
Expression of SGK1 is higher in the
cancerous tissue of patients with AEG compared with non-cancerous
tissue and the healthy control group
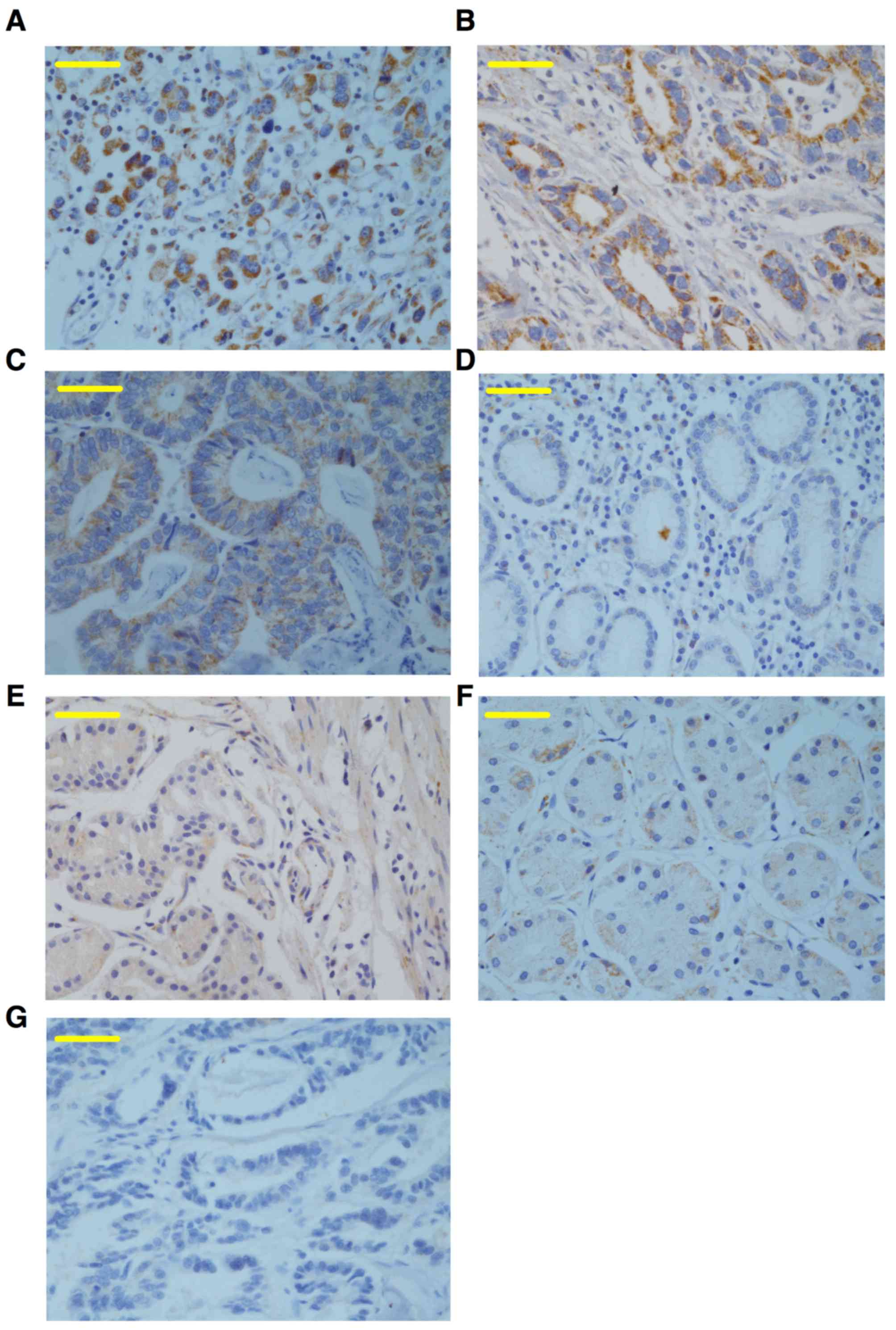
To determine SGK1 expression in cancerous, adjacent
and healthy control tissue samples, immunohistochemical staining
for SGK1 was performed on samples from 60 cases of
paraffin-embedded AEG and healthy control tissues. The results
revealed that SGK1 was positively expressed as brown-yellow
granules, which were primarily localized to the epithelial cell
cytoplasm and weakly expressed in the nuclei (Fig. 1). The expression of SGK1 was
significantly higher in cancerous tissue from patients with AEG
(65%), compared with the adjacent non-cancerous tissues (31.7%) and
healthy controls (10%) (both P<0.0001; Fig. 1; Table
I), indicating that the expression of SGK1 may be a biomarker
for AEG.
 | Table I.Expression of serum- and
glucocorticoid-regulated kinase in the cancerous and adjacent
tissues of patients with AEG and the healthy control group. |
Table I.
Expression of serum- and
glucocorticoid-regulated kinase in the cancerous and adjacent
tissues of patients with AEG and the healthy control group.
| Group | No. of patients | Positive (%) | Negative (%) | P-value |
|---|
| AEG cancerous | 60 | 39 (65) | 21 (35) |
|
| Adjacent control | 60 | 19 (31.7) | 41 (62.3) | <0.0001 |
| Healthy control | 20 | 2 (10) | 18 (90) |
|
Higher expression of SGK1 is
positively correlated with poor differentiation and severe lymph
node metastasis
As the expression of SGK1 was demonstrated to be
higher in the cancerous AEG tissue, the association between the
expression of SGK1 and the progression of AEG was examined.
Clinicopathological characteristics of the patients with AEG are
presented in Table II. Although the
expression of SGK1 was not significantly associated with age,
gender, pathological type, tumor size and tumor stage, SGK1
expression was positively associated with the differentiation and
lymph node metastasis status (both P<0.05; Table II). Positive SGK1 immunostaining was
present in 61.5% (24/39) of the poorly differentiated tissue
samples, which was significantly higher compared with the well
(3/39; 7.7%) or moderately (12/39; 30.8%) differentiated samples
(both P<0.05; Table II).
Furthermore, the percentage of SGK1-positively stained tissue in
the N2-3 lymph node metastasis group was 79.4% (31/39), which was
significantly higher compared with that in the N0-1 group (20.6%)
(P=0.006; Table II). These results
reveal that a higher expression of SGK1 is positively correlated
with poor differentiation and severe lymph node metastasis,
suggesting that higher SGK1 expression may be a novel etiological
agent and potential indicator of poor prognosis for AEG.
 | Table II.Association between the expression of
SGK1 and clinicopathological characteristics of patients with
adenocarcinoma of esophagogastric junction. |
Table II.
Association between the expression of
SGK1 and clinicopathological characteristics of patients with
adenocarcinoma of esophagogastric junction.
| Clinicopathological
characteristic | Null/low SGK1
expression | Medium SGK1
expression | Strong SGK1
expression | No. of patients | χ2 | P-value |
|---|
| Gender |
|
|
|
| 0.277 | 0.87 |
| Male | 11 | 10 | 13 | 34 |
|
|
|
Female | 8 | 6 | 12 | 26 |
|
|
| Age, years |
|
|
|
| 0.371 | 0.543 |
| ≤62 | 12 | 8 | 11 | 31 |
|
|
|
>62 | 9 | 8 | 12 | 29 |
|
|
|
Differentiation |
|
|
|
| 9.746 | 0.045a |
|
Well | 6 | 1 | 2 | 9 |
|
|
|
Middle | 9 | 3 | 9 | 21 |
|
|
|
Low | 6 | 12 | 12 | 30 |
|
|
| Lymph node
status |
|
|
|
| 7.447 | 0.006a |
|
N0-N1 | 13 | 3 | 5 | 21 |
|
|
|
N2-N3 | 8 | 13 | 18 | 39 |
|
|
| Tumor size |
|
|
|
| 3.313 | 0.191 |
| T2 | 8 | 10 | 8 | 26 |
|
|
| T3 | 13 | 6 | 15 | 34 |
|
|
|
Tumor-node-metastasis |
|
|
|
| 3.504 | 0.061 |
|
I–II | 9 | 3 | 4 | 16 |
|
|
|
III | 12 | 13 | 19 | 44 |
|
|
| Pathological
type |
|
|
|
| 0.196 | 0.658 |
|
Protruding | 2 | 0 | 6 | 8 |
|
|
|
Ulcerative | 12 | 9 | 9 | 30 |
|
|
| Ulcer
infiltrating | 3 | 2 | 3 | 8 |
|
|
|
Infiltrate | 4 | 5 | 5 | 14 |
|
|
SGK1 expression is negatively
correlated with overall AEG survival rate
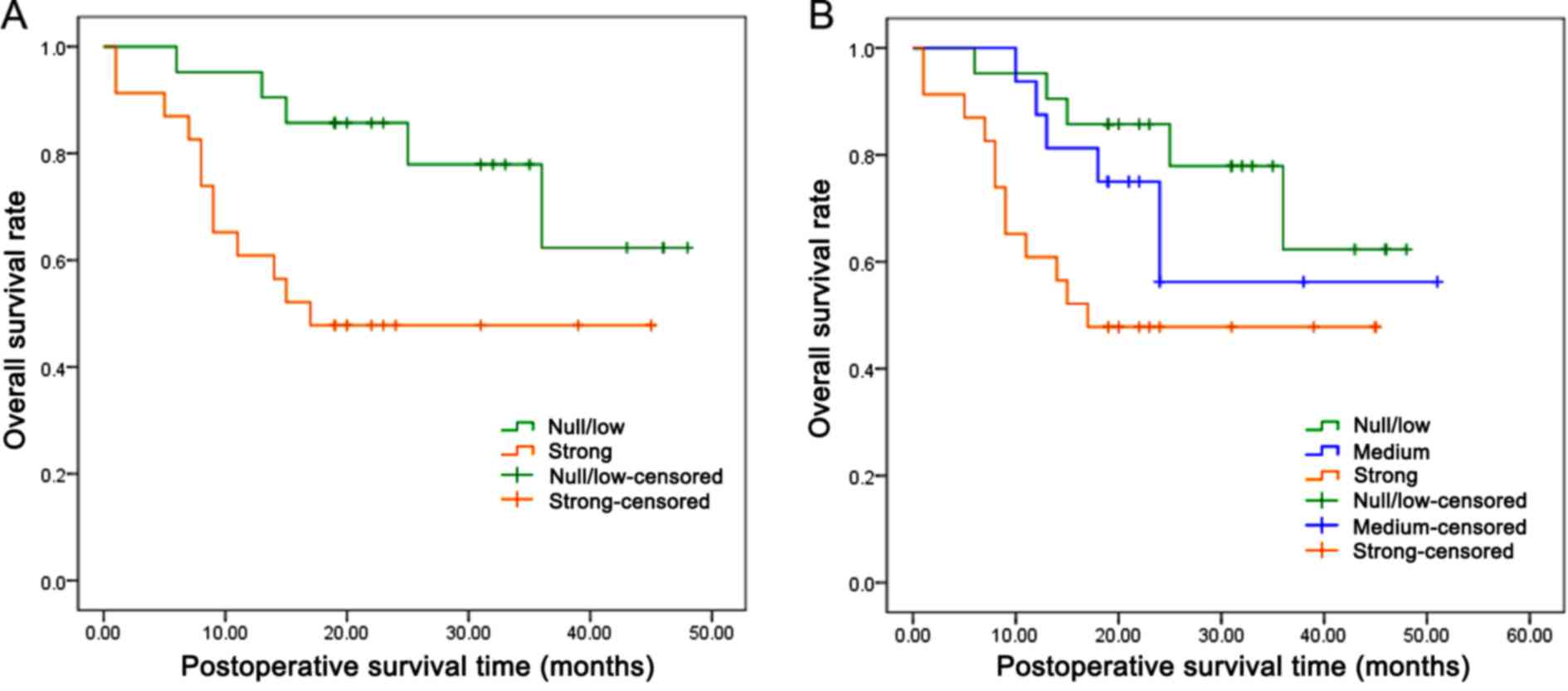
In order to assess the potential consequence of
higher SGK1 expression on patients with AEG, the overall cumulative
survival rate in patients was compared with different levels of
SGK1 expression. A total of 58 patients (2 patients were not
available) were followed up for survival analysis over a period of
60 months. Because of the limited follow-up time, the survival rate
of the two groups was >25% and the median survival time could
not be calculated. However, the mean survival time for patients
with AEG with strong SGK1-positive staining was 26.08 months,
significantly lower compared with the SGK1-negative group (39.10
months) (P=0.027; Fig. 2A, Table III). Additionally, further analysis
demonstrated that the patients with weak and moderate SGK1-positive
staining also exhibited a significantly shorter mean survival time
of 36.50 months, as compared with the SGK1-negative group (P=0.049;
Fig. 2B, Table III). In addition, univariate and
multivariate prognosis analyses were performed using the Cox's
proportional hazards regression model to analyze if higher
expression of SGK1 was an independent prognostic factor for the
overall survival of patients. As illustrated in Table IV, univariate prognosis analysis
revealed that the overall survival of patients with AEG was
significantly associated with a higher expression level of SGK1
(P=0.023) and poorer lymph node stage (P=0.020). However, the
multivariate Cox's proportional hazards model revealed no
significant association between overall survival and the lymph node
status (P=0.074) or the expression of SGK1 (P=0.135) (Table IV), suggesting that the status of
SGK1 expression is not an independent prognostic factor in patients
with AEG.
 | Table III.Means and medians for the survival
time (months) of patients with adenocarcinoma of esophagogastric
junction with expression of SGK1. |
Table III.
Means and medians for the survival
time (months) of patients with adenocarcinoma of esophagogastric
junction with expression of SGK1.
|
|
|
| Mean 95% confidence
interval | Median |
|
|---|
|
|
|
|
|
|
|
|---|
| SGK1 |
Estimationa | SEa | Lower bound | Upper bound |
Estimationa | SEa | Lower bound | Upper bound | P-value |
|---|
| Null/low | 39.100 | 3.371 | 32.493 | 45.706 | N/A | N/A | N/A | N/A |
|
| Strong | 26.087 | 3.845 | 18.551 | 33.623 | 17.000 | N/A | N/A | N/A |
|
| Overall | 32.862 | 2.881 | 27.216 | 38.508 | N/A | N/A | N/A | N/A | 0.027b |
| Null/low | 39.100 | 3.371 | 32.493 |
| N/A | N/A | N/A | N/A |
|
| Medium | 36.500 | 5.533 | 25.654 |
| N/A | N/A | N/A | N/A |
|
| Strong | 26.087 | 3.845 | 18.551 |
| 17.000 | N/A | N/A | N/A |
|
| Overall | 35.218 | 2.685 | 29.955 |
| N/A | N/A | N/A | N/A | 0.049c |
 | Table IV.Univariate and multivariate analysis
showing overall survival in patients with adenocarcinoma of
esophagogastric junction. |
Table IV.
Univariate and multivariate analysis
showing overall survival in patients with adenocarcinoma of
esophagogastric junction.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Covariates | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Gender | 1.676
(0.567–4.567) | 0.350 |
|
|
| Age | 0.997
(0.429–2.429) | 0.994 |
|
|
| Pathological
type | 0.659
(0.401–1.401) | 0.112 |
|
|
|
Differentiation | 1.659
(0.867–3.867) | 0.127 |
|
|
| Invasion depth | 1.804
(0.694–4.694) | 0.226 |
|
|
| Lymph node | 2.066
(1.121–3.121) | 0.020* | 1.366
(0.461–4.461) | 0.074 |
| TNM | 4.231
(0.987–18.987) | 0.052* |
|
|
| SGK1 | 1.838
(1.088–3.088) | 0.023* | 1.598
(0.872–2.872) | 0.135 |
Discussion
To the best of our knowledge, the present study is
the first to examine the expression of SGK1 and investigate its
association with the clinicopathological characteristics and
prognosis of patients with AEG. In the present study, it was
established that SGK1 expression is significantly higher in the
cancerous tissue of patients with AEG as compared with adjacent
tissue and the healthy control group. Furthermore, the analysis
indicates that a higher expression level of SGK1 was correlated
with multiple clinicopathological factors including cancer cell
differentiation, lymph node metastasis and the overall survival
rate of patients with AEG. These results provide the first evidence
that high SGK1 expression may be a novel risk factor for the
progression of AEG, and may also serve as a prognostic biomarker
for this increasingly prevalent cancer.
SGK1 has been demonstrated to share >50% sequence
similarity with another serine/threonine kinase protein kinase B
(Akt), a downstream target of the PI3K signaling pathway (18,19).
Additionally, SGK1 is able to be fully activated through
phosphorylation at Thr256 by phosphoinositide dependent protein
kinase-1 (PDK) 1 and Ser422 by PDK2 similar to Akt, suggesting that
SGK1 may be involved in the parallel signaling pathways that Akt is
involved in (20). Considering the
extensively distributed and essential function of PI3K-AKT pathway
in the progression of various cancers (21), the aberrant expression of SGK1 in
cancerous tissues was expected. For the first time, the results of
the present study have demonstrated that SGK1 is highly expressed
in AEG, which is similar to its expression in colon, breast and
lung cancer (10–12). However, decreased expression of SGK1
has also been reported in certain cancers, including prostate and
ovarian cancer, and adrenocortical tumor (14–16). These
controversial results may be due to the specificity of different
types of cancer, particularly considering that these three cancer
types with a low expression of SGK1 are all associated with the
endocrinal system, which may contribute to the lower expression of
SGK1 through certain unidentified mechanisms. These controversial
results also encourage further investigations in other cancer types
from the same body system, such as esophageal and gastric cancer,
which may be helpful to clarify the expression profile of SGK1 and
its significance in the progression of cancer.
In the present study, it was identified that a
higher expression level of SGK1 is associated with the poor
differentiation and worse prognosis of patients with AEG. This
result is consistent with the majority of previous studies that
demonstrated that SGK1 was increased in the cancerous tissue of
multiple cancers such as lung, breast and colorectal cancer. Using
SGK1 knockout mice, Nasir et al (10) demonstrated that SGK1 deficiency
increased the resistance of mice to chemical-induced
carcinogenesis. Combined with the results from the study performed
by Abbruzzese et al (12),
which revealed that higher SGK1 mRNA expression was associated with
the prognostic indicators of lung cancer, including tumor size and
clinical stage as classified in non-small cell lung cancer, the
results of the present study suggest that a higher expression of
SGK1 is an etiological agent and/or prognosis indicator of AEG.
In the current study, the correlation of SGK1
expression with the overall survival of patients with AEG was also
analyzed. The results demonstrated that elevated SGK1 staining was
associated with a poorer prognosis in patients. Thus, SGK1
upregulation may be a poor prognosis indicator in patients with
AEG. An increase in postoperative visitation for patients with high
SGK1 expression may identify the recurrence and metastasis of AEG
at an early stage, which has important significance for increasing
the detection rate and early treatment of AEG. Survival follow-up
outcome analysis identified that the mortality rate for patients
with high expression of SGK1 was almost 0% after 2 years. It was
also noted that ~50% of the patients were followed for <36
months, thus further follow-up is required and more significant
results may be achieved once the clinical data is more
complete.
Although the molecular mechanism underlying the
association between higher SGK1 expression and poor differentiation
and worse prognosis has not been demonstrated in the present study,
a number of aspects of activated SGK1 provide a plausible basis for
the potential higher risk of increased SGK1 in cancerous tissue.
Firstly, it has been previously demonstrated that SGK1 is
associated with the progression of inflammation (22–24), and
inflammation per se has been demonstrated to be associated
with the development of multiple types of cancer (25,26). A
previous study demonstrated that active SGK1 decreases the
production of pro-inflammatory cytokines, such as tumor necrosis
factor, interleukin (IL)-12, and IL-1 (23). Due to the essential roles of the
pro-inflammatory cytokines in the modulation of cancer progression,
such as the role of IL-1 in priming interferon γ-producing
tumor-antigen-specific CD8+ T cells, higher expression
of SGK1 could inhibit the activation of specific immune responses
to cancer cells, promoting immune evasion. Secondly, the activation
of SGK1 has been revealed to phosphorylate forkhead box protein O3
(FoxO3), an established potent tumor suppressor, through inducing
cell cycle arrest and apoptosis in multiple cancers (27–29).
Higher expression of SGK1-phosphorylated FoxO3 results in its
export from the nucleus to the cytoplasm, possibly deactivating the
anti-tumorigenesis property of FoxO3 (30). Thirdly, activation of SGK1 has been
reported to promote the expression of pro-matrix metalloproteinase
9 expression (31), which may aid in
explaining the pro-metastasis effects of higher expressed SGK1 in
patients with AEG. Lastly, activated SGK1 has been demonstrated to
promote epithelial cell survival under conditions of cellular
stress, such as serum-deprivation and chemotherapy treatment
(30,32), increased SGK1 expression may promote
the progression of AEG through this mechanism.
In conclusion, the present study has demonstrated
for the first time that the expression of SGK1 is significantly
increased in cancerous AEG tissue, and that upregulated SGK1 is
associated with a more aggressive and poor prognosis in patients
with AEG. Although the pathogenesis mechanism underlying the role
of SGK1 in AEG warrants further investigation in vivo and
in vitro, the results of the current study indicate that
SGK1 is a novel potential target for the early clinical diagnosis,
intervention therapy, and prognosis of AEG.
Acknowledgements
The present study was supported by the Program for
the Innovation Team of Science and Technology in Universities of
Henan Province (grant no. 15IRTSTHN024), the Development Program
for the Key Laboratories of Henan Province (grant no. 2015SPT003)
and the National Institute of Dental and Craniofacial Research
(grant no. DE023633; received by HW).
References
|
1
|
Engstrom PF, Arnoletti JP, Benson AB III,
Chen YJ, Choti MA, Cooper HS, Covey A, Dilawari RA, Early DS,
Enzinger PC, et al: NCCN clinical practice guidelines in oncology:
Colon cancer. J Natl Compr Canc Netw. 7:778–831. 2009.PubMed/NCBI
|
|
2
|
Liu K, Zhang W, Chen X, Chen X, Yang K,
Zhang B, Chen Z, Zhou Z and Hu J: Comparison on clinicopathological
features and prognosis between esophagogastric junctional
adenocarcinoma (Siewert II/III Types) and distal gastric
adenocarcinoma: Retrospective cohort study, a single institution,
high volume experience in china. Medicine (Baltimore).
94:e13862015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vial M, Grande L and Pera M: Epidemiology
of adenocarcinoma of the esophagus, gastric cardia, and upper
gastric third. Recent Results Cancer Res. 182:1–17. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Buas MF and Vaughan TL: Epidemiology and
risk factors for gastroesophageal junction tumors: Understanding
the rising incidence of this disease. Semin Radiat Oncol. 23:3–9.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Siewert JR, Feith M and Stein HJ: Biologic
and clinical variations of adenocarcinoma at the esophago-gastric
junction: Relevance of a topographic-anatomic subclassification. J
Surg Oncol. 90:139–146. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lang F, Böhmer C, Palmada M, Seebohm G,
Strutz-Seebohm N and Vallon V: (Patho) physiological significance
of the serum-and glucocorticoid-inducible kinase isoforms. Physiol
Rev. 86:1151–1178. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lang F and Cohen P: Regulation and
physiological roles of serum-and glucocorticoid-induced protein
kinase isoforms. Sci STKE. 2001:re172001.PubMed/NCBI
|
|
8
|
Brunet A, Park J, Tran H, Hu LS, Hemmings
BA and Greenberg ME: Protein kinase SGK mediates survival signals
by phosphorylating the forkhead transcription factor FKHRL1
(FOXO3a). Mol Cell Biol. 21:952–965. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lang F, Henke G, Embark HM, Waldegger S,
Palmada M, Böhmer C and Vallon V: Regulation of channels by the
serum and glucocorticoid-inducible kinase-implications for
transport, excitability and cell proliferation. Cell Physiol
Biochem. 13:41–50. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nasir O, Wang K, Föller M, Gu S, Bhandaru
M, Ackermann TF, Boini KM, Mack A, Klingel K, Amato R, et al:
Relative resistance of SGK1 knockout mice against chemical
carcinogenesis. IUBMB Life. 61:768–776. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sommer EM, Dry H, Cross D, Guichard S,
Davies BR and Alessi DR: Elevated SGK1 predicts resistance of
breast cancer cells to Akt inhibitors. Biochem J. 452:499–508.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Abbruzzese C, Mattarocci S, Pizzuti L,
Mileo AM, Visca P, Antoniani B, Alessandrini G, Facciolo F, Amato
R, D'Antona L, et al: Determination of SGK1 mRNA in non-small cell
lung cancer samples underlines high expression in squamous cell
carcinomas. J Exp Clin Cancer Res. 31:42012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Talarico C, D'Antona L, Scumaci D, Barone
A, Gigliotti F, Fiumara CV, Dattilo V, Gallo E, Visca P, Ortuso F,
et al: Preclinical model in HCC: The SGK1 kinase inhibitor SI113
blocks tumor progression in vitro and in vivo and synergizes with
radiotherapy. Oncotarget. 6:37511–37525. 2015.PubMed/NCBI
|
|
14
|
Rauhala HE, Porkka KP, Tolonen TT,
Martikainen PM, Tammela TL and Visakorpi T: Dual-specificity
phosphatase 1 and serum/glucocorticoid-regulated kinase are
downregulated in prostate cancer. Int J Cancer. 117:738–745. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chu S, Rushdi S, Zumpe ET, Mamers P, Healy
DL, Jobling T, Burger HG and Fuller PJ: FSH-regulated gene
expression profiles in ovarian tumours and normal ovaries. Mol Hum
Reprod. 8:426–433. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ronchi CL, Sbiera S, Leich E, Tissier F,
Steinhauer S, Deutschbein T, Fassnacht M and Allolio B: Low SGK1
expression in human adrenocortical tumors is associated with
ACTH-independent glucocorticoid secretion and poor prognosis. J
Clin Endocrinol Metab. 97:E2251–E2260. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pirker R, Pereira JR, von Pawel J,
Krzakowski M, Ramlau R, Park K, de Marinis F, Eberhardt WE,
Paz-Ares L, Störkel S, et al: EGFR expression as a predictor of
survival for first-line chemotherapy plus cetuximab in patients
with advanced non-small-cell lung cancer: Analysis of data from the
phase 3 FLEX study. Lancet Oncol. 13:33–42. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Webster MK, Goya L, Ge Y, Maiyar AC and
Firestone GL: Characterization of sgk, a novel member of the
serine/threonine protein kinase gene family which is
transcriptionally induced by glucocorticoids and serum. Mol Cell
Biol. 13:2031–2040. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Park J, Leong ML, Buse P, Maiyar AC,
Firestone GL and Hemmings BA: Serum and glucocorticoid-inducible
kinase (SGK) is a target of the PI3-kinase-stimulated signaling
pathway. EMBO J. 18:3024–3033. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kobayashi T and Cohen P: Activation of
serum-and glucocorticoid-regulated protein kinase by agonists that
activate phosphatidylinositide 3-kinase is mediated by
3-phosphoinositide-dependent protein kinase-1 (PDK1) and PDK2.
Biochem J. 339:319–328. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mundi PS, Sachdev J, McCourt C and
Kalinsky K: AKT in cancer: New molecular insights and advances in
drug development. Br J Clin Pharmacol. 82:943–956. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wu C, Yosef N, Thalhamer T, Zhu C, Xiao S,
Kishi Y, Regev A and Kuchroo VK: Induction of pathogenic TH17 cells
by inducible salt-sensing kinase SGK1. Nature. 496:513–517. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou H, Gao S, Duan X, Liang S, Scott DA,
Lamont RJ and Wang H: Inhibition of serum-and
glucocorticoid-inducible kinase 1 enhances TLR-mediated
inflammation and promotes endotoxin-driven organ failure. FASEB J.
29:3737–3749. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gu Z, Lamont GJ, Lamont RJ, Uriarte SM,
Wang H and Scott DA: Resolvin D1, resolvin D2 and maresin 1
activate the GSK3β anti-inflammatory axis in TLR4-engaged human
monocytes. Innate Immun. 22:186–195. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bhatelia K, Singh K and Singh R: TLRs:
Linking inflammation and breast cancer. Cell Signal. 26:2350–2357.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gao S, Brown J, Wang H and Feng X: The
role of glycogen synthase kinase 3-β in immunity and cell cycle:
Implications in esophageal cancer. Arch Immunol Ther Exp (Warsz).
62:131–144. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dehner M, Hadjihannas M, Weiske J, Huber O
and Behrens J: Wnt signaling inhibits Forkhead box O3a-induced
transcription and apoptosis through up-regulation of serum-and
glucocorticoid-inducible kinase 1. J Biol Chem. 283:19201–19210.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Huang H and Tindall DJ: Dynamic FoxO
transcription factors. J Cell Sci. 120:2479–2487. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
You H, Jang YJ, You-Ten AI, Okada H, Liepa
J, Wakeham A, Zaugg K and Mak TW: p53-dependent inhibition of
FKHRL1 in response to DNA damage through protein kinase SGK1. Proc
Natl Acad Sci USA. 101:14057–14062. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Brunet A, Park J, Tran H, Hu LS, Hemmings
BA and Greenberg ME: Protein kinase SGK mediates survival signals
by phosphorylating the forkhead transcription factor FKHRL1
(FOXO3a). Mol Cell Biol. 21:952–965. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Borst O, Schaub M, Walker B, Schmid E,
Münzer P, Voelkl J, Alesutan I, Rodríguez JM, Vogel S, Schoenberger
T, et al: Pivotal role of serum-and glucocorticoid-inducible kinase
1 in vascular inflammation and atherogenesis. Arterioscler Thromb
Vasc Biol. 35:547–557. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mikosz CA, Brickley DR, Sharkey MS, Moran
TW and Conzen SD: Glucocorticoid receptor-mediated protection from
apoptosis is associated with induction of the serine/threonine
survival kinase gene, sgk-1. J Biol Chem. 276:16649–16654. 2001.
View Article : Google Scholar : PubMed/NCBI
|