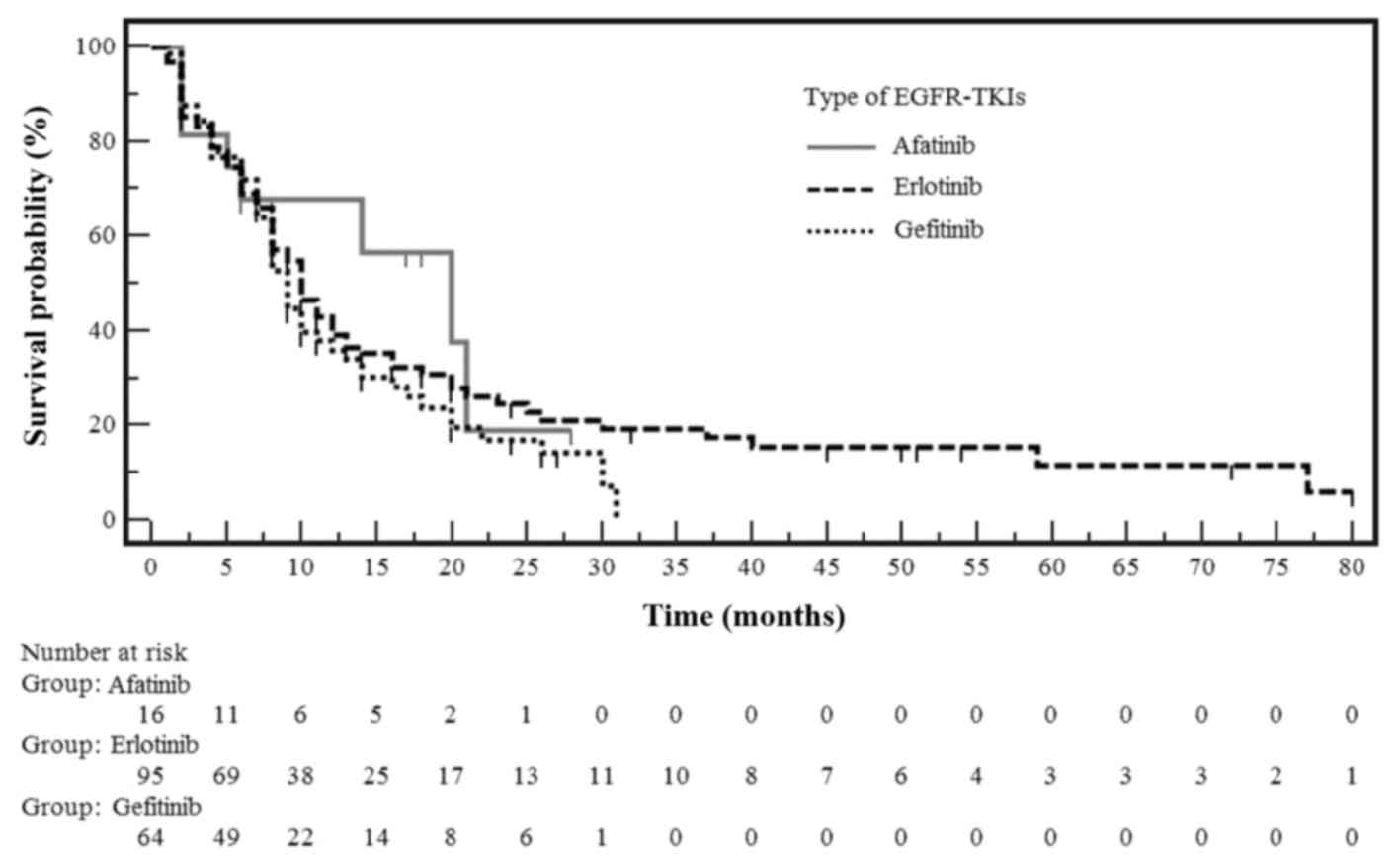
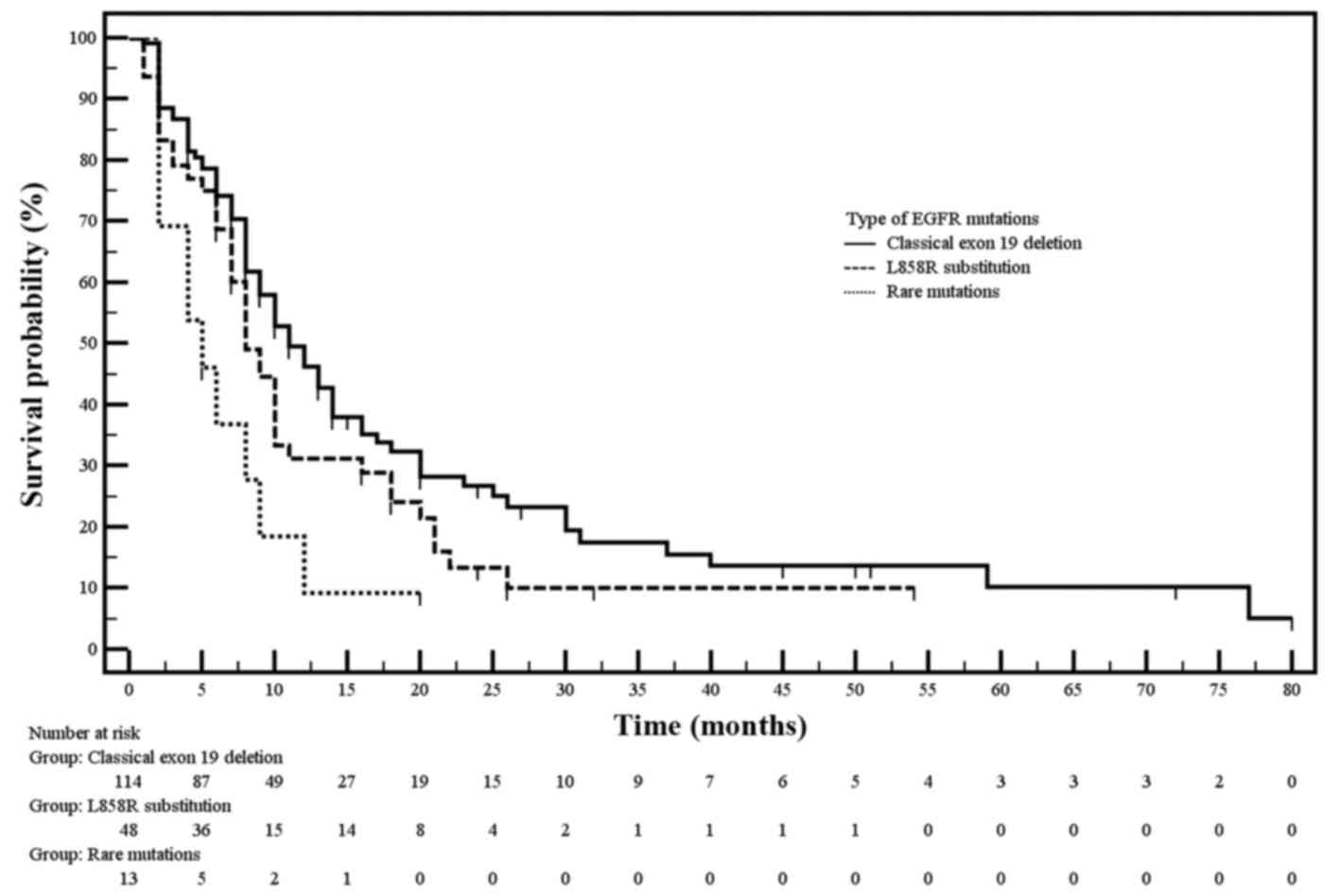
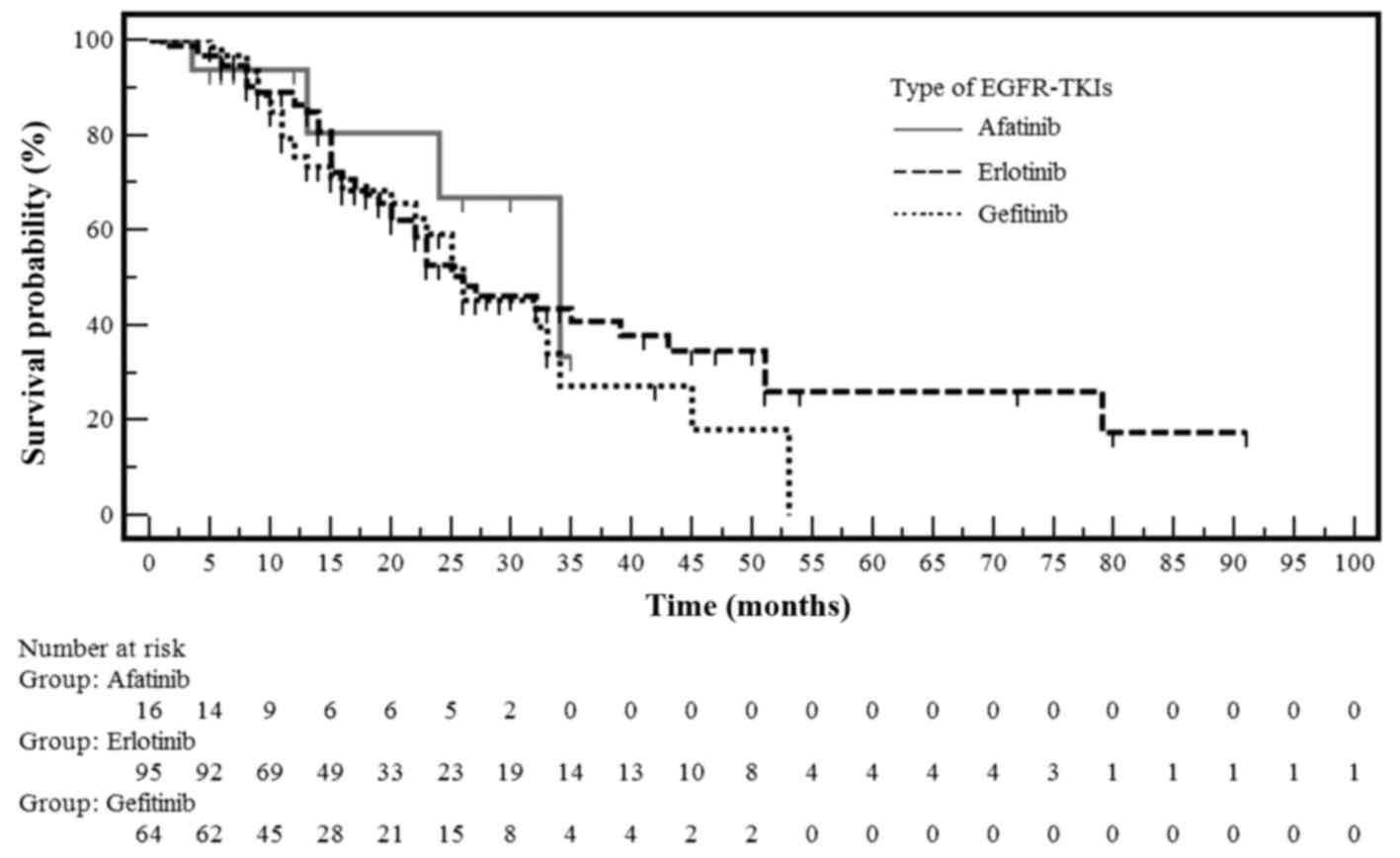
|
1
|
Maemondo M, Inoue A, Kobayashi K, Sugawara
S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I,
et al: Gefitinib or chemotherapy for non-small-cell lung cancer
with mutated EGFR. N Engl J Med. 362:2380–2388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mok TS, Wu YL, Thongprasert S, Yang CH,
Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, et
al: Gefitinib or carboplatin-paclitaxel in pulmonary
adenocarcinoma. N Engl J Med. 361:947–957. 2010. View Article : Google Scholar
|
|
3
|
Rosell R, Carcereny E, Gervais R,
Vergnenegre A, Massuti B, Felip E, Palmero R, Garcia-Gomez R,
Pallares C, Sanchez JM, et al: Erlotinib versus standard
chemotherapy as first-line treatment for European patients with
advanced EGFR mutation-positive non-small-cell lung cancer
(EURTAC): A multicentre, open-label, randomised phase 3 trial.
Lancet Oncol. 13:239–246. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wu WS, Wu CH, Lai SL, Chiu CH, Shih JF,
Lee YC and Chen YM: Erlotinib salvage therapy in pulmonary
adenocarcinoma patients with disease progression after previous
EGFR-TKI treatment. Am J Clin Oncol. 39:556–562. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wu YL, Zhou C, Hu CP, Feng J, Lu S, Huang
Y, Li W, Hou M, Shi JH, Lee KY, et al: Afatinib versus cisplatin
plus gemcitabine for first-line treatment of Asian patients with
advanced non-small-cell lung cancer harbouring EGFR mutations
(LUX-Lung 6): An open-label, randomised phase 3 trial. Lancet
Oncol. 15:213–222. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim ST, Uhm JE, Lee J, Sun JM, Sohn I, Kim
SW, Jung SH, Park YH, Ahn JS, Park K and Ahn MJ: Randomized phase
II study of gefitinib versus erlotinib in patients with advanced
non-small cell lung cancer who failed previous chemotherapy. Lung
Cancer. 75:82–88. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Park K, Tan E-H, Zhang L, Hirsh V, O'Byrne
K and Boyer M: LBA2_PR Afatinib (A) vs gefitinib (G) as first-line
treatment for patients (pts) with advanced non-small cell lung
cancer (NSCLC) harboring activating EGFR mutations: Results of the
global, randomized, open-label, Phase IIb trial LUX-Lung 7 (LL7).
Ann Oncol. 26 Suppl 9:ix161–ix162. 2015. View Article : Google Scholar
|
|
8
|
Ramalingam SS, O'Byrne K, Boyer M, Mok T,
Janne PA, Zhang H, Liang J, Taylor I, Sbar EI and Paz-Ares L:
Dacomitinib versus erlotinib in patients with EGFR-mutated advanced
nonsmall-cell lung cancer (NSCLC): Pooled subset analyses from two
randomized trials. Ann Oncol. 27:423–410. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Karachaliou N, Molina-Vila MA and Rosell
R: The impact of rare EGFR mutations on the treatment response of
patients with non-small cell lung cancer. Expert Rev Respir Med.
9:241–244. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Beau-Faller M, Prim N, Ruppert AM,
Nanni-Metéllus I, Lacave R, Lacroix L, Escande F, Lizard S, Pretet
JL, Rouquette I, et al: Rare EGFR exon 18 and exon 20 mutations in
non-small-cell lung cancer on 10 117 patients: A multicentre
observational study by the French ERMETIC-IFCT network. Ann Oncol.
25:126–131. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Krawczyk P, Reszka K, Ramlau R, PowrÓzek
T, Pankowski J, Wojas-Krawczyk K, Kalinka-Warzocha E, Szczęsna A,
Nicoś M, Jarosz B, et al: Prevalence of rare EGFR gene mutations in
nonsmall-cell lung cancer: A multicenter study on 3856 Polish
Caucasian patients. Ann Oncol. 27:358–359. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liang W, Wu X, Fang W, Zhao Y, Yang Y, Hu
Z, Xue C, Zhang J, Zhang J, Ma Y, et al: Network meta-analysis of
erlotinib, gefitinib, afatinib and icotinib in patients with
advanced non-small-cell lung cancer harboring EGFR mutations. PLoS
One. 9:e852452014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lim SH, Lee JY, Sun JM, Ahn JS, Park K and
Ahn MJ: Comparison of clinical outcomes following gefitinib and
erlotinib treatment in non-small-cell lung cancer patients
harboring an epidermal growth factor receptor mutation in either
exon 19 or 21. J Thorac Oncol. 9:506–511. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Urata Y, Katakami N, Morita S, Kaji R,
Yoshioka H, Seto T, Satouchi M, Iwamoto Y, Kanehara M, Fujimoto D,
et al: Randomized phase III study comparing gefitinib with
erlotinib in patients with previously treated advanced lung
adenocarcinoma: WJOG 5108L. J Clin Oncol. 34:3248–3257. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhou C and Yao LD: Strategies to improve
outcomes of patients with EGFR-mutant non-small cell lung cancer:
Review of the literature. J Thorac Oncol. 11:174–186. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang Y, Sheng J, Kang S, Fang W, Yan Y,
Hu Z, Hong S, Wu X, Qin T, Liang W and Zhang L: Patients with exon
19 deletion were associated with longer progression-free survival
compared to those with L858R mutation after first-line EGFR-TKIs
for advanced non-small cell lung cancer: A meta-analysis. PLoS One.
9:e1071612014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang JC, Wu YL, Schuler M, Sebastian M,
Popat S, Yamamoto N, Zhou C, Hu CP, O'Byrne K, Feng J, et al:
Afatinib versus cisplatin-based chemotherapy for EGFR
mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6):
Analysis of overall survival data from two randomised, phase 3
trials. Lancet Oncol. 16:141–151. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu SG, Liu YN, Tsai MF, Chang YL, Yu CJ,
Yang PC, Yang JC, Wen YF and Shih JY: The mechanism of acquired
resistance to irreversible EGFR tyrosine kinase inhibitor-afatinib
in lung adenocarcinoma patients. Oncotarget. 7:12404–12413.
2016.PubMed/NCBI
|
|
19
|
de Pas T, Toffalorio F, Manzotti M,
Fumagalli C, Spitaleri G, Catania C, Delmonte A, Giovannini M,
Spaggiari L, de Braud F and Barberis M: Activity of epidermal
growth factor receptor-tyrosine kinase inhibitors in patients with
non-small cell lung cancer harboring rare epidermal growth factor
receptor mutations. J Thorac Oncol. 6:1895–1901. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lohinai Z, Hoda MA, Fabian K, Ostoros G,
Raso E, Barbai T, Timar J, Kovalszky I, Cserepes M, Rozsas A, et
al: Distinct epidemiology and clinical consequence of classic
versus rare EGFR mutations in lung adenocarcinoma. J Thorac Oncol.
10:738–746. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Klughammer B, Brugger W, Cappuzzo F,
Ciuleanu T, Mok T, Reck M, Tan EH, Delmar P, Klingelschmitt G, Yin
AY, et al: Examining treatment outcomes with erlotinib in patients
with advanced non-small cell lung cancer whose tumors harbor
uncommon EGFR mutations. J Thorac Oncol. 11:545–555. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang S, Cang S and Liu D: Third-generation
inhibitors targeting EGFR T790M mutation in advanced non-small cell
lung cancer. J Hematol Oncol. 9:342016. View Article : Google Scholar : PubMed/NCBI
|