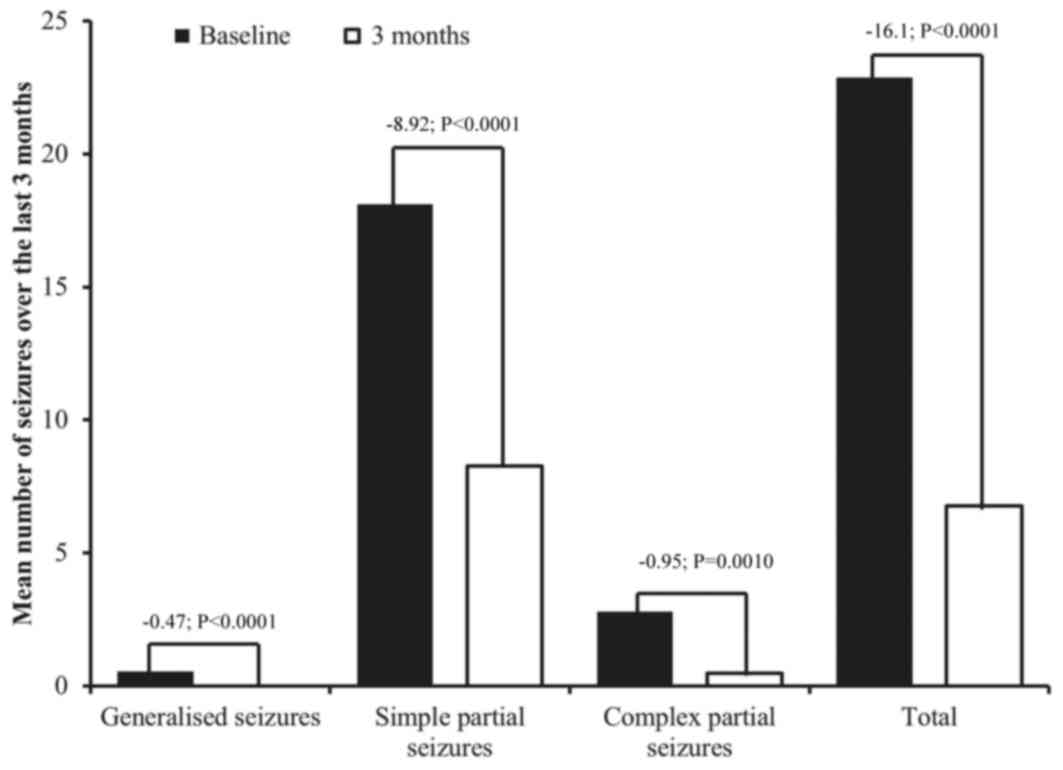
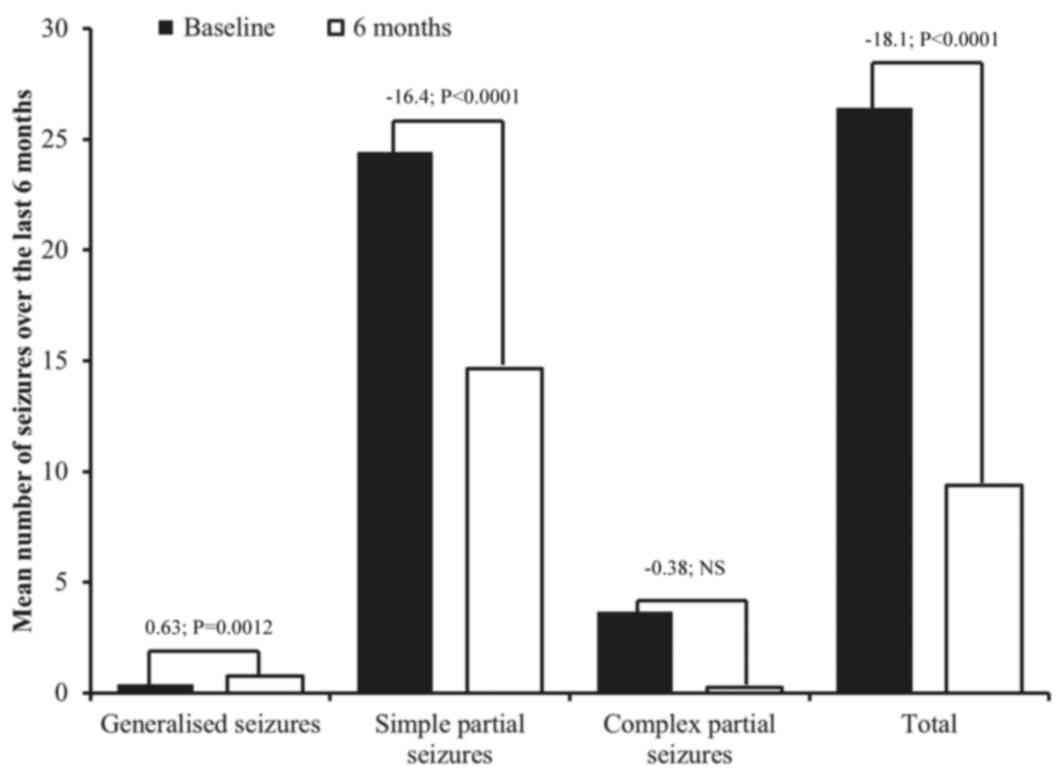
|
1
|
Hildebrand J, Lecaille C, Perennes J and
Delattre JY: Epileptic seizures during follow-up of patients
treated for primary brain tumors. Neurology. 65:212–215. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rossetti AO and Stupp R: Epilepsy in brain
tumor patients. Curr Opin Neurol. 23:603–609. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Singh G, Rees JH and Sander JW: Seizures
and epilepsy in oncological practice: Causes, course, mechanisms
and treatment. J Neurol Neurosurg Psychiatry. 78:342–349. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
van Breemen MS, Wilms EB and Vecht CJ:
Epilepsy in patients with brain tumours: Epidemiology, mechanisms,
and management. Lancet Neurol. 6:421–430. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rudà R and Soffietti R: What is new in the
management of epilepsy in gliomas? Curr Treat Options Neurol.
17:3512015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schiff D, Lee EQ, Nayak L, Norden AD,
Reardon DA and Wen PY: Medical management of brain tumors and the
sequelae of treatment. Neuro Oncol. 17:488–504. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Smith KC: The management of seizures in
brain tumor patients. J Neurosci Nurs. 42:28–39. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Haque N, Thrasher K, Knowles EE Jr, Werk
HC Jr and Sholiton LJ: Studies on dexamethasone metabolism in man:
Effect of diphenylhydantoin. J Clin Endocrinol Metab. 34:44–50.
1972. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gambertoglio JG, Holford NH, Kapusnik JE,
Nishikawa R, Saltiel M, Stanik-Lizak P, Birnbaum JL, Hau T and
Amend WJ Jr: Disposition of total and unbound prednisolone in renal
transplant patients receiving anticonvulsants. Kidney Int.
25:119–123. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chalk JB, Ridgeway K, Brophy T, Yelland JD
and Eadie MJ: Phenytoin impairs the bioavailability of
dexamethasone in neurological and neurosurgical patients. J Neurol
Neurosurg Psychiatry. 47:1087–1090. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Warren RD and Bender RA: Drug interactions
with antineoplastic agents. Cancer Treat Rep. 61:1231–1241.
1977.PubMed/NCBI
|
|
12
|
Rudà R, Bello L, Duffau H and Soffietti R:
Seizures in low-grade gliomas: Natural history, pathogenesis, and
outcome after treatments. Neuro Oncol. 14 Suppl 4:iv55–iv64. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fincham RW and Schottelius DD: Decreased
phenytoin levels in antineoplastic therapy. Ther Drug Monit.
1:277–283. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Perucca E and Pisani F: Pharmacokinetics
and interactions of the new antiepileptic drugs. Epilepsy Res
Suppl. 3:79–88. 1991.PubMed/NCBI
|
|
15
|
Beydoun A, D'Souza J, Hebert D and Doty P:
Lacosamide: Pharmacology, mechanisms of action and pooled efficacy
and safety data in partial-onset seizures. Expert Rev Neurother.
9:33–42. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ben-Menachem E, Biton V, Jatuzis D,
Abou-Khalil B, Doty P and Rudd GD: Efficacy and safety of oral
lacosamide as adjunctive therapy in adults with partial-onset
seizures. Epilepsia. 48:1308–1317. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chung S, Sperling MR, Biton V, Krauss G,
Hebert D, Rudd GD and Doty P; SP754 Study Group, : Lacosamide as
adjunctive therapy for partial-onset seizures: A randomized
controlled trial. Epilepsia. 51:958–967. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Halász P, Kälviäinen R,
Mazurkiewicz-Beldzińska M, Rosenow F, Doty P, Hebert D and Sullivan
T; SP755 Study Group, : Adjunctive lacosamide for partial-onset
seizures: Efficacy and safety results from a randomized controlled
trial. Epilepsia. 50:443–453. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Saria MG, Corle C, Hu J, Rudnick JD,
Phuphanich S, Mrugala MM, Crew LK, Bota DA, Dan Fu B, Kim RY, et
al: Retrospective analysis of the tolerability and activity of
lacosamide in patients with brain tumors: Clinical article. J
Neurosurg. 118:1183–1187. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the eastern cooperative oncology group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pellerino A, Magistrello M, Franchino F,
Nicolotto E, Soffietti R and Rudà R: Efficacy and tolerability of
lacosamide in patients with glioma: A prospective study. Eur J
Neurol. 22 Suppl 1:732015.
|
|
22
|
European Medicines Agency, . Lacosamide
(Vimpat 50 mg tablets): Summary of product characteristics.
http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000863/WC500050338.pdf[Accessed
2016 May 20].
|
|
23
|
US Food and Drug Administration, . Vimpat
(lacosamide) tablets and injection: US prescribing information.
http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/022253lbl.pdf2016
May 20;
|
|
24
|
Klein M, Engelberts NH, van der Ploeg HM,
Kasteleijn-Nolst Trenité DG, Aaronson NK, Taphoorn MJ, Baaijen H,
Vandertop WP, Muller M, Postma TJ and Heimans JJ: Epilepsy in
low-grade gliomas: The impact on cognitive function and quality of
life. Ann Neurol. 54:514–520. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Maschio M, Dinapoli L, Mingoia M, Sperati
F, Pace A, Pompili A, Carapella CM, Vidiri A and Muti P: Lacosamide
as add-on in brain tumor-related epilepsy: Preliminary report on
efficacy and tolerability. J Neurol. 258:2100–2104. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yap KY, Chui WK and Chan A: Drug
interactions between chemotherapeutic regimens and antiepileptics.
Clin Ther. 30:1385–1407. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Landmark C Johannessen and Patsalos PN:
Drug interactions involving the new second- and third-generation
antiepileptic drugs. Expert Rev Neurother. 10:119–140. 2010.
View Article : Google Scholar : PubMed/NCBI
|