Introduction
High intake of fatty food is associated with an
increased risk of cancer, particularly for colorectal cancer (CRC)
(1–3).
A high-fat diet is associated with aggressive prostate cancer and
n-6 fatty acids, including linoleic acid (LA), enhance breast
cancer invasion and metastasis (4).
LA in combination with azoxymethane results in the upregulation of
insulin-like growth factor-I/II receptors (5), Ras (6),
cyclooxygenase-2 (7) and high
mobility group box-1 (8), which act
to accelerate colon carcinogenesis (6,7,9,10).
Prolonged treatment with LA induces a quiescent state in CRC cells
and the dormancy of subcutaneous tumors in mice, which is thought
to be associated with low proliferation of cancer stem cells (CSCs)
(11,12).
Trans fatty acids (TFAs) are reported to increase
the risk of certain types of cancers, including CRC and prostate
cancer (13,14). TFAs, in particular elaidic acid (EA),
enhance cancer cell growth, invasion, survival and metastasis
through activation of the Wnt signaling pathway and the induction
of epithelial-mesenchymal transition (15–17). TFAs
were previously recognized as safe for human consumption; however,
the Food and Drug Administration has proposed that TFAs must be
removed from prepared foods by June 2018 (18).
LA and EA are abundant dietary fatty acids, and are
part of a normal diet eaten by patients with cancer receiving
chemotherapy. The present study focused on the effect of LA and EA
on the action of 5-fluorouracil (5-FU), a common anticancer agent,
with a particular emphasis on cancer stemness.
Materials and methods
Cell culture
The mouse colon cancer CT26 cell line was provided
by Professor Isaiah J. Fidler (MD Anderson Cancer Center, Houston,
TX, USA). The mouse rectal carcinoma CMT93 cell line and the mouse
lung cancer LL2 cell line were purchased from DS Pharma Biomedical
Co., Ltd. (Osaka, Japan). Cells were cultured in Dulbecco's
modified Eagle's medium (Wako Pure Chemical Industries, Ltd.,
Osaka, Japan) supplemented with 10% fetal bovine serum
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at 37°C in a
humidified condition with 5% CO2 atmosphere.
Cells in 6 dishes (1×105 in 3.5
cm-diameter dishes) were treated with LA (50 µg/ml) or EA (20
µg/ml) and concurrently with 5-FU (1 µg/ml) at 37°C for 24 h. For
sequential treatment, cells were seeded in 6 dishes
(1×105 in 3.5 cm-diameter dishes) and treated with LA
(50 µg/ml) or EA (20 µg/ml) at 37°C for 24 h prior to 5-FU
treatment (1 µg/ml at 37°C for 24 h).
The cell viabilities were assessed using a 3-(4,
5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfo-phenyl)-2H-tetrazolium
(MTS) Assay kit (Promega Corporation, Madison, WI, USA). Following
treatment with LA or EA and/or 5-FU, MTS solution was added to each
well for 2 h at 37°C in 5% CO2. The absorbance at 490 nm
was recorded using a microplate reader.
Animals
Male BALB/c mice (4-weeks-old) were purchased from
Japan SLC, Inc. (Shizuoka, Japan). The animals were maintained in
the pathogen-free animal facility with a 12/12 h light/dark cycle
in a temperature (22°C) and humidity-controlled environment,
according to institutional guidelines approved by the Committee for
Animal Experimentation of Nara Medical University (Kashihara,
Japan; approval no. 9559), in accordance with the current
regulations and standards of the Japanese Ministry of Health, Labor
and Welfare.
Animal models
To establish a subcutaneous tumor model, CT26 cancer
cells (1×107) were inoculated into the scapular
subcutaneous tissue of BALB/c mice. Mice were euthanized and
observed using immunohistochemistry 4 weeks following inoculation.
5-FU (10 mg/kg; Wako Pure Chemical Industries, Ltd.) was injected
intraperitoneally twice a week for 2 weeks following inoculation.
LA (Sigma-Aldrich; Merck KGaA) or EA (Wako Pure Chemical
Industries, Ltd.) were administrated at 1.0 and 0.1% w/w,
respectively, through the daily supplementation of a standard CE-2
diet (CLEA Japan, Inc., Tokyo, Japan). The mean intake of LA and EA
was 4.2±0.78 and 0.31±0.07 mg/day, respectively. A total of five
mice were used for per treatment group. The mean weight of the mice
was 21.5±1.4 g at the start of treatment.
Semi-quantitative reverse
transcription-quantification polymerase chain reaction
(RT-qPCR)
Total RNA (1 µg) was extracted using an RNeasy
extraction kit (Qiagen, Inc., Valencia, CA, USA). RNA was extracted
from 1×107 CT26 cells and eluted in 50 µl RNase-free
water. cDNA was synthesized using ReverTra Ace RT-qPCR kit (Toyobo,
Tokyo, Japan). Quantification of PCR products was performed with
QuantiTect Primer Assays using QuantiFast, QuantiTect, Rotor-Gene,
and FastLane kit for SYBR-Green-based detection (Qiagen, Inc.),
according to the manufacturer's protocol. mRNA quantification was
performed according to the 2−ΔΔcq method (19). The number of replicates was 30 cycles.
The primer sets used for amplification were as follows: Mouse CD133
(prominin 1; accession no., BC028286.1) forward,
5′-GAAAAGTTGCTCTGCGAACC-3′ and reverse, 5′-TCTCAAGCTGAAAAGCAGCA-3′;
mouse nucleostemin (NS; accession no. AY181025.1) forward,
5′-CAGGATGCTGACGATCAAGA-3′ and reverse, 5′-TTGATTGCTCAGGTGACAGC-3′;
and mouse β-actin (accession no. NM_007393.4) forward,
5′-AGCCATGTACGTAGCCATCC-3′ and reverse 5′-CTCTCAGCTGTGGTGGTGAA-3′.
The primers were synthesized by Sigma Genosys (Sigma-Aldrich; Merck
KGaA). The PCR conditions were set according to the manufacturer's
protocol. The number of replicates was 30 cycles. PCR products were
electrophoresed on a 2% agarose gel and visualized using ethidium
bromide.
Immunohistochemistry
Consecutive 4 µm tissue sections were
immunohistochemically stained using the immunoperoxidase technique,
as described previously (8). Mouse
CD133 antibody (AG13328; ProteinTech Group, Inc., Chicago, IL, USA)
and mouse nucleostemin antibody (sc-166430; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA) were used at a concentration
of 0.5 µg/ml at 37°C for 2 h. Color development was performed using
3–3′-diaminobenzidine (Dako; Agilent Technologies, Inc.), and the
specimens were counterstained with Mayer's hematoxylin
(Sigma-Aldrich; Merck KGaA) to visualize the nuclei. Following
immunostaining, all slides were assessed to measure the number of
positively stained nuclei using an all-in-one microscope (BZ-X700;
Keyence, Osaka, Japan).
Aldehyde dehydrogenase (ALDH)
activity
ALDH activity was measured using the ALDEFLUOR kit
(Veritas Technologies LLC, Tokyo, Japan) according to the
manufacturer's protocol. The activity was normalized to the
negative control, diethylaminobenzaldehyde.
Statistical analysis
Statistical significance was calculated using the
two-tailed Fisher's exact test, χ2 test and unpaired
Student's t-test, with the assumption of Gaussian distributions, by
Kolmogorov and Smirnov tests Statistical analysis was performed
using InStat software (GraphPad Software, Inc., La Jolla, CA, USA).
The data are presented as the mean ± standard deviation. P<0.05
(two-sided) was considered to indicate a statistically significant
difference.
Results
Effect of LA and EA on the
5-FU-induced reduction of cancer cell viability
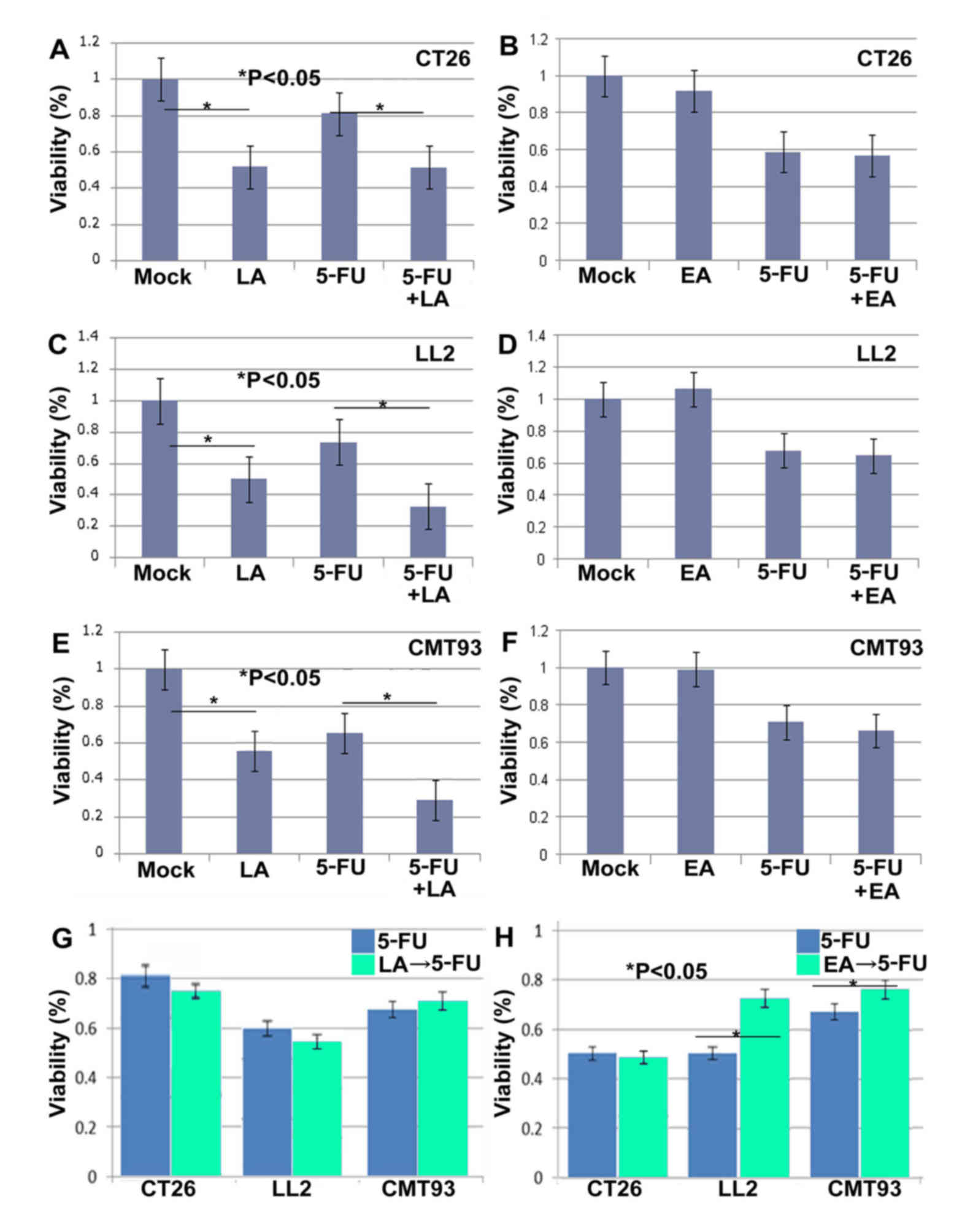
Three cancer cell lines, CT26 (colon cancer), LL2
(lung cancer) and CMT93 (rectal cancer), were concurrently treated
with either LA or EA and 5-FU, or with LA, EA or 5-FU alone.
Treatment with LA alone decreased cell viability in all 3 cell
lines (Fig. 1). LA also augmented the
tumor cell inhibition observed following 5-FU treatment in all 3
cell lines. In contrast, EA alone did not significantly suppress
tumor viability in any of the 3 cell lines (Fig. 1). In addition, concurrent treatment of
EA with 5-FU did not recover or enhance 5-FU-induced growth
inhibition in the three cell lines.
Sequential treatment with LA or EA followed by 5-FU
was then examined (Fig. 1).
Concurrent treatment of 5-FU and LA revealed a more pronounced
decrease of cell viability in the 3 cell lines. In contrast,
temporal treatment of LA to 5-FU demonstrated no additional
decrease of viability compared with that of 5-FU treatment alone.
(Fig. 1). In addition, EA
pretreatment reversed the effect of 5-FU, increasing the viability
of LL2 and CMT93 cells (Fig. 1).
Effect of LA or EA with 5-FU on the
stemness of cancer cells
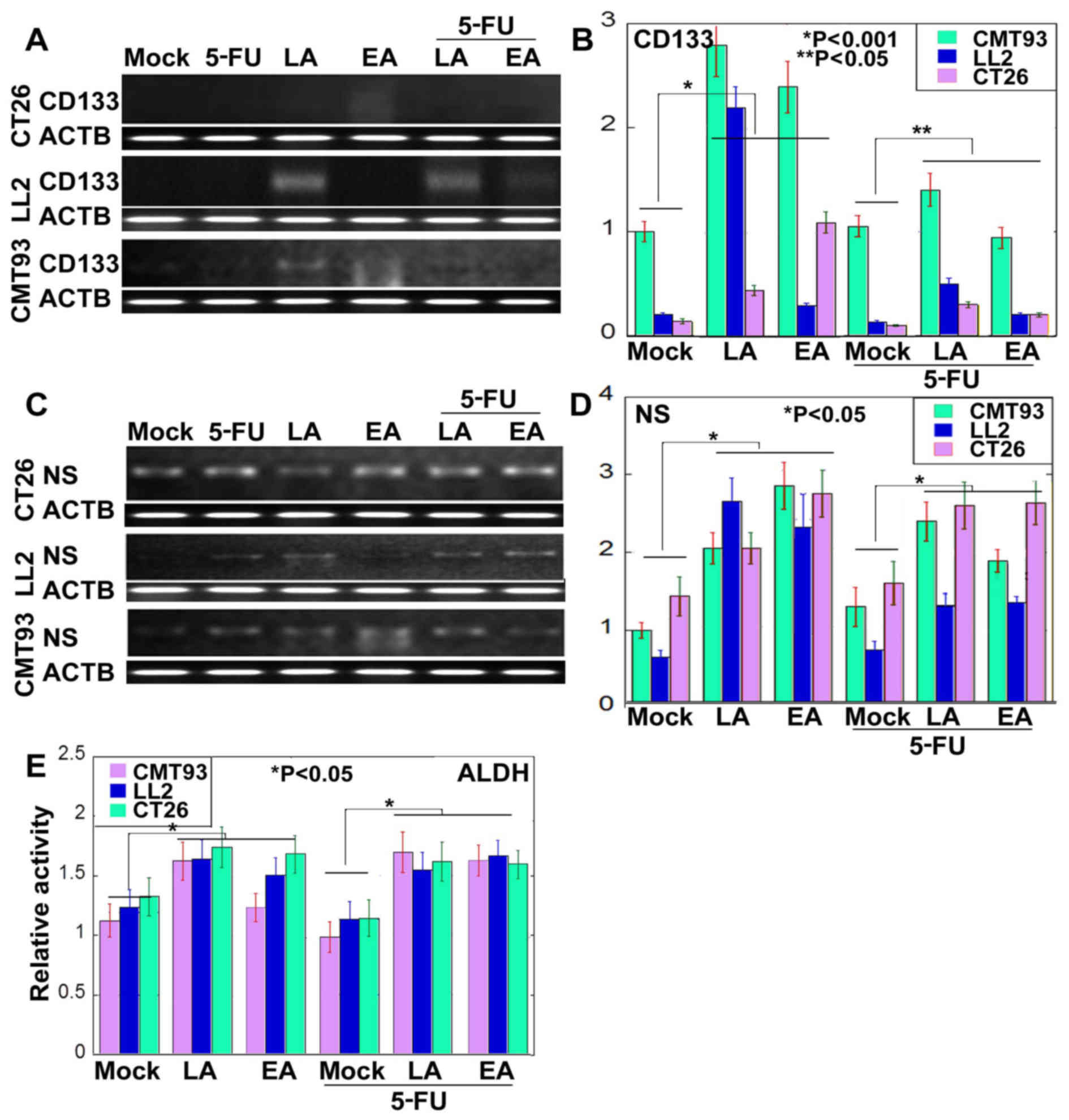
Previous studies have demonstrated that the
resistance of cancer cells to anticancer drugs is associated with
CSCs (20,21). Therefore, alterations of stemness in
LA- or EA-treated cancer cells were examined (Fig. 2). Expression of CD133 and NS was
examined in cells treated with 5-FU alone, or concurrently with LA
or EA (Fig. 2A-D). The expression of
CD133 and NS was increased in cells treated with LA or EA when
compared with untreated cells. Although expression of NS appears to
be higher in cells treated with 5-FU and LA concurrently compared
with cells treated with LA alone in CT26 and CMT93 cells, the
expression of CD133 and NS was lower in cells treated concurrently
with 5-FU and either LA or EA compared with cells treated with LA
or EA alone. However expression was increased in cells treated
concurrently with 5-FU and either LA or EA compared with cells
treated with 5-FU alone. Activity of ALDH, a stem cell-associated
enzyme, was increased in cells treated with LA or EA compared with
in untreated cells (Fig. 2E). ALDH
activity was higher in cells treated concurrently with 5-FU and
either LA or EA compared with cells treated with 5-FU alone
(Fig. 2E).
Effect of LA or EA with 5-FU on tumor
growth
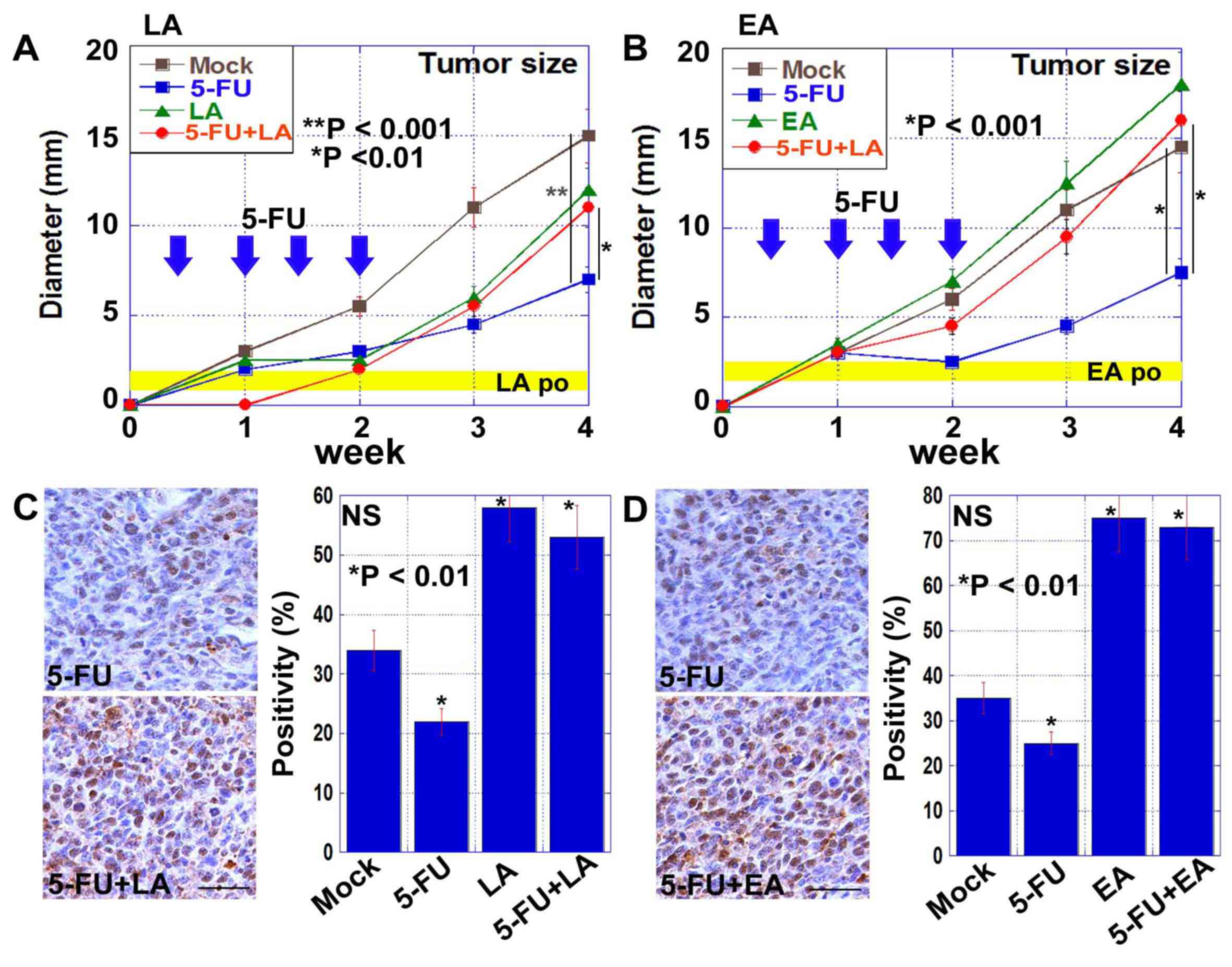
Finally, the effect of oral intake of LA or EA on
the antitumor effects of 5-FU was examined in BALB/c mice
inoculated with CT26 cells (Fig. 3).
Treatment with 5-FU inhibited the growth of CT26-derived tumors,
and this was abrogated by concurrent treatment with either LA or EA
(Fig. 3A and B). The expression of NS
in CT26-derived tumors was examined by immunohistochemistry
(Fig. 3C and D). NS-positive CSCs
were more abundant in tumors treated with 5-FU and either LA or EA
compared with tumors treated with 5-FU alone.
Discussion
Fatty acids are dietary components, which are known
to be involved in colon carcinogenesis (6,7). LA and EA
are known to be risk factors for CRCs (14,22–24),
enhancing inflammation and suppressing mucosal immunity (25–28).
LA-derived prostaglandin E2, produced by cyclooxygenase-2, induces
chronic persistent inflammation, which produces reactive oxygen
species (25,26). Likewise, EA induces secretion of
TNF-α, activation of nuclear factor-κB, and inhibition of cluster
of differentiation 8+ T-lymphocytes (27,28).
LA and EA affect CSCs. LA induces senescence in
cultured CRC cells and dormancy in CRC cells inoculated into mice
(11). Conversely, EA increases the
proliferation of cells in spheres via the activation of epidermal
growth factor receptor and the Wnt signaling pathway, and the
upregulation of stem cell markers (16,17). EA
also induces epithelial-mesenchymal transition and enhances tumor
growth and metastasis (16,17).
Previous studies have revealed that cancer stemness
is responsible for resistance to anticancer chemotherapy (20,21). CSCs
possess refractoriness to chemotherapy through low proliferative
activity, advanced DNA repair and the expulsion of chemotherapeutic
agents from the cytosol (29).
Therefore, factors affecting the stemness of cancer cells may also
affect the efficacy of chemotherapy.
Sequential treatment with LA or EA followed by 5-FU
abrogated growth inhibition by 5-FU. Pretreatment with LA or EA may
select and condense CSCs or increase the stemness of these cells.
ALDH activity, a marker for stem cell activity (30), was enhanced by LA and EA. Upregulation
of CD133 and NS by LA or EA varied by cell line. CD133 expression
is associated with stem cells (31,32),
whereas NS expression is associated with stem cells and progenitor
cells (33). Therefore, LA and EA may
affect populations of stem cells and progenitors in a cell or
tissue-dependent manner (34).
The anti-chemotherapeutic effect of ingested LA or
EA was confirmed using a mouse subcutaneous tumor model. Oral
intake of LA or EA abrogated the antitumor effects of 5-FU. This
result may be an example of the anti-chemotherapeutic properties of
dietary components. The anti-chemotherapeutic effects of LA or EA
were observed not only in CT26 and CMT93 CRC cells, but also in LL2
lung cancer cells. LA and EA are not thought to be risk factors for
lung cancer; however, the present data indicated that the
anti-chemotherapeutic effects of LA or EA may affect the efficacy
of treatment of various malignancies. The effect of dietary
components on chemotherapeutic effects, or on cancer stemness,
should be the focus of wide ranging studies concerning human
cancers.
Acknowledgements
The authors would like to thank Ms. Tomomi Masutani
(Nara Medical University) for expert assistance with the
preparation of this manuscript. The present study was supported by
the Ministry of Education, Culture, Sports, Science and Technology
KAKENHI (grant nos. 13200228, 14478268, 13394212, 13209774 and
16675788).
References
|
1
|
Pietrzyk L, Torres A, Maciejewski R and
Torres K: Obesity and obese-related chronic low-grade inflammation
in promotion of colorectal cancer development. Asian Pac J Cancer
Prev. 16:4161–4168. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Uzunlulu M, Caklili O Telci and Oguz A:
Association between metabolic syndrome and cancer. Ann Nutr Metab.
68:173–179. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cappellani A, Zanghi A, Di Vita M,
Cavallaro A, Piccolo G, Veroux P, Lo Menzo E, Cavallaro V, De Paoli
P, Veroux M and Berretta M: Strong correlation between diet and
development of colorectal cancer. Front Biosci (Landmark Ed).
18:190–198. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rose DP: Dietary fatty acids and cancer.
Am J Clin Nutr. 66 4 Suppl:998S–1003S. 1997.PubMed/NCBI
|
|
5
|
Zhang W, Thornton WH and MacDonald RS:
Insulin-like growth factor-I and II receptor expression in rat
colon mucosa are affected by dietary lipid intake. J Nutr.
128:158–165. 1998.PubMed/NCBI
|
|
6
|
Singh J, Hamid R and Reddy BS: Dietary
fish oil inhibits the expression of farnesyl protein transferase
and colon tumor development in rodents. Carcinogenesis. 19:985–989.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Singh J, Hamid R and Reddy BS: Dietary fat
and colon cancer: Modulation of cyclooxygenase-2 by types and
amount of dietary fat during the postinitiation stage of colon
carcinogenesis. Cancer Res. 57:3465–3470. 1997.PubMed/NCBI
|
|
8
|
Ohmori H, Luo Y, Fujii K, Sasahira T,
Shimomoto T, Denda A and Kuniyasu H: Dietary linoleic acid and
glucose enhances azoxymethane-induced colon cancer and metastases
via the expression of high-mobility group box 1. Pathobiology.
77:210–217. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhou S, Wang G, Chen B and Wang P: Effect
of dietary fatty acids on tumorigenesis of colon cancer induced by
methylnitrosourea in rats. J Environ Pathol Toxicol Oncol.
19:81–86. 2000.PubMed/NCBI
|
|
10
|
Rao CV, Hirose Y, Indranie C and Reddy BS:
Modulation of experimental colon tumorigenesis by types and amounts
of dietary fatty acids. Cancer Res. 61:1927–1933. 2001.PubMed/NCBI
|
|
11
|
Ohmori H, Sasahira T, Fujii K, Yi L,
Shimomoto T and Kuniyasu H: Linoleic acid-induced growth
suppression induces quiescent cancer cell nests in nude mice.
Pathobiology. 75:226–232. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Luo Y, Chihara Y, Fujimoto K, Sasahira T,
Kuwada M, Fujiwara R, Fujii K, Ohmori H and Kuniyasu H: High
mobility group box 1 released from necrotic cells enhances regrowth
and metastasis of cancer cells that have survived chemotherapy. Eur
J Cancer. 49:741–751. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
King IB, Kristal AR, Schaffer S,
Thornquist M and Goodman GE: Serum trans-fatty acids are associated
with risk of prostate cancer in beta-Carotene and Retinol Efficacy
Trial. Cancer Epidemiol Biomarkers Prev. 14:988–992. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Vinikoor LC, Schroeder JC, Millikan RC,
Satia JA, Martin CF, Ibrahim J, Galanko JA and Sandler RS:
Consumption of trans-fatty acid and its association with colorectal
adenomas. Am J Epidemiol. 168:289–297. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kuniyasu H, Ohmori H, Fujii K, et al:
Significance of trans fatty acids in colorectal cancerCurrent
Research in Cancer. 9. Sunitha M: Research Media; Kerala, India:
pp. 21–29. 2015
|
|
16
|
Ohmori H, Fujii K, Kadochi Y, Mori S,
Nishiguchi Y, Fujiwara R, Kishi S, Sasaki T and Kuniyasu H: Elaidic
acid, a trans fatty acid, enhances the metastasis of colorectal
cancer cells. Pathobiology. 84:144–151. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fujii K, Luo Y, Fujiwara-Tani R, Kishi S,
He S, Yang S, Sasaki T, Ohmori H and Kuniyasu H: Pro-metastatic
intracellular signaling of elaidic trans fatty acid. Int J Oncol.
50:85–92. 2017.PubMed/NCBI
|
|
18
|
U.S. Food and Drug Administration, . Final
Determination Regarding Partially Hydrogenated Oils. U.S. Food and
Drug Administration; Silver Spring, MD: 2015
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Leirós GJ and Balañá ME: Metastatic cancer
stem cells: New molecular targets for cancer therapy. Curr Pharm
Biotechnol. 12:1909–1922. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gaur P, Sceusi EL, Samuel S, Xia L, Fan F,
Zhou Y, Lu J, Tozzi F, Lopez-Berestein G, Vivas-Mejia P, et al:
Identification of cancer stem cells in human gastrointestinal
carcinoid and neuroendocrine tumors. Gastroenterology.
141:1728–1737. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bultman SJ: Interplay between diet, gut
microbiota, epigenetic events and colorectal cancer. Mol Nutr Food
Res. 61:2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rao CV and Reddy BS: Modulating effect of
amount and types of dietary fat on ornithine decarboxylase,
tyrosine protein kinase and prostaglandins production during colon
carcinogenesis in male F344 rats. Carcinogenesis. 14:1327–1333.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Slattery ML, Benson J, Ma KN, Schaffer D
and Potter JD: Trans-fatty acids and colon cancer. Nutr Cancer.
39:170–175. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gupta RA and Dubois RN: Colorectal cancer
prevention and treatment by inhibition of cyclooxygenase-2. Nat Rev
Cancer. 1:11–21. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Moonen HJ, Dommels YE, van Zwam M, van
Herwijnen MH, Kleinjans JC, Alink GM and de Kok TM: Effects of
polyunsaturated fatty acids on prostaglandin synthesis and
cyclooxygenase-mediated DNA adduct formation by heterocyclic
aromatic amines in human adenocarcinoma colon cells. Mol Carcinog.
40:180–188. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Rao YP, Kumar PP and Lokesh BR: Molecular
mechanisms for the modulation of selected inflammatory markers by
dietary rice bran oil in rats fed partially hydrogenated vegetable
fat. Lipids. 51:451–467. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dlouhý P, Kucera P, Kraml P, Pompachová A,
Potocková J, Smejkalová V, Mokrejs P, Jacek M and Andel M:
Short-term dietary intake of C18:1 trans fatty acids decreases the
function of cellular immunity in healthy young men. Ann Nutr Metab.
53:129–136. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
McCubrey JA, Abrams SL, Fitzgerald TL,
Cocco L, Martelli AM, Montalto G, Cervello M, Scalisi A, Candido S,
Libra M and Steelman LS: Roles of signaling pathways in drug
resistance, cancer initiating cells and cancer progression and
metastasis. Adv Biol Regul. 57:75–101. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Meng E, Mitra A, Tripathi K, Finan MA,
Scalici J, McClellan S, da Silva L Madeira, Reed E, Shevde LA,
Palle K and Rocconi RP: ALDH1A1 maintains ovarian cancer stem
cell-like properties by altered regulation of cell cycle checkpoint
and DNA repair network signaling. PLoS One. 9:e1071422014.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ren F, Sheng WQ and Du X: CD133: A cancer
stem cells marker, is used in colorectal cancers. World J
Gastroenterol. 19:2603–2611. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sun Y, Kong W, Falk A, Hu J, Zhou L,
Pollard S and Smith A: CD133 (Prominin) negative human neural stem
cells are clonogenic and tripotent. PLoS One. 4:e54982009.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu SJ, Cai ZW, Liu YJ, Dong MY, Sun LQ,
Hu GF, Wei YY and Lao WD: Role of nucleostemin in growth regulation
of gastric cancer, liver cancer and other malignancies. World J
Gastroenterol. 10:1246–1249. 2004.PubMed/NCBI
|
|
34
|
Haraguchi N, Inoue H, Tanaka F, Mimori K,
Utsunomiya T, Sasaki A and Mori M: Cancer stem cells in human
gastrointestinal cancers. Hum Cell. 19:24–29. 2006. View Article : Google Scholar : PubMed/NCBI
|