Introduction
The reduced expression in immortalized cells (REIC)
gene was isolated using the subtractive hybridization method, as
its expression is reduced in a number of human immortalized and
neoplastic tumor cells (1).
Investigation into the sequence homology of REIC using a
computerized scientific database revealed that REIC and Dickkopf-3
(Dkk-3) are identical. Dkk-3 belongs to the Dkks gene family
(1,2).
Therefore, REIC was designated as REIC/Dkk-3. Previous studies by
our group have demonstrated that overexpression of REIC/Dkk-3 by an
adenovirus (Ad)-REIC vector resulted in a therapeutic effect
against numerous human cancer subtypes via a mechanism triggered by
endoplasmic reticulum (ER) stress-mediated c-Jun N-terminal kinase
(JNK) activation (3–6).
To obtain improved therapeutic effects of Ad-REIC
against cancer, our group previously developed a powerful gene
expression system; the C-TSC cassette [cytomegalovirus (CMV)-RU5′
located upstream (C); another promoter unit composed of triple
tandem promoters, human telomerase reverse transcriptase (hTERT),
simian virus 40 (SV40) and CMV, located downstream of the cDNA
(TSC); plus a polyadenylation (polyA) signal], which exhibited a
higher efficacy of REIC/Dkk-3 expression compared with that of
conventional vector systems (7,8).
Adaptation of the C-TSC cassette and the cargo REIC/Dkk-3 cDNA to
an adenovirus vector resulted in Ad-C-TSC-REIC, which induced
apoptotic cell death to a greater extent in more types of cancer
cells than the usual CMV promoter-driven Ad-REIC (7,8). However,
due to the higher efficiency of virus-mediated REIC/Dkk-3
expression levels by Ad-C-TSC-REIC, we have identified that the
viral production rate is markedly mitigated in HEK293 cells, which
are commonly used in adenovirus expansion as a production
donor.
Therefore, the present study aimed to improve the
Ad-C-TSC-REIC vector by making it more permissive in donor HEK293
cells, without losing its high expression level in cancer cells.
Thus, the C-T cassette was developed by replacing the 3′-TSC tandem
promoter unit in the C-TSC cassette with a single hTERT promoter
(T). This novel cassette provided comparable expression levels in
cancer cells and demonstrated significantly mitigated cell death of
donor cells used for adenoviral production, resulting in an
increased adenoviral titer. The data obtained in the present study
indicated that the improved adenovirus vector carrying REIC/Dkk-3,
termed Ad-C-T-REIC, may have the potential to become a more useful
tool for its application in cancer gene therapy.
Materials and methods
Cells
The embryonic kidney cell line HEK293 [American Type
Culture Collection (ATCC); Manassas, VA, USA], embryonic kidney
cell line HEK293T (ATCC), prostate adenocarcinoma cell line PC-3
(ATCC) and glioblastoma cell line T98 (ATCC) were cultivated in
DMEM/F12 medium (Invitrogen; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS;
Thermo Fisher Scientific, Inc.) at 37°C with 5% CO2.
Human umbilical vein endothelial cells (HUVEC; Kurabo Industries
Ltd., Osaka, Japan) were grown in Humedia-EG medium (Kurabo
Industries Ltd.) supplemented with 2% FBS, fibroblast growth
factor-B (5 ng/ml), epidermal growth factor (10 ng/ml),
hydrocortisone (1 µg/ml) and heparin (10 µg/ml) at 37°C with 5%
CO2.
Preparation of plasmid and adenovirus
vectors
The compositions of a series of novel, constructed
expression plasmids are presented in Fig.
1A. pIDT-SMART (C-TSC) was previously reported (7). A 189-base pair element of the hTERT
promoter (T) [accession no. DQ264729 (1618–1806) from GenBank] was
used for modification of pIDT-SMART (C-TSC). Full-length cDNA of
human REIC/Dkk-3 was amplified by reverse transcription-polymerase
chain reaction and inserted into the indicated vectors.
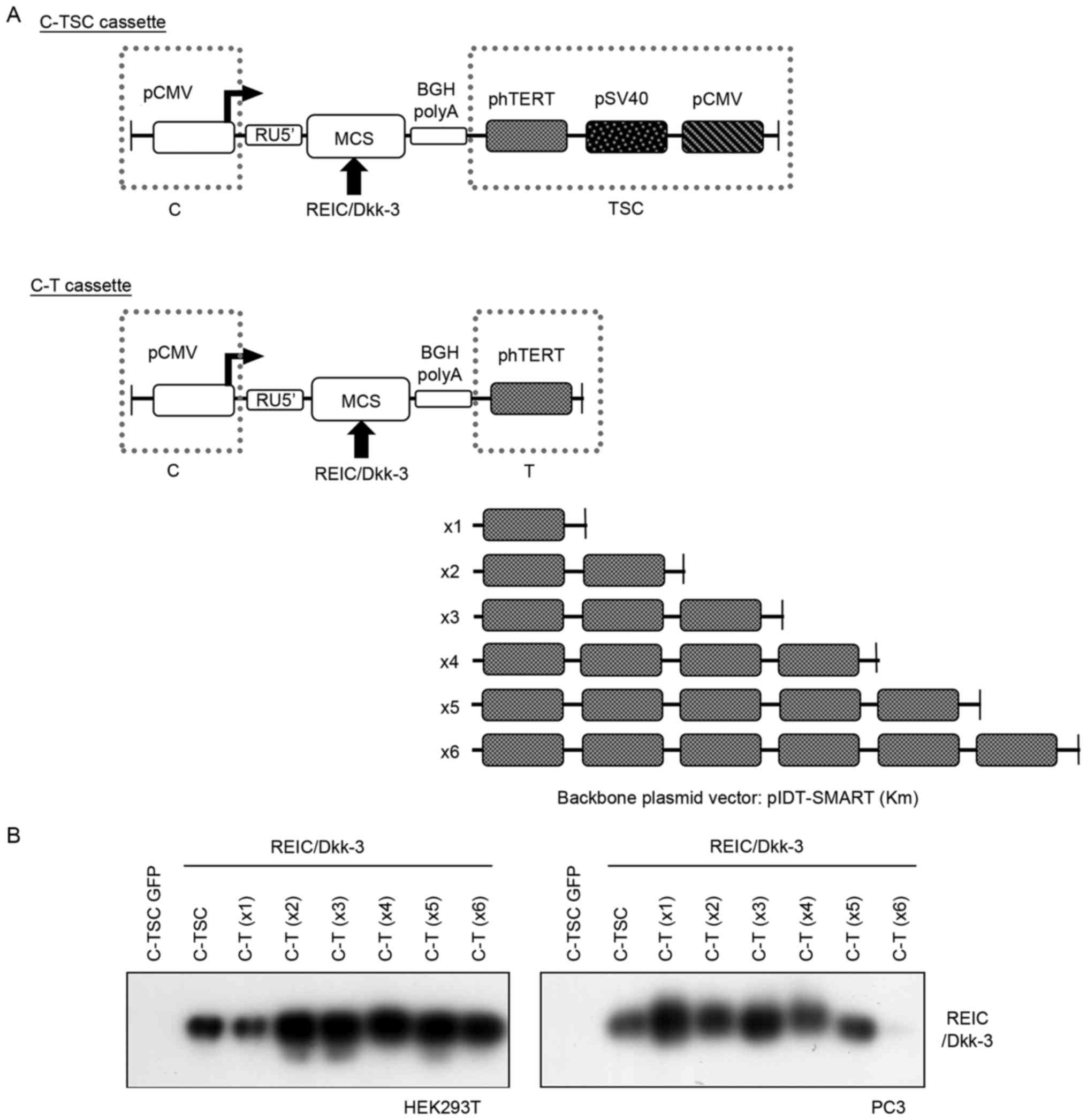 | Figure 1.Schematic diagram of the improved gene
expression system and assessment of its gene expression. (A) A
series of indicated plasmids were constructed on the basis of the
promoter-less pIDT-SMART (Km) vector. (B) Expression of REIC/Dkk-3
protein was assessed by western blotting following transfection
with the indicated plasmids in HEK293T and PC3 cells. C-TSC,
cassette containing cytomegalovirus-RU5′ upstream, human telomerase
reverse transcriptase, simian virus 40 and cytomegalovirus
downstream, with a polyadenylation signal; CMV, cytomegalovirus;
MCS, multi-cloning site; BGH, bovine growth hormone; polyA,
polyadenylation; p, phosphorylated; hTERT, human telomerase reverse
transcriptase; REIC, reduced expression in immortalized cells;
Dkk-3, Dickkopf-3; GFP, green fluorescent protein C-T, cassette
containing cytomegalovirus-RU5′ upstream, human telomerase reverse
transcriptase downstream and a polyadenylation signal. |
The improved C-T unit with REIC/Dkk-3 cDNA was
cloned into a pShuttle plasmid vector (Takara Biotechnology Co.,
Ltd., Dalian, China). The resulting donor plasmid was used for
constructing an adenovirus vector according to the manufacturer's
protocol.
Transfection
Cells were transiently transfected with plasmid
vectors using FuGENE-HD (Promega Corporation, Madison, WI, USA)
according to the manufacturer's protocol. A negative control used
FuGENE-HD alone. Cells were infected with adenovirus vectors at the
indicated multiplicity of infection (MOI), as previously described
(3–6).
Ad-LacZ (Takara Biotechnology Co., Ltd.) was used as a control
virus for assessments of apoptosis and to determine infection
efficiency.
Western bloting
Cells (1×105 cells/well in 6-well plates)
were transiently transfected with plasmid vectors using FuGENE-HD.
After 24 h, the transfected cells were collected, treated with
M-PER lysis reagent (Invitrogen; Thermo Fisher Scientific, Inc.)
and kept on ice for 10 min. Cell debris was then removed by
centrifugation at 20,000 × g at 4°C for 20 min. The protein
concentration of the supernatant was determined by a Bio-Rad
Protein Quantification assay (Bio-rad Laboratories, Inc., Hercules,
CA, USA). Samples of the extracted protein (10 µg) were separated
with 10% SDS-PAGE. The proteins on the gel were transferred onto a
polyvinylidene membrane, which was blocked with 10% skimmed milk in
phosphate-buffered saline with Tween-20 (PBS-T) for 30 min at room
temperature, then incubated with primary antibodies overnight at
4°C.
The following primary antibodies were used, at a
dilution of 1:1,000: Rabbit anti-human REIC/Dkk-3 antibody, which
was generated in the Department of Cell Biology, Okayama University
Graduate School of Medicine, Dentistry and Pharmaceutical Sciences
(Okayama, Japan) (3–6); rabbit anti-human GRP78 [binding
immunoglobulin protein (BiP)] antibody (cat. no. ab21685; Abcam,
Cambridge, MA, USA); rabbit anti-human stress-activated protein
kinases (SAPK)/JNK (cat. no. 9252); mouse anti-human phosphorylated
(phosphor)-SAPK/JNK (Thr183/Tyr185) (cat. no. 9255); rabbit
anti-human p38 (cat. no. 9212); rabbit anti-human phospho-p38
(Thr180/Tyr182) antibodies (cat. no. 4511; all from Cell Signaling
Technology, Inc.) and mouse anti-human tubulin antibody (cat. no.
T5168; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany).
The membrane was then washed in PBS-T three times
for 10 min and incubated with a secondary antibody diluted to
1:1,000 (anti-rabbit IgG, HRP-linked antibody: cat. no. 7074 or
anti-mouse IgG, HRP-linked antibody: cat. no. 7076; Cell Signaling
Technology, Inc.) for 1 h at room temperature. Protein detection
was performed with Pierce Western Blotting substrate Plus (Thermo
Fisher Scientific, Inc.). All experiments were repeated in
triplicate.
Apoptosis assay
Apoptotic cells were analyzed as per a previously
described method (9). Cells
(1×105 cells/well) were seeded in 6-well plates and
incubated for 24 h at 37°C. At 48 h after transfection, Hoechst
33342 (2 µg/ml) was added into the culture medium and the cells
were incubated for a further 30 min. Apoptotic cells were detected
using fluorescence microscopy (magnification, ×400). Cell counts
were manually performed from 5 fields of view for each well; the
experiment was performed in triplicate.
X-gal staining
Cells (1×105 cells/well in 12-well
plates) were seeded and incubated at 37°C for 24 h. At 48 h after
infection at the indicated MOI, X-gal staining was performed with a
β-gal Staining kit (cat. no. K1465-01; Thermo Fisher Scientific,
Inc.), according to the manufacturer's protocol. Using optical
microscopy (BZ-X710; Keyence Co., Osaka, Japan; magnification,
×100), the number of infected cells were manually counted from 3
fields of view for each well. The experiment was repeated in
triplicate.
Titration of adenovirus
production
Adenovirus production was assessed in HEK293 cells.
HEK293 donor cells (2×105 cells in 10-cm dishes) were
seeded and incubated for 24 h at 37°C. Ad-C-TSC-REIC and
Ad-C-T-REIC were infected at 2 MOI into HEK293 cells at 37°C for 48
h. Subsequent to infection, cells were collected and ruptured with
3 freeze and thaw cycles. Purification of the adenovirus from the
collected cell extracts was performed using a commercially
available small column, ViraBind Adenovirus Miniprep kit (Cell
Biolabs, Inc., San Diego, CA, USA), according to the manufacturer's
protocol. The titer of the purified virus was determined using an
Adeno-X Rapid Titer kit (Takara Biotechnology Co., Ltd.) according
to the manufacturer's protocol. Using optical microscopy as
previously described, infected cells were identified. Infected cell
counts were performed for 20 visual fields per well at ×100
magnification, to evaluate the number of plaque-forming units.
Statistical analysis
Results are expressed as the mean ± standard
deviation, unless otherwise indicated. Unpaired Student's t-tests
were performed for statistical analyses using Microsoft Excel 2016
(Microsoft Corporation, Redmond, WA, USA). P<0.05 was considered
to indicate a statistically significant difference.
Results
Assessment of the plasmids for gene
expression levels and induction of apoptotic cell death
In order to improve the Ad-C-TSC-REIC vector and
make it more permissive in donor HEK293 cells, without losing its
high expression level in cancer cells, the present study modified
the 3′-TSC (phTERT-pSV40-pCMV) region in the C-TSC cassette, as the
3′-region is known to affect gene expression level in vectors
(7). To further simplify and weaken
the 3′-TSC, the cancer-specific hTERT promoter (phTERT:T) was
selected among the components of the 3′-TSC construct, since either
pSV40 or pCMV was expected to demonstrate higher promoter activity
compared with that of phTERT, based on previous studies (7,8). The
present study first aimed to make a series of improved constructs
using phTERT as the basis of the C-TSC vector structure. To
determine the optimal number of phTERT at the 3′-side of the cDNA,
six constructs were prepared, termed C-T (×1 to ×6), where phTERT
was serially repeated up to six (Fig.
1A). Following insertion of REIC/Dkk-3 into the multi-cloning
site of the indicated plasmid constructs, temporal transfection
experiments were performed using the newly constructed C-T vectors
and the C-TSC vector, and the performance of each of the vectors
for expression of the REIC/Dkk-3 gene was assessed. Western
blotting revealed that the expression level of C-T (×1) in HEK293T
cells was comparable to or slightly lower compared with that of
C-TSC, whereas the remaining C-T constructs (×2-x6) demonstrated
further increases in expression to almost the same levels.
Conversely, in prostate cancer PC3 cells, C-T (×1) exhibited the
highest expression level of REIC/Dkk-3 in the tested constructs
(Fig. 1B). Of note, C-T (×6) did not
induce any appreciable level of REIC/Dkk-3 expression in PC3
cells.
Under the same experimental conditions, the present
study then assessed whether apoptotic cell death was initiated
using the newly constructed plasmids, since overexpression of
REIC/Dkk-3 is able to induce apoptosis in a various types of cancer
cell (3–6). Hoechst staining is a conventional and
reliable method for detecting apoptotic cell death in which cells
express bright fluorescence, particularly in the apoptosis-mediated
condensed nuclei, and therefore this method was used to assess
effects of the indicated constructs (Fig.
2A). Combined with the results of quantification (Fig. 2B), it was revealed that despite the
higher expression levels of REIC/Dkk-3 derived from C-T (×1–6)
plasmids, apoptosis was hardly detected in HEK293 cells, whereas a
high rate of apoptosis was observed in PC3 cells transfected with
C-T (×1–4), but not in those transfected with C-T (×5 and ×6). The
greatest effect was induced by the C-T (×1) construct. Repeated
experiments using the C-T constructs together with the C-TSC
construct also demonstrated that, although REIC/Dkk-3 produced by
the C-TSC construct induced a high rate of apoptotic cell death in
HEK293T and PC3 cells, the C-T (×1, ×3 and ×4) constructs
selectively induced high rates of apoptotic cell death in PC3 cells
but not in HEK293T cells (Fig. 3). As
a result, the C-T (×1) construct was selected for further analysis,
and termed ‘C-T’ due to its simpler vector structure.
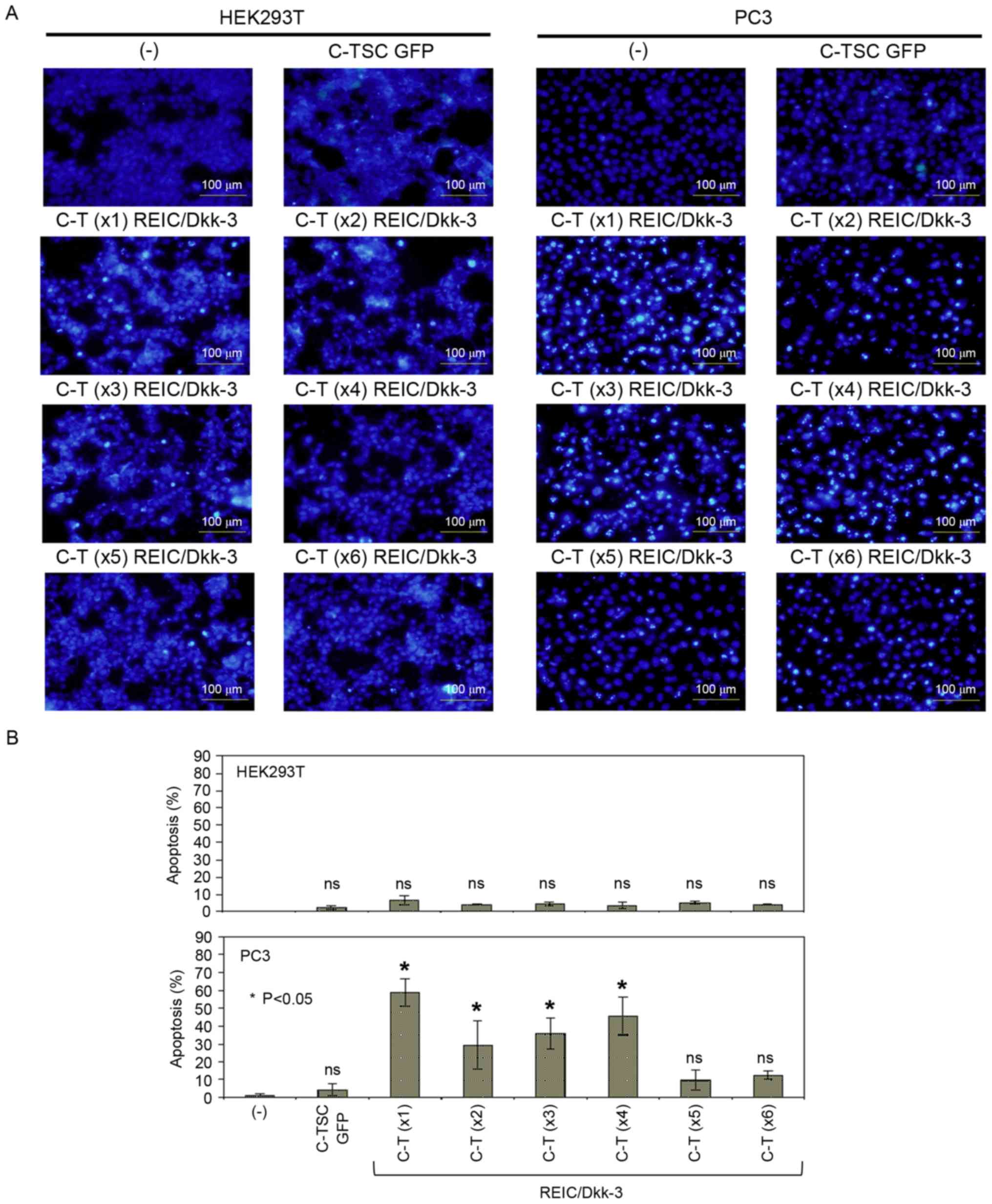 | Figure 2.Assessment of newly designed plasmid
vectors for their apoptosis-inducing capabilities. (A) Cells
transfected with the indicated plasmids were incubated for 48 h and
subsequently stained with Hoechst 33342. (B) Hoechst-positive cells
were counted as an index of apoptotic cells. All treatments were
performed in triplicate and cell counts were performed from the sum
of five images from each well with fluorescence microscopy and
averaged from three wells. C-TSC, cassette containing
cytomegalovirus-RU5′ upstream, human telomerase reverse
transcriptase, simian virus 40 and cytomegalovirus downstream, with
a polyadenylation signal; GFP, green fluorescent protein; C-T,
cassette containing cytomegalovirus-RU5′ upstream, human telomerase
reverse transcriptase downstream and a polyadenylation signal;
REIC, reduced expression in immortalized cells; Dkk-3, Dickkopf-3;
ns, not significant. |
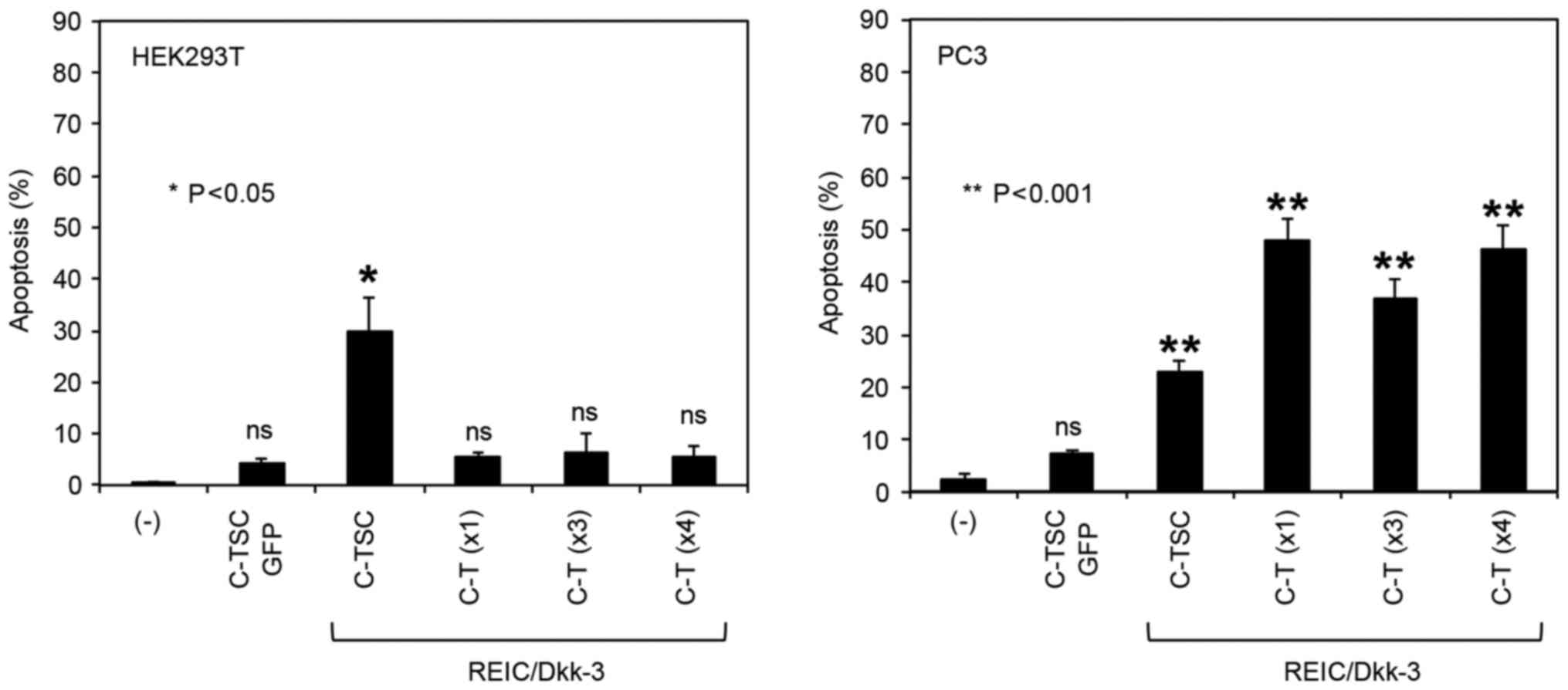 | Figure 3.Repeat assessment of the
apoptosis-inducing capabilities of the novel plasmid vectors, in
comparison with the C-TSC plasmid. Following 48-h transfection of
cells with the indicated plasmids, the cells were stained with
Hoechst 33342 and the number of apoptotic cells were counted. All
treatments were performed in triplicate and cell counts were
performed from the sum of five images from each well and averaged
from three wells. C-TSC, cassette containing cytomegalovirus-RU5′
upstream, human telomerase reverse transcriptase, simian virus 40
and cytomegalovirus downstream, with a polyadenylation signal; GFP,
green fluorescent protein; C-T, cassette containing
cytomegalovirus-RU5′ upstream, human telomerase reverse
transcriptase downstream and a polyadenylation signal; REIC,
reduced expression in immortalized cells; Dkk-3, Dickkopf-3; ns,
not significant. |
Transfection with C-TSC and C-T
plasmids results in similar levels of REIC/Dkk-3 signaling, and BiP
protein was elevated in cells transfected with C-TSC only
The present study next investigated why the rates of
apoptosis in HEK293T cells and PC3 cells were different. One
potential reason may be that ER stress-associated apoptosis is
induced by REIC/Dkk-3 overexpression, which induces JNK activation,
which in turn is linked to cancer-specific cell death (3–6). In
general, ER stress is affected by the folding speed of a newly
synthesized protein; therefore, the present study examined the
speed of expression of the REIC/Dkk-3 protein in HEK293T cells
transfected with C-TSC and C-T plasmids. However, no difference was
observed in REIC/Dkk-3 protein expression levels across a time
course experiment in cells transfected with the two vectors
(Fig. 4A). The elevation of the ER
stress marker BiP protein, which was linked to the activation of
JNK in a parallel manner in C-TSC-transfected cells, was confirmed
(Fig. 4B). Notably, this did not
occur in C-T-transfected cells, which suggested the presence of an
unknown mechanism, or mechanisms, independent of simple REIC/Dkk-3
protein expression triggering ER stress, followed by JNK-mediated
cell death.
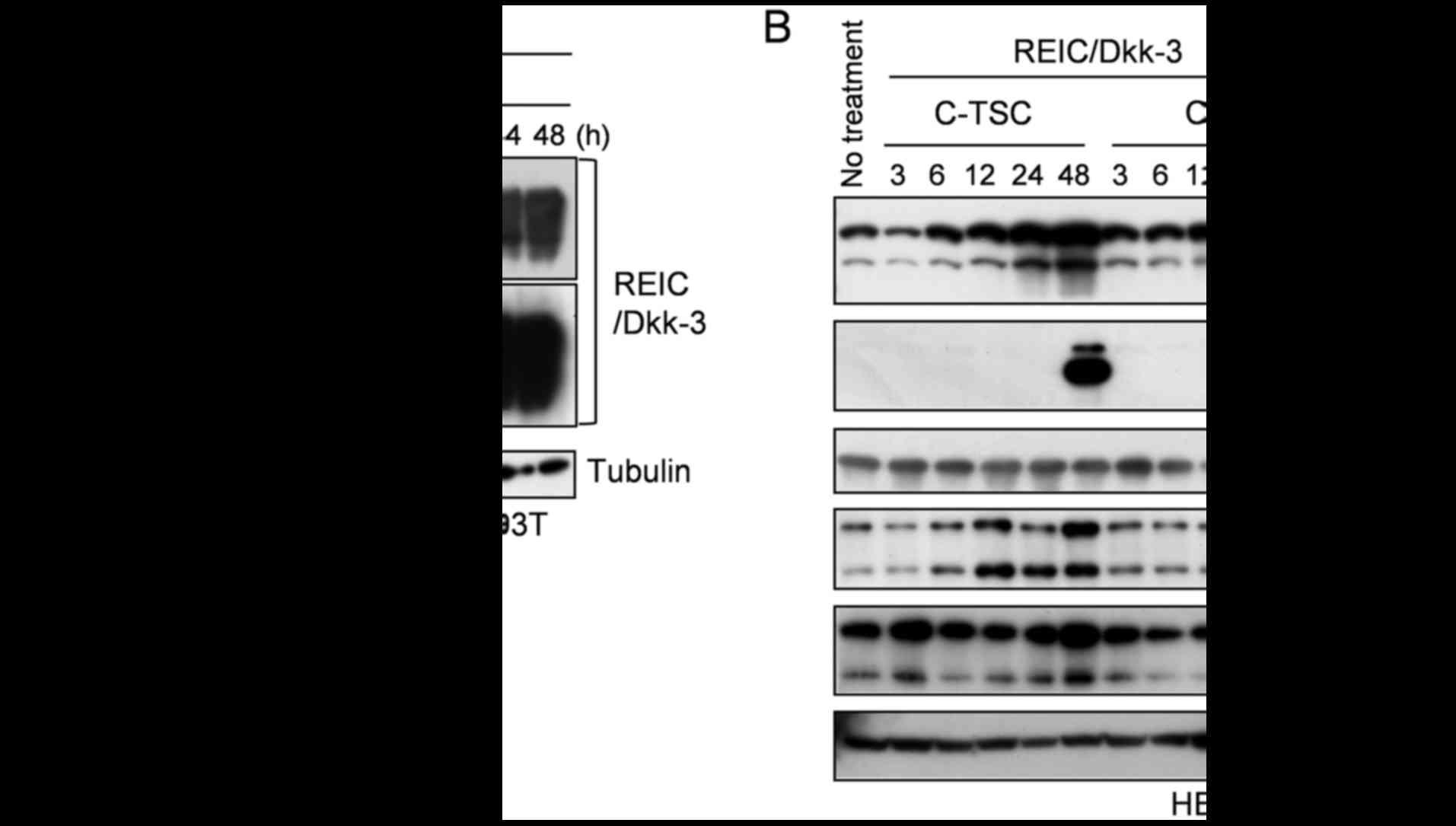 | Figure 4.Time course analysis of REIC/Dkk-3
expression levels and associated intracellular signaling. (A) Time
course evaluation of REIC/Dkk-3 expression levels from REIC/Dkk-3
cDNA-carrying C-TSC and C-T plasmids was performed in HEK293T cells
by western blotting. (B) Protein expression levels of the
endoplasmic reticulum stress marker BiP and activation levels of
p38 and JNK were analyzed by western blotting. REIC, reduced
expression in immortalized cells; Dkk-3, Dickkopf-3; C-TSC,
cassette containing cytomegalovirus-RU5′ upstream, human telomerase
reverse transcriptase, simian virus 40 and cytomegalovirus
downstream, with a polyadenylation signal; C-T, cassette containing
cytomegalovirus-RU5′ upstream, human telomerase reverse
transcriptase downstream and a polyadenylation signal; BiP, binding
immunoglobulin protein; p, phosphorylated; JNK, c-Jun N-terminal
kinase. |
Adaptation of the C-T cassette to the
conventional Ad-REIC
To further validate the utility of the C-T
construct, the C-T composition was adapted to an adenovirus vector
and an improved Ad-C-T-REIC adenovirus vector was developed. The
REIC/Dkk-3 expression levels were compared by western blotting in
cells transfected with the newly produced Ad-C-T-REIC and the
common CMV and C-TSC cassette-driven adenovirus vectors
(Ad-CMV-REIC and Ad-C-TSC-REIC; Fig.
5). In comparison with the common Ad-CMV-REIC, Ad-C-T-REIC
significantly enhanced the expression of REIC/Dkk-3 in PC3 cancer
cells and normal HUVEC cells. These expression levels were
comparable to those of Ad-C-TSC-REIC in PC3 cells, whereas the
expression levels tended to further increase in HUVEC cells
(Fig. 5A). To investigate the
therapeutic utility of Ad-C-T-REIC in human cancer cells, the
present study then assessed the in vitro apoptotic effects
of the vector under the same conditions as those presented in
Fig. 5A. Notably, the Ad-C-T-REIC
exhibited markedly higher induction of apoptosis in PC3 cells with
MOI 100 (Fig. 5B). Similar results
were also obtained in an aggressive human glioblastoma cell line,
T98 (data not shown). The present study confirmed that there was no
notable difference in the infectious efficiency of the adenovirus
vector between PC3 and HUVEC cells (Fig.
5C). Finally, it was demonstrated that viral production was
~2-fold higher with Ad-C-T-REIC compared with Ad-C-TSC-REIC
(Fig. 6; Table I). Taken together, the results
indicated that the improved C-T cassette enhanced gene expression
to a level comparable to the C-TSC cassette, and increased
adenoviral production in HEK293 cells. Hence, the Ad-C-T-REIC
vector has potential to become a useful tool for cancer gene
therapy.
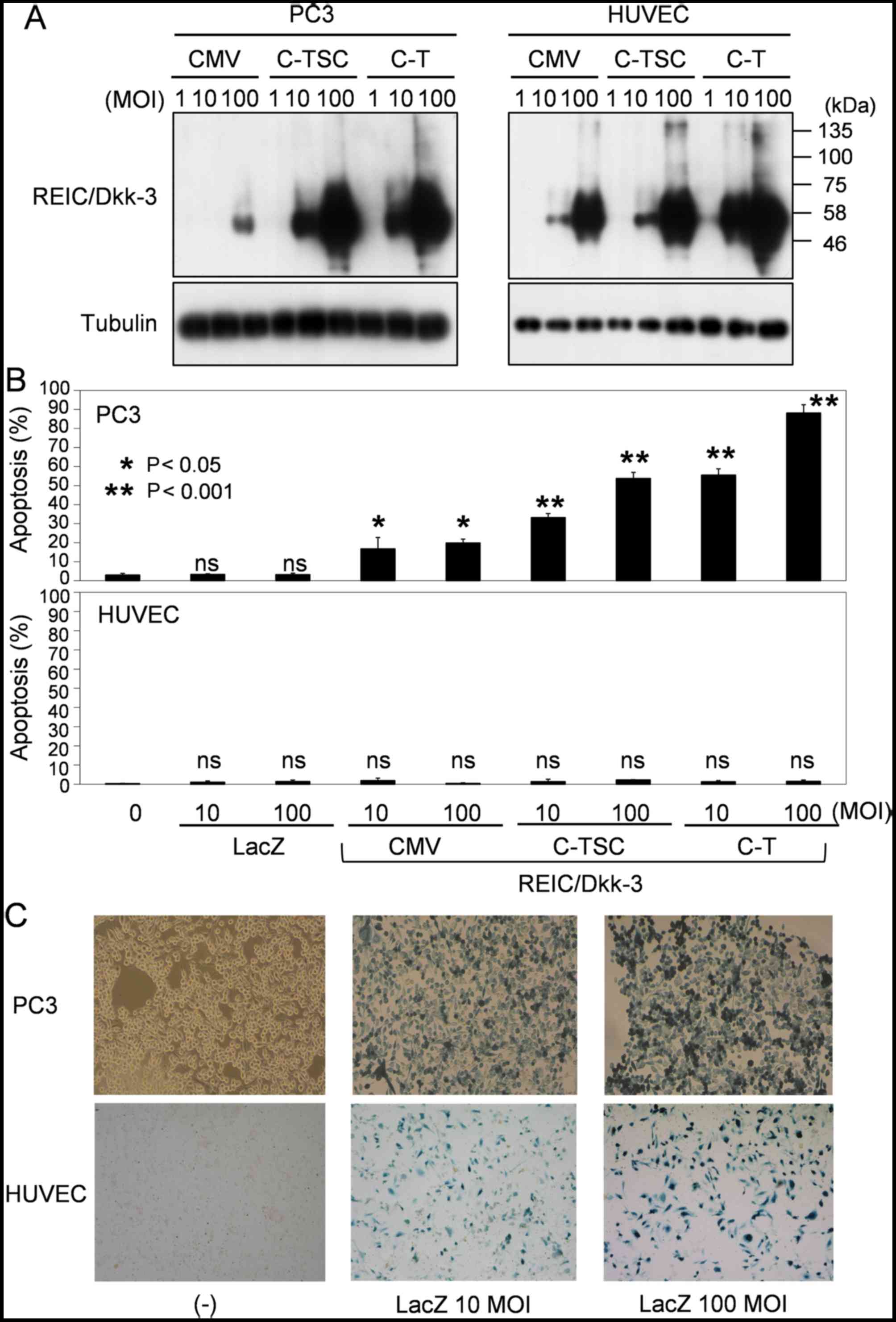 | Figure 5.Adaptation of the C-T cassette to the
conventional Ad-REIC and evaluation of the adapted vector,
Ad-C-T-REIC, for gene expression and induction of apoptosis. (A)
Expression levels of REIC/Dkk-3 from the indicated adenovirus
vectors was determined in a human prostate cancer cell line (PC-3)
and normal HUVECs. Tubulin was used as a control for loaded amounts
of protein. (B) Induction of apoptosis in PC3 and HUVEC cells by
the indicated adenovirus vectors carrying REIC/Dkk-3. (C)
Assessment of infection efficiency in PC3 and HUVEC cells by
infection with the conventional Ad-LacZ at MOIs of 10 and 100.
Expression of the LacZ gene was assessed by X-gal staining. C-T,
cassette containing cytomegalovirus-RU5′ upstream, human telomerase
reverse transcriptase downstream and a polyadenylation signal; Ad,
adenovirus; REIC, reduced expression in immortalized cells; Dkk-3,
Dickkopf-3; HUVECs, human umbilical vein endothelial cells; LacZ,
β-galactosidase; MOI, multiplicity of infection; C-TSC, cassette
containing cytomegalovirus-RU5′ upstream, human telomerase reverse
transcriptase, simian virus 40 and cytomegalovirus downstream, with
a polyadenylation signal; CMV, cytomegalovirus; ns, not
significant. |
 | Figure 6.Evaluation of viral production using
Ad-C-TSC-REIC and Ad-C-T-REIC. HEK293 cells were infected with
Ad-C-TSC-REIC and Ad-C-T-REIC. Cells were lysed and the virus was
purified from the cell extracts. The titer of the purified virus
was determined using an Adeno-X Rapid Titer kit; using optical
microscopy, infected cells were counted from 20 images per each
well. Magnification, ×100. Ad, adenovirus; C-TSC, cassette
containing cytomegalovirus-RU5′ upstream, human telomerase reverse
transcriptase, simian virus 40 and cytomegalovirus downstream, with
a polyadenylation signal; REIC, reduced expression in immortalized
cells; C-T, cassette containing cytomegalovirus-RU5′ upstream,
human telomerase reverse transcriptase downstream and a
polyadenylation signal. |
 | Table I.PFU of the stained HEK293 cells. |
Table I.
PFU of the stained HEK293 cells.
| Parameter | Ad-C-TSC-REIC | Ad-C-T-REIC |
|---|
| Hexon-positive cells,
n | 35.8±6.9 | 62.2±14.0 |
| Titer, PFU/ml |
8.62±1.7×109 |
15±3.4×109 |
Discussion
The previously developed adenovirus vector carrying
REIC/Dkk-3, Ad-C-TSC-REIC, is expected to become an innovative
cancer therapeutic tool due to its notably powerful gene expression
(7,8).
However, the significantly reduced rate of adenoviral production by
HEK293 cells due to the higher expression level of REIC/Dkk-3, or
for an unknown reason, with the Ad-C-TSC-REIC vector remains an
issue that requires consideration. Therefore, the present study
aimed to establish a method for resolving this issue. The present
study attempted to identify a solution via modification of the
original Ad-C-TSC-REIC construct. The 3′-TSC region was replaced
with a single hTERT promoter, resulting in the creation of the
Ad-C-T-REIC vector. The improved vector demonstrated significantly
higher expression of REIC/Dkk-3, which was comparable to that of
the Ad-C-TSC-REIC vector, but with attenuation of apoptotic cell
death in donor HEK293 cells. This resulted in a two-fold elevation
of viral production compared with that observed in the case of
Ad-C-TSC-REIC, suggesting that the improved Ad-C-T-REIC vector may
be valuable for application to cancer gene therapy.
The hTERT promoter is generally used to provide the
cargo gene within a certain vector for cancer-specific expression;
however, its insufficient gene expression activity remains a
serious issue. Therefore, it has been reported that the combination
of the hTERT promoter with multiple enhancer elements from other
promoters may significantly enhance the gene expression in
comparison to that of the control plasmid vectors (10). Notably, in the present study it was
revealed that positioning the hTERT promoter at the 3′-side of the
cDNA alone was sufficient to produce a significant effect on gene
expression level without the need for a serial combination with
other promoters or enhancers. Nevertheless, the 3′-side of the
hTERT promoter may not exhibit cancer specificity, since strong
expression of REIC/Dkk-3 was observed in normal human HUVEC cells,
which may in part be due to the 5′-side of the CMV promoter.
At present, it remains difficult to explain the
difference in apoptosis that was observed between the C-TSC and C-T
systems in HEK293T cells. As indicated by the results of the
present study, this was not only dependent on the REIC/Dkk-3
expression level and speed. Our previous study demonstrated that
overexpression of REIC/Dkk-3 induced cancer-specific apoptosis by
triggering certain ER stress markers and subsequent JNK activation
(3–6).
Notably, ER stress and JNK activation were mitigated by the C-T
vector compared with transfection with the C-TSC vector. In this
respect, a reasonable explanation remains to be elucidated, as the
molecular mechanism underlying gene expression levels in the newly
developed vector require detailed clarification (7). Further, more extensive studies are
required to clarify the perplexity of the expression principle and
intracellular events that may have been triggered by numerous
materials in the present study (for example, vector-derived
untranslated RNAs that may function as microRNAs or others),
including the REIC/Dkk-3 protein produced by the novel system.
Further studies in this area will ultimately result in an improved
understanding of the vector principles required to produce higher
and more specific levels of gene expression dependent on cell
types, and contribute to the development of therapeutic approaches
for a number of subtypes of cancer.
Acknowledgements
The present study was supported by the Japanese
Society for the Promotion of Science (KAKENHI; grant nos.
JP26290039 and JP15K14382). The authors declare the following
conflicts of interest, Okayama University is applying for patents
for the new vector systems. Dr Masakiyo Sakaguchi, Dr Masami
Watanabe, Dr Nam-ho Huh and Dr Hiromi Kumon are the designated
inventors for the patents. Okayama University and Momotaro-Gene,
Inc. (Okayama, Japan) are working together for the development of
the Ad-REIC agent; Momotaro-Gene, Inc. holds the patents for the
Ad-REIC agent. Dr Masakiyo Sakaguchi, Dr Masami Watanabe, Dr Nam-ho
Huh, Dr Yasutomo Nasu and Dr Hiromi Kumon own stock in
Momotaro-Gene, Inc.
References
|
1
|
Tsuji T, Miyazaki M, Sakaguchi M, Inoue Y
and Namba M: A REIC gene shows down-regulation in human
immortalized cells and human tumor-derived cell lines. Biochem
Biophys Res Commun. 268:20–24. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tsuji T, Nozaki I, Miyazaki M, Sakaguchi
M, Pu H, Hamazaki Y, Iijima O and Namba M: Antiproliferative
activity of REIC/Dkk-3 and its significant down-regulation in
non-small-cell lung carcinomas. Biochem Biophys Res Commun.
289:257–263. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Abarzua F, Sakaguchi M, Takaishi M, Nasu
Y, Kurose K, Ebara S, Miyazaki M, Namba M, Kumon H and Huh NH:
Adenovirus-mediated overexpression of REIC/Dkk-3 selectively
induces apoptosis in human prostate cancer cells through activation
of c-Jun-NH2-kinase. Cancer Res. 65:9617–9622. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kashiwakura Y, Ochiai K, Watanabe M,
Abarzua F, Sakaguchi M, Takaoka M, Tanimoto R, Nasu Y, Huh NH and
Kumon H: Down-regulation of inhibition of differentiation-1 via
activation of activating transcription factor 3 and Smad regulates
REIC/Dickkopf-3-induced apoptosis. Cancer Res. 68:8333–8341. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sakaguchi M, Kataoka K, Abarzua F,
Tanimoto R, Watanabe M, Murata H, Than SS, Kurose K, Kashiwakura Y,
Ochiai K, et al: Overexpression of REIC/Dkk-3 in normal fibroblasts
suppresses tumor growth via induction of interleukin-7. J Biol
Chem. 284:14236–14244. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kawasaki K, Watanabe M, Sakaguchi M,
Ogasawara Y, Ochiai K, Nasu Y, Doihara H, Kashiwakura Y, Huh NH,
Kumon H and Date H: REIC/Dkk-3 overexpression downregulates
P-glycoprotein in multidrug-resistant MCF7/ADR cells and induces
apoptosis in breast cancer. Cancer Gene Ther. 16:65–72. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sakaguchi M, Watanabe M, Kinoshita R, Kaku
H, Ueki H, Futami J, Murata H, Inoue Y, Li SA, Huang P, et al:
Dramatic increase in expression of a transgene by insertion of
promoters downstream of the cargo gene. Mol Biotechnol. 56:621–630.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Watanabe M, Sakaguchi M, Kinoshita R, Kaku
H, Ariyoshi Y, Ueki H, Tanimoto R, Ebara S, Ochiai K, Futami J, et
al: A novel gene expression system strongly enhances the anticancer
effects of a REIC/Dkk-3-encoding adenoviral vector. Oncol Rep.
31:1089–1095. 2014.PubMed/NCBI
|
|
9
|
Abarzua F, Kashiwakura Y, Takaoka M,
Watanabe M, Ochiai K, Sakaguchi M, Iwawaki T, Tanimoto R, Nasu Y,
Huh NH and Kumon H: An N-terminal 78 amino acid truncation of
REIC/Dkk-3 effectively induces apoptosis. Biochem Biophys Res
Commun. 375:614–618. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim SJ, Lee HS, Shin JH, Kim CG, Jeong S,
Park K, Choe H and Lee H: Preferentially enhanced gene expression
from a synthetic human telomerase reverse transcriptase promoter in
human cancer cells. Oncol Rep. 16:975–979. 2006.PubMed/NCBI
|




















