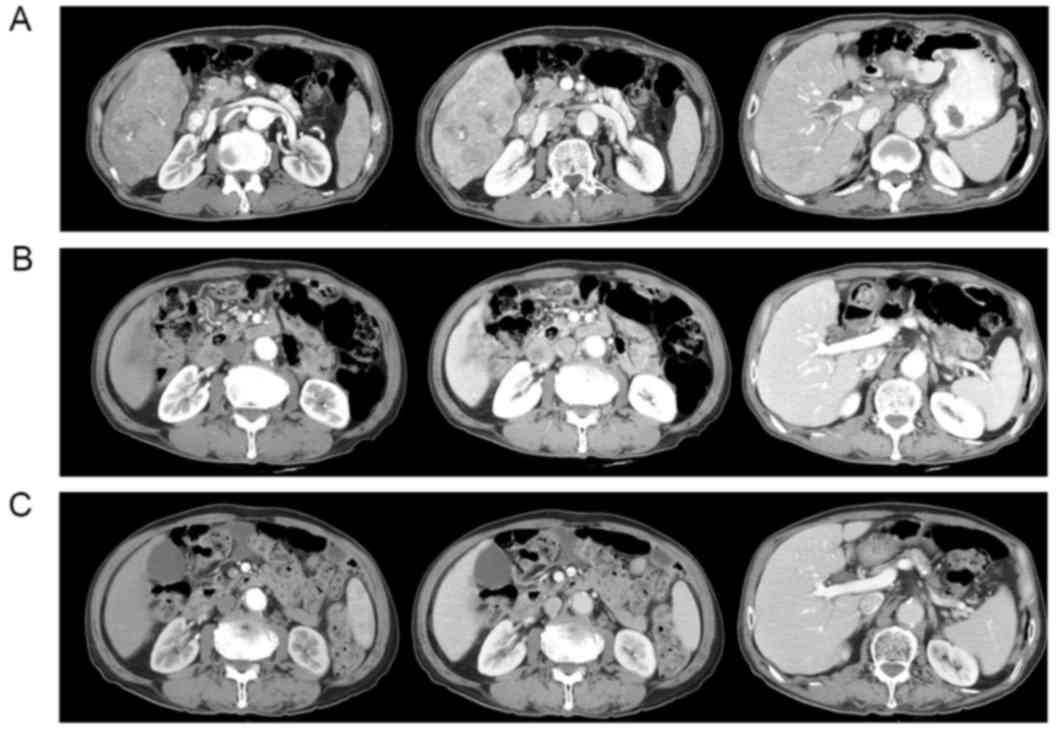
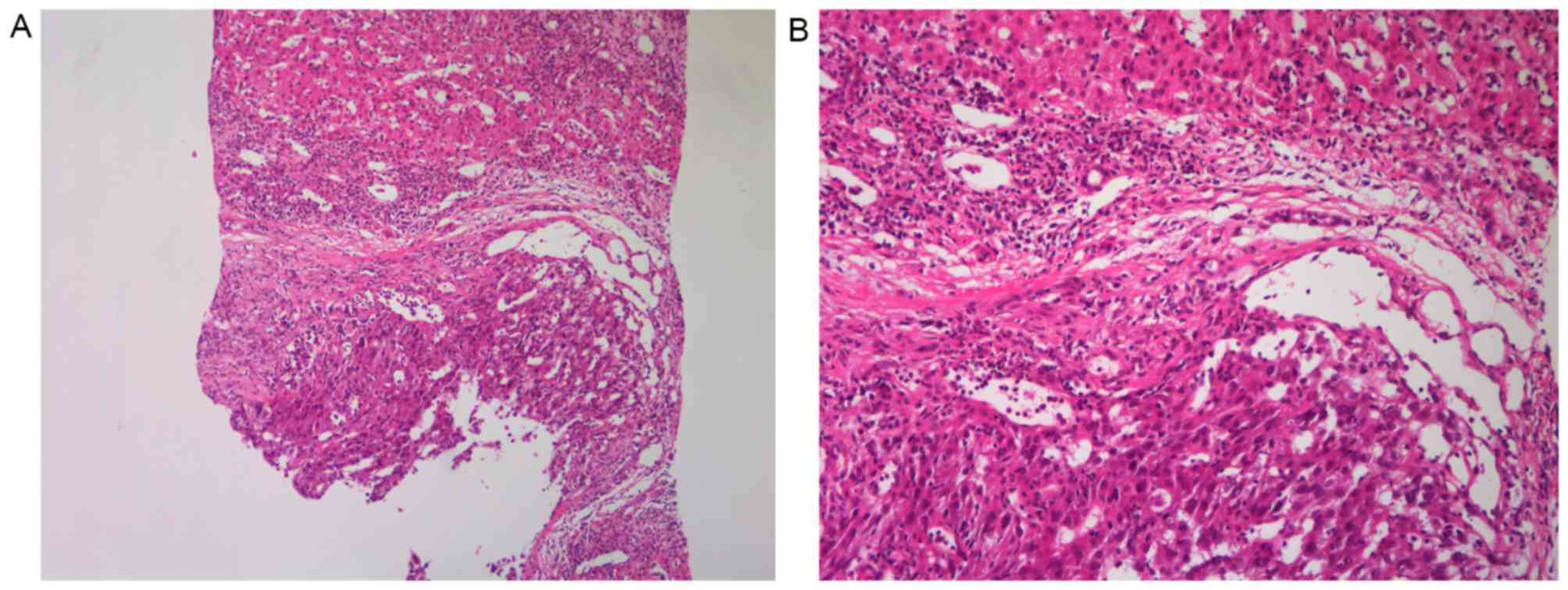
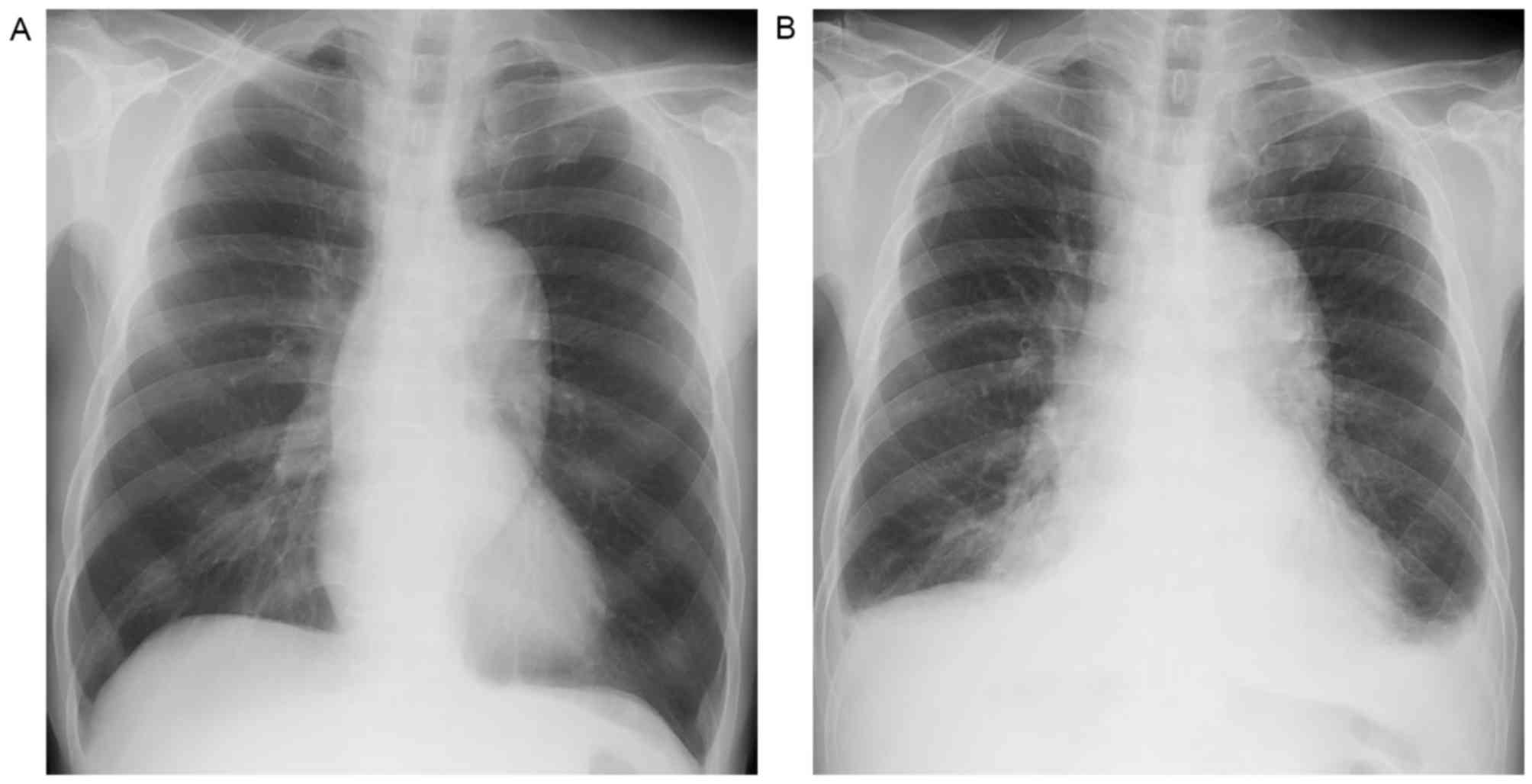
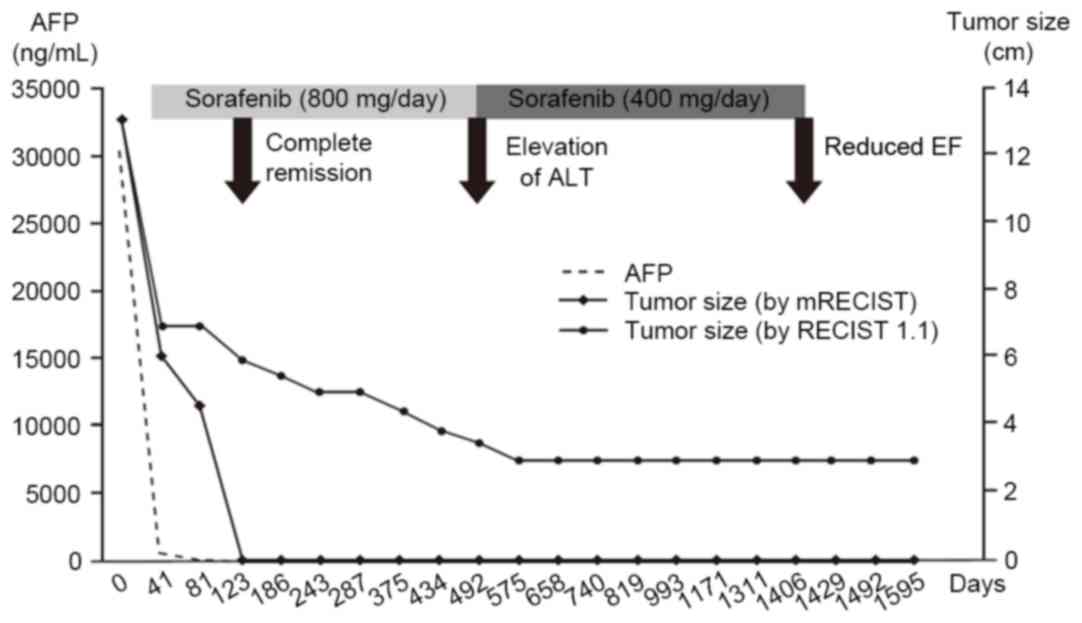
|
1
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Llovet JM, Burroughs A and Bruix J:
Hepatocellular carcinoma. Lancet. 362:1907–1917. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
European Association For The Study Of The
Liver1; European Organisation For Research And Treatment Of Cancer:
EASL-EORTC clinical practice guidelines: Management of
hepatocellular carcinoma. J Hepatol. 56:908–943. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wilhelm SM, Carter C, Tang L, Wilkie D,
McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M, et al:
BAY 43–9006 exhibits broad spectrum oral antitumor activity and
targets the RAF/MEK/ERK pathway and receptor tyrosine kinases
involved in tumor progression and angiogenesis. Cancer Res.
64:7099–7109. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wilhelm SM, Adnane L, Newell P, Villanueva
A, Llovet JM and Lynch M: Preclinical overview of sorafenib, a
multikinase inhibitor that targets both Raf and VEGF and PDGF
receptor tyrosine kinase signaling. Mol Cancer Ther. 7:3129–3140.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Llovet JM, Ricci S, Mazzaferro V, Hilgard
P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A,
et al: Sorafenib in advanced hepatocellular carcinoma. N Engl J
Med. 359:378–390. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S,
Kim JS, Luo R, Feng J, Ye S, Yang TS, et al: Efficacy and safety of
sorafenib in patients in the Asia-Pacific region with advanced
hepatocellular carcinoma: A phase III randomised, double-blind,
placebo-controlled trial. Lancet Oncol. 10:25–34. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ahn SY, Lee HS, Kweon YO, Tak WY and Park
SY: Sustained remission over 36 months of advanced hepatocellular
carcinoma after short-term sorafenib therapy. Dig Dis Sci.
58:1428–1432. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Park JG: Long-term outcomes of patients
with advanced hepatocellular carcinoma who achieved complete
remission after sorafenib therapy. Clin Mol Hepatol. 21:287–294.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim MS, Jin YJ, Lee JW, Lee JI, Kim YS,
Lee SY and Chae MH: Complete remission of advanced hepatocellular
carcinoma by sorafenib: A case report. World J Gastrointest Oncol.
5:38–42. 2013.PubMed/NCBI
|
|
11
|
Liu D, Liu A, Peng J, Hu Y and Feng X:
Case analysis of complete remission of advanced hepatocellular
carcinoma achieved with sorafenib. Eur J Med Res. 20:122015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bruix J and Llovet JM: Prognostic
prediction and treatment strategy in hepatocellular carcinoma.
Hepatology. 35:519–254. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cancer Therapy Evaluation Program 3 (1998)
Common Toxicity Criteria. Version 2.0 DCTD, NCI, NIH, DHHS.
Revised: 23. 1998.
|
|
15
|
Yau T, Yao TJ, Chan P, Wong H, Pang R, Fan
ST and Poon RT: The significance of early alpha-fetoprotein level
changes in predicting clinical and survival benefits in advanced
hepatocellular carcinoma patients receiving sorafenib. Oncologist.
16:1270–1279. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hutson TE, Bellmunt J, Porta C, Szczylik
C, Staehler M, Nadel A, Anderson S, Bukowski R, Eisen T and
Escudier B: Sorafenib TARGET Clinical Trial Group: Long-term safety
of sorafenib in advanced renal cell carcinoma: Follow-up of
patients from phase III TARGET. Eur J Cancer. 46:2432–2440. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Adjei AA, Blumenschein GR Jr, Mandrekar S,
Hillman S, Gatzemeier U and Heigener D: Long-term safety and
tolerability of sorafenib in patients with advanced non-small-cell
lung cancer: A case-based review. Clin Lung Cancer. 12:212–217.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Abdel-Rahman O and Fouad M: Risk of
cardiovascular toxicities in patients with solid tumors treated
with sorafenib: An updated systematic review and meta-analysis.
Future Oncol. 10:1981–1992. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Schmidinger M, Zielinski CC, Vogl UM,
Bojic A, Bojic M, Schukro C, Ruhsam M, Hejna M and Schmidinger H:
Cardiac toxicity of sunitinib and sorafenib in patients with
metastatic renal cell carcinoma. J Clin Oncol. 26:5204–5212. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Duran JM, Makarewich CA, Trappanese D,
Gross P, Husain S, Dunn J, Lal H, Sharp TE, Starosta T, Vagnozzi
RJ, et al: Sorafenib cardiotoxicity increases mortality after
myocardial infarction. Circ Res. 114:1700–1712. 2014. View Article : Google Scholar : PubMed/NCBI
|