Introduction
Urothelial cancer of the bladder is the 4th most
commonly diagnosed cancer in men worldwide (1). Patients with metastatic disease are
often treated with a combination chemotherapy containing
gemcitabine and cisplatin as a standard of care (2,3). However,
the treatment success is limited, resulting in a median survival of
12–15 months. Treatment failure is commonly caused by acquired
resistance after primary response (2,3).
Therefore, efficient second line chemotherapies are urgently
needed.
Integrins have been identified to play an important
role in the development of resistance to chemotherapy in bladder
cancer (4). These molecules are
transmembrane receptors with two different chains, an α (alpha) and
a β (beta) subunit. Integrins are bridges for cell-cell and
cell-extracellular matrix (ECM) interactions. Cell-matrix contact
plays a fundamental role in the metastatic potential of tumors
(5). Alterations of integrin
expression may result in an enhanced adhesive behavior in bladder
cancer (6). Moreover, the expression
patterns of integrin subtypes are known to be important mediators
of tumor cell de-differentiation and tumor proliferation (7). Furthermore, it was shown that integrins
were involved in the development of metastasis and recurrence of
urothelial cancer (6,8,9).
Drug-adapted cancer cell lines have been
successfully used to study cancer cell resistance mechanisms
(10,11). To reflect the heterogeneity of
individual bladder cancer patients and to enable a systematic
evaluation of the role of integrins concerning resistance
acquisition, we used a panel of 12 urothelial cancer cell lines
consisting of 4 parental chemosensitive cell lines and their
sublines with acquired resistance to gemcitabine or cisplatin
(12,13).
Materials and methods
Drugs
Cisplatin was purchased from Gry-Pharma
(Kirchzarten, Germany), gemcitabine from Lilly (Bad Homburg,
Germany).
Cell lines
The cell lines 5637, T24, HT1376, and TCC-SUP were
obtained from the American Type Culture Collection (Manassas, VA,
USA). The following drug-resistant sublines were established by
continuous exposure to increasing drug concentrations as described
previously (12,14) and are part of the Resistant Cancer
Cell Line (RCCL) collection (http://www.kent.ac.uk/stms/cmp/RCCL/RCCLabout.html):
5637rCDDP1000 (cisplatin-resistant, 1,000 ng
cisplatin/ml), 5637rGEMCI20
(gemcitabine-resistant, 20 ng gemcitabine/ml),
T24rCDDP1000,
T24rGEMCI20,
HT1376rCDDP1000,
HT1376rGEMCI20,
TCC-SUPrCDDP1000, and
TCC-SUPrGEMCI20. All cell lines were grown in
Iscove's modified Dulbecco's medium supplemented with 10% fetal
calf serum (FCS; Gibco, Karlsruhe, Germany), 100 IU/ml penicillin,
and 100 µg/ml streptomycin at 37°C. Cell line authentication was
performed by short tandem repeat profiling.
Cell adhesion to extracellular matrix
components
24-well plates were coated with extracellular matrix
components (Matrigel; Corning, Amsterdam, The Netherlands)
overnight. Plates were washed with 1% bovine serum albumin (BSA) in
phosphate buffered saline (PBS) to block nonspecific cell adhesion.
Thereafter, 0.5×106 tumor cells were added to each well
for 60 min. Subsequently, non-adherent tumor cells were washed off.
The adherent cells were fixed with 1% glutaraldehyde and counted in
five different fields using a microscope (20× objective) to
calculate the mean cellular adhesion rate.
Chemotaxis
Serum induced cell migration was examined using
6-well transwell chambers (Greiner, Frickenhausen, Germany) with 8
µm pores. To evaluate cell migration, cells were placed in the
upper chamber for 20 h in serum-free medium. The lower chamber
contained 10% serum. After incubation, the upper surface of the
transwell membrane was wiped gently with a cotton swab to remove
non-migrating cells. Cells migrating to the lower surface of the
membrane were stained using hematoxylin and counted. Cells
migrating into the lower chamber were counted separately under the
microscope.
Blocking study
Cells were preincubated for 60 min with a
function-blocking anti-integrin β1 monoclonal antibody (20 mg/l)
(MAB 2253Z; MerckMillipore, Darmstadt, Germany). Controls remained
untreated. Adhesion and chemotaxis was tested as indicated
above.
Flow cytometry
Cells were washed in blocking solution (PBS, 0.5%
BSA) and then incubated for 60 min at 4°C with phycoerythrin
(PE)-conjugated monoclonal antibodies directed against the
following integrin subtypes: Anti-α3 (IgG1; clone C3II.1), anti-α5
(IgG1; clone IIA1), anti-α6 (IgG2b, clone MP 4F10), anti-β1 (IgG1;
clone MAR4), anti-β3 (IgG1; clone VI-PL2) or anti-β4 (IgG2a; clone
439-9B; all: BD Biosciences, Heidelberg, Germany). Integrin
expression was measured by flow cytometry (FACSCalibur; BD
Biosciences, Heidelberg, Germany). Mouse IgG1-PE (MOPC-21) or mouse
IgG2a-PE (G155-178; all: BD Biosciences) antibodies were used as
isotype control.
Immunohistochemistry
33 cases of invasive and non-invasive bladder
cancers as well es corresponding normal urothelium were taken from
the archive of the Dr. Senckenberg Institute of Pathology in
Frankfurt. Tissue sections were stained for Integrin β1, (D2E5)
Rabbit mAb, Cell Signaling Technology (Waltham, MA, USA), dilution
1:100. In brief, 4 µm sections were cut and pretreated with
Trilogy™, Cell Marcque (Rocklin, CA, USA), incubated with the
antibody, antigen retrieval was performed at pH 6 in a microwave
oven using the Peroxidase-FLEX EnVision kit (Dako, Jena,
Germany).
A pathologist, who was blinded to clinical history
and therapeutic response, scored the immunohistochemical staining
using a five-stage staining score: 0=negative; 1=weak; 2=moderate;
3=strong; 4=very strong.
Images were acquired using a digital slide scanner
(ScanScope XT; Aperio, Vista, CA, USA).
Statistical analysis
Results are expressed as mean ± SD of at least three
independent experiments. For statistical analysis student's t-test,
analysis of variance (ANOVA), and Student-Newman-Keuls-Test were
performed whenever applicable. P<0.05 was considered to indicate
a statistically significant difference.
Results
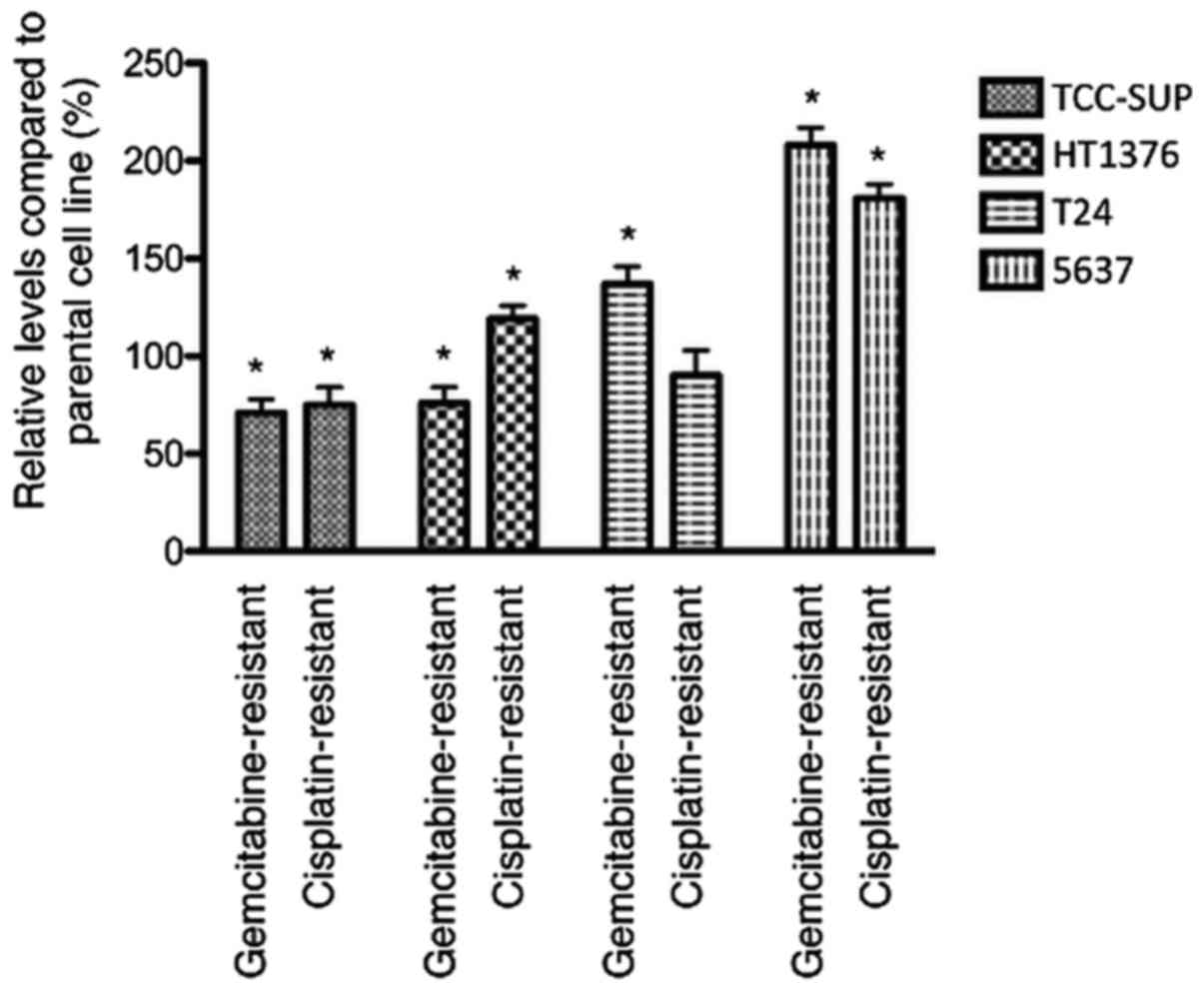
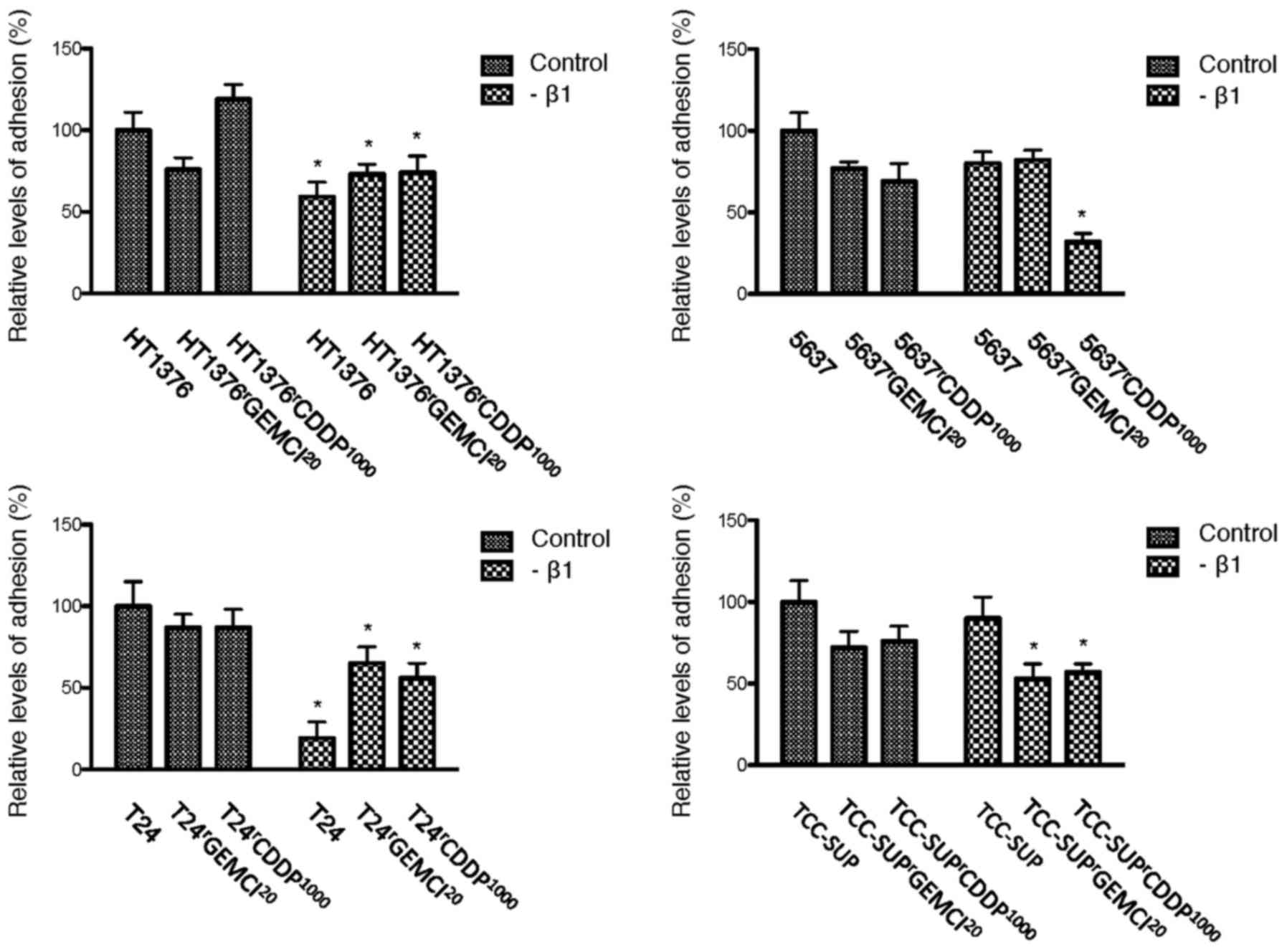
Influence of acquired resistance on
adhesion to extracellular matrix components
In untreated cells, adhesion to extracellular matrix
components was decreased in 2 of 4 gemcitabine-resistant sublines
(HT1376rGEMCI20 and
TCC-SUPrGEMCI20) and upregulated in 2 of 4
cell lines (T24rGEMCI20 and
5637rGEMCI20) compared to parental cells. In
cisplatin-resistant sublines, adhesion was decreased in 1 of 4
cisplatin-resistant sublines
(TCC-SUPrCDDP1000) and enhanced in 2 cell
lines (HT1376rCDDP1000 and
5637rCDDP1000) (Fig. 1).
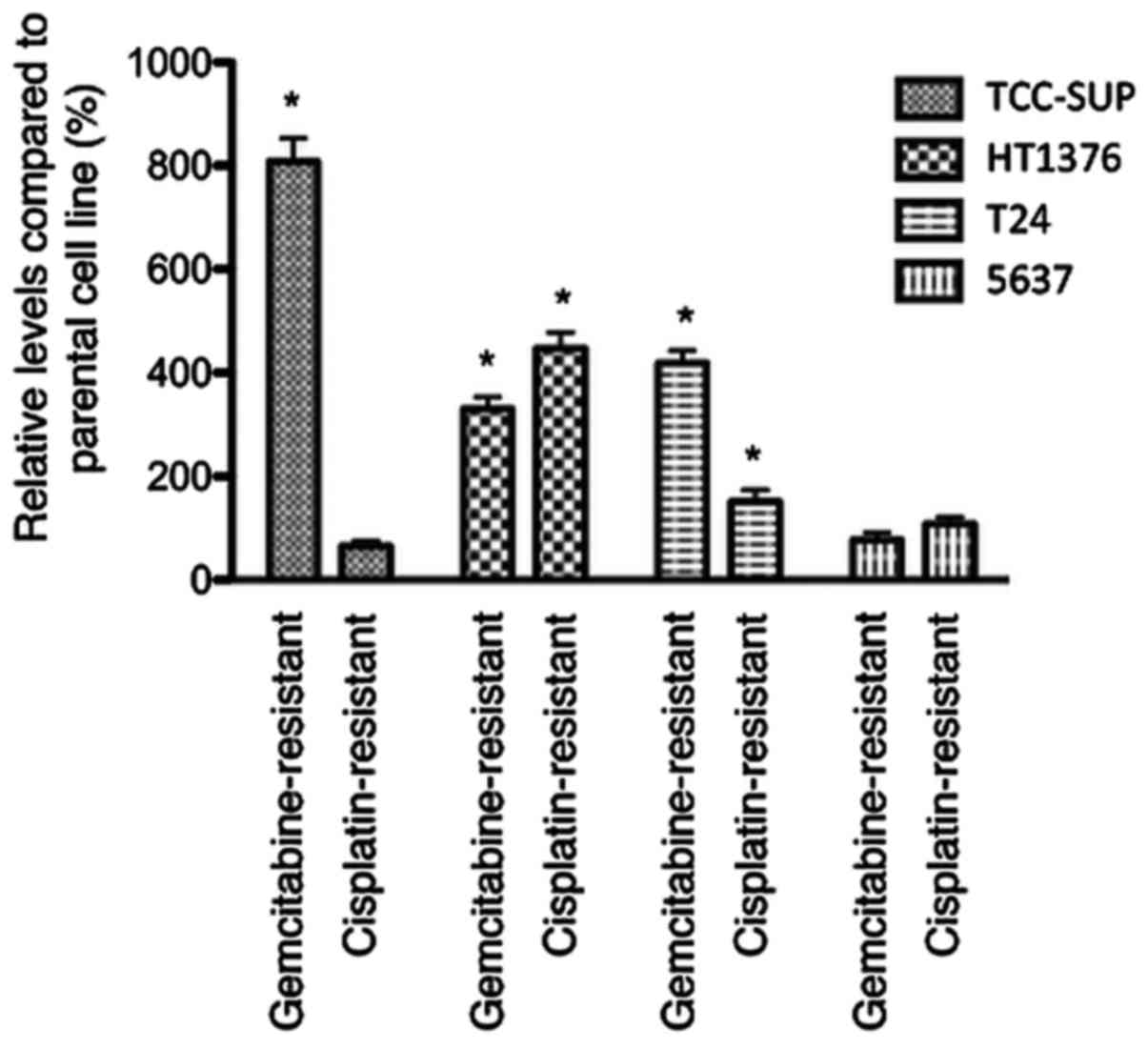
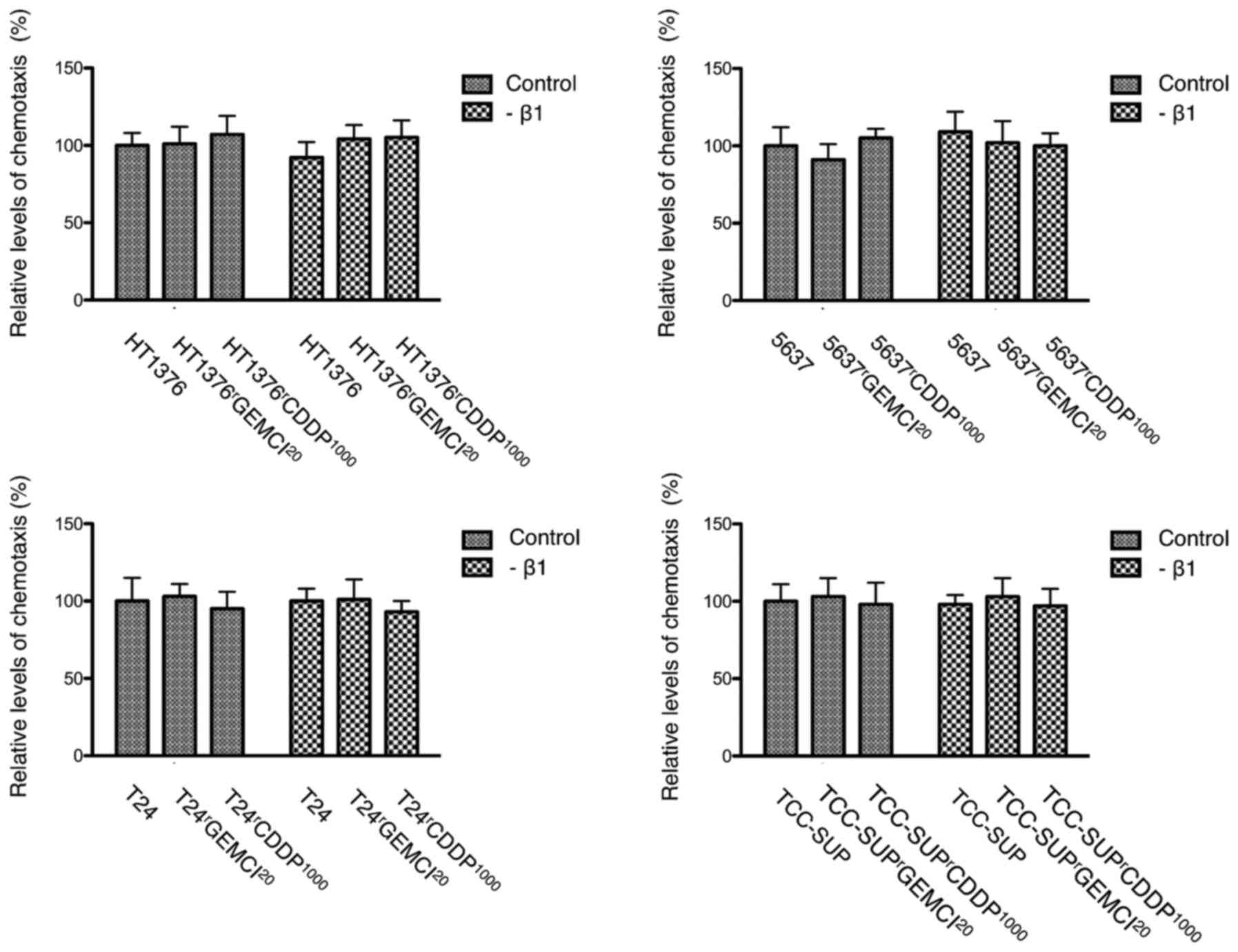
Influence of acquired chemoresistance
on chemotaxis
Chemotaxis was enhanced in 3 of 4
gemcitabine-resistant urothelial cancer cell lines
(gemcitabine-resistant sublines of TCC-SUP, HT1376 and T24). In
cisplatin-resistant sublines, chemotaxis was enhanced in
HT1376rCDDP1000 and
T24rCDDP1000 compared to parental cell lines
(Fig. 2).
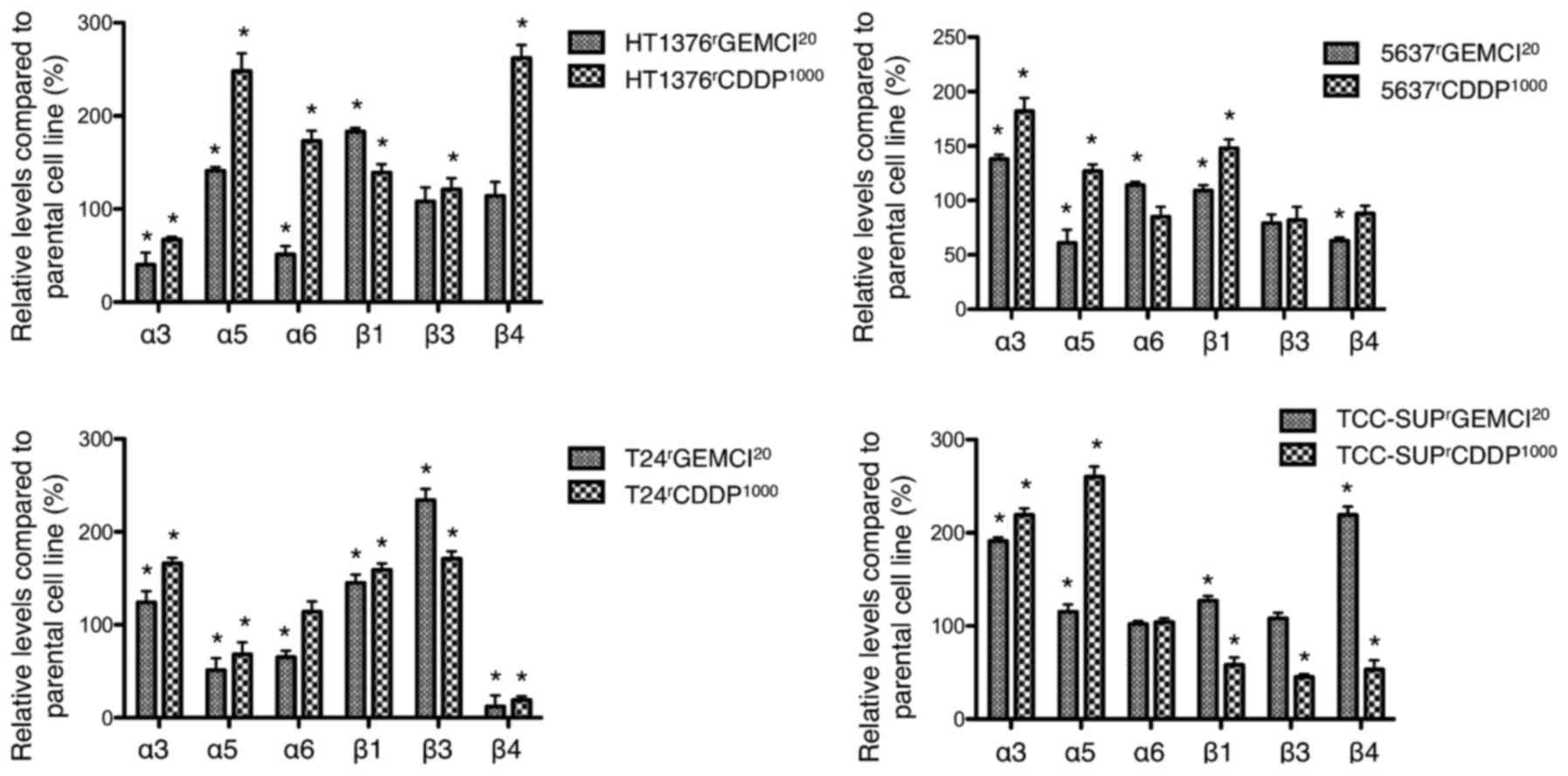
Differential expression of cell
surface integrins
Expression of integrins on the cell surface was
analyzed by flow cytometry (Fig. 3).
In gemcitabine-resistant sublines, the expression of integrin α3
was enhanced in 3 sublines (gemcitabine-resistant sublines of T24,
5637, and TCC-SUP) and diminished in
HT1376rGEMCI20 compared to parental cell
lines. Integrin β1 expression was upregulated in all
gemcitabine-resistant sublines compared to parental cells. Integrin
β4 expression was enhanced in TCC-SUPrGEMCI20
and diminished in T24rGEMCI20 and in
5637rGEMCI20.
Comparing cisplatin-resistant cell lines, Integrin
α3 expression was upregulated in 3 of 4 sublines
(cisplatin-resistant sublines of T24, 5637, and TCC-SUP) and
downregulated in HT1376rCDDP1000. Integrin α5
was upregulated in 3 of 4 sublines (cisplatin-resistant sublines of
5637, TCC-SUP, and HT1376). Integrin β1 expression was upregulated
in 3 of 4 sublines (cisplatin-resistant sublines of HT1376, T24,
and 5637) and downregulated in
TCC-SUPrCDDP1000. Integrin β4 was upregulated
in HT1376rCDDP1000 and downregulated in all
other tested sublines (cisplatin-resistant sublines of T24, 5637,
and TCC-SUP).
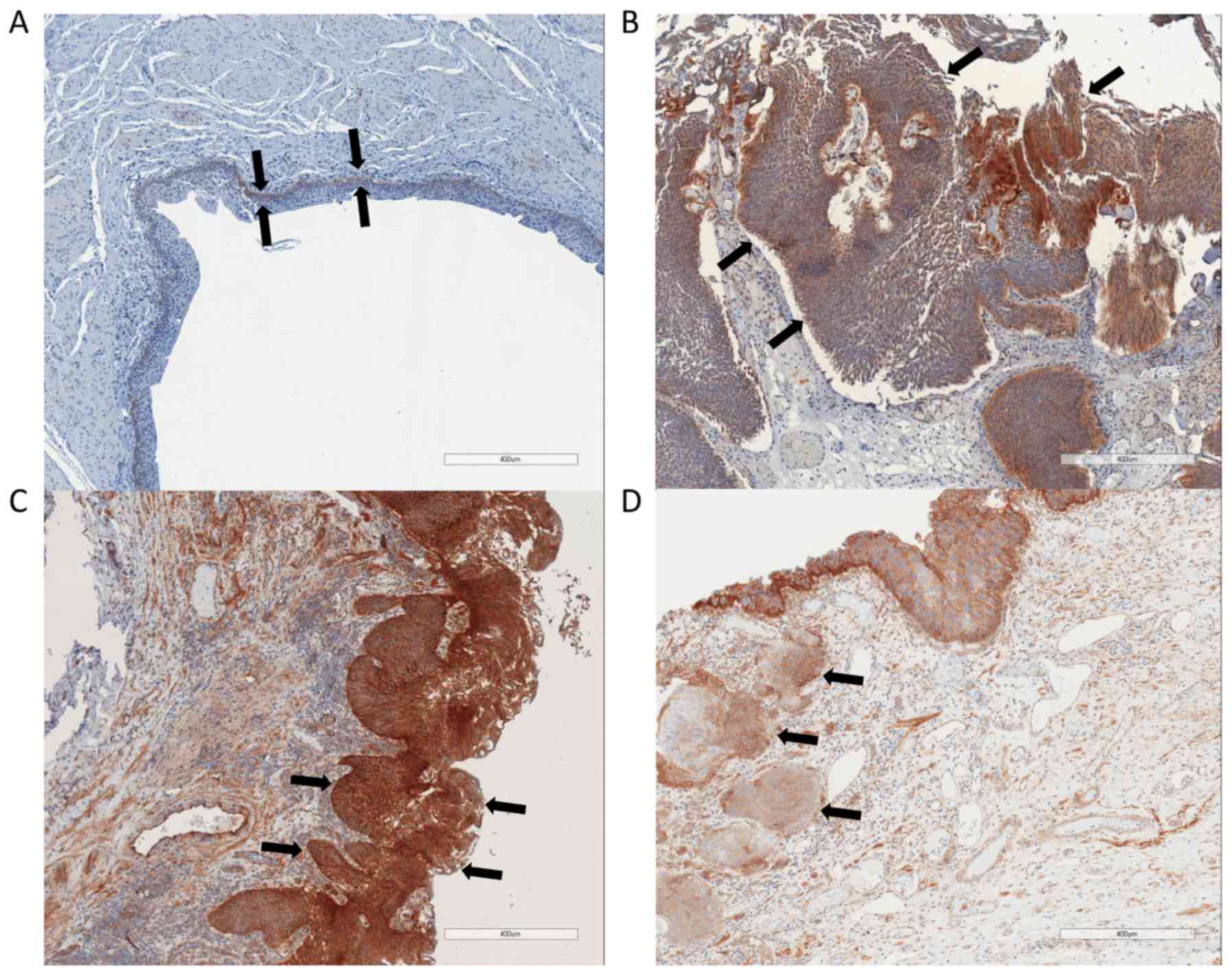
Immunohistochemical staining of
integrin β1
In non-malignant tissue samples, integrin β1 was
only visible in the basal layer of the urothelium. The medium
staining score for normal urothelium was between ‘negative’ to
‘weak’ (0.88±0.33). In bladder cancer samples, the mean integrin β1
staining score was between ‘moderate’ and ‘strong’ (2.48±1.42).
There was no significant difference of integrin β1 staining between
low grade tumors or high grade tumors and no significant difference
between non-muscle invasive tumors and muscle invasive tumors
(Fig. 4).
Influence of blocking integrin β1 on
adhesion and chemotaxis
Functional blocking of integrin β1 resulted in a
reduced adhesion in 2 of 4 parental urothelial cancer cell lines
(HT1376 and T24). In gemcitabine-resistant cells, adhesion was
downregulated in 3 of 4 cell lines (gemcitabine-resistant sublines
of HT1376, T24 and TCC-SUP). In cisplatin-resistant cells, adhesion
was downregulated in all 4 tested cell lines (Fig. 5). We could not detect an influence on
chemotaxis after blocking integrin β1 (Fig. 6).
Discussion
In the present study, we used a well-established
panel of urothelial cancer cell lines with acquired resistance to
gemcitabine or cisplatin, the standard therapeutics for metastatic
urothelial cancer of the bladder (2,3). Cell line
panels seem to be necessary to reflect the heterogeneity of
different patient-derived cancer cell lines. Although the complex
scenario of metastatic colonization is not fully understood, there
is strong evidence that alterations of tumor-matrix contact are
necessary to allow motile crawling into the surrounding tissue
(15).
In several cancer cells, resistance to gemcitabine
seems to be connected with integrins and associated proteins
(16–18). In addition, resistance to cytotoxic
drugs and proliferation regulation was shown to be dependent on
extracellular matrix proteins (19).
In this study, acquisition of resistance to gemcitabine or
cisplatin showed a changed adhesive behavior with some resistant
sublines showing an enhanced adhesive behavior and other resistant
sublines being less adhesive (Fig.
1). In contrast, the influence on chemotaxis was more uniform,
with 5 of 8 sublines showing an enhanced chemotaxis and no subline
with a significantly diminished chemotaxis after acquisition of
resistance (Fig. 2). This is in line
with Ploenes et al who reported about an enhanced chemotaxis
in lung cancer cell lines with an increased chemoresistance
(20).
Since integrins seem to be involved in the
development of resistance to chemotherapy in bladder cancer
(4) and alterations of integrin
expression change adhesive and invasive behavior of bladder cancer
cells (6), we aimed to elucidate the
role of integrins in this context.
Integrin α3 might be involved in resistance
acquisition, since it was upregulated in 3 of 4
gemcitabine-resistant and also in 3 of 4 cisplatin-resistant
sublines in this study (Fig. 3).
Litynska et al (21) tried to
analyze the role of integrin α3 in bladder cancer by blocking its
function. They described that adhesion was up- or downregulated
after blocking integrin α3 depending on the tested cell line. The
cell line specific effects that can be triggered after acquisition
of resistance show the heterogeneity between independent cell lines
and underline the importance of using a panel of cell lines for a
better interpretation.
It was reported that integrin α5 contributes to a
more malignant phenotype in urothelial bladder cancer (22). In our study, this integrin subunit was
overexpressed in most of the tested chemoresistant sublines what
might underline the more malignant phenotype of the chemoresistant
sublines (Fig. 3).
We observed that most chemoresistant sublines showed
a diminished expression of integrin β4 compared to their parental
counterparts (Fig. 3). Therefore, a
downregulation of integrin β4 could be connected with a more
malignant behavior. This is in line with reports that an
overexpression of integrin β4 inhibits growth and migration in
bladder cancer cell lines and plays an anti-tumoral role (23,24).
In all gemcitabine-resistant and in 3 of 4
cisplatin-resistant sublines, surface expressed integrin β1 was
upregulated compared to parental cell lines (Fig. 3). Since chemotaxis was frequently
enhanced after acquisition of resistance, these results might
support the conclusions of Chakraborty et al (25) who postulated that blockade of
β1-integrin with a specific antibody could result in alteration of
multiple signaling pathways related to adhesion and migration.
Interestingly, Zhang and coworkers showed that they could reverse
chemoresistance to mitomycin c by blocking integrin β1 (4). Integrin β1 was overexpressed in most of
the tested sublines and it was reported to contribute to a more
malignant phenotype in urothelial bladder cancer (4,25). We
could confirm in this study that overexpression of integrin β1 is
associated with a malignant phenotype since we detected a stronger
expression in malignant tissue samples compared to normal
urothelium. Nevertheless, there was no different expression
comparing low grade with high grade tumors or between non-muscle
invasive bladder cancer and muscle invasive bladder cancer
(Fig. 4).
To further analyze the role of integrin β1, we
suppressed the function of integrin β1 and meassured adhesion and
chemotaxis afterwards. There was an influence on adhesion after
blocking integrin β1 with a reduced adhesion in 2 of 4 parental and
3 of 4 gemcitabine-resistant sublines. In cisplatin-resistant
cells, adhesion was even downregulated in all 4 tested cell lines
(Fig. 5).
We could not show an influence on chemotaxis after
blocking integrin β1 (Fig. 6). If
there is no influence on chemotaxis or if the used transwell
migration assay is not able to reflect the impact on chemotaxis is
not clear. An explanation for the latter might be that effects that
influence invasion after blocking integrin β1 are delayed and
therefore not detectable with the used transwell migration
assay.
Discussing the role of different integrins and the
influence on adhesive and invasive behavior, we should be aware
that differentially guided adhesive behavior of different tumor
cell lines has been previously observed (21). Blocking integrin β1 inhibited
cell-matrix interactions in HCV29 and BC3726 cell lines, whereas
binding of the bladder cancer cell lines T24 and Hu456 was enhanced
(21). Each cell line therefore may
possess a characteristic receptor set and long-term treatment with
chemotherapy may influence integrin subfamilies differently.
Therefore systematic analysis of cell line panels is fundamental
(6).
We did not investigate the relevance of each
integrin member in detail that was used here. To provide a complete
picture of the role of integrin subtypes in gemcitabine- and
cisplatin-resistant bladder cancer ongoing studies are necessary.
Particularly, blocking experiments using integrins α3, α5, and β4
would be of interest. In addition, these findings are limited to
bladder cancer cell lines and bladder tissue. The role of integrins
in chemoresistant bladder cancer should be further evaluated in
vivo in an animal model.
Overall, evidence is presented here that acquired
resistance to gemcitabine or cisplatin frequently enhances
chemotaxis, what might be a surrogate for an increased invasive
behavior in chemoresistant bladder cancer cell lines. Since
overexpression of integrin β1 seems to be frequently upregulated in
chemoresistant urothelial bladder cancer cell lines, further in
vivo studies should evaluate downregulation of integrin β1 as a
potential therapeutic target especially in chemotherapy refractory
cases.
Acknowledgements
The work was supported by the charity Hilfe für
krebskranke Kinder Frankfurt e.V., its trust Frankfurter Stiftung
für krebskranke Kinder, the Patenschaftsmodell program of the
University Hospital Frankfurt and the Kent Cancer Trust.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pectasides D, Pectasides M and
Economopoulos T: Systemic chemotherapy in locally advanced and/or
metastatic bladder cancer. Cancer Treat Rev. 32:456–470. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
von der Maase H, Sengelov L, Roberts JT,
Ricci S, Dogliotti L, Oliver T, Moore MJ, Zimmermann A and Arning
M: Long-term survival results of a randomized trial comparing
gemcitabine plus cisplatin, with methotrexate, vinblastine,
doxorubicin, plus cisplatin in patients with bladder cancer. J Clin
Oncol. 23:4602–4608. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang CJ, Shen ZJ, Pan CW, Zhong S, Li T
and Zhang MG: Engagement of integrinβ1 induces resistance of
bladder cancer cells to mitomycin-C. Urology. 79:638–643. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Canel M, Serrels A, Frame MC and Brunton
VG: E-cadherin-integrin crosstalk in cancer invasion and
metastasis. J Cell Sci. 126:393–401. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Juengel E, dos Santos Meyer S, Schneider
T, Makarevic J, Hudak L, Bartsch G, Haferkamp A, Wiesner C and
Blaheta RA: HDAC inhibition suppresses bladder cancer cell adhesion
to collagen under flow conditions. Exp Biol Med (Maywood).
238:1297–1304. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Danen EH: Integrins: Regulators of tissue
function and cancer progression. Curr Pharm Des. 11:881–891. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yamasaki M, Soda S, Sakakibara Y, Suiko M
and Nishiyama K: The importance of 1,2-dithiolane structure in
α-lipoic acid for the downregulation of cell surface β1-integrin
expression of human bladder cancer cells. Biosci Biotechnol
Biochem. 78:1939–1942. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Behnsawy HM, Miyake H, Abdalla MA, Sayed
MA, Ahmed Ael-F and Fujisawa M: Expression of integrin proteins in
non-muscle-invasive bladder cancer: Significance of intravesical
recurrence after transurethral resection. BJU Int. 107:240–246.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sharma SV, Haber DA and Settleman J: Cell
line-based platforms to evaluate the therapeutic efficacy of
candidate anticancer agents. Nat Rev Cancer. 10:241–253. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Domingo-Domenech J, Vidal SJ,
Rodriguez-Bravo V, Castillo-Martin M, Quinn SA, Rodriguez-Barrueco
R, Bonal DM, Charytonowicz E, Gladoun N, de la Iglesia-Vicente J,
et al: Suppression of acquired docetaxel resistance in prostate
cancer through depletion of notch- and hedgehog-dependent
tumor-initiating cells. Cancer Cell. 22:373–388. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vallo S, Michaelis M, Rothweiler F,
Bartsch G, Gust KM, Limbart DM, Rödel F, Wezel F, Haferkamp A and
Cinatl J Jr: Drug-resistant urothelial cancer cell lines display
diverse sensitivity profiles to potential second-line therapeutics.
Transl Oncol. 8:210–216. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mani J, Vallo S, Rakel S, Antonietti P,
Gessler F, Blaheta R, Bartsch G, Michaelis M, Cinatl J, Haferkamp A
and Kögel D: Chemoresistance is associated with increased
cytoprotective autophagy and diminished apoptosis in bladder cancer
cells treated with the BH3 mimetic (−)-Gossypol (AT-101). BMC
Cancer. 15:2242015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Michaelis M, Rothweiler F, Barth S, Cinatl
J, van Rikxoort M, Löschmann N, Voges Y, Breitling R, von Deimling
A, Rödel F, et al: Adaptation of cancer cells from different
entities to the MDM2 inhibitor nutlin-3 results in the emergence of
p53-mutated multi-drug resistant cancer cells. Cell Death Dis.
2:e2432011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yilmaz M and Christofori G: Mechanisms of
motility in metastasizing cells. Mol Cancer Res. 8:629–642. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Huanwen W, Zhiyong L, Xiaohua S, Xinyu R,
Kai W and Tonghua L: Intrinsic chemoresistance to gemcitabine is
associated with constitutive and laminin-induced phosphorylation of
FAK in pancreatic cancer cell lines. Mol Cancer. 8:1252009.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jia Z: Role of integrin-linked kinase in
drug resistance of lung cancer. Onco Targets Ther. 8:1561–1565.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Duxbury MS, Ito H, Benoit E, Waseem T,
Ashley SW and Whang EE: RNA interference demonstrates a novel role
for integrin-linked kinase as a determinant of pancreatic
adenocarcinoma cell gemcitabine chemoresistance. Clin Cancer Res.
11:3433–3438. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Miyamoto H, Murakami T, Tsuchida K, Sugino
H, Miyake H and Tashiro S: Tumor-stroma interaction of human
pancreatic cancer: Acquired resistance to anticancer drugs and
proliferation regulation is dependent on extracellular matrix
proteins. Pancreas. 28:38–44. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ploenes T, Scholtes B, Krohn A, Burger M,
Passlick B, Müller-Quernheim J and Zissel G: CC-chemokine ligand 18
induces epithelial to mesenchymal transition in lung cancer A549
cells and elevates the invasive potential. PLoS One. 8:e530682013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lityńska A, Przybyło M, Pocheć E and
Laidler P: Adhesion properties of human bladder cell lines with
extracellular matrix components: The role of integrins and
glycosylation. Acta Biochim Pol. 49:643–650. 2002.PubMed/NCBI
|
|
22
|
Saito T, Kimura M, Kawasaki T, Sato S and
Tomita Y: Correlation between integrin alpha 5 expression and the
malignant phenotype of transitional cell carcinoma. Br J Cancer.
73:327–331. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kim SY, Bachman NJ, Nair TS, Goldsmith S,
Liebert M, Grossman HB, Lomax MI and Carey TE: Beta 4 integrin
transfection of UM-UC-2 (human bladder carcinoma) cells: Stable
expression of a spontaneous cytoplasmic truncation mutant with
rapid loss of clones expressing intact beta 4. Cancer Res.
57:38–42. 1997.PubMed/NCBI
|
|
24
|
Harabayashi T, Kanai Y, Yamada T, Sakamoto
M, Ochiai A, Kakizoe T, Koyanagi T and Hirohashi S: Reduction of
integrin beta4 and enhanced migration on laminin in association
with intraepithelial spreading of urinary bladder carcinomas. J
Urol. 161:1364–1371. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chakraborty A, White SM and Guha S:
Granulocyte colony-stimulating receptor promotes
beta1-integrin-mediated adhesion and invasion of bladder cancer
cells. Urology. 68:208–213. 2006. View Article : Google Scholar : PubMed/NCBI
|