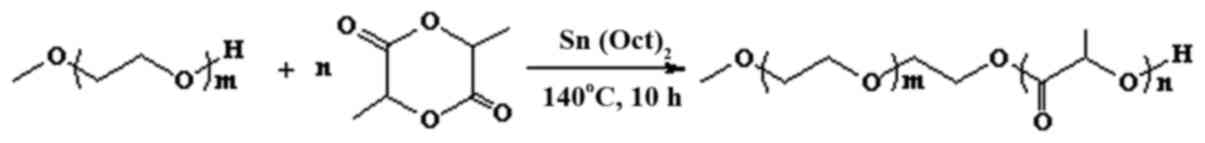
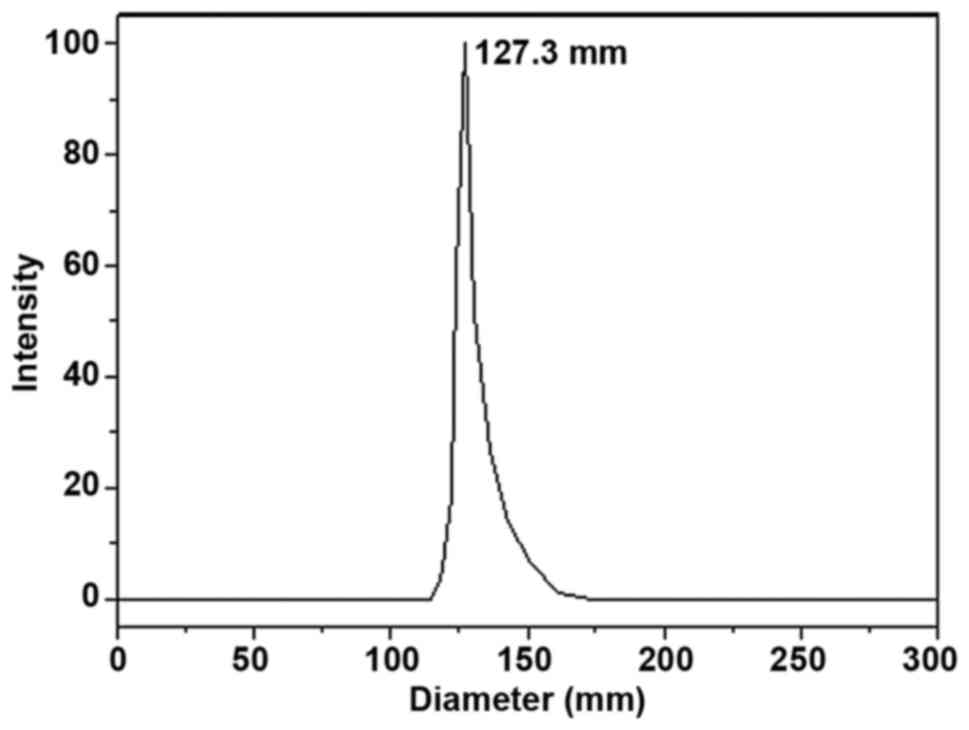
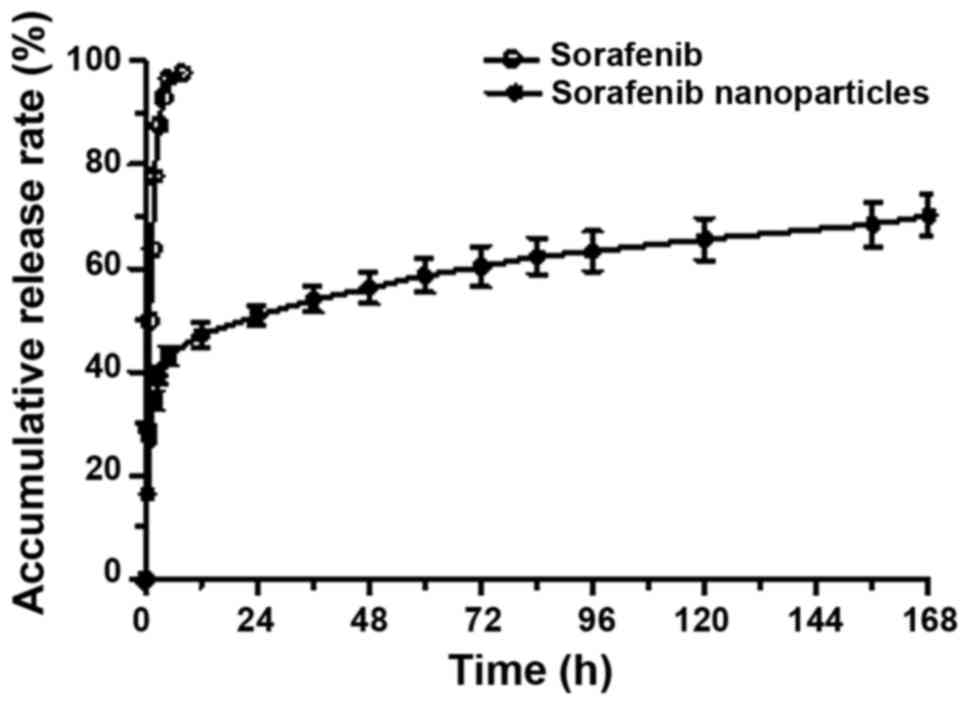
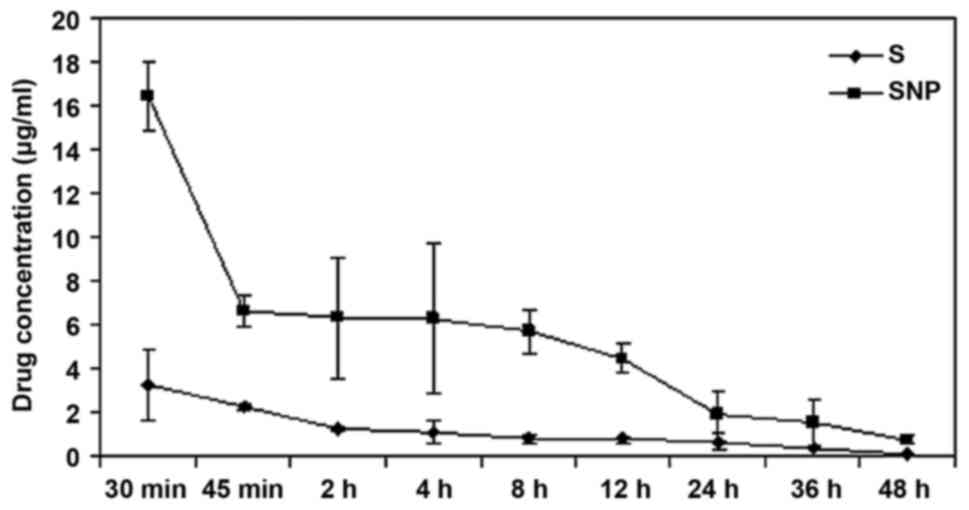
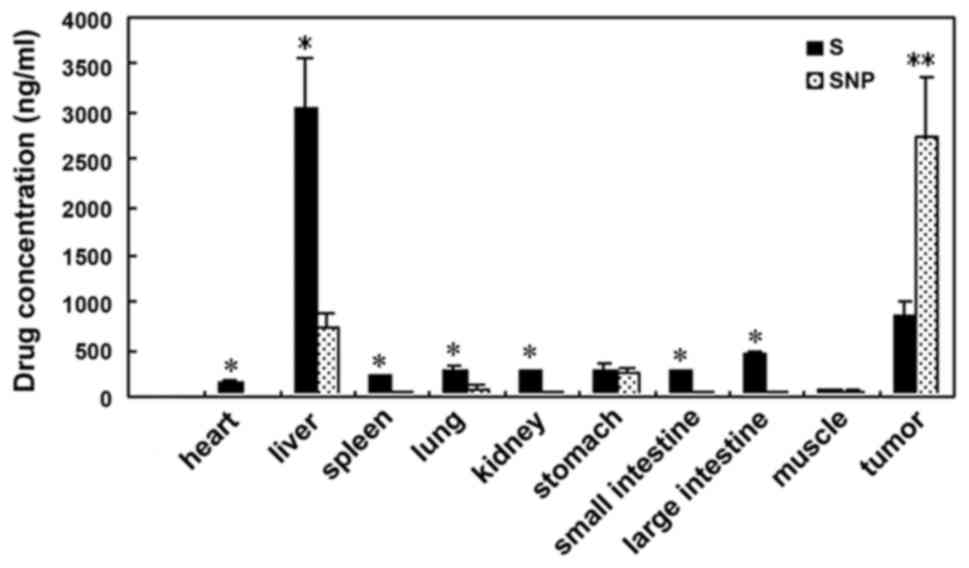
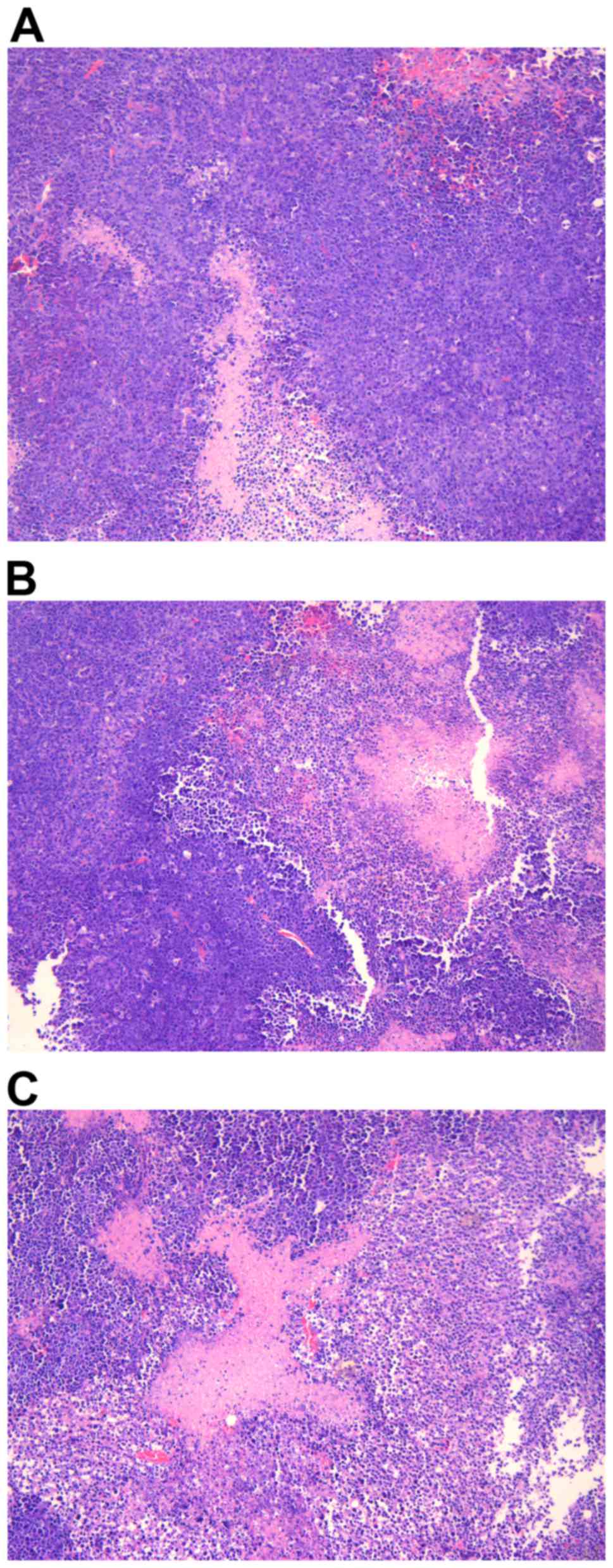
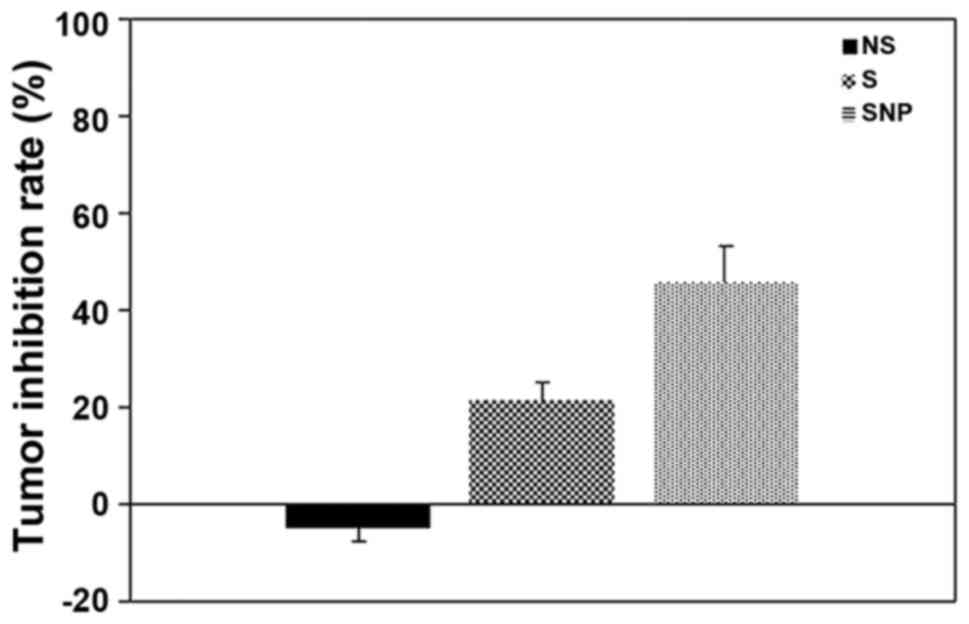
|
1
|
Matsuda Y, Ichida T and Fukumoto M:
Hepatocellular carcinoma and liver transplantation: Clinical
perspective on molecular targeted strategies. Med Mol Morphol.
44:117–124. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tang ZY, Ye SL, Liu YK, Qin LX, Sun HC, Ye
QH, Wang L, Zhou J, Qiu SJ, Li Y, et al: A decade's studies on
metastasis of hepatocellular carcinoma. J Cancer Res Clin Oncol.
130:187–196. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lyons JF, Wilhelm S, Hibner B and Bollag
G: Discovery of a novel Raf kinase inhibitor. Endocr Relat Cancer.
8:219–225. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wilhelm SM, Carter C, Tang L, Wilkie D,
McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M, et al:
BAY 43–9006 exhibits broad-spectrum oral antitumor activity and
targets the RAF/MEK/ERK pathway and receptor tyrosine kinases
involved in tumor progression and angiogenesis. Cancer Res.
64:7099–7109. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Clark JW, Eder JP, Ryan D, Lathia C and
Lenz HJ: Safety and pharmacokinetics of the dual action Raf kinase
and vascular endothelial growth factor receptor inhibitor,
BAY43-9006, in patients with advanced, refractory solid tumors.
Clin Cancer Res. 11:5472–5480. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Luo XN, Lu LG, Shao PJ, Hu BS, Li Y, Yu XY
and He X: Relationship between sorafenib-associated hand-food skin
reaction and efficacy in treatment of advanced hepatocellular
carcinoma. Zhonghua Yi Xue Za Zhi. 92:889–893. 2012.(In Chinese).
PubMed/NCBI
|
|
8
|
Chu D, Lacouture ME, Fillos T and Wu S:
Risk of hand-foot skin reaction with sorafenib: A systematic review
and meta-analysis. Acta Oncol. 47:176–186. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Stmmberg D, Awada A, Hirte H, Clark JW,
Seeber S, Piccart P, Hofstra E, Voliotis D, Christensen O,
Brueckner A and Schwartz B: Pooled safety analysis of BAY 43–9006
(sorafenib) monotherapy in patients with advanced solid tumours: Is
rash associated with treatment outcome? Eur J Cancer. 42:548–556.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Robert C, Sofia JC, Spatz A, Le Cesne A,
Malka D, Pautier P, Wechsler J, Lhomme C, Escudier B, Boige V, et
al: Cutaneous side effects of kinase inhibitors and blocking
antibodies. Lancet Oncol. 6:491–500. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang CH, Lin WC, Chuang CK, Chang YC, Pang
ST, Lin YC, Kuo TT, Hsieh JJ and Chang JW: Hand-foot skin reaction
in patients treated with sorafenib: A clinicopathological study of
cutaneous manifestations due to multitargeted kinase inhibitor
therapy. Br J Demmtol. 158:592–596. 2008. View Article : Google Scholar
|
|
12
|
Sun GT, Qiu WH, Shi MM and Chen Z:
Sorafenib inhibits human hepatoma cell line HepG2 autophagy. J Surg
Concepts Pract. 15:51–55. 2010.(In Chinese).
|
|
13
|
Liu L, Cao Y, Chen C, Zhang X, McNabola A,
Wilkie D, Wilhelm S, Lynch M and Carter C: Sorafenib blocks the
RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor
cell apoptosis in hepatocellular carcinoma model PLC/PRF/5. Cancer
Res. 66:11851–11858. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang CP, Lu YY, Gao XD, Wang Z, Bai WL, Qu
JH, Zeng Z, Zhang M, Chang XJ, Yang YP and Jie FJ: Efficacy of
sorafenib for advanced hepatocellular carcinoma and prognostic
factors. Chin J Clin Oncol. 39:587–592. 2012.(In Chinese).
|
|
15
|
Abou-Alfa GK, Schwartz L, Ricci S, Amadori
D, Santoro A, Figer A, De Greve J, Douillard JY, Lathia C, Schwartz
B, et al: Phase II study of sorafenib in patients with advanced
hepatocellular carcinoma. J Clin Onco1. 24:4293–4300. 2006.
View Article : Google Scholar
|
|
16
|
Liovet J, Ricci S, Mazzaferro V, Hilgard
P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A,
et al: Sorafenib in advanced hepatocellular carcinoma. N Engl J
Med. 359:378–390. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S,
Kim JS, Luo R, Feng J, Ye S, Yang TS, et al: Efficacy and safety of
sorafenib in patients in the Asia-Pacific region with advanced
hepatocellular carcinoma: A phase III randomized, double-blind,
placebo-controlled trial. Lancet Oncol. 10:25–34. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hu AB, He XS, Tai Q, Zhu XF, Ma Y, Wang
DP, Wang GD, Wu LW, Ju WQ and Huang JF: Efficacy and safety of
sorafenib in the prevention and treatment of hepatocellular
carcinoma recurrences after liver transplantation. Zhonghua Yi Xue
Za Zhi. 92:1264–1267. 2012.(In Chinese). PubMed/NCBI
|
|
19
|
Qin JM, Zhang YD, Wang HY, et al:
Nanotechnology usage in diagnosis and treatment of liver diseases.
Chin J Mod Med. 13:49–52. 2003.
|
|
20
|
Qin JM, Zhang YD, Wang HY and Wu MC:
Application and progress of nano-drugs in liver disease. Chin J
Hepatobiliary Surg. 10:646–648. 2004.(In Chinese).
|
|
21
|
Qin JM, Yin PH, Li Q, Sa ZQ, Sheng X, Yang
L, Huang T, Zhang M, Gao KP, Chen QH, et al: Anti-tumor effects of
brucine immuno-nanoparticles on hepatocellular carcinoma. Int J
Nanomedicine. 7:369–379. 2012. View Article : Google Scholar : PubMed/NCBI
|