Introduction
Lung cancer is the main cause of mortalities,
accounting for 18% of all cancer-associated mortalities (1). Lung cancer has the highest incidence and
mortality rates amongst all malignancies worldwide (2,3). The
pathogenesis of lung cancer remains unclear. However, an
inflammatory microenvironment consisting of infiltrating
lymphocytes and secretary cytokines is recognized to be a key
inducer of tumorigenesis and malignancy (4–6).
As a major cyclooxygenase-2-derived metabolite,
prostaglandin E2 (PGE2) is well known as an important inflammatory
factor, which is able to induce tumor growth and suppress immune
functions by secreting into the tumor microenvironment (7,8). It has
been revealed that PGE2 inhibits immune responses by upregulating
the level of forkhead transcription factor 3 expression, which is
known to promote the development of cluster of differentiation
(CD)4+ and CD25+ regulatory T cells (Treg)
(9).
Programmed cell death 1 (PD-1) belongs to the CD-28
family and is expressed on T cells, dendritic cells, natural killer
cells, macrophages and B-cells. Programmed cell death ligand
(PD-L)1 and PD-L2 are two major ligands of PD-1. PD-L1 is reported
to be produced by T-cells, B cells and myeloid dendritic cells, and
at low levels in the lungs, kidney, liver and heart. The activation
of PD-1/PD-L1 signaling in tumors can inhibit T cell function and
weaken the immune response, leading to a poor prognosis (10–12).
Blocking a combination of PGE2 and PD-1 signaling has been reported
to be therapeutic in chronic lymphocytic choriomeningitis virus
infection by augmenting the numbers of functional virus-specific
cytotoxic T lymphocytes via PGE2 receptors, EP2 and EP4 (9,13).
However, whether a direct association exists between PGE2 and PD-1
remains unclear.
In the present study, the level of PD-1 expression
in infiltrating CD4+ and CD8+ T cells
isolated from lung cancer tissues was analyzed and whether the
level of PD-1 expression in T cells may be directly regulated by
PGE2 in lung cancer tissue homogenate, which my lead to immune
inhibition, was investigated. Clarifying the immune tolerance of
infiltrating lymphocytes would be useful for improving the
immunotherapy of lung cancer.
Patients and methods
Patients
A total of 75 patients with lung cancer were
recruited for the present study from The Third Hospital of Southern
Medical University (Guangzhou, China) between August 2014 and
October 2015. Patients with lung cancer enrolled in the present
study were classified into four groups [stages I (n=20), II (n=25),
III (n=17) and IV (n=13)], according to the pathological
tumor-node-metastasis stage based upon the 8th edition of the
American Joint Committee on Cancer Staging Manual (14). Any participants with systemic
disorders or viral infections were excluded from the study.
Written informed consent was obtained from all
patients prior to enrollment in the present study, and the
experimental protocol was approved by the Ethics Committee of the
Third Hospital of Southern Medical University. The demographic and
clinical characteristics of patients are presented in Table I.
 | Table I.Basic clinical characteristics of the
patients in the present study. |
Table I.
Basic clinical characteristics of the
patients in the present study.
| Parameters | Stage I, n=20 | Stage II, n=25 | Stage III,
n=17 | Stage IV, n=13 |
|---|
| TNM stage | T1aN0M0 (11) | T1bN1M0 (7) | T1N2M0 (6) | T2N2M1a (8) |
| (no. of
patients) | T1bN0M0 (9) | T2aN1M0 (10) | T2N2M0 (6) | T3N2M1a (5) |
|
|
| T2bN0M0 (8) | T3N1M0 (5) |
|
| Age,
yearsa | 56 (42–63) | 58 (46–67) | 57 (44–65) | 56 (45–68) |
| Sex |
| Female/male | 14/6 | 17/8 | 12/5 | 9/4 |
Tissues and isolation of
lymphocytes
Fresh lung cancer tissue samples were cut from the
center of the tumor block (40–50 mg). Following washing in ice-cold
PBS, the tumor tissues were cut into 1 mm3 sections. A
mechanical trituration method was used to obtain tissue homogenate.
Single cells from lung cancer tissue homogenate were prepared by
filtrating using a 100-mesh sieve. Subsequently, the cells were
suspended in Dulbecco's modified Eagle's medium (DMEM; Thermo
Fisher Scientific Inc., Waltham, MA, USA). Ficoll-Paque Plus (GE
Healthcare, Chicago, IL, USA) was then added to the cell suspension
to isolate infiltrating lymphocytes in lung cancer tissues using a
density-gradient centrifugation method.
Fluorescein isothiocyanate (FITC)-conjugated
anti-CD4 (cat. no. 555346; dilution, 1:500), phycoerythrin
(PE)-conjugated anti-CD8 (cat. no. 557086; dilution, 1:500) and
isotype-matched controls (dilution, 1:500; all from BD Biosciences,
San Jose, CA, USA) were used to stain the isolated lymphocytes at
37°C for 30 min. CD4+ T cells and CD8+ T
cells were sorted using FACSAria II (BD Biosciences). All data were
analyzed using Flow Jo software (version 7.6.2; Tree Star, Inc.,
Ashland, OR, USA).
Cell culture and ELISA
In order to investigate the association between PGE2
and PD-1, sorted infiltrating CD4+ and CD8+ T
cells were cultured at a density of 1×105 cells/well in
24-well plates (Corning Incorporation NY, USA), were pre-coated
with CD3/CD28 monoclonal antibodies and were maintained in DMEM
supplemented with 10% FBS, 100 U/ml penicillin and 0.1 mg/ml
streptomycin. All cell culture media and supplements were purchased
from Gibco (Thermo Fisher Scientific, Inc.). EP1 antagonist
ONO-8711 (Cayman Chemical Company, Ann Arbor, MI, USA), EP2
antagonist ONO-AE1-259-01, EP3 antagonist ONO-AE5-599 (both from
ONO Pharmaceutical Co., Osaka, Japan) and EP4 antagonist GW627368
(MedChem Express Co., Shanghai, China) were used to inhibit the
corresponding signaling pathways.
The concentration of PGE2 in the supernatant of lung
cancer tissue homogenate was determined by ELISA, according to the
manufacturer's instructions (Uscn Life Sciences, Inc., Wuhan,
China).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was isolated from the T cell subsets of
patients with lung cancer using TRIzol® (Invitrogen;
Thermo Fisher Scientific, Inc.), according to the manufacturer's
instructions. Reverse transcription was performed using the
RevertAid First Strand cDNA Synthesis kit (Thermo Fisher Scientific
Inc.). The thermocycling conditions were as follows: 37°C for 30
min and 65°C for 10 min.
Gene-specific PCR amplification was performed using
Power SYBR Green Master Mix (Thermo Fisher Scientific Inc.). The
thermocycling conditions for qPCR were as follows: 95°C for 10 min,
40 cycles of 95°C for 15 sec and 60°C for 1 min. The relative level
of gene expression was evaluated using the 2−ΔΔCq method
(15) following normalization to the
level of GAPDH expression. The primers used are presented in
Table II.
 | Table II.Primer sequences used in quantitative
polymerase chain reaction. |
Table II.
Primer sequences used in quantitative
polymerase chain reaction.
| Primer | Primer sequences
(5′-3′) |
|---|
| PD-1 |
|
Forward |
AAGCTTATGTGGGTCCGGC |
|
Reverse |
GGATCCTCAAAGAGGCC |
| EP1 |
|
Forward |
TCGCTTCGGCCTCCACCTTCTTTG |
|
Reverse |
CGTTGGGCCTCTGGTTGTGCTTAG |
| EP2 |
|
Forward |
CCACGATGCTCCTGCTGCTT |
|
Reverse |
TCCACAAAGGTCAGTCTGTTT |
| EP3 |
|
Forward |
CGGGGCTACGGAGGGGATGC |
|
Reverse |
ATGGCGCTGGCGATGAACAACGAG |
| EP4 |
|
Forward |
GGTCATCTTACTCATCGCCACCTCTC |
|
Reverse |
TCCCACTAACCTCATCCACCAACAG |
| GAPDH |
|
Forward |
GGTGGTCTCCTCTGACTTCAACA |
|
Reverse |
GTGGTCGTTGAGGGCAATG |
Western blotting
The cells were lysed in RIPA buffer with protease
inhibitors (Roche Diagnostics, Basel, Switzerland) on ice for 30
min and centrifuged for 10 min at 12,000 × g at 4°C. Protein
concentration was determined using a bicinchoninic acid protein
assay kit (Thermo Fisher Scientific, Inc.). Subsequently, 30 µg
protein/well were separated on a 10% SDS-PAGE gel and transferred
onto polyvinylidene difluoride membranes (GE Healthcare). Membranes
were blocked with 5% skimmed dry milk for 30 min at room
temperature and were incubated overnight at 4°C with rabbit
anti-human PD-1 monoclonal antibody (cat. no. ab214421; dilution,
1:1,000), rabbit anti-human EP-2 polyclonal antibody (cat. no.
ab117270; dilution, 1:1,000), rabbit anti-human EP-4 polyclonal
antibody (cat. no. ab45295; dilution, 1:1,000) and rabbit
anti-human GAPDH monoclonal antibody (cat. no. ab181602; dilution,
1:2,000; all from Abcam, Cambridge, UK). The membranes were
subsequently incubated with a goat anti-rabbit horseradish
peroxidase-conjugated secondary antibody (cat. no. sc-2004;
dilution, 1:1,500; Santa Cruz Biotechnology, Inc., Dallas, TX, USA)
for 2 h at 37°C. The blots were visualized using the enhanced
chemiluminescence detection system (Beyotime Institute of
Biotechnology, Haimen, China). The densitometry score was
determined using Quantity One software (version 4.6; Bio-Rad
Laboratories, Inc., Hercules, CA, USA).
Statistical analysis
All data were expressed as the mean ± standard
deviation. The Kruskal-Wallis and Dunn's multiple comparison tests
were used to compare three or more groups of sample data. The
comparison between two groups was analyzed by the Mann-Whitney
non-parametric test. The correlations between variables were
evaluated by the Spearman's rank correlation test. All statistical
analyses were performed using GraphPad Prism software (version 5.0;
GraphPad Software, Inc., La Jolla, CA, USA). Two-sided P<0.05
was considered to indicate a statistically significant difference.
All results were repeated three times.
Results
Expression of PD-1 in infiltrating
CD8+ T lymphocytes is associated with the level of PGE2 in lung
cancer tissue homogenate
It was revealed that the normal functions of T
lymphocytes were suppressed in numerous types of solid tumors,
resulting in reduced antitumor immunity and evasion of host immune
surveillance (16,17). As an inflammatory factor, PD-1
exhibited marked immunosuppressive effects in a number of different
types of tumors (18,19). In order to determine the change in the
number of PD-1+ T cells during the progression of lung
cancer, the levels of PD-1 expression in infiltrating
CD4+ T cells and CD8+ T cells was determined
in patients with lung cancer at four disease stages: Stage I
(n=20), II (n=25), III (n=17) and IV (n=13).
Following isolation of tumor infiltrating
lymphocytes from patient tissues, FITC-conjugated anti-CD4 and
PE-conjugated anti-CD8 were used to label CD4+ and
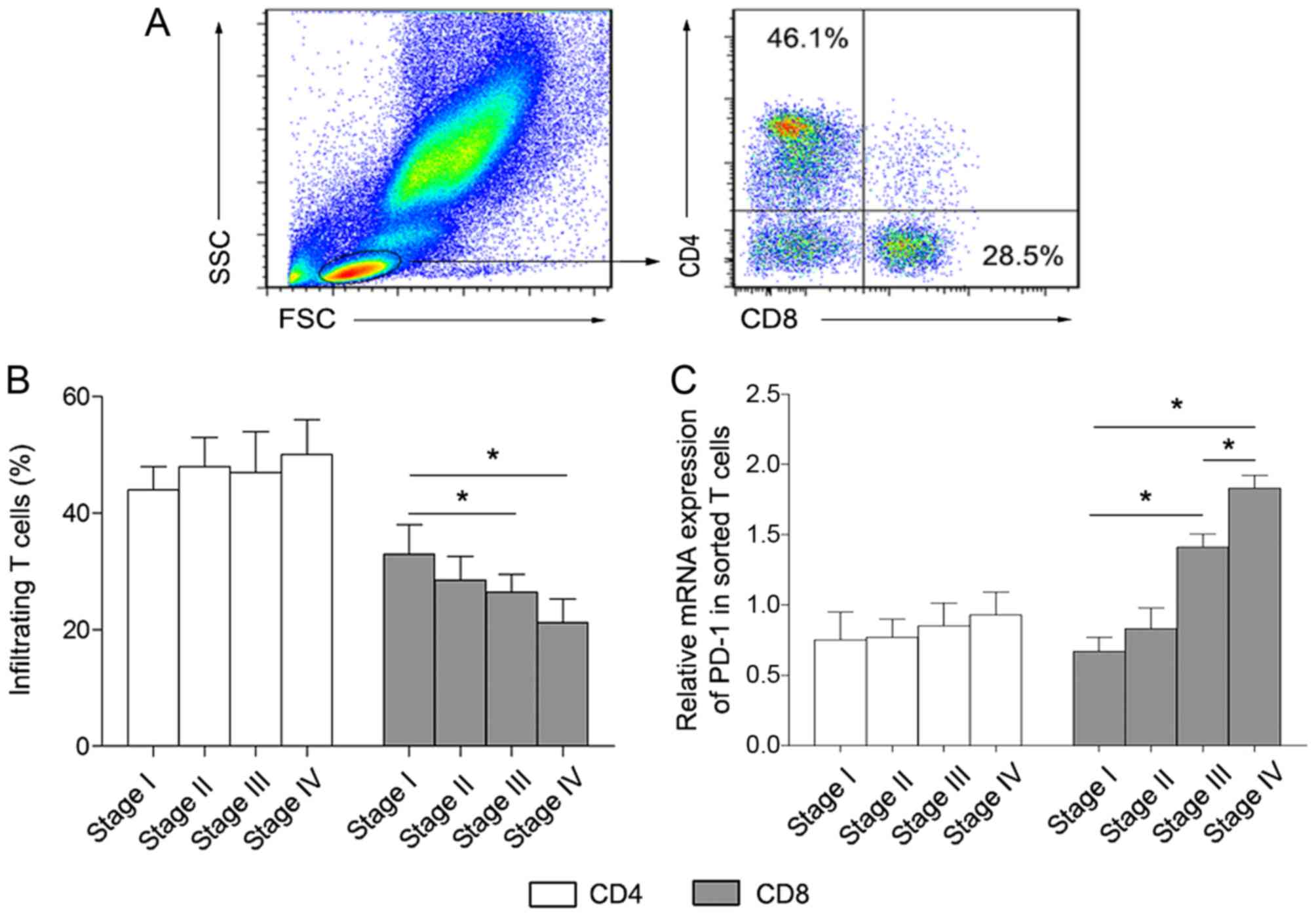
CD8+ T cells, respectively. As presented in Fig. 1A and B, the percentage of
CD4+ T cells was not significantly altered in the four
lung cancer groups. By contrast, the proportion of CD8+
T cells gradually decreased along with lung cancer development.
Subsequently, the level of PD-1 expression in sorted
CD4+ and CD8+ T cells was analyzed.
The PD-1 expression level in sorted CD8+
T cells was significantly increased in stages III and IV compared
with stages I and II of disease (Fig.
1C). By contrast, there were no marked changes between the
stages of disease in sorted CD4+ T cells (Fig. 1C). These results revealed that a major
feature of the immune microenvironment in lung cancer tissues was
the inhibition of CD8+ T cells rather than
CD4+ T cells, and this immunosuppression may be mediated
by the increased level of PD-1 expression on CD8+ T
cells.
As a key pro-inflammatory factor, PGE2 is highly
expressed in numerous types of solid tumors, including liver,
prostate and lung cancer, mediating tumor proliferation and
metastasis by secreting into the tumor microenvironment (7–9). To
clarify whether PGE2 secreted by lung cancer cells directly
affected PD-1 expression, the concentration of PGE2 in lung cancer
tissue homogenate was detected using the ELISA method in the
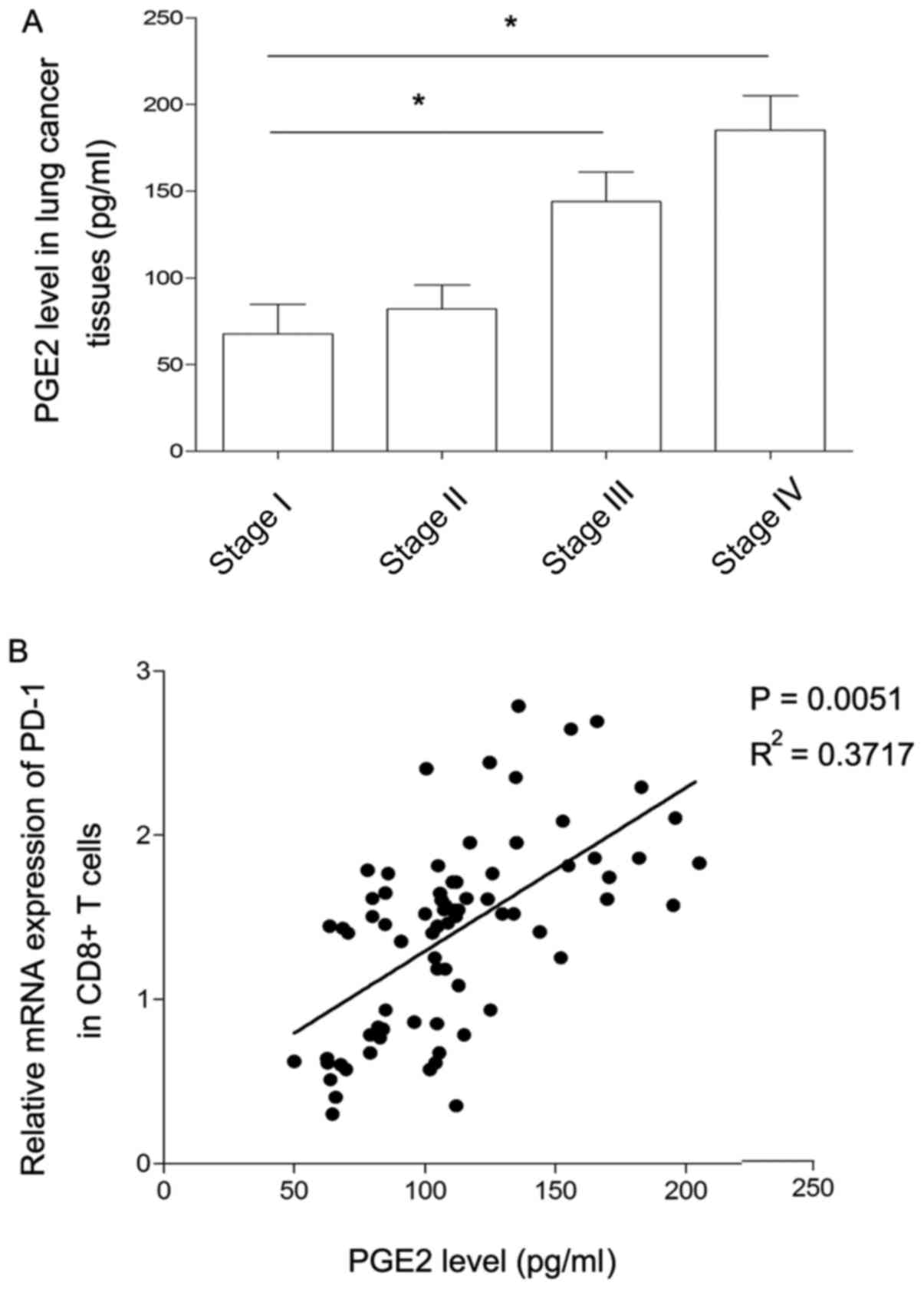
present study. As presented in Fig.
2A, the level of PGE2 expression was at similarly low levels at
stages I and II of disease, but expression was significantly
increased at stage III compared with stage I. Similarly, there was
a marked increase in the level of PGE2 expression at stage IV
compared with stage III. Considering that the pattern of changes in
the levels of PGE2 was similar to the changes observed in the level
of PD-1 expression in CD8+ T cells, the association
between PGE2 and PD-1 expression was further analyzed. As presented
in Fig. 2B, the level of PGE2
produced by lung cancer cells was positively correlated with the
level of PD-1 expression in CD8+ T cells. This finding
indicated that PGE2 may induce the inhibition of CD8+ T
cells by promoting the level of PD-1 in the lung cancer
microenvironment.
Expression level of PD-1 was regulated
by PGE2 via EP2- and EP4-associated signaling pathways
The cellular effects of PGE2 are mediated by a
family of G-protein-coupled receptors designated EP1, −2, −3 and
−4. Of the four EP receptor subtypes, EP1 and EP3 generally elicit
excitatory actions, whereas EP2 and EP4 elicit inhibitory actions
on cellular function (20–22).
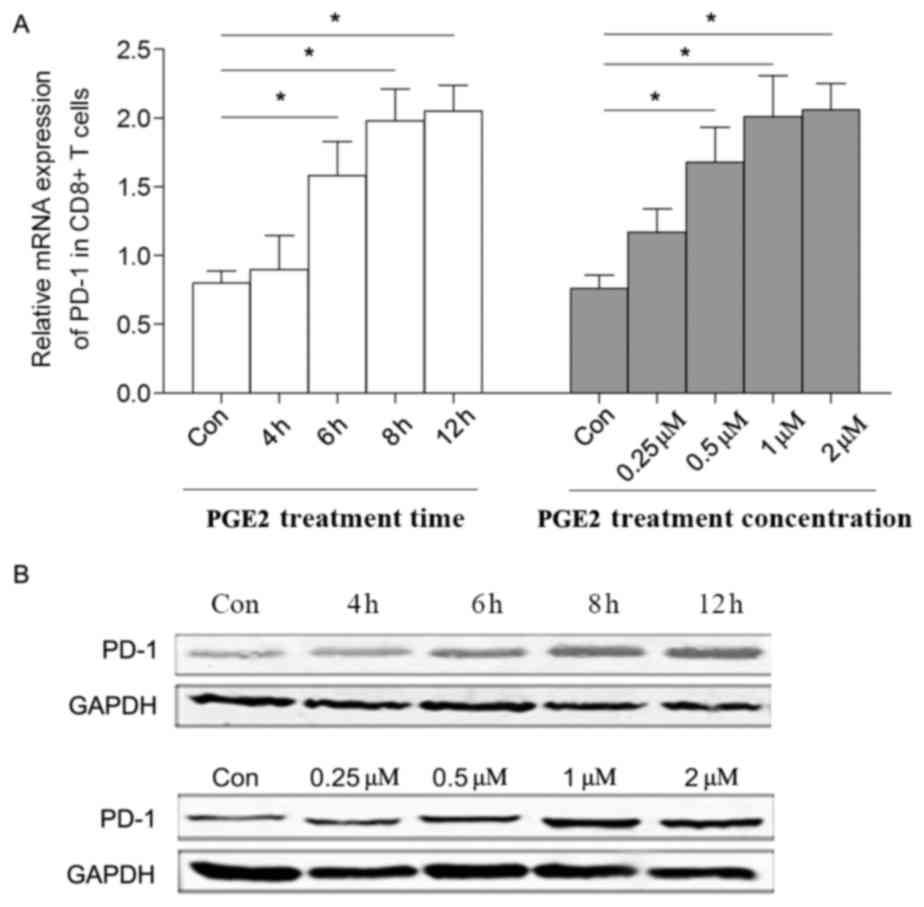
In the present study, sorted CD8+ T cells
were stimulated by PGE2 in vitro, and the level of PD-1
expression was demonstrated to be upregulated in a time- and
dose-dependent manner (Fig. 3A and
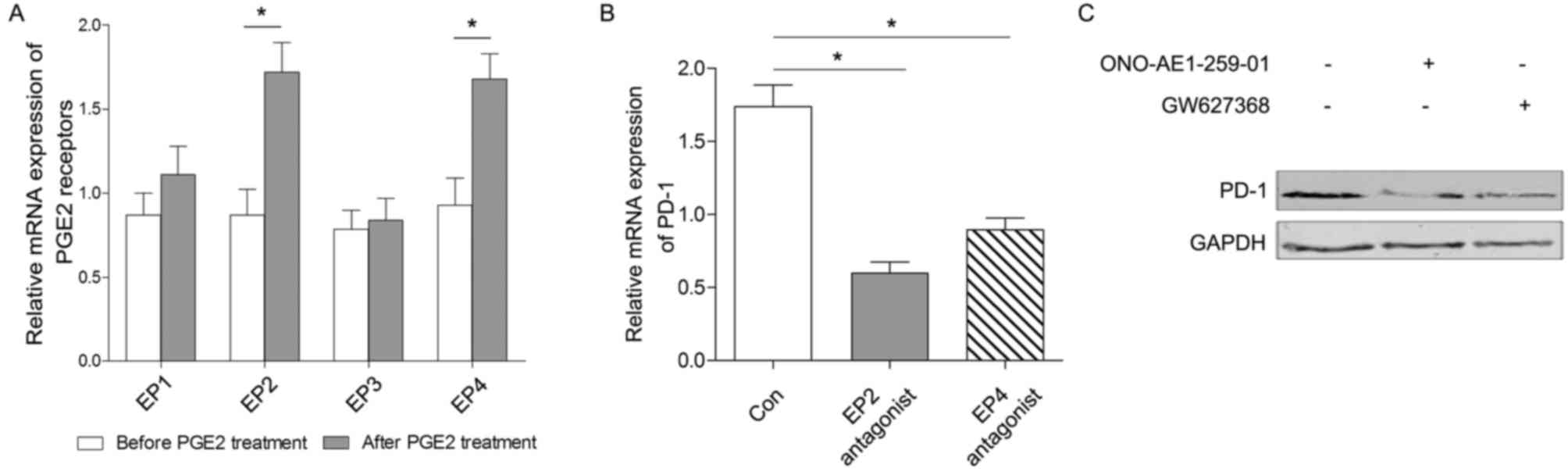
B). In the present study, it was indicated that the levels of
mRNA expression of EP2 and EP4 were all increased following PGE2
treatment, whereas there was no statically significant change in
the production of EP1 or EP3 following treatment with 0.5 µM PGE2
for 6 h (Fig. 4A).
To investigate the specific pathways through which
PGE2 mediates its effects, the medium was supplemented with EP2 and
EP4 antagonists for an additional 6 h to block the corresponding
signaling pathways. The results demonstrated that the level of PD-1
expression was closely associated with the PGE2/EP2 and PGE2/EP4
signaling pathways (Fig. 4B and C).
Therefore, the level of PD-1 expression in CD8+ T cells
may be regulated by PGE2 via the EP2 and EP4 signaling
pathways.
Notably, although there was no association between
PD-1 expression in CD4+ T cells and the level of PGE2,
it was indicated that the activation of PGE2 signaling may also
increase the level of PD-1 expression in CD4+ T cells
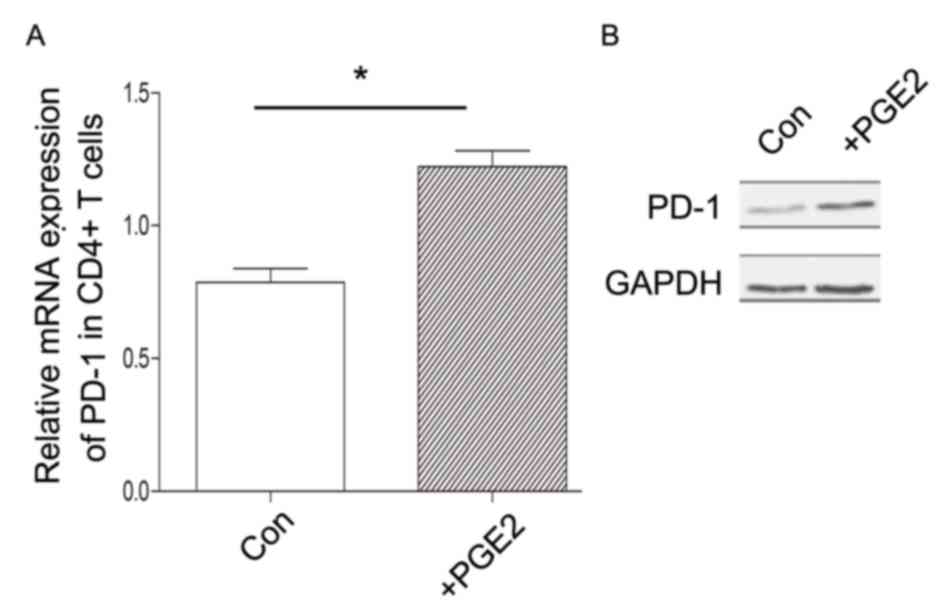
in vitro (Fig. 5). Therefore,
complex regulatory mechanisms for PD-1 expression may exist in
CD4+ T cells in vivo, which may involve other
cytokines or signaling transduction pathways.
Discussion
Cancer immunotherapy targeting the inflammatory
microenvironment is a promising strategy in numerous types of solid
tumors, including lung cancer. As a key factor widely expressed in
the immune system and malignant tumor cells, PD-1 facilitates an
inhibitory immune response. PD-L1 on lung cancer cells was revealed
to be able to increase the apoptosis of antigen-specific T cells
and to inhibit the activation of CD4+ and
CD8+ T cells via PD-1, resulting in reduced antitumor
immunity and evasion of host immune surveillance (23,24). In
the present study, the level of PD-1 expression in infiltrating
CD4+ and CD8+ T cells in lung cancer was
investigated. Although the percentage of CD8+ T cells
gradually decreased as the disease stage increased, there was an
increased ratio of PD-1 expression in this subset of T cells.
Considering the immunosuppressive signaling induced
by PD-1, it was hypothesized that the decreased percentage of
CD8+ T cells was potentially mediated by high PD-1
expression. Conversely, there were no marked changes in the
percentage of CD4+ T cells between different stages of
disease, which may be due to the presence of various
CD4+ T subsets which would lead to different and even
opposite immune effects. Regulatory T cells (CD4+
CD25+) and other Th2-type T cells always act as immune
inhibitors, whereas Th1-type T cells are able to enhance immune
responses (25,26). Therefore, the PD-1 level of each
different CD4+ T cell subset requires analyzing in order
to determine its association with lung cancer development in future
studies.
To investigate how lung cancer cells regulate the
level of PD-1 expression, the present study focused on the
inflammatory cytokine, PGE2, which is highly expressed in lung
cancer tissues (27,28). It was revealed that the level of PGE2
expression in the lung cancer tissue homogenates was positively
correlated with the level of PD-1 expression in CD8+ T
cells. To clarify the potential mechanism, PGE2-associated
signaling pathways were analyzed in the present study.
The signaling initiated from PGE2 is primarily
mediated by its four receptors, EP1, EP2, EP3 and EP4. The role of
EP1 is reported to be associated with intracellular calcium
concentration, and promoter analysis of the EP2 and EP4 genes
indicated the presence of several consensus sequences associated
with inflammatory stimuli, including interleukin-6, nuclear
factor-κB and activator protein 2 (29,30). A
previous study revealed that EP4 is able to mediate PGE2-induced
migration of A549 lung cancer cells (31). EP3 is distinct as it has multiple
isoforms generated by alternative mRNA splicing, and EP3 has also
been found to be crucial for tumor stroma formation and tumor
growth (32,33). The functions of various PGE2 receptors
in regulating the level of PD-1 expression were distinguished.
By treating the sorted T cells with PGE2 and the
antagonists of PGE2 receptors in vitro in the present study,
it was observed that the activation of EP2- and EP4-signaling was
able to promote the level of PD-1 expression. Treatment with PGE2
was able to markedly promote PD-1 expression in CD4+ and
CD8+ T cells in vitro. However, the level of PD-1
expression in CD4+ T cells was not correlated with the
concentration of PGE2 in the tissue homogenates. Since
CD4+ T cells contain multiple subsets with differential
functions, the present study considered that more complex
regulatory mechanisms may exist in order to regulate PD-1
expression in various CD4+ T subsets in vivo,
which may be associated with the synergistic effect between PGE2
and other cytokines, as well as the crosstalk between
CD4+ T cells and lung cancer cells via direct
contact.
In conclusion, the results of the present study
revealed that the level of PD-1 expression in the infiltrating
CD8+ T cells of patients with lung cancer at various
disease phases was positively regulated by PGE2 via the EP2- and
EP4-associated signaling pathways. More thorough studies should be
performed to reveal the characteristics of various T-cell subsets
in the tumor microenvironment in order to be able to reverse immune
tolerance and improve the immunotherapy of lung cancer.
Acknowledgements
The present study was supported by the Innovation
Project from the Department of Education of Guangdong (grant no.
2014KTSCX043), the Norman Bethune Program of Jilin University
(grant no. 2015328), and the Natural Science Foundation of
Guangdong (grant no. 2015A030313264).
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sun S, Schiller JH, Spinola M and Minna
JD: New molecularly targeted therapies for lung cancer. J Clin
Invest. 117:2740–2750. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rolfo C, Sortino G, Smits E, Passiglia F,
Bronte G, Castiglia M, Russo A, Santos ES, Janssens A, Pauwels P
and Raez L: Immunotherapy: Is a minor god yet in the pantheon of
treatments for lung cancer? Expert Rev Anticancer Ther.
14:1173–1187. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Fazilleau N, Mark L, McHeyzer-Willams LJ
and McHeyzer-Williams MG: Follicular helper T cells: Lineage and
location. Immunity. 30:324–335. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Inoue S, Leitner WW, Golding B and Scott
D: Inhibitory effects of B cells on antitumor immunity. Cancer Res.
66:7741–7747. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lee-Chang C, Bodogai M, Martin-Montalvo A,
Wejksza K, Sanghvi M, Moaddel R, de Cabo R and Biragyn A:
Inhibition of breast cancer metastasis by resveratrol-mediated
inactivation of tumor-evoked regulatory B cells. J Immunol.
191:4141–4151. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
O'Callaghan G and Houston A: Prostaglandin
E2 and the EP receptors in malignancy: Possible therapeutic
targets? Br J Pharmacol. 172:5239–5250. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pai R, Soreghan B, Szabo IL, Pavelka M,
Baatar D and Tarnawski AS: Prostaglandin E2 transactivates EGF
receptor: A novel mechanism for promoting colon cancer growth and
gastrointestinal hypertrophy. Nat Med. 8:289–293. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Baratelli F, Lin Y, Zhu L, Yang SC,
Heuzé-Vourc'h N, Zeng G, Reckamp K, Dohadwala M, Sharma S and
Dubinett SM: Prostaglandin E2 induces FOXP3 gene expression and T
regulatory cell function in human CD4+ T cells. J Immunol.
175:1483–1490. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chemnitz JM, Parry RV, Nichols KE, June CH
and Riley JL: SHP-1 and SHP-2 associate with immunoreceptor
tyrosine-based switch motif of programmed death 1 upon primary
human T cell stimulation, but only receptor ligation prevents T
cell activation. J Immunol. 173:945–954. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Park JJ, Omiya R, Matsumura Y, Sakoda Y,
Kuramasu A, Augustine MM, Yao S, Tsushima F, Narazaki H, Anand S,
et al: B7-H1/CD80 interaction is required for the induction and
maintenance of peripheral T-cell tolerance. Blood. 116:1291–1298.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dong H, Strome SE, Salomao DR, Tamura H,
Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, et al:
Tumor-associated B7-H1 promotes T-cell apoptosis: A potential
mechanism of immune evasion. Nat Med. 8:793–800. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen JH, Perry CJ, Tsui YC, Staron MM,
Parish IA, Dominguez CX, Rosenberg DW and Kaech SM: Prostaglandin
E2 and programmed cell death 1 signaling coordinately impair CTL
function and survival during chronic viral infection. Nat Med.
21:327–334. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Rami-Porta R, Bolejack V, Giroux DJ,
Chansky K, Crowley J, Asamura H and Goldstraw P; International
Association for the Study of Lung Cancer Staging and Prognostic
Factors Committee, Advisory Board Members and Participating
Institutions, : The IASLC lung cancer staging project: The new
database to inform the eighth edition of the TNM classification of
lung cancer. J Thorac Oncol. 9:1618–1624. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Garnelo M, Tan A, Her Z, Yeong J, Lim CJ,
Chen J, Lim KH, Weber A, Chow P, Chung A, et al: Interaction
between tumour-infiltrating B cells and T cells controls the
progression of hepatocellular carcinoma. Gut. 66:342–351. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pagès F, Galon J, Dieu-Nosjean MC, Tartour
E, Sautès-Fridman C and Fridman WH: Immune infiltration in human
tumors: A prognostic factor that should not be ignored. Oncogene.
29:1093–1102. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lin Z, Chen X, Li Z, Luo Y, Fang Z, Xu B
and Han M: PD-1 antibody monotherapy for malignant melanoma: A
systematic review and meta-analysis. PLoS One. 11:e01604852016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang X, Bao Z, Zhang X, Li F, Lai T, Cao
C, Chen Z, Li W, Shen H and Ying S: Effectiveness and safety of
PD-1/PD-L1 inhibitors in the treatment of solid tumors: A
systematic review and meta-analysis. Oncotarget. 8:59901–59914.
2017.PubMed/NCBI
|
|
20
|
Yang L, Yamagata N, Yadav R, Brandon S,
Courtney RL, Morrow JD, Shyr Y, Boothby M, Joyce S, Carbone DP and
Breyer RM: Cancer-associated immunodeficiency and dendritic cell
abnormalities mediated by the prostaglandin EP2 receptor. J Clin
Invest. 111:727–735. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nataraj C, Thomas DW, Tilley SL, Nguyen
MT, Mannon R, Koller BH and Coffman TM: Receptors for prostaglandin
E(2) that regulate cellular immune responses in the mouse. J Clin
Invest. 108:1229–1235. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
von der Emde L, Goltz D, Latz S, Müller
SC, Kristiansen G, Ellinger J and Syring I: Prostaglandin receptors
EP1-4 as a potential marker for clinical outcome in urothelial
bladder cancer. Am J Cancer Res. 4:952–962. 2014.PubMed/NCBI
|
|
23
|
Han L, Liu F, Li R, Li Z, Chen X, Zhou Z,
Zhang X, Hu T, Zhang Y, Young K, et al: Role of programmed death
ligands in effective T-cell interactions in extranodal natural
killer/T-cell lymphoma. Oncol Lett. 8:1461–1469. 2014.PubMed/NCBI
|
|
24
|
Zitvogel L and Kroemer G: Targeting
PD-1/PD-L1 interactions for cancer immunotherapy. Oncoimmunology.
1:1223–1225. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sakaguchi S, Vignali DA, Rudensky AY, Niec
RE and Waldmann H: The plasticity and stability of regulatory T
cells. Nat Rev Immunol. 13:461–467. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yun X, Shang Y and Li M: Effect of
Lactobacillus salivarius on Th1/Th2 cytokines and the number of
spleen CD4+ CD25+ Foxp3+ Treg in asthma Balb/c mouse. Int J Clin
Exp Pathol. 8:7661–7674. 2015.PubMed/NCBI
|
|
27
|
Che D, Zhang S, Jing Z, Shang L, Jin S,
Liu F, Shen J, Li Y, Hu J, Meng Q, et al: Macrophages induce EMT to
promote invasion of lung cancer cells through the IL-6-mediated
COX-2/PGE2/β-catenin signalling pathway. Mol Immunol. 90:197–210.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fan Y, Wang Y and Wang K: Prostaglandin E2
stimulates normal bronchial epithelial cell growth through
induction of c-Jun and PDK1, a kinase implicated in oncogenesis.
Respir Res. 16:1492015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Quan Y, Jiang J and Dingledine R: EP2
receptor signaling pathways regulate classical activation of
microglia. J Biol Chem. 288:9293–9302. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cho JS, Han IH, Lee HR and Lee HM:
Prostaglandin E2 Induces IL-6 and IL-8 Production by the EP
Receptors/Akt/NF-κB pathways in nasal polyp-derived fibroblasts.
Allergy Asthma Immunol Res. 6:449–457. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kim JI, Lakshmikanthan V, Frilot N and
Daaka Y: Prostaglandin E2 promotes lung cancer cell migration via
EP4-betaArrestin1-c-Src signalsome. Mol Cancer Res. 8:569–577.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Israel DD and Regan JW: EP(3) prostanoid
receptor isoforms utilize distinct mechanisms to regulate ERK 1/2
activation. Biochim Biophys Acta. 1791:238–245. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Amano H, Hayashi I, Endo H, Kitasato H,
Yamashina S, Maruyama T, Kobayashi M, Satoh K, Narita M, Sugimoto
Y, et al: Host prostaglandin E(2)-EP3 signaling regulates
tumor-associated angiogenesis and tumor growth. J Exp Med.
197:221–232. 2003. View Article : Google Scholar : PubMed/NCBI
|