Introduction
The CDw75 epitope, also referred as CD75s (1) or CD75 (2),
is a B/T-cell sialylated antigen located at the terminal position
of poly-N-acetyl-lactosamine residues (3). The sequence of this glycosylated motif
is Neu5Acα(2,6)Galβ(1,4)GlcNAc, and
it is a part of the glucidic moiety of N- and O-glycans,
particularly of N-glycoproteins (4).
The final step of its synthesis is catalyzed by the enzyme
β-galactoside α(2,6) sialyltransferase (ST6Gal I; EC 2.4.99.1)
(4).
The CDw75 antigen is a ligand for the leukocyte
adhesion receptor cluster of differentiation (CD)22, whose
activation leads to the inhibition of intracellular signaling
pathways (5). CD22 is located on the
B cell membrane and specifically recognizes α(2,6)-linked
sialic acid (6,7). Given that ST6Gal I knockout mice show
low serum B cell and immunoglobulin (Ig)M levels (8), CDw75 must have an important role in B
cell proliferation and activation.
Although CDw75 was initially discovered in the B
lymphocyte cell membrane, where its expression stops when
differentiating into plasma cells (9), several reports have studied the
expression of the CDw75 epitope in various epithelial carcinomas,
including breast (10), stomach
(11) or pancreatic cancer (12).
Colorectal cancer (CRC) is one of the leading types
of cancer worldwide, particularly in developed countries. In CRC,
ST6Gal I activity affects the adherence (13) and invasion (14) ability of tumor cells. ST6Gal I
activity is also associated with the acquisition of the neoplastic
phenotype (15), as well as the
resistance to irinotecan (16) and
Fas-mediated apoptosis (17).
Furthermore, in CRC patients, ST6Gal I activity is greater in tumor
than in healthy tissue (18,19), particularly in metastatic tumors
(20). The CDw75 antigen is expressed
in the majority of colorectal tumors, where its expression is
augmented with regard to the healthy mucosa (21,22).
Additionally, CDw75 expression is particularly high in non-polypoid
tumors and advanced or metastatic stages of the disease, although
results from clinicopathological studies suggest it could represent
a good prognosis marker for 3-year overall survival (23). However, the expression of CDw75 in
tumor tissue from CRC patients showed no prognostic potential for
predicting disease-free survival (23).
Taking into account all the knowledge here presented
that link the expression of the CDw75 antigen to CRC development,
the authors considered that some of the glycoproteins that bear
this epitope could be implicated in the molecular pathogenesis and
progression of the disease and therefore, might be used as
biomarkers or even as therapeutic targets for this neoplasia.
Therefore, the aim of the present study was to identify proteins
with the CDw75 epitope in human colorectal tissue.
Materials and methods
Patients, tissue specimens and cell
lines
Colorectal tissue specimens from 26 CRC patients,
who underwent surgery at the University Hospital Complex of Ourense
(CHUO, Ourense, Spain) between April 2003 and October 2010 were
included in the present study. The 26 patients were employed in the
following way: 9 patients for the identification of proteins with
the CDw75 epitope (mean age 71.9±5.2 years; age range, 65–78 years)
and 23 patients for the verification of these proteins (mean age
74.8±9.1 years; age range, 57–91 years). A total of 6 patients were
used in both assays. Apart from the tumor specimens, paired healthy
tissues, resected ≥10 cm away from the primary carcinoma, were also
employed. Samples were obtained during surgical resection of the
tumors and were immediately stored at −80°C until use. The present
study received the approval from the Ethical Committee of Clinical
Research of Galicia (Health Department, Xunta de Galicia, Spain),
and clinical information of patients was treated anonymously. All
procedures performed in studies involving human participants were
in accordance with the ethical standards of the institutional
and/or national research committee and with the 1964 Helsinki
declaration and its later amendments or comparable ethical
standards.
For the purpose of this work, two CRC cell lines
were also employed: Caco-2 [American Type Culture Collection
(ATCC), HTB-37] and HT-29 (ATCC, HTB-38), who were donated by Dr.
Craig Murdoch (School of Clinical Dentistry, University of
Sheffield, UK). Caco-2 cells were cultured in Dulbecco's modified
Eagle's medium (Thermo Fisher Scientific, Inc., Waltham, MA, USA)
supplemented with 10% fetal bovine serum (Sigma-Aldrich; Merck
KGaA, Darmstadt, Germany) and 1% penicillin/streptomycin
(Sigma-Aldrich; Merck KGaA), whereas HT-29 cells were grown in
McCoy's 5A medium (Sigma-Aldrich; Merck KGaA) supplemented with 10%
fetal bovine serum, 1% penicillin/streptomycin and 2 mM L-glutamine
(Sigma-Aldrich; Merck KGaA). The two cell lines were maintained at
37°C and 5% CO2, and harvested when the cells reached
confluence. Caco-2 cells were also left for 15 days following
confluence to spontaneously differentiate, and the medium was
changed daily.
Sample processing
Tissue specimens were subjected to a modified
version of a previously published protocol (24), with the goal of obtaining subcellular
fractions enriched in membrane-associated proteins or in cytosolic
species. All procedures were performed on ice bath or alternatively
in thermostatic centrifuges. Briefly, the biopsies were
mechanically homogenized in 0.01 M Tris-HCl buffer (pH 7.4) with
0.25 M sucrose and a protease inhibitor cocktail (Complete Mini;
Roche Diagnostics, Basel, Switzerland). Non-homogenized material
was removed by centrifugation at 750 × g for 10 min at 4°C. The
resulting supernatant was subsequently centrifuged at 33,000 × g
for 60 min at 4°C. In this way, the total cellular membranes were
pelleted, and the supernatant contained the crude cytosolic
fraction. The two fractions were further centrifuged at 145,000 × g
for 45 min at 4°C to eliminate the sucrose. Lastly, final pellets
were resuspended in 0.01 M Tris-HCl buffer (pH 7.4) and kept at
−20°C.
Caco-2 and HT-29 cell lysates were obtained from 75
cm2 flasks at confluence in 500 µl RIPA buffer (0.1%
SDS, 150 mM NaCl, 50 mM Tris-HCl (pH 8.5), 0.5% deoxycholic acid,
1% NP-40, 2 mM Na3VO4 and 4 mM NaF)
supplemented with a protease inhibitor cocktail (Complete Mini;
Roche Diagnostics). The resultant cell-free homogenate was
incubated on ice for 15 min and centrifuged at 9,600 × g for 10 min
at 4°C to discard cell debris. The subsequent supernatant was
preserved at −20°C. Finally, total protein measurements were
performed using the bicinchoninic acid method, following
manufacturer's instructions (Sigma-Aldrich; Merck KGaA).
Two-dimensional western blotting
Given that the CDw75 epitope has been described as a
membrane-associated antigen and alterations in the cell membrane
glycoconjugates is a well-known tumor characteristic (25), only the preparations enriched in
membrane-associated proteins were used for two-dimensional (2D)
western blotting. Prior to 2D electrophoresis, the samples were
subjected to a desalting step by ultrafiltration through 10 kDa
molecular weight cut-off Amicon© Ultra-4 centrifugal
filter units (Sigma-Aldrich; Merck KGaA), three times at 2,700 × g
for 30 min at 4°C. The final retentate (~25 µl) was resuspended in
a solubilization buffer composed of 7 M urea, 2 M thiourea and 4%
3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonate hydrate
(CHAPS) to a final volume of 300 µl. The protein concentration of
the resulting solution was calculated by a modified Bradford assay
as previously described (26).
Each experiment consisted of two 2D separations
performed at the same time. One gel was involved protein
identification by mass spectrometry (MS), whereas the other was
used for CDw75 immunodetection by western blotting (WB). In the
first dimension, proteins (100 for MS gels or 30 µg for WB gels)
were subjected to isoelectric focusing (pH, 4.7–5.9) using
ReadyStrip™ IPG strips (Bio-Rad Laboratories, Inc., Hercules, CA,
USA). Afterwards, the strips were washed consecutively in a
solution composed of 6 M urea, 50 mM Tris (pH 8.8), 2% SDS and 30%
glycerol and supplemented with 1% dithiothreitol or 2.5%
iodoacetamide for protein reduction or alkylation. The second
dimension consisted of a denaturing separation in 10% SDS-PAGE
gels.
Following 2D electrophoresis, the proteins were
transferred for 80 min at 120 V to polyvinylidene difluoride (PVDF)
membranes (Hybond P 0.45; GE Healthcare, Chicago, IL, USA). The
membranes were blocked with 5% non-fat dry milk in PBS for 1 h at
room temperature and then incubated with an anti-CDw75 monoclonal
antibody (1:1,250; catalog no. MA5-11900; Thermo Fisher Scientific,
Inc., Waltham, MA, USA) overnight at 4°C.
Following several washes with PBS, the membranes
were incubated with an alkaline phosphatase-conjugated secondary
antibody for 1 h (1:15,000, catalog no. A9688; Sigma-Aldrich; Merck
KGaA). Finally, the membranes were incubated in darkness with
5-bromo-4-chloro-3-indolyl-phosphate/nitro blue tetrazolium
(Sigma-Aldrich; Merck KGaA) until CDw75 spots appeared. All
incubations were performed at room temperature.
One-dimensional western blotting
A standard one-dimensional (1D) WB workflow was also
employed. Briefly, 20 µg protein from crude cytosol- and
membranes-enriched fractions were separated in 10% SDS-PAGE gels,
transferred to PVDF membranes, blocked with 5% non-fat dry milk in
PBS for 1 h at room temperature and probed for several
antigens/proteins with monoclonal antibodies: Anti-CDw75 (the same
as aforementioned), anti-Hp (1:500; catalog no. ab13429, Abcam,
Cambridge, UK), anti-K8 (1:1,250, catalog no. sc-8020, Santa Cruz
Biotechnology, Inc., Dallas, TX, USA) and anti-K18 (1:1,250,
catalog no. sc-51582, Santa Cruz Biotechnology, Inc.). The
membranes were incubated with the primary antibodies for 1 h. After
several washes with PBS, CDw75-labelled membranes were incubated
with the same secondary antibody used in 2D WB, whereas Hp, K8 and
K18 membranes were incubated with a goat anti-mouse IgG horseradish
peroxidase-conjugated antibody for 1 h (1:15,000, catalog no.
ab97040; Abcam). All antibody incubations were performed at room
temperature.
The development of the membranes was either
chromogenic (for CDw75 detection) or chemiluminescent (for
detection of Hp, K8 and K18). CDw75-labelled membranes were handled
as aforementioned, whereas Hp, K8 and K18 membranes were incubated
with chemiluminescent reagents according to manufacturer's
instructions (Clarity™ Western ECL Substrate, Bio-Rad Laboratories,
Inc.), and visualized using a ChemiDoc™ XRS+system (Bio-Rad
Laboratories, Inc.). With the aim to reuse the membranes for the
detection of other epitopes, in some occasions the membranes were
stripped with a solution (pH 2.2) of 200 mM glycine, 0.1% SDS and
1% Tween-20 (twice for 10 min).
Silver staining, spot selection and
protein identification
2D gels were silver-stained for spot visualization.
For that purpose, the gels were fixed overnight in 40% ethanol and
10% acetic acid, sensitized in 30% ethanol, 6.8% sodium acetate and
0.3% sodium thiosulfate for 30 min, impregnated in 0.1% silver
nitrate for 20 min, developed with 3% sodium carbonate and 0.01%
formaldehyde, and stopped in 1.5% EDTA for 10 min.
Spots containing CDw75-carrying proteins were
selected after the matching of the paired gel and membrane images
with the assistance of the 2D-gel imaging and analysis PDQuest
software (version 7.4.0; Bio-Rad Laboratories, Inc.). The selected
spots were sent for MS analysis to the Scientific and Technical
Support Centre for Research (CACTI, University of Vigo, Spain).
Proteins were digested in gel with trypsin using a
DP Chemical 96 kit (Bruker Corporation, Billerica, MA, USA).
Tryptic peptides were analyzed by matrix-assisted laser
desorption/ionization-time of flight tandem mass spectrometry
(MALDI-TOF/TOF) with an Autoflex III smartbeam (Bruker
Corporation). The data was acquired and analyzed automatically
using the flexControl 3.0 and flexAnalysis 3.0 software (Bruker
Corporation), respectively. When possible, the major peaks obtained
by MALDI-TOF were selected to be further characterized by TOF/TOF
analyses. m/z data were submitted for database searching
against SwissProt 56.6 (http://web.expasy.org/docs/swiss-prot_guideline.html).
Search parameters were set as follows: Taxonomy Homo
sapiens, enzyme trypsin, allowance of one missed cleavage site,
carbamidomethyl of cystein as fixed modification, oxidation of
methionine as variable modification, monoisotopic mass values, 100
ppm of mass tolerance for precursor ions, 0.5 Da mass tolerance for
fragment ions and protein mass unrestricted.
For greater certainty, protein identifications were
carefully studied to select the candidates that, in addition to
being statistically significant, i) had similar molecular masses
and isoelectric points compared with those of the spots in which
they were identified; ii) were expressed in colorectal tissue and
iii) were known to be glycoproteins. For this step, the following
databases were used: UniProt (http://www.uniprot.org/), Human Protein Reference
Database (http://www.hprd.org/query), The Human
Protein Atlas (http://www.proteinatlas.org/), Ensembl (http://www.ensembl.org/index.html) and
SWISS-2DPAGE (http://www.expasy.org/swiss-2dpage?de).
Statistical analysis
All statistical analyses were performed with the IBM
SPSS Statistics software (version, 19.0; IBM Corp., Armonk, NY,
USA). In order to compare the expression of the assayed proteins
among different tissues and subcellular fractions, the Wilcoxon
signed-rank test was employed. To find out whether the expression
of the proteins was correlated with the expression of the CDw75
antigen, the Pearson's correlation test was used. P<0.05 was
considered to indicate a statistically significant difference.
Results
Identification of glycoproteins that
contain the CDw75 sequence
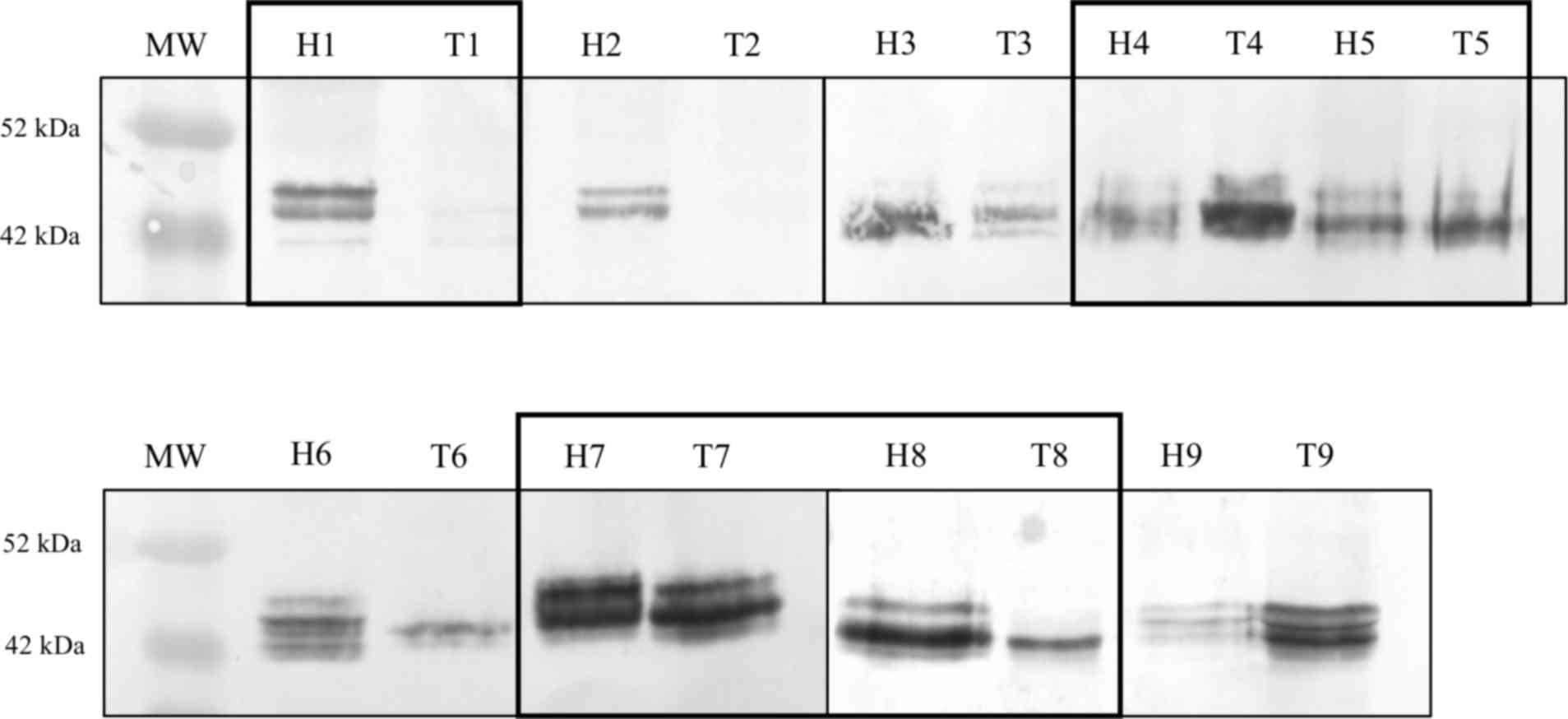
Prior to the 2D protein separation, 9 CRC patients
were screened by 1D WB in order to select samples with marked CDw75
expression and/or notable differences between healthy and tumor
tissue (Fig. 1). According to these
criteria, 5 patients (patients 1, 4, 5, 7 and 8) were selected for
the separation, selection and identification of proteins with the
CDw75 epitope. In 2D CDw75 immunoblots, the epitope appeared as 4–8
positive spots located on a narrow area of the membranes, between
44–50 kDa in molecular mass and 4.9–5.1 of isoelectric point
(Fig. 2). Differences in CDw75 spot
pattern between healthy and tumor tissue or association with tumor
stage were not detected. A summary of the MS findings is shown in
Table I.
 | Table I.Summary of the results from the 2-D
electrophoresis separation and mass spectrometry identification of
CDw75-carrying proteins in colorectal tissue from colorectal cancer
patients. |
Table I.
Summary of the results from the 2-D
electrophoresis separation and mass spectrometry identification of
CDw75-carrying proteins in colorectal tissue from colorectal cancer
patients.
| Sample | Tissue | Protein | UniProt accession
number | Spots MS | Spots MS/MS |
|---|
| 1 | Healthy | Keratin 1 | P04264 | 1 | – |
|
|
| Keratin 8 | P05787 | 4 | 1 |
|
|
| Keratin 18 | P05783 | 12 | – |
|
|
| Keratin 19 | P08727 | 1 | – |
|
|
| Haptoglobin | P00738 | 4 | 2 |
|
|
| Seroalbumin | P02768 | 1 | – |
|
|
| Actin |
| 3 | 3 |
|
|
| ATP synthase
subunit β | P06576 | 1 | – |
| 4 | Healthy | Keratin 8 | P05787 | 1 | – |
|
|
| Keratin 18 | P05783 | 2 | – |
|
| Tumor | Actin | – | 1 | 4 |
|
|
| Seroalbumin | P02768 | 1 | – |
|
|
| Cytochrome b-c1
complex subunit 1, mitochondrial | P31930 | – | 1 |
|
|
| Keratin 18 | P05783 | 1 | 1 |
| 5 | Healthy | Keratin 8 | P05787 | 2 | – |
|
| Tumor | Keratin 8 | P05787 | 1 | – |
|
|
| Keratin 19 | P08727 | 3 | 1 |
|
|
| LRMP | Q12912 | – | 1 |
| 7 | Healthy | Keratin 18 | P05783 | 2 | – |
|
|
| Keratin 19 | P08727 | 2 | – |
|
|
| RPSA | P08865 | 1 | 1 |
|
|
| GRP-78 | P11021 | 1 | – |
|
| Tumor | RPSA | P08865 | 2 | 2 |
|
|
| Keratin 8 | P05787 | – | 1 |
| 8 | Healthy | Keratin 1 | P04264 | 2 | – |
|
|
| Keratin 9 | P35527 | 1 | – |
|
|
| HSP70-1A | P0DMV8 | 1 | – |
|
| Tumor | Actin | – | 1 | 1 |
|
|
| ERp44 | Q9BS26 | 1 | 1 |
|
|
| Haptoglobin | P00738 | – | 1 |
Following careful consideration of the candidates
included in Table I, only three
proteins (Hp, K8 and K18) were found to fulfill all the
requirements established for selection: Correspondence in molecular
mass and isoelectric point with CDw75-positive spots, as well as
known colorectal expression and glycoprotein structure.
Verification of proteins with the
CDw75 epitope
Several 1D WB assays were performed to check whether
the CDw75 expression pattern was coincident with the bands
generated by Hp, K8 and K18 immunostaining. For this purpose,
detection of each candidate protein in membrane- and
cytoplasmic-enriched fractions from 23 CRC patients was performed.
The membranes were then stripped and assayed for the presence of
CDw75. The exact number of specimens employed for each assay were
as follows: 5 for Hp, 18 for K8 and 17 for K18. The majority of the
specimens were analyzed for more than one protein.
The Hp signal appeared as a single and blurred 40–45
kDa band, whereas the expression of CDw75 obtained in the same
patients was heterogeneous, ranging from null to four intense 42–47
kDa bands (Fig. 3A). The expression
of Hp and CDw75 was more abundant in the cytosol compared with the
membrane fraction (P=0.043 for Hp in healthy and tumor tissue; not
significant for CDw75). The expression of both molecules was
significantly correlated in the membrane fraction from healthy
tissue (n=5, P=0.018).
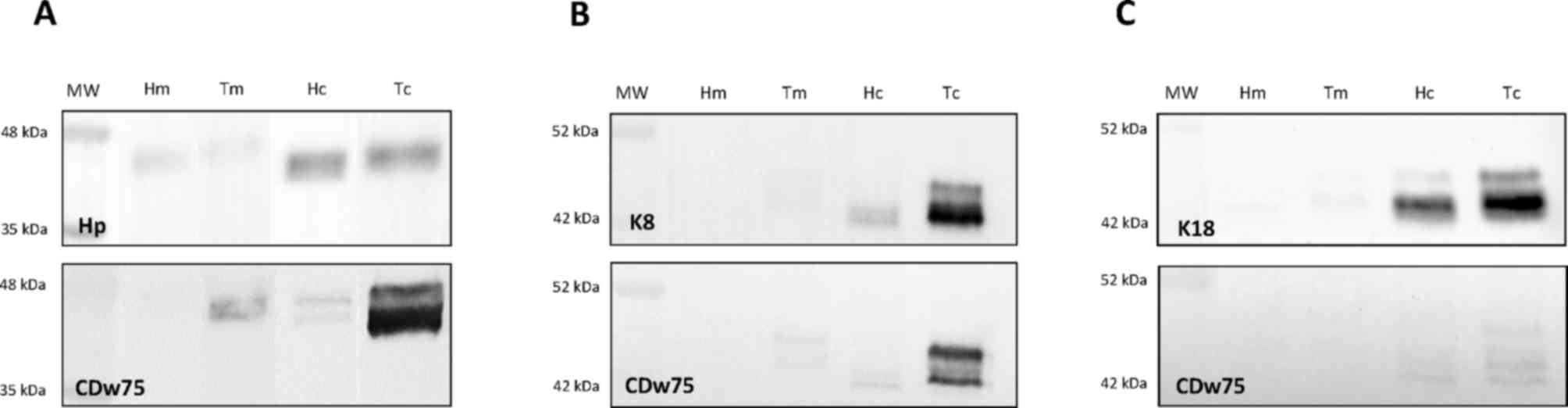 | Figure 3.Representative immunoblot images for
the expression of (A) haptoglobin, (B) keratin 8 and (C) keratin 18
in crude cytosolic- and total membranes-enriched fractions and
comparison with their corresponding immunoblots for the expression
of CDw75 (lower panels). A total of 23 colorectal tissue specimens
(5 for haptoglobin, 18 for keratin 8 and 17 for keratin 18) were
analyzed in this assay, of which only 3 are portrayed in this
figure. MW, molecular mass marker; Hc, cytoplasmic fraction from
healthy tissue; Hm, membrane-enriched fraction from healthy tissue;
Tc, cytoplasmic fraction from tumor tissue; Tm, membrane-enriched
fraction from tumor tissue; Hp, haptoglobin; K8, keratin 8; K18,
keratin 18. |
The expression of K8 was detected as two or three
bands, ranging from 42 to 47 kDa, and showed a considerable
heterogeneity. The CDw75 signal in the same patients was also
diverse, with up to four bands in the range of 42–46 kDa (Fig. 3B). The expression of K8 and CDw75 was
more intense in the cytoplasmic compared with in the membrane
fraction (only in healthy tissue, P=0.043 for K8 and P=0.042 for
CDw75). The expression level of both molecules was correlated in
the cytoplasmic fractions from healthy (n=18, P=0.002) and tumor
(P=0.050) tissue.
The expression of K18 also displayed a substantial
heterogeneity, with zero to three bands ranging from 43 to 47 kDa
(Fig. 3C). A similar pattern was
observed for CDw75, with up to four bands with 42–46 kDa that
exhibited a great variability depending on the patient. The
expression of K18 and CDw75 was more intense in the cytoplasmic
compared with in the membrane fraction (only in healthy tissue,
P=0.043 for K18 and P=0.042 for CDw75). The expression level of
both molecules was significantly correlated in the cytoplasmic
fraction from healthy tissue (n=17, P=0.019).
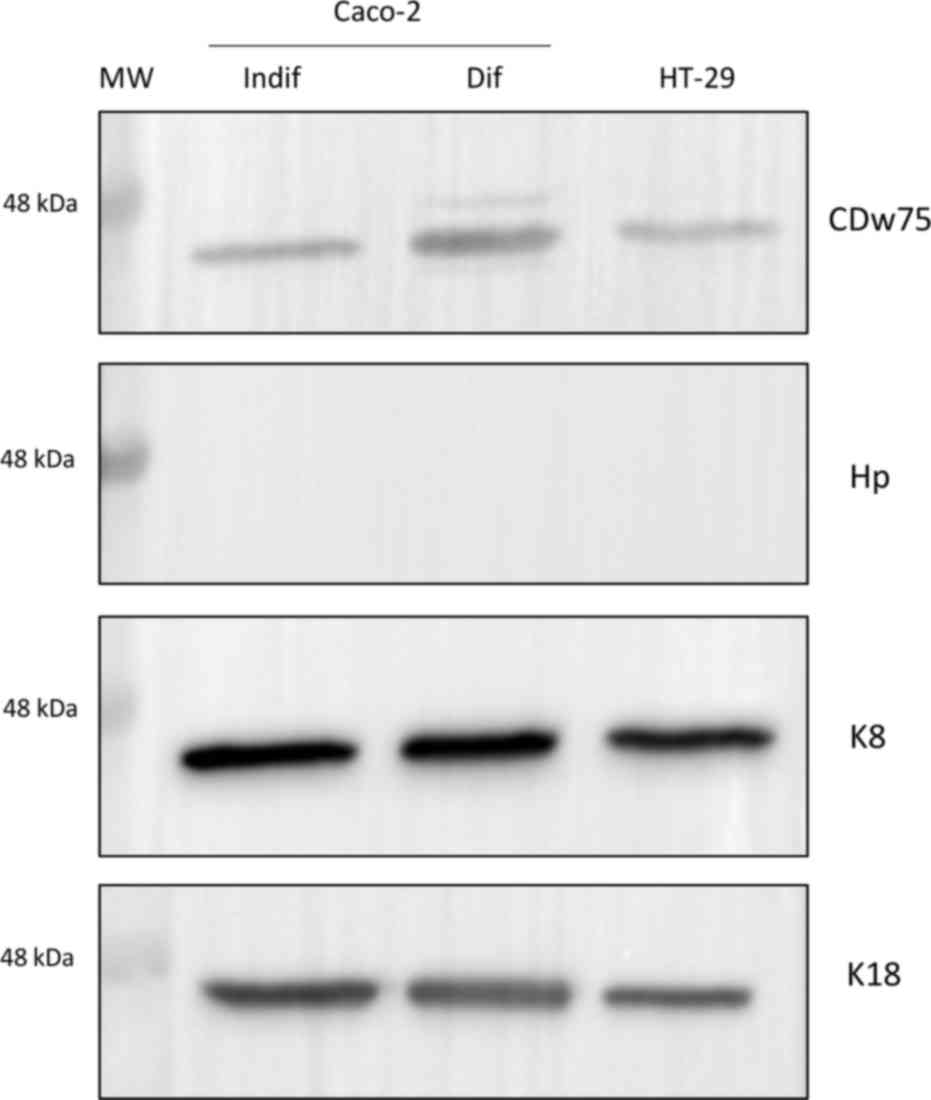
To complete the study of colorectal tissue from CRC
patients, Caco-2 and HT-29 cell lysates were also screened with the
same experimental strategy (Fig. 4).
In the WB assay performed, the CDw75 epitope appeared as a 46 kDa
band, accompanied by a 48 kDa band in differentiated Caco-2 cells.
Hp was not detected in any of the two replicates performed, whereas
K8 and K18 were detected as a single 46 kDa band in HT-29 cells and
in both differentiation states of Caco-2 cells.
Discussion
CDw75 is an α(2,6) sialylated
antigen abnormally expressed in different epithelial tumors.
Although this epitope is more abundant in the tumor tissue from CRC
patients and its expression levels seem to correlate with the
development of the neoplasia, the fact that most colorectal tumors
express the molecule implies that CDw75 expression has a poor
prognostic value (23). Additionally,
it is unknown if the greater expression of the epitope observed in
tumor tissue is a genuine molecular characteristic of CRC or just
an epiphenomenon accompanying the malignization of the colorectal
mucosa and, therefore, a poor discriminative marker. If the former
assumption is true, the identification of overexpressed proteins
with CDw75 would contribute to the clarification of the
pathogenesis of CRC. Consequently, the objective of the present
study was to identify the protein species that have the CDw75 motif
in human CRC.
Several different candidates were initially
identified by MS (Table I) in spots
that were selected by its coincidence in molecular mass and
isoelectric point with CDw75-positive spots in 2D WB. However, the
present study considered that a spatial coincidence was
insufficient to state conclusively that these proteins have the
CDw75 epitope. Therefore, each of these proteins was searched in
the aforementioned databases and thus discarded or accepted as
potential CDw75 bearers in CRC.
Keratins 1 and 9 are characteristically expressed in
the skin epithelium. Thus, the presence of these proteins in
samples other than the skin is usually regarded as a sign of
experimental contamination (27).
Likewise, the detection of seroalbumin is most likely due to
vascular infiltration of the tissue specimens.
Keratin 19, actins, lymphoid-restricted membrane
protein (LRMP), 40S ribosomal protein SA (RPSA), 78 kDa
glucose-regulated protein (GRP-78), heat shock 70 kDa protein 1A
(HSP70-1A) and endoplasmic reticulum resident protein 44 (ERp44).
It was not possible to determine the isoform(s) of actin present.
All these proteins are known to be expressed in the colorectal
epithelium (28–33), but to date there is no reported
evidence of their glycoprotein nature. Therefore, even though some
of the proteins have been associated with CRC and other types of
neoplasia, including keratin 19 (28), LRMP (29), RPSA (30), GRP-78 (31) and ERp44 (33), these proteins were not further
analyzed in subsequent studies.
The ATP synthase subunit β (34) and the cytochrome b-c1 complex subunit
1 (35) are mitochondrial proteins
associated with ATP production. Consequently, both proteins
(36,37) are virtually expressed in all tissues
and cells, which probably would make them very unspecific markers.
In addition, the two proteins are not known to be glycosylated.
K8 and K18 are well-known single-layered epithelia
cytoskeletal proteins (38), which
may explain the presence of these proteins in colorectal specimens.
Although a total membrane-associated protein fraction was employed,
the interaction of K8 and K18 with desmosomes and hemidesmosomes
(K8 and K18 constitute the intermediate filaments) (39) could explain the presence of these
cytoplasmic proteins in membrane preparations. Furthermore, K8 and
K18 are known to be glycosylated (40) and were identified in five (K8) and
four (K18) of the specimens analyzed in the present study.
Therefore, K8 and K18 were considered to be good candidates to have
CDw75 in their glycan antennae.
Hp, which was identified in two different tissue
specimens in the present study, is an acute-phase protein (a serum
polypeptide synthesized by hepatocytes) (41). Nonetheless, the colorectal epithelium
expression of Hp has also been previously reported (42,43). The
Hp molecule is composed of two α and two β subunits, of which only
β subunits are glycosylated (44).
These reasons led to the selection of Hp as a putative
CDw75-bearing protein.
Although it is likely that Hp, K8 and K18 are not
the only proteins that have the CDw75 antigen (in fact, the 48 kDa
CDw75 band present in differentiated Caco-2 cells does not coincide
with any of the probed proteins by 1D WB), the existing knowledge
on their glycosylation status, tissue expression and the
association of Hp, K8 and K18 with CRC pathogenesis led us to the
continuation of their characterization in the present study. The WB
screening revealed that the expression pattern of Hp, K8 and K18
was partially coincident in molecular mass with some of the
CDw75-positive bands developed on the same blots.
The unique and wide Hp band observed in WB suggests
the presence of one polypeptide, but with high microheterogeneity
(possibly due to several glycoforms). Furthermore, a part of the
broad band was coincident with CDw75 signals. However, Hp cannot be
the only protein that bears this epitope as there are heavier bands
present in immunoblots of CDw75 compared with immunoblots of Hp. In
previous studies, the sequence Neu5Ac-Gal-GlcNAc has been detected
among the multiple carbohydrate determinants that are attached to
serum Hp (45,46). Given the complex histological
composition of the CRC specimens employed in the present study, and
the fact that it was not possible to detect any signal of the
protein in Caco-2 and HT-29 cell lines by WB, the exact origin of
the Hp detected by MS cannot be elucidated, although, as stated
previously the protein is known to be expressed in the colorectal
epithelium (42,43). In this sense, further evidence is
necessary to assess a potential CDw75-glycosylated form of
colorectal Hp.
In the present study, the K8/K18 band pattern in
immunoblots appeared almost identical to that of CDw75 in the same
CRC specimens, with a similar relative abundance of positive bands
in the subcellular fractions compared with the epitope. Regarding
CRC cell lines, the unique K8 and K18 bands were also coincident
with one of the CDw75-positive bands. These proteins are the main
keratin pair in simple epithelia, such as the colorectal
epithelium, where K8 and K18 are crucial for tissue homeostasis
(47). In addition, there are
numerous evidence for abnormalities of K8/K18 expression in CRC
(48–52). Despite K8 and K18 being traditionally
described as cytoskeletal proteins, several studies have reported
the presence of both keratins on the cell membrane in breast
carcinoma (53), K18 in healthy and
tumor hepatocytes (54) and K8 in CRC
cells but not in their healthy counterparts (55). Therefore, K8 and K18 might have a cell
membrane-associated glucide, such as the CDw75 epitope. In any
case, the presence of glycoproteins in the cytoplasm and even in
the nucleus is well-documented (56,57). In
addition, it is worth mentioning that in the present study the
expression of K8 and CDw75 was associated in healthy and tumor
tissues, whereas the expression of K18 and CDw75 were only
associated in the healthy tissues. Although these findings call for
further research, they also suggest K8/K18 have different
glycosylation dynamics in relation to CRC, which could indicate a
dissimilar implication of these proteins in the colorectal
carcinogenesis. In this sense, it is well known that glycosylation
affects protein functionality (58).
In summary, the present study presents several
direct and indirect evidence that suggest that Hp, K8 and K18 are
CDw75-carrying proteins in colorectal tissue. All of these proteins
were identified due to their coincidence in molecular mass and
isoelectric point with CDw75-positive spots in 2D WB. Furthermore,
these proteins are expressed in the colorectal epithelium and are
known to be glycosylated. Since it is unknown whether these
proteins normally bear this epitope or it is the result of the
malignant transformation of the colorectal epithelium, more studies
are necessary to unveil the role of Hp, K8 and K18 in the
development and progression of CRC.
Acknowledgements
The present study was funded by the Consellería de
Cultura, Educación e Ordenación Universitaria (Xunta de Galicia,
Spain) and FEDER (European Union): CINBIO (grant number ED431G/02,
2016–2019), XUGA (grant no. PGIDIT02BTF30101PR) and Research Groups
Program (grant no. 2007/00035-0). The funding sources had no
involvement in any step of the present study. The authors of the
present study wish to thank Dr Craig Murdoch (School of Clinical
Dentistry, University of Sheffield, UK), for his donation of the
Caco-2 and HT-29 cell lines and for his technical advice in
relation to cell culture.
An abstract containing part of this study was
presented at the II International Congress on Analytical
Proteomics, held in Ourense (Spain) on the 18th-20th July 2011. The
abstract was included in the II ICAP 2011-Book of abstracts as
contribution no. P14, pp 160.
Glossary
Abbreviations
Abbreviations:
|
CRC
|
colorectal cancer
|
|
1D
|
one-dimensional
|
|
2D
|
two-dimensional
|
|
ERp44
|
endoplasmic reticulum resident protein
44
|
|
GRP-78
|
78 kDa glucose-regulated protein
|
|
Hp
|
haptoglobin
|
|
HSP70-1A
|
heat shock 70 kDa protein 1A
|
|
K8/K18
|
keratin 8/18
|
|
LRMP
|
lymphoid-restricted membrane
protein
|
|
MS
|
mass spectrometry
|
|
RPSA
|
40S ribosomal protein SA
|
|
ST6Gal I
|
β-galactoside α(2,6)
sialyltransferase
|
|
WB
|
western blotting
|
|
PVDF
|
polyvinylidene difluoride
|
References
|
1
|
Eichler W: Characteristics of two
CD75-related cell-surface expressed antigens of human lymphocytes.
Mol Immunol. 44:2047–2055. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Carbone A and Gloghini A: CD75: A B-cell
marker which should not be forgotten in lymphocyte predominant
Hodgkin lymphoma. Am J Hematol. 89:4492014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Harduin-Lepers A, Vallejo-Ruiz V,
Krzewinski-Recchi MA, Samyn-Petit B, Julien S and Delannoy P: The
human sialyltransferase family. Biochimie. 83:727–737. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dall'Olio F: The
sialyl-alpha2,6-lactosaminyl structure: Biosynthesis and functional
role. Glycoconj J. 17:669–676. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nitschke L: The role of CD22 and other
inhibitory co-receptors in B-cell activation. Curr Opin Immunol.
17:290–297. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stamenkovic I, Sgroi D, Aruffo A, Sy MS
and Anderson T: The B-lymphocyte adhesion molecule CD22 interacts
with leukocyte common antigen CD45RO on T cells and alpha 2–6
sialyltransferase, CD75, on B cells. Cell. 66:1133–1144. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sgroi D, Varki A, Braesch-Andersen S and
Stamenkovic I: CD22, a B cell-specific immunoglobulin superfamily
member, is a sialic acid-binding lectin. J Biol Chem.
268:7011–7018. 1993.PubMed/NCBI
|
|
8
|
Hennet T, Chui D, Paulson J and Marth JD:
Immune regulation by the ST6Gal sialyltransferase. Proc Natl Acad
Sci USA. 95:pp. 4504–4509. 1998; View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dorken B, Moller P, Pezzutto A,
Schwartz-Albiez R and Moldenhauer G: B-cell antigens:
CDw75Leukocyte Typing IV. White Cell Differentiation Antigens.
Knapp W, Dorken B, Gilks WR, Rieber EP, Schmidt RE, Stein H and von
den Borne AEPGK: Oxford University Press; Oxford: pp. 109–110.
1989
|
|
10
|
Reed W, Erikstein BK, Funderud S, Lilleng
R, Tvedt K and Nesland JM: CDw75 antigen expression in breast
lesions. Path Res Pract. 189:394–398. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shen L, Li HX, Luo HS, Shen ZX, Tan SY,
Guo J and Sun J: CDw75 is a significant histopathological marker
for gastric carcinoma. World J Gastroenterol. 10:1682–1685. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Distler U, Souady J, Hülsewig M,
Drmić-Hofman I, Haier J, Denz A, Grützmann R, Pilarsky C, Senninger
N, Dreisewerd K, et al: Tumor associated CD75s and
CD75s-gangliosides are potential targets for adjuvant therapy in
pancreatic cancer. Mol Cancer Ther. 7:2464–2475. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dall'Olio F, Malagolini N, Di Stefano G,
Ciambella M and Serafini-Cessi F: Alpha 2,6 sialylation of
N-acetyllactosaminic sequences in human colorectal cancer cell
lines. Relationship with non-adherent growth. Int J Cancer.
47:291–297. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhu Y, Srivatana U, Ullah A, Gagneja H,
Berenson CS and Lance P: Suppression of a sialyltransferase by
antisense DNA reduces invasiveness of human colon cancer cells in
vitro. Biochim Biophys Acta. 1536:148–160. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chiricolo M, Malagolini N, Bonfiglioli S
and Dall'Olio F: Phenotypic changes induced by expression of
beta-galactoside alpha2,6 sialyltransferase I in the human colon
cancer cell line SW948. Glycobiology. 16:146–154. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Swindall AF, Londoño-Joshi AI, Schultz MJ,
Fineberg N, Buchsbaum DJ and Bellis SL: ST6Gal-I protein expression
is upregulated in human epithelial tumors and correlates with stem
cell markers in normal tissues and colon cancer cell lines. Cancer
Res. 73:2368–2378. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Swindall AF and Bellis SL: Sialylation of
the Fas death receptor by ST6Gal-I provides protection against
Fas-mediated apoptosis in colon carcinoma cells. J Biol Chem.
286:22982–22990. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dall'Olio F, Malagolini N, di Stefano G,
Minni F, Marrano D and Serafini-Cessi F: Increased CMP-NeuAc: Gal
beta 1,4GlcNAc-R alpha 2,6 sialyltransferase activity in human
colorectal cancer tissues. Int J Cancer. 44:434–439. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Vázquez-Martín C, Gil-Martín E and
Fernández-Briera A: Elevation of ST6Gal I activity in malignant and
transitional tissue in human colorectal cancer. Oncology.
69:436–444. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gessner P, Riedl S, Quentmaier A and
Kemmner W: Enhanced activity of CMP-neuAc: Gal beta 1-4GlcNAc:alpha
2,6-sialyltransferase in metastasizing human colorectal tumor
tissue and serum of tumor patients. Cancer Lett. 75:143–149. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Elpek GO, Gelen T, Karpuzoğlu G,
Karpuzoğlu T, Aksoy NH and Keles N: Clinicopathologic evaluation of
CDw75 antigen expression in colorectal adenocarcinomas. Pathol
Oncol Res. 8:175–182. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Costa-Nogueira C, Villar-Portela S, Cuevas
E, Gil-Martín E and Fernández-Briera A: Synthesis and expression of
CDw75 antigen in human colorectal cancer. BMC Cancer. 9:4312009.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Villar-Portela S, Muinelo-Romay L, Cuevas
E, Gil-Martín E and Fernández-Briera A: Disease-free survival of
colorectal cancer patients in relation to CDw75 antigen expression.
Pathobiology. 78:201–209. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lopez LC, Maillet CM, Oleszkowicz K and
Shur BD: Cell surface and Golgi pools of
beta-1,4-galactosyltransferase are differentially regulated during
embryonal carcinoma cell differentiation. Mol Cell Biol.
9:2370–2377. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Smets LA and Van Beek WP: Carbohydrates of
the tumour cell surface. Biochim Biophys Acta. 738:237–249.
1984.PubMed/NCBI
|
|
26
|
Ramagli LS and Rodriguez L: Quantitation
of microgram amounts of protein in two-dimensional polyacrylamide
gel electrophoresis sample buffer. Electrophoresis. 6:559–563.
1985. View Article : Google Scholar
|
|
27
|
Rappsilber J, Ryder U, Lamond AI and Mann
M: Large-scale proteomic analysis of the human spliceosome. Genome
Res. 12:1231–1245. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang X and Zheng PS: Expression and
significance of CK7 and CK19 in colon cancer. Xi Bao Yu Fen Zi Mian
Yi Xue Za Zhi. 26:157–158. 2010.(In Chinese). PubMed/NCBI
|
|
29
|
Wu C, Jin X, Tsueng G, Afrasiabi C and Su
AI: BioGPS: Building your own mash-up of gene annotations and
expression profiles. Nucl Acids Res. 44:D313–D316. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
DiGiacomo V and Meruelo D: Looking into
laminin receptor: Critical discussion regarding the non-integrin
37/67-kDa laminin receptor/RPSA protein. Biol Rev Camb Philos Soc.
91:288–310. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chang YJ, Chen WY, Huang CY, Liu HH and
Wei PL: Glucose-regulated protein 78 (GRP78) regulates colon cancer
metastasis through EMT biomarkers and the NRF-2/HO-1 pathway. Tumor
Biol. 36:1859–1869. 2015. View Article : Google Scholar
|
|
32
|
Black JD and Rezvani K: Heat shock protein
70s as potential molecular targets for colon cancer therapeutics.
Curr Med Chem. 23:3171–3188. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chang Y, Wu Y, Liu W and Ji G: Knockdown
of ERp44 leads to apoptosis via activation of ER stress in HeLa
cells. Biochem Biophys Res Commun. 463:606–611. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Weber J and Senior AE: ATP synthesis
driven by proton transport in F1F0-ATP synthase. FEBS Lett.
545:61–70. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mitchell P: Protonmotive redox mechanism
of the cytochrome b-c1 complex in the respiratory chain:
Protonmotive ubiquinone cycle. FEBS Lett. 56:1–6. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hong S and Pedersen PL: ATP synthase and
the actions of inhibitors utilized to study its roles in human
health, disease, and other scientific areas. Microbiol Mol Biol
Rev. 72:590–641. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xia D, Esser L, Tang WK, Zhou F, Zhou Y,
Yu L and Yu CA: Structural analysis of cytochrome bc1 complexes:
Implications to the mechanism of function. Biochim Biophys Acta.
1827:1278–1294. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Moll R, Divo M and Langbein L: The human
keratins: Biology and pathology. Histochem Cell Biol. 129:705–733.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fuchs E and Cleveland DW: A structural
scaffolding of intermediate filaments in health and disease.
Science. 279:514–519. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chou CF, Smith AJ and Omary MB:
Characterization and dynamics of O-linked glycosylation of human
cytokeratin 8 and 18. J Biol Chem. 267:3901–3906. 1992.PubMed/NCBI
|
|
41
|
Giblett ER: The haptoglobin system. Ser
Haematol. 1:3–2012. 1968.
|
|
42
|
Uhlén M, Björling E, Agaton C, Szigyarto
CA, Amini B, Andersen E, Andersson AC, Angelidou P, Asplund A,
Asplund C, et al: A human protein atlas for normal and cancer
tissues based on antibody proteomics. Mol Cell Proteomics.
4:1920–1932. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Harvey S, Kohga S, Sait SN, Markus G, Hurd
TC, Martinick M, Geradts J, Saxena R and Gibbs JF: Co-expression of
urokinase with haptoglobin in human carcinomas. J Surg Res.
152:189–197. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Nakano M, Nakagawa T, Ito T, Kitada T,
Hijoka T, Kasahara A, Tajiri M, Wada Y, Taniguchi N and Miyoshi E:
Site-specific analysis of N-glycans on haptoglobin in sera of
patients with pancreatic cancer: A novel approach for the
development of tumor markers. Int J Cancer. 122:2301–2309. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Okuyama N, Ide Y, Nakano M, Nakagawa T,
Yamanaka K, Moriwaki K, Murata K, Ohigashi H, Yokoyama S, Eguchi H,
et al: Fucosylated haptoglobin is a novel marker for pancreatic
cancer: A detailed analysis of the oligosaccharide structure and a
possible mechanism for fucosylation. Int J Cancer. 118:2803–2808.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Park SY, Yoon SJ, Jeong YT, Kim JM, Kim
JY, Bernert B, Ullman T, Itzkowitz SH, Kim JH and Hakomori SI:
N-glycosylation status of beta-haptoglobin in sera of patients with
colon cancer, chronic inflammatory diseases and normal subjects.
Int J Cancer. 126:142–155. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Majumdar D, Tiernan JP, Lobo AJ, Evans CA
and Corfe BM: Keratins in colorectal epithelial function and
disease. Int J Exp Pathol. 93:305–318. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Modjtahedi N, Frebourg T, Fossar N,
Lavialle C, Cremisi C and Brison O: Increased expression of
cytokeratin and ferritin-H genes in tumorigenic clones of the SW
613-S human colon carcinoma cell line. Exp Cell Res. 201:74–82.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mouat MF, Kolli K, Orlando R, Hargrove JL
and Grider A: The effects of quercetin on SW480 human colon
carcinoma cells: A proteomic study. Nutr J. 4:112005. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Knösel T, Emde V, Schlüns K, Schlag PM,
Dietel M and Petersen I: Cytokeratin profiles identify diagnostic
signatures in colorectal cancer using multiplex analysis of tissue
microarrays. Cell Oncol. 28:167–175. 2006.PubMed/NCBI
|
|
51
|
Polley AC, Mulholland F, Pin C, Williams
EA, Bradburn DM, Mills SJ, Mathers JC and Johnson IT: Proteomic
analysis reveals field-wide changes in protein expression in the
morphologically normal mucosa of patients with colorectal
neoplasia. Cancer Res. 66:6553–6562. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Koelink PJ, Sier CF, Hommes DW, Lamers CB
and Verspaget HW: Clinical significance of stromal apoptosis in
colorectal cancer. Br J Cancer. 101:765–773. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Godfroid E, Geuskens M, Dupressoir T,
Parent I and Szpirer C: Cytokeratins are exposed on the outer
surface of established human mammary carcinoma cells. J Cell Sci.
99:595–607. 1991.PubMed/NCBI
|
|
54
|
Wells MJ, Hatton MW, Hewlett B, Podor TJ,
Sheffield WP and Blajchman MA: Cytokeratin 18 is expressed on the
hepatocyte plasma membrane surface and interacts with
thrombin-antithrombin complexes. J Biol Chem. 272:28574–28581.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gires O, Andratschke M, Schmitt B, Mack B
and Schaffrik M: Cytokeratin 8 associates with the external leaflet
of plasma membranes in tumor cells. Biochem Biophys Res Commun.
328:1154–1162. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hart GW, Haltiwanger RS, Holt GD and Kelly
WG: Glycosylation in the nucleus and cytoplasm. Annu Rev Biochem.
58:841–874. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Greis KD and Hart GW: Nuclear and
cytoplasmic glycoproteinsGlycoproteins II. Montreuil J,
Vliegenthart JFG and Schachter H: Elsevier Science; Amsterdam: pp.
33–51. 1997, View Article : Google Scholar
|
|
58
|
Lee HS, Qi Y and Im W: Effects of
N-glycosylation on protein conformation and dynamics: Protein Data
Bank analysis and molecular dynamics simulation study. Sci Rep.
5:89262015. View Article : Google Scholar : PubMed/NCBI
|