Introduction
Cancer stem cells (CSCs), or tumor-initiating cells,
are a rare population of tumor cells with stem cell-like
characteristics and self-renewing abilities that are responsible
for cancer maintenance (1,2). CSCs are tumorigenic and metastatic, and
are more resistant to chemotherapy and radiotherapy compared with
differentiated tumor cells (3,4).
Furthermore, CSCs persist in tumors, resulting in relapse following
chemotherapy. Thus, developing novel target drugs in order to
diminish CSC resistance to chemotherapy or radiotherapy may result
in improving therapeutic efficacy and patient prognosis (4).
Most cancer-associated mortalities are caused by
metastasis (5), and numerous
processes and molecules are involved in this malignant phenotype.
Cancer cells disseminate from the primary site to another organ
site via the blood, the lymphatic system or seeding. When
metastasis is initiated, epithelial cells lose their cell-cell
adhesion capacity and cell polarity, and gain migratory and
invasive properties, forming mesenchymal cells, a process termed
the epithelial-mesenchymal transition (EMT) (6). EMT is characterized by the
downregulation of epithelial markers including epithelial
(E)-cadherin, cytokeratin (CK)-8, CK-18, CK-19, claudins, occludins
and the upregulation of mesenchymal markers including neural
(N)-cadherin, fibronectin, vimentin and tenascin C (6). EMT is also regulated by transcription
factors including Twist family BHLH transcription factor 1, Snail
family transcriptional repressor (Snail)1, Snail2 (also known as
Slug), Zinc finger E-Box binding homeobox (ZEB)1 and ZEB2 (7). EMT is a crucial process which
contributes to the development of drug resistance and cancer
stemness, and the expression level of EMT-associated markers
predicts patient prognosis and therapeutic effectiveness (8–10).
Oral cancer is one of the most frequently occurring
cancers globally, and is particularly prevalent in Southeast Asia
(11). Surgery, radiotherapy,
chemotherapy or combinations of these methods are the main
therapeutic methods used for oral cancer (12). However, the 5-year survival rates of
patients with stage I, II, III and IV oral cavity cancer are
72–90%, 39–85%, 27–70% and 12–50%, respectively (13–15). The
overall prognosis of advanced-stage oral cancer has not
substantially improved in the past two decades (13,15).
Therefore, identifying a novel therapeutic method is
imperative.
Piperlongumine (PL), a natural product of the plant
Piper longum, is a potent anti-inflammatory,
antiatherosclerotic and antitumor agent. PL suppresses the
production of tumor necrosis factor-α and interleukin-6, and
inhibits the activation of nuclear factor-κB (NF-κB) against
proinflammatory responses (16,17).
Furthermore, PL prevents plaque formation and inhibits vascular
smooth muscle cell migration and invasion via the inhibition of
NF-κB activation in atherosclerotic lesions (18). The selective tumoricidal effect of PL
against cancer cells, but not normal cells, has been demonstrated
in various types of cancer including breast cancer, hepatocellular
carcinoma, colon cancer, leukemia, glioblastoma, head and neck
cancer and prostate cancer (19–22). PL
induces the accumulation of reactive oxygen species (ROS),
resulting in the apoptosis of cancer cells (19,23–25). This
occurs through various mechanisms including the activation of
caspases, the inhibition of cyclin dependent kinase (CDK)1, CDK2
and cyclin D1, and the downregulation of antiapoptotic genes
including B-cell lymphoma 2 (BCL2), Raf-1 proto-oncogene,
serine/threonine kinase and surviving (25,26). In
addition, PL inactivates the phosphatidylinositol-3-kinase/protein
kinase B (Akt)/mammalian target of rapamycin, mitogen-activated
protein kinase 14/c-Jun N-terminal kinase, NF-κB, and signal
transducer and activator of transcription 3 (STAT3) pathways
(20,21,25). These
pathways serve functions in the regulation of self-renewal and
differentiation of CSCs (27–29).
The present study examined whether PL regulates the
properties of CSCs which are associated with tumor malignancy and
the therapeutic resistance of cancer cells. It was demonstrated
that PL suppressed CSC-forming ability and the expression of the
CSC-associated markers SRY-box 2 (SOX2), POU class 5 homeobox 1
(Oct-4), and Nanog homeobox (NANOG). PL inhibited cell migration
and invasion, and regulated EMT, in addition to increasing chemo-
and radiosensitivity and inhibiting tumor growth in vitro
and in vivo. These results suggest that PL inhibits numerous
malignant phenotypes via the suppression of cancer stemness in oral
cancer.
Materials and methods
Cell culture
The oral cancer cell lines SAS and CGHNC8 (30) were used. SAS cells were kindly
provided by Professor S.C. Lin, Yang Ming University (Taipei,
Taiwan) and CGHNC8 cells were derived from oral cancer squamous
cell carcinomas of patients from Chang Gung Memorial Hospital
(Taoyuan, Taiwan). These cells were cultured in Dulbecco's modified
Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS;
Biological Industries, Cromwell, CT, USA) and antibiotics (100 U/ml
penicillin, 100 U/ml streptomycin and 0.25 g/ml amphotericin B)
(Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C in 5%
CO2.
Chemicals and antibodies
PL (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany)
was dissolved in 100% dimethyl sulfoxide stock solution and
subsequently diluted using the culture medium. The following
primary antibodies were used in the present study: Anti-E-cadherin
(cat. no. 24E10, dilution, 1:1,000), anti-N-cadherin (cat. no.
D4R1HN, dilution, 1:1,000), anti-vimentin (cat. no. D21H3,
dilution, 1:1,000; Cell Signaling Technology, Inc., Danvers, MA,
USA), anti-Snail (cat. no. SC-28199, dilution, 1:1,000), anti-Slug
(cat. no. SC-10436, dilution, 1:1,000), anti-Oct-4 (cat. no.
SC-9081, dilution, 1:1,000), and anti-CK18 (cat. no. SC-6259,
dilution, 1:1,000; Santa Cruz Biotechnology, Inc., Dallas, TX,
USA), anti-NANOG (cat. no. ab109250, dilution, 1:1,000; Abcam,
Cambridge, UK), anti-SOX2 (cat. no. AB5603, dilution, 1:1,000; EMD
Millipore, Billerica, MA, USA), and anti-GAPDH (cat. no. GTX100118,
dilution, 1:8,000; GeneTex, Inc., Irvine, CA, USA).
Tumor sphere formation
For tumor sphere formation, cells were cultured in
DMEM/nutrient mixture F-12 (F12) serum-free medium (Gibco; Thermo
Fisher Scientific, Inc.) containing 10 ng/ml epidermal growth
factor, 10 ng/ml human recombinant basic fibroblast growth factor,
and B27 supplement (Invitrogen; Thermo Fisher Scientific, Inc.). A
diluted cell suspension (3,000 cells/ml) was seeded in ultralow
attachment plates (Corning Incorporated, Corning, NY, USA) then
incubated at 37°C for 2 weeks and fresh medium was added to each
well every 4 days. After 2 weeks, the tumor spheres were visualized
and enumerated through a Nikon ECLIPSE Ti inverted light
microscope.
Cell migration and invasion
assays
Cell migration ability was evaluated using an in
vitro wound healing assay. Cells were seeded in
ibidi® culture inserts (Applied BioPhysics, Inc., Troy,
NY, USA) on 6-well plates. Following incubation at 37°C for 8 h,
the culture insert was detached, forming a 500 µm cell-free gap in
the cell monolayer. Subsequently, the medium was replaced with new
medium containing 1% FBS and 2.5 and 5 µM PL, and cell migration
towards the gap area was photographed every 5 h by Nikon ECLIPSE Ti
inverted light microscope. The cell invasion assay was performed
using BD BioCoat Matrigel invasion chambers (BD Biosciences,
Franklin Lakes, NJ, USA) and Millicell invasion chambers (EMD
Millipore). The membrane of the Millicell upper chamber insert
(pore size, 8 mm) was placed in a 24-well plate and coated with
Matrigel. Cells in DMEM with 1% FBS were seeded in the upper
chambers. The lower chambers contained DMEM with 10% FBS in order
to trap the invading cells. Cells (1×105) were seeded in
the upper chambers, with 2.5 and 5 µM concentrations of PL for 24 h
and incubated at 37°C. Cell invasion ability was determined by
staining the cells with crystal violet for 30 min at room
temperature that successfully passed through the Matrigel-coated
membrane to the opposite chamber and invaded cells was photographed
by Nikon ECLIPSE Ti light microscope.
Radio- and chemosensitivity
assays
To examine whether PL serves as a radiosensitizer, a
clonogenic assay was conducted. A total of 103 cells
were seeded in 6-well dishes and incubated for 16 h. Following
treatment with 0.625 µM PL for 24 h at 37°C, the cells were exposed
to 2 Gy of radiation. Subsequently, the medium was replaced with
DMEM with 10% FBS. The cells were incubated at 37°C for 5–7 days
and stained using crystal violet for 30 min at room temperature.
The results of the radiosensitivity assay were similar to those of
the clonogenic survival assay. The number of surviving colonies
(defined as a colony with ≥50 cells) were counted. The survival
percentage was calculated using the following equation: Survival
percentage = (number of colonies of treated cells)/(number of
colonies of control cells) ×100. The colonies were counted using
ImageJ software (version 2.0; National Institutes of Health,
Bethesda, MD, USA). To determine chemosensitivity, a total of 3,000
cells/well were seeded in 96-well plates. After 16 h, the medium
was removed, and 0.03 µM 5-fluorouracil (5-FU) (Sigma-Aldrich;
Merck KGaA) was added alone or in combination with 2.5 µM PL in the
DMEM medium. After 48 h incubated at 37°C, cell viability was
determined using the MTS assay (Promega Corporation, Madison, WI,
USA) according to the manufacturer's protocol and measured the
absorbance at 490 nm. The relative cell viability was calculated
using the following equation: Cell viability = (absorbance of
treated cells)/(absorbance of control cells) ×100.
Cell growth and colony formation
assays
Cells were seeded at a density of 5×104
in 6-well plates for 16 h and subsequently treated with 5 µM PL for
24 h and 48 h at 37°C. The extent of cell growth was determined
daily using a hemocytometer. For colony formation analysis,
103 cells were seeded in 6-well plates for 16 h, treated
with 0.625 and 2.5 µM PL, and allowed to grow uninterrupted for 7
days at 37°C in DMEM containing 20% FBS. Cell colonies were counted
following staining with 5% crystal violet for 15 min at room
temperature.
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted using the Quick-RNA MiniPrep
kit (Zymo Research Corp., Irvine, CA, USA) according to the
manufacturer's protocol. The concentration, purity, and amount of
total RNA were quantified using the Nano-Drop ND-1000 ultraviolet
spectrophotometer (NanoDrop Technologies; Thermo Fisher Scientific,
Inc.). Reverse transcription was conducted as previously described
(31). qPCR was performed using the
Fast SYBR®-Green Master Mix (Invitrogen; Thermo Fisher
Scientific, Inc.) on the ABI Step One Real-Time PCR system (ABI
Corporation, Lee's Summit, MO, USA), according to the
manufacturer's protocol. The thermal cycling conditions were an
initial denaturation step at 95°C for 10 min, 40 cycles at 95°C for
15 sec and 60°C for 1 min. A comparison RT-PCR method was used to
assay the relative expression of the genes in the control and PL
treated groups, the expression level of each gene was determined
using the ΔΔCq method as described previously (32). The following oligonucleotide sequences
were used: SOX2 forward, 5′-GCCGAGTGGAAACTTTTGTCG-3′ and reverse,
5′-GGCAGCGTGTACTTATCCTTCT-3′; NANOG forward,
5′-TTTGTGGGCCTGAAGAAAACT-3′ and reverse,
5′-AGGGCTGTCCTGAATAAGCAG-3′; Oct-4 forward,
5′-CTTGAATCCCGAATGGAAAGGG-3′ and reverse,
5′-GTGTATATCCCAGGGTGATCCTC-3′; and GAPDH forward,
5′-CGGAGTCAACGGATTTGGTCGTATG-3′ and reverse,
5′-AGCCTTCTCCATGGTGGTGAAGA-3′. GAPDH expression was used as a
reference.
Western blotting
Protein extraction and western blotting were
performed as previously described (33). Cells were collected and lysed in CHAPS
lysis buffer (10 mM Tris, pH 7.4, 1 mM MgCl2, 1 mM EGTA,
150 mM NaCl, 0.5% CHAPS and 10% glycerol; Sigma-Aldrich; Merck
KGaA) containing a protease inhibitor. For protein separation, 30
µg of protein was electrophoresed on 10% sodium dodecyl
sulfate-polyacrylamide gel and transferred onto a polyvinylidene
difluoride membrane (Bio-Rad Laboratories, Inc., Hercules, CA,
USA). The membranes were blocked and subsequently hybridized with
specific primary antibodies, followed by incubation with
horseradish peroxidase-conjugated goat anti-rabbit IgG secondary
antibody (cat. no. 21234; dilution, 1:5,000) or goat anti-mouse IgG
secondary antibody (cat. no. 31430; dilution, 1:5,000) (Invitrogen;
Thermo Fisher Scientific, Inc.).
Proteins were visualized using Western Blotting Plus
Chemiluminescence reagents (Thermo Fisher Scientific, Inc.) and
autoradiography. The density of each protein band was determined
following normalization with the GAPDH control band by using a gel
image system and ImageJ software (version 2.0; National Institutes
of Health).
Mouse xenografts
Animal procedures were ethically approved by the
Institutional Animal Care and Use Committee of Chang Gung
University (Taoyuan, Taiwan) and conformed to the guidelines of the
research council for the care and use of laboratory animals at our
institution. All animals were allowed free access to chlorinated
water and irradiated food. The mice were housed in individually
ventilated cages with a ventilation rate of 65 exchanges of fresh
air per hour, a controlled temperature of 22°C and a 12/12-h
light-dark cycle. The human end points for tumor model was approved
by IACUC in this study included: Tumor volume reaching 2,500
mm3 and more than 10% body weight loss. A total of 5
five-week-old male BALB/cAnN.Cg-Foxn1nu/CrlNarl mice
(18–22 g; National Laboratory Animal Center, Taipei, Taiwan) were
used, and 106 SAS cells were subcutaneously injected
into the upper portion of the hind limb. After 7 days, PL (2.4
mg/kg) was intraperitoneally administered daily for 13 consecutive
days. The tumor size was monitored, and the volume was calculated
using the following equation: Tumor volume: (Short length of the
tumor2 × long length of the tumor)/2. Mice were
sacrificed 6 weeks after the first PL administration, and the
xenograft tumors were removed and weighed. Xenografted tumors were
fixed in 3.7% formaldehyde solution for 24 h at room temperature
and embedded in paraffin. Each sample was counterstained with a
hematoxylin and eosin stain (H&E) according to the
manufacturer's protocol (Zymed; Thermo Fisher Scientific,
Inc.).
Statistical analyses
Student's unpaired t-test was used to compare the
difference between two groups. Statistical comparisons between
multiple groups were performed using a one-way analysis of variance
and Tukey's post hoc test. The results were presented as the mean ±
the standard deviation (SD). All of the figures were plotted using
GraphPad Prism (version 6.0; GraphPad Software Inc., La Jolla, CA,
USA). P<0.05 was considered to indicate a statistically
significant difference.
Results
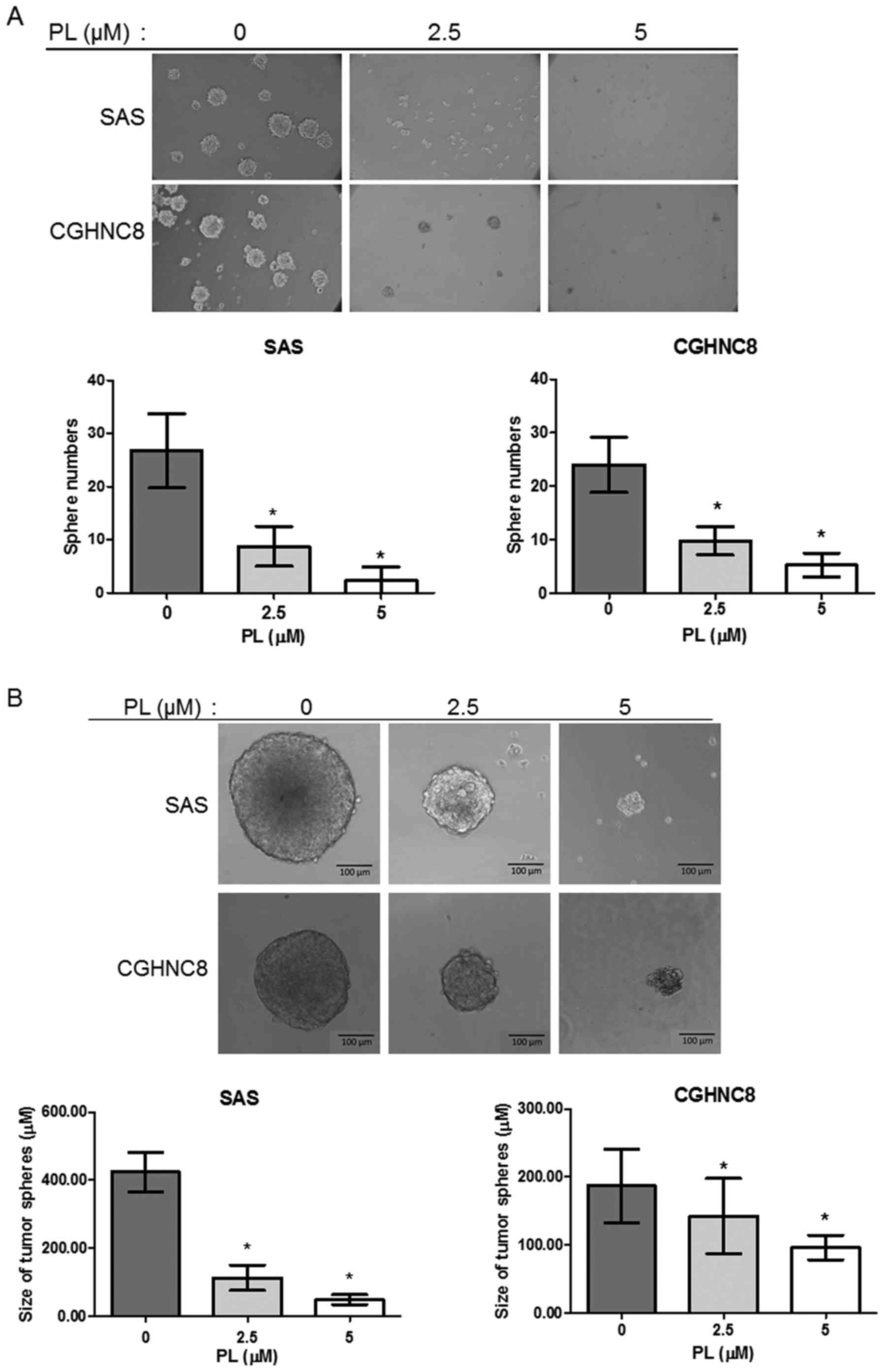
PL inhibits CSC properties
In the present study, a tumorsphere formation assay
was used to investigate the function of PL in regulating CSC
properties following treatment of SAS and CGHNC8 cells with PL
(0.0, 2.5 and 5.0 µM). PL inhibited the number and size of tumor
spheres formed by the tested cell lines in a
concentration-dependent manner. Following treatment with 2.5 and
5.0 µM PL, the number of tumorspheres was reduced to ~67.28 and
91.58%, respectively, compared with control cells, and similar
effects were observed in the CGHNC8 cells (Fig. 1A). The average diameters of the
tumorspheres were 418.33 and 190.88 µm in the control SAS and
CGHNC8 cells, respectively. By contrast, treatment with 5 µM PL
induced the formation of small spheres with diameters of 48.27 and
95.62 µm in the SAS and CGHNC8 cells, respectively (Fig. 1B). Furthermore, the present study
examined the alteration of the expression of CSC-associated genes
and proteins following PL treatment. Transcription factors
including Oct-4, NANOG and SOX2 regulate the pluripotency of
embryonic stem cells, and CK18 participates in cell differentiation
(34,35). Following PL treatment, the mRNA levels
of SOX2, NANOG, and Oct-4 decreased to 42, 60 and 36% respectively
in the SAS cells and to 86, 96 and 87% respectively in the CGHNC8
cells, compared with control cells (Fig.
2A). Furthermore, following PL treatment, the CK18 expression
levels increased 1.6 and 1.8 times in the SAS and CGHNC8 cells,
respectively. Similarly, PL reduced Oct-4, NANOG, and SOX2 protein
levels and increased CK18 protein levels (Fig. 2B). These results suggest that PL
inhibits tumor sphere-forming ability via the suppression of Oct-4,
NANOG, and SOX2 expression and promoting cell differentiation.
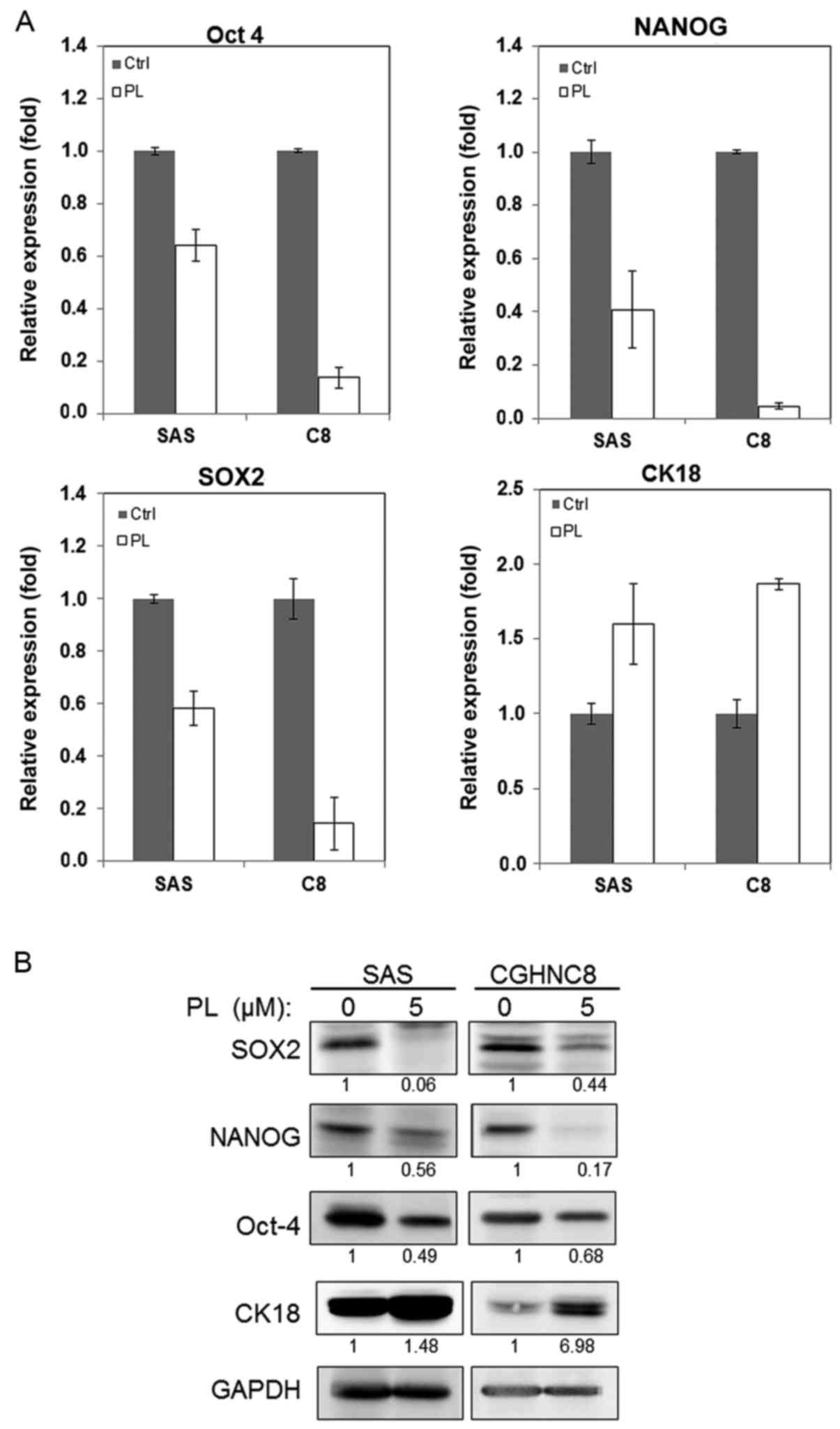 | Figure 2.PL reduced the expression of the
cancer stem cell markers SOX2, Oct-4, and NANOG but increased the
expression of the differentiation marker CK18. SAS and CGHNC8 cells
were treated with 5.0 µM PL for 48 h; subsequently, the cells were
harvested and analyzed. (A) mRNA expression levels of SOX2, NANOG,
and Oct-4 and CK18 were measured using reverse
transcription-quantitative polymerase chain reaction. (B) Cellular
protein levels were determined using western blotting (control
cells, 0.1% DMSO). Experiments were performed in triplicate. PL,
piperlongumine; SOX2, SRY-box 2; Oct-4, POU class 5 homeobox 1;
NANOG, NANOG homeobox; CK18, cytokeratin 18. |
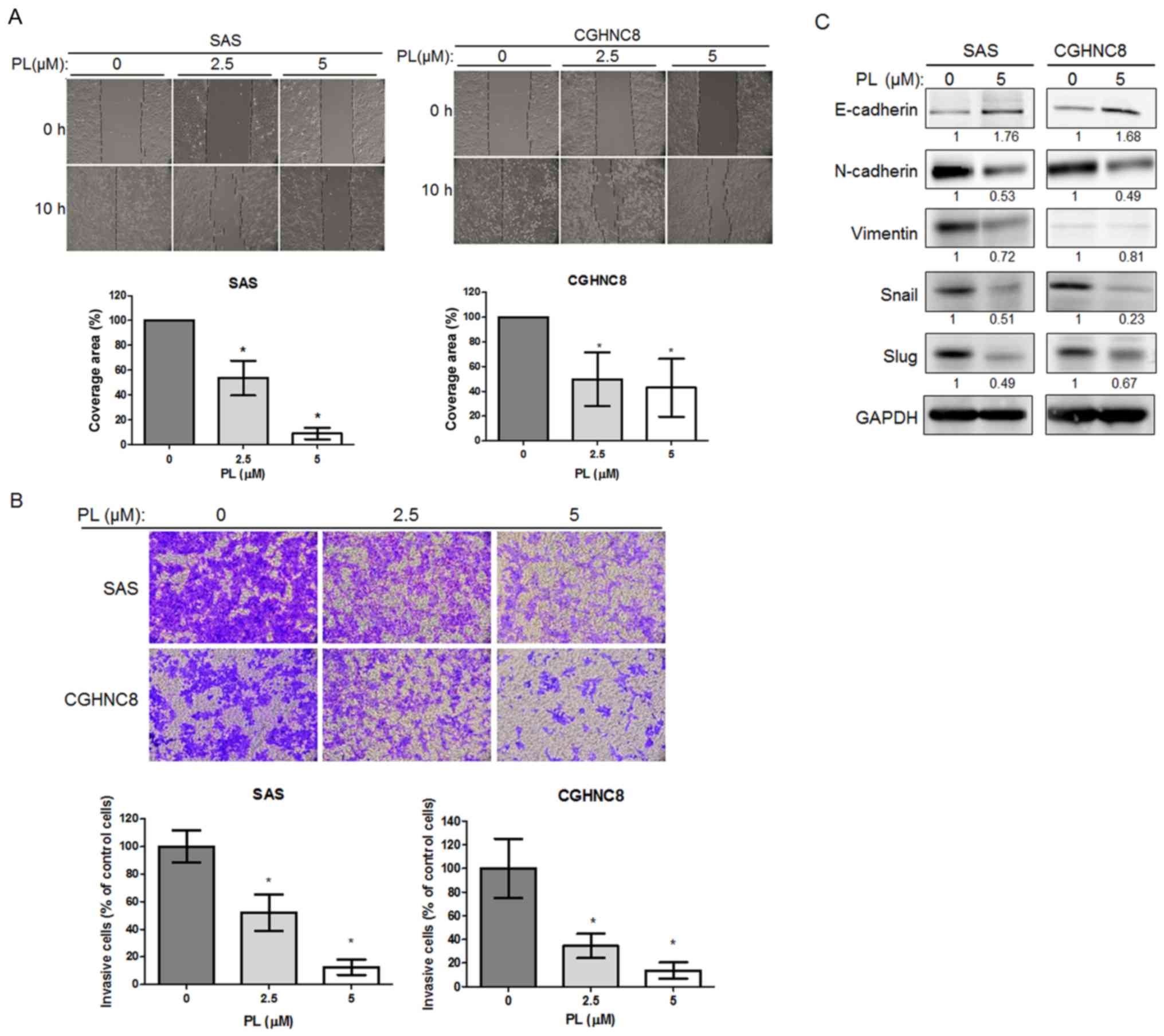
PL reduces cell migration and invasion
ability and EMT
As CSCs are invasive, the present study examined the
potential effect of PL on cell invasion. Cell migration and
invasion were determined using wound healing and Matrigel Transwell
assays, respectively. Although different cell lines migrated at
different rates, the control cells covered the majority of the
wound area; in comparison, fewer PL-treated cells migrated to the
wound area (8% in the SAS cells and 61% in the CGHNC8 cells) after
10 h (Fig. 3A). The Matrigel invasion
assay revealed that after 18 h of PL treatment, the number of
invasive cells for the SAS and CGHNC8 cell lines decreased compared
with that observed for the control cells (Fig. 3B). PL reduced the cell migration and
invasion ability of the two cell lines in a concentration-dependent
manner. EMT is another biological process that enables the
metastasis of tumor cells through the bloodstream. Similar to the
cancer stemness pathway, this process mechanistically initiates
cancer metastasis and confers treatment resistance (36). Therefore, the function that PL serves
in EMT was investigated. Compared with the control cells, the
PL-treated cells exhibited an increased expression level of
E-cadherin but a decreased expression level of N-cadherin and
vimentin. PL reduced the levels of transcription factors, including
Snail and Slug that participate in EMT (Fig. 3C). These results suggest that PL
suppressed cell migration and invasion ability, and so regulated
EMT in oral cancer cells.
PL increases radio- and
chemosensitivity
Surgery, radiotherapy, chemotherapy or combinations
of these methods are the main therapeutic methods for oral cancer.
The main reason for therapeutic failure is acquired resistance,
which is attributed to CSCs. Therefore, whether PL serves as a
sensitizer was investigated in order to improve therapeutic
efficacy. The clonogenic survival assay was used to examine the
radiosensitization effect of PL following the treatment of cells
with PL alone, radiation alone or a combination of PL and
radiation. The radiation sensitivity levels of SAS and CGHNC8 cells
treated with the combination of PL and radiation increased by 47.5
and 25.63%, respectively (Fig. 4A).
5-FU, which is commonly used for treating oral cavity cancers, was
selected to test chemosensitivity. As presented in Fig. 4B, ~60 and 70% of SAS and CGHNC8 cells,
respectively, were killed following treatment with 5-FU for 48 h.
The same effect was observed in PL-treated cells. The SAS and
CGHNC8 cells that were treated with 5-FU and PL exhibited enhanced
drug sensitivity (Fig. 4B). Thus, PL
enhanced the radio- and chemosensitivity levels of oral cancer
cells.
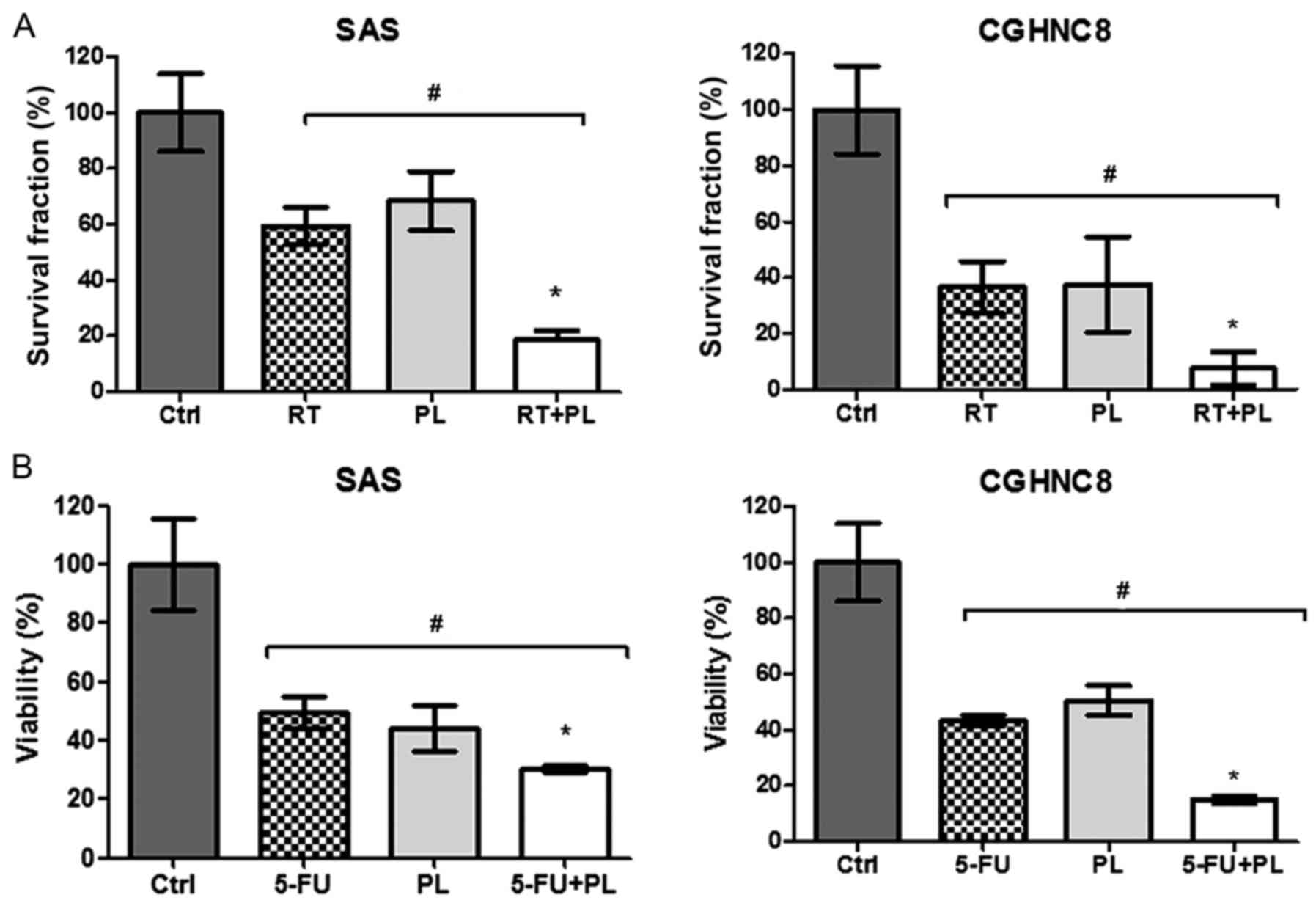 | Figure 4.PL increased the radio- and
chemosensitivity levels of oral cancer cells. (A) Clonogenic
survival assays were used to determine the effect of PL on the
radiosensitivity of SAS and CGHNC8 cells. Cells were treated with
either PL alone (0.625 µM), radiation alone (2 Gy) or a combination
of the two treatments. (B) PL increased chemosensitivity, as
determined by the cytotoxic assay. Cells were treated with 0.03 µM
5-FU alone, 2.50 µM PL alone, or a combination of the two
treatments for 48 h. Cell survival was examined using an MTS assay.
The data of the cytotoxic assay were compared with the data of
control cells. Experiments were performed in triplicate. *P<0.05
vs. the ctrl or RT/5-FU treated groups, #P<0.05 vs.
the RT/5-FU or RT+PL/5-FU+PL treated groups, with comparisons
indicated by lines. PL, piperlongumine; Ctrl, control; RT,
radiotherapy; 5-FU, 5-fluorouracil. |
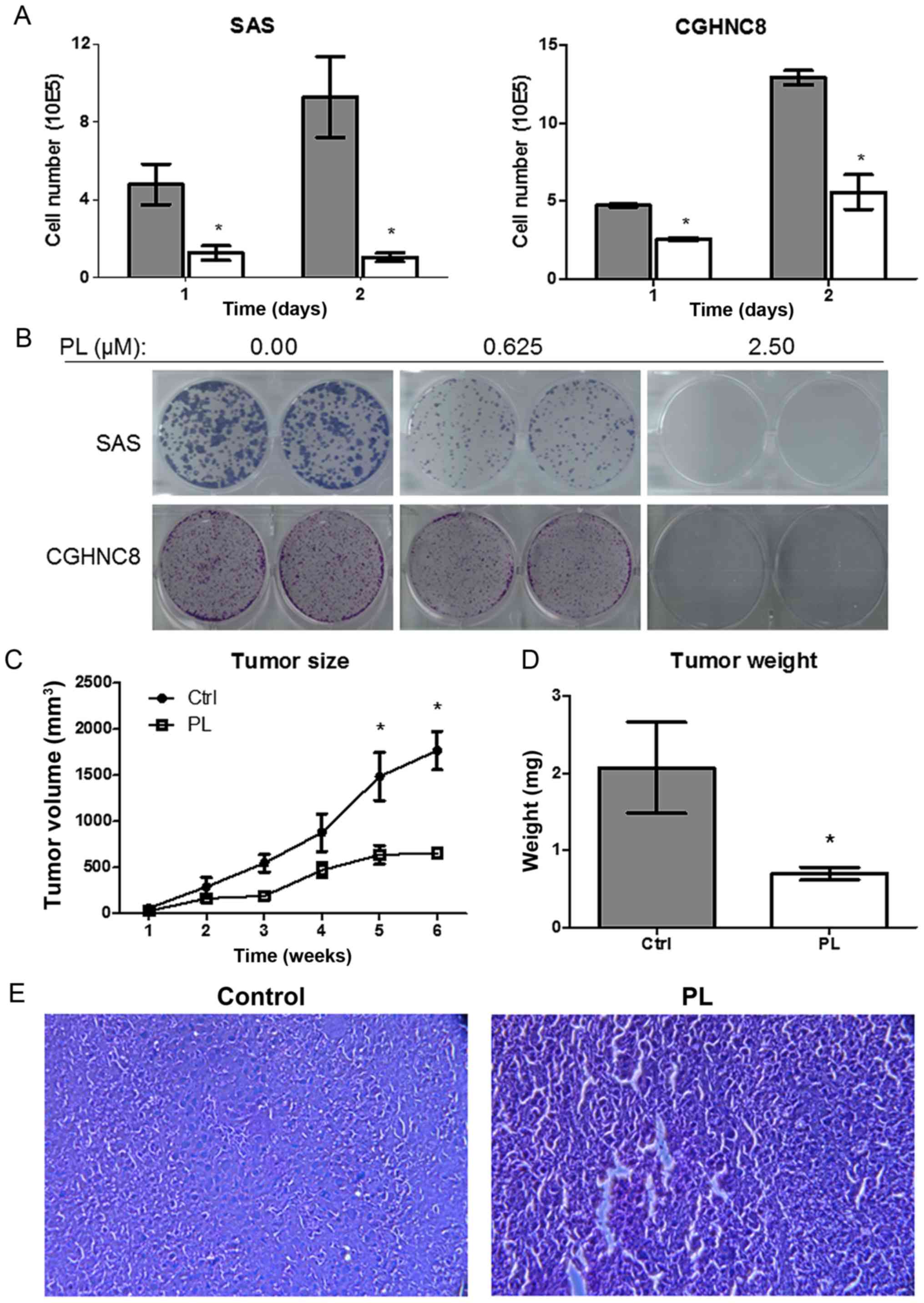
PL inhibits tumor growth ability in
vitro and in vivo
PL induces apoptosis in various types of cancer
(19,20). Therefore, whether PL regulates the
tumor growth ability of oral cancer cells was investigated. PL
treatment (5 µM) for 48 h reduced the growth of the SAS and CGHNC8
cells to 11.2 and 21.9%, respectively, compared with the control
cells (Fig. 5A). The colony formation
assay was used to examine the long-term effect of PL by treating
cells with PL (0.625 and 2.500 µM) for 7 days. Consistently, PL
significantly suppressed colony formation, and no or few colonies
were formed following PL treatment (2.5 µM; Fig. 5B). Furthermore, PL reduced the growth
of oral cancer cells and their colony-forming ability. In order to
investigate the effects of PL on tumor growth in vivo,
xenograft SAS tumors were established in BALB/c nude mice.
Following injection with SAS tumor cells, the mice (5 per treatment
group) were intraperitoneally administered with PL or control
saline and monitored for 6 consecutive weeks. Tumor growth was
significantly slower in the PL-treated groups than in the control
groups (Fig. 5C). On day 42, tumor
growth decreased by 63% in the PL-treated group (P<0.05). Once
the mice were sacrificed, the tumor weight was revealed to be
decreased by 66% in the PL-treated group (Fig. 5D). Tumor tissues were confirmed using
H&E staining (Fig. 5E). The
results indicate that PL inhibited tumor growth in vitro and
in vivo.
Discussion
CSCs serve a pivotal function not only in the
therapy resistance phenotype but also in local recurrence and
metastasis (8). CSCs may generate
tumors through the stem cell processes of self-renewal and
differentiation (4,37). Therefore, eliminating CSCs may provide
a therapeutic strategy for cancer. In the present study, PL was
demonstrated to suppress the tumor sphere-forming ability of the
oral cancer cells SAS and CGHNC8 (Fig.
1) and suppress the expression of the stem cell-associated
genes SOX2, Oct-4, and NANOG. It was demonstrated to increase the
expression of the differentiation gene CK18 (Fig. 2). Furthermore, PL inhibited cell
migration and invasion and regulated the expression of
EMT-associated molecules (Fig. 3). PL
also increased chemo- and radiosensitivity (Fig. 4) and inhibited tumorigenesis in
vitro and in vivo (Fig.
5). Oct-4, NANOG, and SOX2 are essential for early embryonic
development and pluripotency maintenance in embryonic stem cells
(38,39). These factors regulate the cancer
stemness phenotype and are upregulated in multiple types of cancer.
They thus serve a critical function in cancer development and drug
resistance (34,40,41). PL
has been identified as an inhibitor of signal transducer and
activator of transcription 3 (STAT3) from the screening of a
chemical library (22). PL inhibits
STAT3 nuclear translocation, suppresses ligand-induced and
constitutive STAT3 phosphorylation and affects STAT3-regulated gene
expression in breast cancer cells (22). STAT3 is crucial for embryonic stem
cell pluripotency and is essential for the maintenance of inner
cell mass lineages through the downstream effectors Oct-4 and NANOG
(42,43). Therefore, PL may suppress cancer
stemness by inhibiting STAT3. The results of the present study
demonstrate that PL inhibited CSC formation and SOX2, Oct-4 and
NANOG expression, supporting the observation that PL suppresses CSC
properties in oral cancer.
A direct link exists between EMT and CSCs. For
example, EMT may induce a more CSC-like phenotype in various types
of cancer (8,9,44).
Furthermore, CSC markers are associated with tumorigenesis,
metastasis and recurrence. For example, oct-4 is highly expressed
in invasive oral cancer cells, and this expression is accompanied
by alterations in the expression of EMT-associated markers, low
E-cadherin expression and high N-cadherin expression (44). The coexpression of Oct-4 and NANOG
enhances CSC-like properties, modulates STAT3 activation and
subsequently regulates Snail expression and promotes cell
metastatic ability in hepatocellular carcinoma (45). Snail and Slug are the major
transcription factors that repress epithelial marker genes and
activate mesenchymal markers to promote cell mobility. Snail- and
Slug-induced EMT triggers CSC-like phenotypes in multiple types of
cancer (46–49). In the present study, PL was
demonstrated to reduce cancer stemness by inhibiting tumor sphere
formation and suppressing SOX2, Oct-4 and NANOG expression;
however, PL increased the expression of the differentiation marker
CK18 (Figs. 1 and 2). Furthermore, PL inhibited cell mobility,
which was accompanied by the regulation of the expression of
EMT-associated molecules. PL-treated cells exhibited a high
expression of E-cadherin and a low expression of mesenchymal
markers including N-cadherin and vimentin. In addition, PL reduced
the levels of transcription factors Snail and Slug (Fig. 3). PL was demonstrated to suppress
cancer stemness, eliminate EMT processes and suppress the
differential expression of EMT-associated molecules.
For oral cancer therapy, radiotherapy alone may be
used to treat small or early-stage tumors. Radiotherapy is commonly
used following surgery, either alone or in combination with
chemotherapy, in order to treat advanced tumors. ROS are critical
mediators of ionization-induced cell death, and lower ROS levels
have been observed in CSCs exhibiting greater resistance to
radiotherapy (50). Therefore,
modulating ROS levels may be a useful method to improve the
efficacy of radiotherapy. A number of studies have indicated that
PL increases ROS generation in cancer cells, but not in normal
cells, and enhanced ROS levels are associated with cancer cell
death (19,23,51). Thus,
the increased ROS production by PL suggests that PL may serve as a
radiosensitizer in order to improve the therapeutic efficacy of
radiotherapy. In the present study, it was demonstrated that
pretreatment with PL increased the antitumor effect of radiotherapy
(Fig. 4). Similar results were
observed in a breast cancer model. The cell apoptosis rate
increased following combined treatment of PL with radiation, and
the level of intracellular ROS was higher in the combination group
than in the group treated with PL or radiation alone (17). These results may be attributed to the
decreased expression level of BCL2 and the increased expression
level of BCL2 associated X, apoptosis regulator.
Drug resistance may be acquired due to the presence
of CSCs and elevated CSC markers (3,4,40). PL has been demonstrated to increase
5-FU sensitivity in oral cancer (Fig.
4). PL exhibits a synergistic effect with various
chemotherapeutic drugs, including cisplatin and paclitaxel, in
ovarian, head and neck cancer (24,26,52). PL
suppressed the expression of P-glycoprotein, ATP binding cassette
subfamily B member 1, ATP binding cassette subfamily C member 1,
survivin, and phospho-Akt, as well as the transcriptional
activities of NF-κB and TWIST, which reversed doxorubicin
resistance in a breast cancer cell line (53).
In conclusion, PL increases therapeutic sensitivity
and suppresses the malignant phenotype, namely via the suppression
of migration, invasion, EMT and proliferation in vitro and
in vivo by inhibiting cancer stemness and the expression of
stem cell regulatory proteins. This therefore indicates that PL is
a potential therapeutic agent for oral cancer.
Acknowledgements
The present study was supported by the Taipei
Medical University Hospital (grant no. 102TMU-TMUH-06) and the
Ministry of Science and Technology, Taiwan, R.O.C (grant no. MOST
104-2320-B-038-032).
References
|
1
|
Prince ME, Sivanandan R, Kaczorowski A,
Wolf GT, Kaplan MJ, Dalerba P, Weissman IL, Clarke MF and Ailles
LE: Identification of a subpopulation of cells with cancer stem
cell properties in head and neck squamous cell carcinoma. Proc Natl
Acad Sci USA. 104:pp. 973–978. 2007; View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Visvader JE and Lindeman GJ: Cancer stem
cells in solid tumours: Accumulating evidence and unresolved
questions. Nat Rev Cancer. 8:755–768. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rich JN: Cancer stem cells in radiation
resistance. Cancer Res. 67:8980–8984. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Khan IN, Al-Karim S, Bora RS, Chaudhary AG
and Saini KS: Cancer stem cells: A challenging paradigm for
designing targeted drug therapies. Drug Discov Today. 20:1205–1216.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gupta GP and Massagué J: Cancer
metastasis: Building a framework. Cell. 127:679–695. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Thiery JP and Sleeman JP: Complex networks
orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell
Biol. 7:131–142. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sampieri K and Fodde R: Cancer stem cells
and metastasis. Semin Cancer Biol. 22:187–193. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Scheel C and Weinberg RA: Cancer stem
cells and epithelial-mesenchymal transition: Concepts and molecular
links. Semin Cancer Biol. 22:396–403. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Barbazán J, Muinelo-Romay L, Vieito M,
Candamio S, Díaz-López A, Cano A, Gómez-Tato A, Casares de Cal Mde
L, Abal M and López-López R: A multimarker panel for circulating
tumor cells detection predicts patient outcome and therapy response
in metastatic colorectal cancer. Int J Cancer. 135:2633–2643. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Warnakulasuriya S: Global epidemiology of
oral and oropharyngeal cancer. Oral Oncol. 45:309–316. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huang SH and O'Sullivan B: Oral cancer:
Current role of radiotherapy and chemotherapy. Med Oral Patol Oral
Cir Bucal. 18:e233–e240. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chang JT, Chen IH, Liao CT, Wang HM, Hsu
YM, Hung KF, Lin CJ, Hsieh LL and Cheng AJ: A reverse transcription
comparative real-time PCR method for quantitative detection of
angiogenic growth factors in head and neck cancer patients. Clin
Biochem. 35:591–596. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen YJ, Chang JT, Liao CT, Wang HM, Yen
TC, Chiu CC, Lu YC, Li HF and Cheng AJ: Head and neck cancer in the
betel quid chewing area: Recent advances in molecular
carcinogenesis. Cancer Sci. 99:1507–1514. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liao CT, Chang JT, Wang HM, Ng SH, Hsueh
C, Lee LY, Lin CH, Chen IH, Huang SF, Cheng AJ and Yen TC: Analysis
of risk factors of predictive local tumor control in oral cavity
cancer. Ann Surg Oncol. 15:915–922. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee W, Yoo H, Kim JA, Lee S, Jee JG, Lee
MY, Lee YM and Bae JS: Barrier protective effects of
piperlonguminine in LPS-induced inflammation in vitro and in vivo.
Food Chem Toxicol. 58:149–157. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yao L, Chen HP and Ma Q: Piperlongumine
alleviates lupus nephritis in MRL-Fas(lpr) mice by regulating the
frequency of Th17 and regulatory T cells. Immunol Lett. 161:76–80.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Son DJ, Kim SY, Han SS, Kim CW, Kumar S,
Park BS, Lee SE, Yun YP, Jo H and Park YH: Piperlongumine inhibits
atherosclerotic plaque formation and vascular smooth muscle cell
proliferation by suppressing PDGF receptor signaling. Biochem
Biophys Res Commun. 427:349–354. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Raj L, Ide T, Gurkar AU, Foley M, Schenone
M, Li X, Tolliday NJ, Golub TR, Carr SA, Shamji AF, et al:
Selective killing of cancer cells by a small molecule targeting the
stress response to ROS. Nature. 475:231–234. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bezerra DP, Pessoa C, de Moraes MO,
Saker-Neto N, Silveira ER and Costa-Lotufo LV: Overview of the
therapeutic potential of piplartine (piperlongumine). Eur J Pharm
Sci. 48:453–463. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Randhawa H, Kibble K, Zeng H, Moyer MP and
Reindl KM: Activation of ERK signaling and induction of colon
cancer cell death by piperlongumine. Toxicol In Vitro.
27:1626–1633. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Bharadwaj U, Eckols TK, Kolosov M,
Kasembeli MM, Adam A, Torres D, Zhang X, Dobrolecki LE, Wei W,
Lewis MT, et al: Drug-repositioning screening identified
piperlongumine as a direct STAT3 inhibitor with potent activity
against breast cancer. Oncogene. 34:1341–1353. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jin HO, Lee YH, Park JA, Lee HN, Kim JH,
Kim JY, Kim B, Hong SE, Kim HA, Kim EK, et al: Piperlongumine
induces cell death through ROS-mediated CHOP activation and
potentiates TRAIL-induced cell death in breast cancer cells. J
Cancer Res Clin Oncol. 140:2039–2046. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Roh JL, Kim EH, Park JY, Kim JW, Kwon M
and Lee BH: Piperlongumine selectively kills cancer cells and
increases cisplatin antitumor activity in head and neck cancer.
Oncotarget. 5:9227–9238. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shrivastava S, Kulkarni P, Thummuri D,
Jeengar MK, Naidu VG, Alvala M, Redddy GB and Ramakrishna S:
Piperlongumine, an alkaloid causes inhibition of PI3K/Akt/mTOR
signaling axis to induce caspase-dependent apoptosis in human
triple-negative breast cancer cells. Apoptosis. 19:1148–1164. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang XQ, Wang YC, Guo YT and Tang X:
Effect of piperlongumine on drug resistance reversal in human
retinoblastoma HXO-RB44/VCR and SO-Rb50/CBP cell lines. Int J Clin
Exp Pathol. 8:2525–2534. 2015.PubMed/NCBI
|
|
27
|
Bleau AM, Hambardzumyan D, Ozawa T,
Fomchenko EI, Huse JT, Brennan CW and Holland EC: PTEN/PI3K/Akt
pathway regulates the side population phenotype and ABCG2 activity
in glioma tumor stem-like cells. Cell Stem Cell. 4:226–235. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shostak K and Chariot A: NF-κB, stem cells
and breast cancer: The links get stronger. Breast Cancer Res.
13:2142011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Matsuda K, Sato A, Okada M, Shibuya K,
Seino S, Suzuki K, Watanabe E, Narita Y, Shibui S, Kayama T and
Kitanaka C: Targeting JNK for therapeutic depletion of stem-like
glioblastoma cells. Sci Rep. 2:5162012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lu YC, Chen YJ, Wang HM, Tsai CY, Chen WH,
Huang YC, Fan KH, Tsai CN, Huang SF, Kang CJ, et al: Oncogenic
function and early detection potential of miRNA-10b in oral cancer
as identified by microRNA profiling. Cancer Prev Res (Phila).
5:665–674. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen CY, Lin YS, Chen CL, Chao PZ, Chiou
JF, Kuo CC, Lee FP, Lin YF, Sung YH, Lin YT, et al: Targeting
annexin A2 reduces tumorigenesis and therapeutic resistance of
nasopharyngeal carcinoma. Oncotarget. 29:26946–26959. 2015.
View Article : Google Scholar
|
|
32
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen YJ, Lee LY, Chao YK, Chang JT, Lu YC,
Li HF, Chiu CC, Li YC, Li YL, Chiou JF and Cheng AJ: DSG3
facilitates cancer cell growth and invasion through the
DSG3-plakoglobin-TCF/LEF-Myc/cyclin D1/MMP signaling pathway. PLoS
One. 8:e640882013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Guo Y, Liu S, Wang P, Zhao S, Wang F, Bing
L, Zhang Y, Ling EA, Gao J and Hao A: Expression profile of
embryonic stem cell-associated genes Oct4, Sox2 and Nanog in human
gliomas. Histopathology. 59:763–775. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chiu CC, Lee LY, Li YC, Chen YJ, Lu YC, Li
YL, Wang HM, Chang JT and Cheng AJ: Grp78 as a therapeutic target
for refractory head-neck cancer with CD24(−)CD44(+) stemness
phenotype. Cancer Gene Ther. 20:606–615. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Brabletz T: EMT and MET in metastasis:
Where are the cancer stem cells? Cancer Cell. 22:699–701. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Clarke MF, Dick JE, Dirks PB, Eaves CJ,
Jamieson CH, Jones DL, Visvader J, Weissman IL and Wahl GM: Cancer
stem cells-perspectives on current status and future directions:
AACR Workshop on cancer stem cells. Cancer Res. 66:9339–9344. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Boyer LA, Lee TI, Cole MF, Johnstone SE,
Levine SS, Zucker JP, Guenther MG, Kumar RM, Murray HL, Jenner RG,
et al: Core transcriptional regulatory circuitry in human embryonic
stem cells. Cell. 122:947–956. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Loh YH, Wu Q, Chew JL, Vega VB, Zhang W,
Chen X, Bourque G, George J, Leong B, Liu J, et al: The Oct4 and
Nanog transcription network regulates pluripotency in mouse
embryonic stem cells. Nat Genet. 38:431–440. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Tsai LL, Yu CC, Chang YC, Yu CH and Chou
MY: Markedly increased Oct4 and Nanog expression correlates with
cisplatin resistance in oral squamous cell carcinoma. J Oral Pathol
Med. 40:621–628. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ling GQ, Chen DB, Wang BQ and Zhang LS:
Expression of the pluripotency markers Oct3/4, Nanog and Sox2 in
human breast cancer cell lines. Oncol Lett. 4:1264–1268.
2012.PubMed/NCBI
|
|
42
|
Niwa H, Burdon T, Chambers I and Smith A:
Self-renewal of pluripotent embryonic stem cells is mediated via
activation of STAT3. Genes Dev. 12:2048–2060. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Do DV, Ueda J, Messerschmidt DM,
Lorthongpanich C, Zhou Y, Feng B, Guo G, Lin PJ, Hossain MZ, Zhang
W, et al: A genetic and developmental pathway from STAT3 to the
OCT4-NANOG circuit is essential for maintenance of ICM lineages in
vivo. Genes Dev. 27:1378–1390. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Siu A, Lee C, Dang D and Ramos DM: Stem
cell markers as predictors of oral cancer invasion. Anticancer Res.
32:1163–1166. 2012.PubMed/NCBI
|
|
45
|
Yin X, Zhang BH, Zheng SS, Gao DM, Qiu SJ,
Wu WZ and Ren ZG: Coexpression of gene Oct4 and Nanog initiates
stem cell characteristics in hepatocellular carcinoma and promotes
epithelial-mesenchymal transition through activation of Stat3/Snail
signaling. J Hematol Oncol. 8:232015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Fan F, Samuel S, Evans KW, Lu J, Xia L,
Zhou Y, Sceusi E, Tozzi F, Ye XC, Mani SA and Ellis LM:
Overexpression of snail induces epithelial-mesenchymal transition
and a cancer stem cell-like phenotype in human colorectal cancer
cells. Cancer Med. 1:5–16. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
47
|
Guo W, Keckesova Z, Donaher JL, Shibue T,
Tischler V, Reinhardt F, Itzkovitz S, Noske A, Zürrer-Härdi U, Bell
G, et al: Slug and Sox9 cooperatively determine the mammary stem
cell state. Cell. 148:1015–1028. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yasui K, Shimamura M, Mitsutake N and
Nagayama Y: SNAIL induces epithelial-to-mesenchymal transition and
cancer stem cell-like properties in aldehyde
dehydroghenase-negative thyroid cancer cells. Thyroid. 23:989–996.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Masui T, Ota I, Yook JI, Mikami S, Yane K,
Yamanaka T and Hosoi H: Snail-induced epithelial-mesenchymal
transition promotes cancer stem cell-like phenotype in head and
neck cancer cells. Int J Oncol. 44:693–699. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Diehn M, Cho RW, Lobo NA, Kalisky T, Dorie
MJ, Kulp AN, Qian D, Lam JS, Ailles LE, Wong M, et al: Association
of reactive oxygen species levels and radioresistance in cancer
stem cells. Nature. 458:780–783. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yao JX, Yao ZF, Li ZF and Liu YB:
Radio-sensitization by Piper longumine of human breast adenoma
MDA-MB-231 cells in vitro. Asian Pac J Cancer Prev. 15:3211–3217.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Han JG, Gupta SC, Prasad S and Aggarwal
BB: Piperlongumine chemosensitizes tumor cells through interaction
with cysteine 179 of IkappaBalpha kinase, leading to suppression of
NF-κB-regulated gene products. Mol Cancer Ther. 13:2422–2435. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kang Q and Yan S: Piperlongumine reverses
doxorubicin resistance through the PI3K/Akt signaling pathway in
K562/A02 human leukemia cells. Exp Ther Med. 9:1345–1350. 2015.
View Article : Google Scholar : PubMed/NCBI
|